Abstract
BACKGROUND:
Bone metastases affect 50% to 70% of breast cancer (BC) patients and have a high mortality rate. Adipose tissue loss plays a pivotal role in the progression of cancer.
OBJECTIVE:
This study aims to evaluate the prognostic value of adipose tissue for bone metastasis in BC patients.
METHODS:
517 BC patients were studied retrospectively. Patients’ characteristics before the surgery were collected. Quantitative measurements of the subcutaneous fat index (SFI) were performed at the level of the eleventh thoracic vertebra. In order to adjust for the heterogeneity between the low SFI and high SFI groups, propensity score matching (PSM) was used. The Kaplan-Meier method was used to estimate the 5-year bone metastatic incidence. The prognostic analysis was performed with the Cox regression models.
RESULTS:
Compared with the patients without bone metastasis, the patients with bone metastasis had reduced SFI levels. In addition, Kaplan-Meier analysis revealed that patients with low SFI were more likely to develop bone metastases. The independent predictive value of SFI for bone metastases was confirmed by Cox regression analysis. The survival analysis was repeated after PSM with a 1:1 ratio, yielding similar results (
CONCLUSIONS:
SFI is an independent predictor of bone metastasis in BC patients.
Keywords
Introduction
Breast cancer (BC) is the most frequently diagnosed cancer and is the main reason for cancer-related mortality in women [1]. Most of the deaths from BC are due to distant metastases [2]. The bone is the most common site of metastasis, which has been shown to occur in between 50% and 70% of BC patients [3]. Patients with bone metastasis are accompanied by excessive, osteoclast-mediated bone destruction and have a 5-year overall survival (OS) rate of 22.8% [4, 5]. Early detection of bone metastasis is crucial for improving BC survival. However, the predictive factors for bone metastasis have not been sufficiently elucidated. Therefore, there is an urgent need for identifying novel markers that might provide new clues for prevention.
Recently, researchers have shown an increased interest in adipose tissue. Loss of adipose tissue is crucial to the development and progression of cancer [6, 7]. Adipose tissue is anatomically dispersed throughout the body in various compartments, and its function varies according to the region concerned. Subcutaneous adipose tissue (SAT) and visceral adipose tissue (VAT) are physically distinct types of white adipose tissue [8]. The VAT is currently thought of as a metabolic and endocrine organ that can affect body weight balance and systemic immune conditions. SAT, a metabolic storehouse, is linked to the accumulation of visceral fat [9]. In a number of tumor types, including BC, colorectal cancer, hepatocellular carcinoma, and gastric cancer, elevated VAT has been associated with a lower OS rate [10, 11, 12]. Similarly, the SAT index can also be used to predict the outcomes of cancers, such as head and neck cancer [13], BC [14], and prostate cancer [15]. Visceral to subcutaneous fat area ratio (VSR) is an independent prognostic factor for poor prognosis in type 1 endometrial cancer and gastric cancer [16, 17]. Recent research has recognized the crucial role that interactions between adipocytes and BC cells play in the development of BC and associated bone metastases [18].
Conventionally, the third lumbar vertebra (L3) is the standard level at which computed tomography (CT)-measured body composition is assessed [19]. However, abdominal CT scans are not routinely performed on BC patients for assessment and follow-up. A recent study in solid tumors suggests strong correlations between body tissue components (including VAT and SAT) at both the intervertebral space from the 12th thoracic to first lumbar vertebrae (T12-L1) and L3 imaging levels [20]. In addition, in previous studies where L3 was not available, such as lung cancer, the level of the eleventh thoracic vertebra (T11) was used to assess body tissue components [21]. Therefore, we used measurements from the T11 anatomical level in this study.
The present study aimed to determine whether subcutaneous fat could be used as a prognostic parameter for bone metastasis in BC patients.
Materials and methods
Patient characteristics and specimens
The medical records of 517 consecutive female BC patients at Harbin Medical University Cancer Hospital from January 1
The pathologic stage was classified based on the 8th edition of the American Joint Committee on Cancer (AJCC). All BC patients were screened for metastases before operation. No patients had metastases at the time of confirmation of BC. Bone metastases are first defined by emission computed tomography (ECT) scans and then confirmed by X-ray, CT, and magnetic resonance imaging (MRI) scans. Bone metastasis-free survival was calculated from the date of surgery to the date of bone metastasis, or the last follow-up. Patients were followed up every three months for the first two years following surgery and every six months for the following three to five years. The last follow-up was on December 31, 2020. The median follow-up time was 65 months.
The data included characteristics such as age, menstrual status, tumor size, histopathological type, lymph node metastasis, lymphovascular invasion, proliferation index (Ki-67) expression, estrogen receptor (ER) status, progesterone receptor (PR) status, clinical stage, molecular classification, and postoperative treatment. Venous blood samples were collected one week before the operation. Immunohistochemistry (IHC) and/or fluorescence in situ hybridization (FISH) were used to detect the ER, PR, human epidermal growth factor receptor-2 (HER-2), and Ki-67 status. HER-2 positivity was defined as IHC 3+ or FISH positive.
This study was approved by the Ethics Committee of the Harbin Medical University Cancer Hospital (KY2022-10). Informed consent was obtained from each participant.
Adipose tissue imaging analysis
The fat tissue was evaluated using preoperative chest CT scans. The areas of visceral and subcutaneous fat tissues were quantified in a single slice using Image J software version 1.53a (Wayne Rasband, National Institutes of Health, USA). Hounsfield Units (HU) thresholds between -190 and -30 were used to determine subcutaneous fat, while radiodensities between -150 and -50 HU were used to calculate visceral fat [22, 23]. Visceral fat index (VFI) and subcutaneous fat index (SFI) were calculated by dividing the corresponding fat area by the square of the patient’s height [12, 19]. The VSR was calculated by dividing the visceral fat tissue area (VFA) by the subcutaneous fat tissue area (SFA). Two independent radiologists who were blind to the patients’ information performed the CT scans within two weeks before surgery. The two measures’ mean was applied. The intra-observer coefficient of variation was less than 1.2%.
Statistical analysis
All statistical analyses were carried out using SPSS 25.0 software. The data were presented as percentages, or mean
Results
Baseline characteristics of BC patients according to bone metastasis status
Baseline characteristics of BC patients according to bone metastasis status
BC, breast cancer; BMI, body mass index; SFA, subcutaneous fat tissue area; SFI, subcutaneous fat index; VFA, visceral fat tissue area; VFI, visceral fat index; VSR, visceral fat tissue to subcutaneous fat tissue ratio; Ki-67, proliferation index; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; TNBC, triple-negative breast cancer.
This study enrolled 517 female patients with BC. The average age was 50.5
Baseline clinico-pathological parameters of BC patients according to SFI levels before propensity matching
Baseline clinico-pathological parameters of BC patients according to SFI levels before propensity matching
BC, breast cancer; BMI, body mass index; SFI, subcutaneous fat index; Ki-67, proliferation index; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; TNBC, triple-negative breast cancer.
We performed a receiver operating characteristic (ROC) analysis to evaluate the predictive ability of SFI for bone metastasis. The optimal cutoff value of SFI was 30.87 [AUC
Baseline clinico-pathological parameters of BC patients according to SFI levels after propensity matching
Baseline clinico-pathological parameters of BC patients according to SFI levels after propensity matching
BC, breast cancer; BMI, body mass index; SFI, subcutaneous fat index; Ki-67, proliferation index; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; TNBC, triple-negative breast cancer.
In order to adjust for the heterogeneity between the low SFI and high SFI groups, propensity score matching (PSM) was used. After 1:1 PSM, 67% of the low SFI group (
An optimized cut-off value was determined for SFI using ROC curve analysis. The ROC curve identified the optimal cutoff value of SFI as 30.87 cm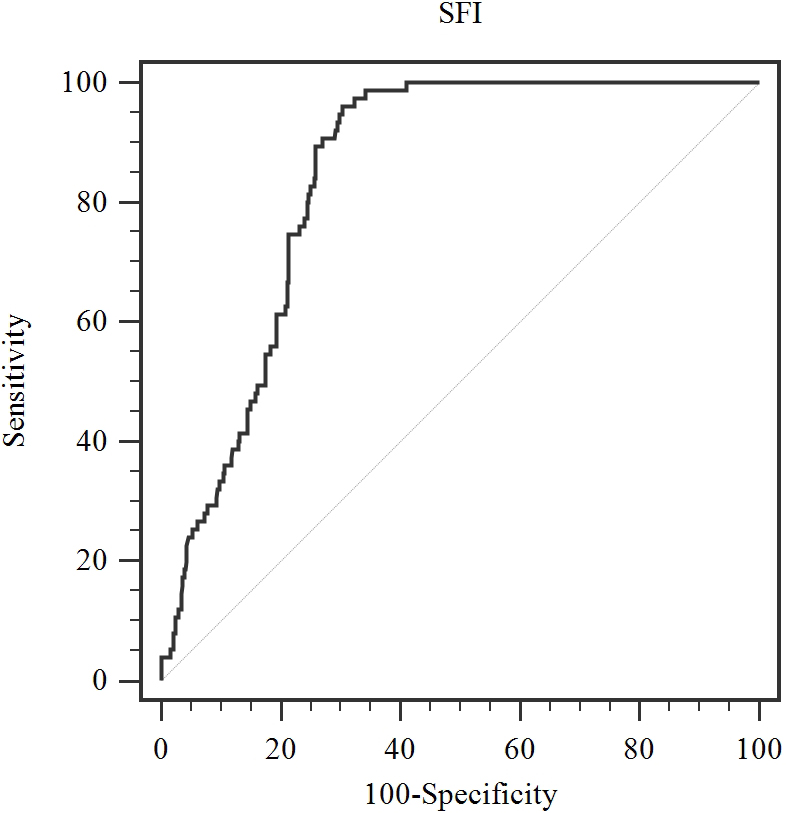
The predictors of bone metastases in patients with breast cancer
BC, breast cancer; BMI, body mass index; SFI, subcutaneous fat index; VFI, visceral fat index; VSR, visceral fat tissue to subcutaneous fat tissue ratio; Ki-67, proliferation index; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; CI, confidence interval.
The possible predictive variables for bone metastases are examined using Cox univariate and multivariate proportional regression models. Before propensity matching, age, SFI, VFI, VSR, histologic type, multifocal disease, tumor size, lymph node status, clinical stage, PR, HER-2 status, and adjuvant hormonal therapy were significantly associated with bone metastasis in univariate analysis. SFI, histologic type, and adjuvant hormonal therapy were independently predictive factors of bone metastasis in multivariate analysis (Table 4). After PSM, the univariate analysis indicated that SFI, VFI, VSR, histologic type, and tumor size were related to bone metastasis. The multivariate analysis revealed that SFI and histologic type were independently predictive factors of bone metastasis (Table 4). The predictive power of SFI in the molecular subtype of BC is provided in supplementary figures (Figs S1–4).
In this paper, we calculated the impact of preoperative SFI on bone metastasis in BC patients. By accurately evaluating SFI in chest CT scans at the T11 level, the results revealed that low SFI presented as a potential predictor for bone metastasis in BC patients. These results remind clinicians to pay close attention to adipose tissue in BC patients.
To our knowledge, no researcher has considered the relationship between SFI and postoperative bone metastases in BC patients. Consistent with our results, previous reports have shown that low SFI is independently associated with increased mortality and shorter survival in cancer patients [24]. Similarly, Dong et al. found that low subcutaneous fat was a risk factor for the prognosis of gastric cancer, including OS and disease-free survival [25]. Black et al. reported that low levels of subcutaneous fat are associated with reduced survival for patients with primary operable colorectal cancer [7]. Moreover, a recent study confirmed that lower SFI was an independent predictor of poor OS in hepatocellular carcinoma [6]. Another study suggests that high SFI and VFI were independently associated with better OS in patients with bone metastases [26]. However, Bradshaw et al. observed that elevated SAT was associated with worse survival among women with non-metastatic BC [14]. The conflicting data may be the result of different study populations, different inclusion criteria, and different indicators of SAT. Thus, further study is needed to clarify the role of SAT in BC.
To date, potential explanations for the predictive effect of SFI on bone metastases have not been clearly identified. SAT is the major source of adiponectin and leptin, which play crucial roles in regulating bone health and bone metastasis in BC [27]. Adiponectin has pro-apoptotic and anti-proliferative properties in human BC cells [28, 29]. It has been reported that adiponectin inhibits the metastatic process via suppression of the adhesion, invasion, and migration of BC cells through activation of the AMPK/S6K axis and upregulation of live kinase B1 (LKB1) [28]. Overall, the prevailing evidence indicates that low levels of adiponectin in the circulation portend a poorer BC risk and prognosis [30]. Adipose tissue mass directly correlates with leptin synthesis and plasma levels, improving lipid metabolism and insulin sensitivity [31]. Leptin helps regulate bone health by modulating bone density, growth, and adiposity. Some studies found lower serum leptin levels in premenopausal BC patients compared with healthy controls [32]. In addition, leptin was also associated with a better prognosis in patients with colorectal cancer [33]. Therefore, adipose tissue-derived adipokines and leptin affect different stages of the bone metastatic process. However, more research is needed to confirm whether and how SAT protects against bone metastasis in BC.
Incidence of bone metastasis based on SFI levels before propensity score matching. The 5-year cumulative hazard of bone metastasis in patients with high SFI (blue, 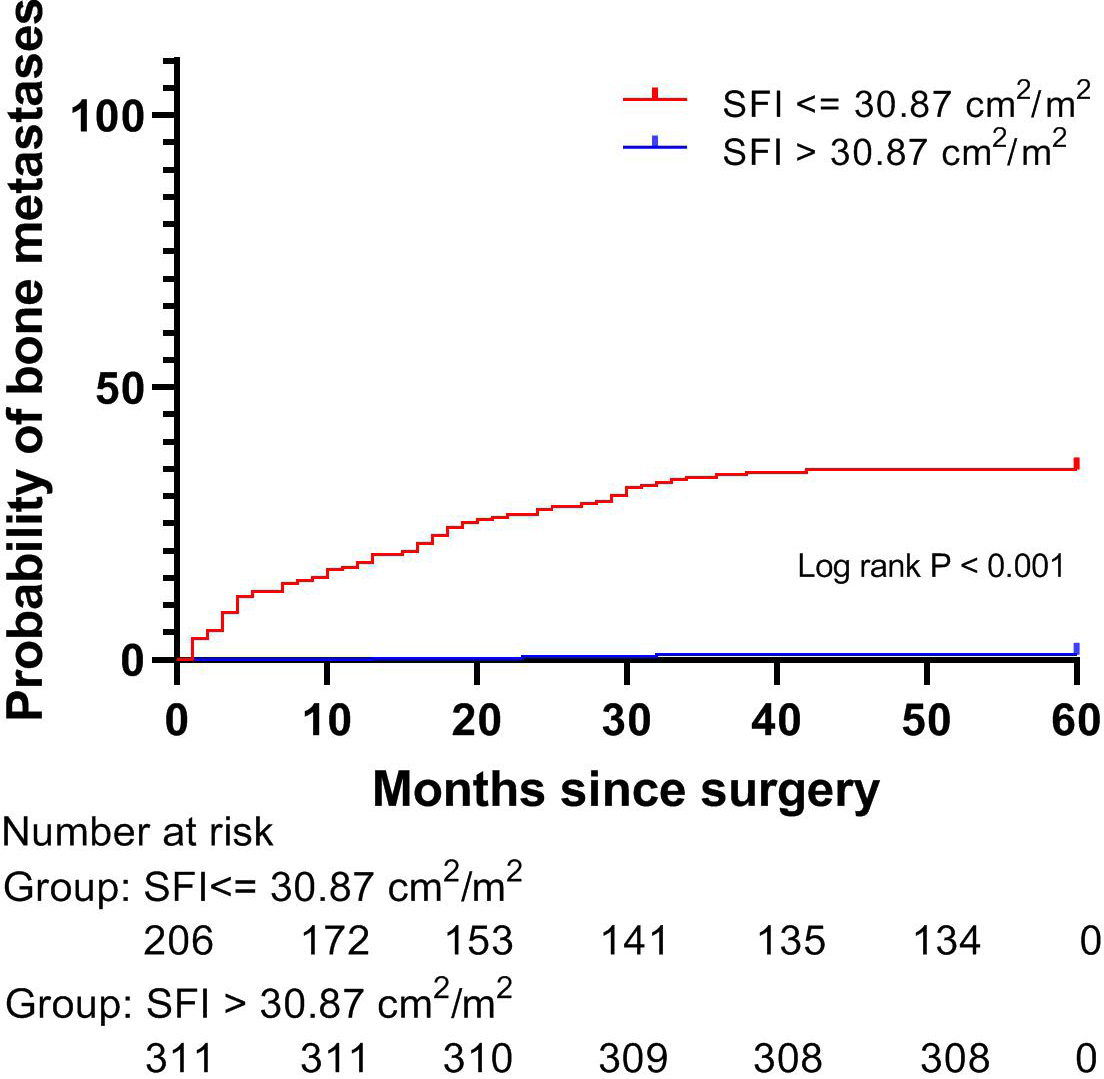
Incidence of bone metastasis based on SFI levels after propensity score matching. After 1:1 propensity score matching, the lower SFI group presented with a higher incidence of bone metastasis (11.4% vs. 2.9%, respectively; 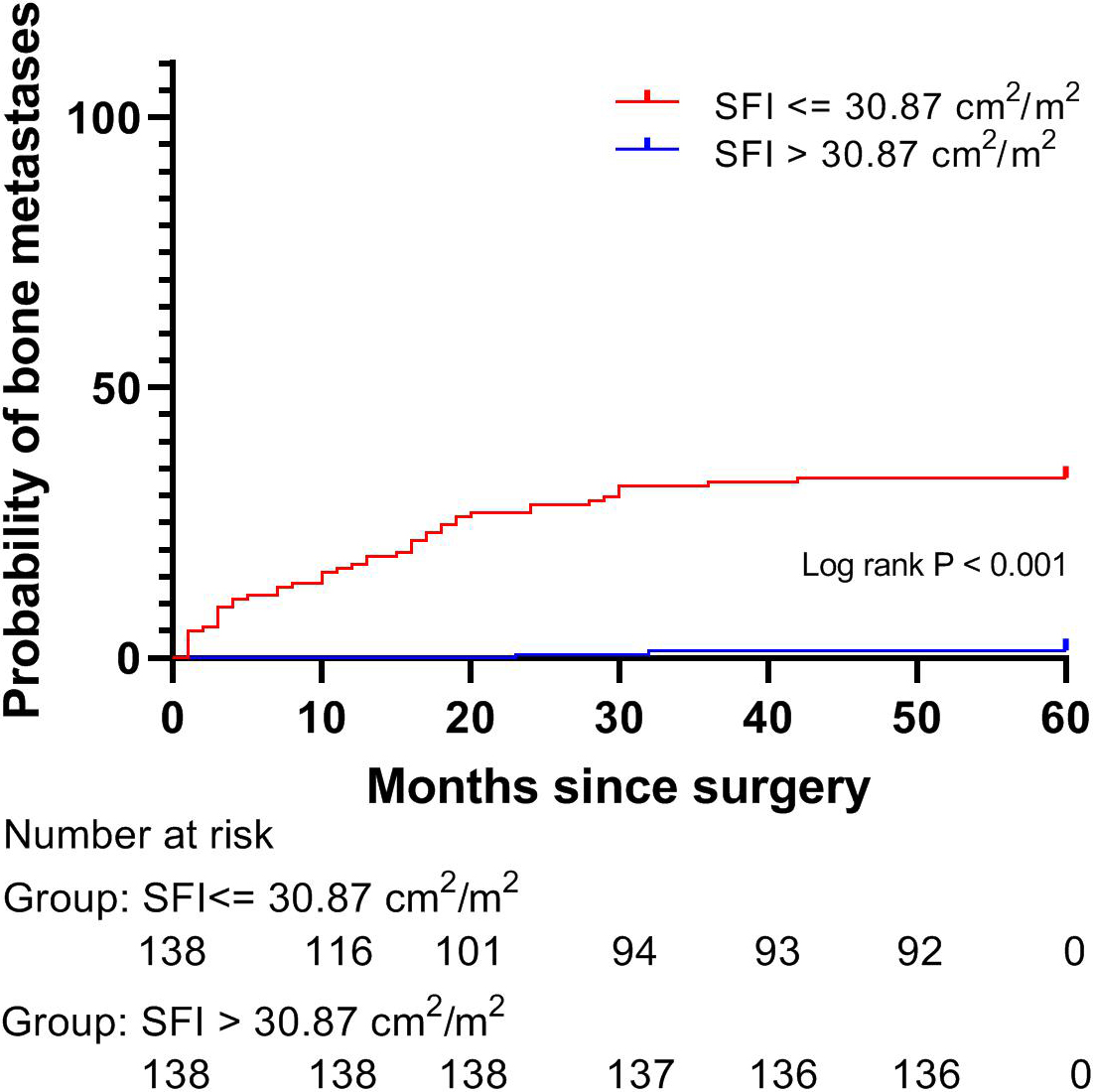
Meanwhile, no association between body mass index (BMI) and bone metastasis was observed in our study. The relationship between BMI and postoperative outcomes in cancer patients has been a subject of controversy. A previous report revealed that the association between BMI and cancer survival is not consistent across all cancer types and stages [34]. The difference in the prognostic value of BMI may be partly explained by the fact that individuals with similar BMI may have different body compositions (e.g., more fat or muscle in one patient than in another) [35]. In other words, BMI cannot distinguish between fat and fat-free mass or different fat stores for proper assessment of body composition [36]. In this study, we used regional measures of obesity, such as SFI and VFI, which reduced the risk of this error estimate and enhanced the consistency of the findings.
Because our findings showed that patients with lower SFI are more likely to develop bone metastasis, SFI may be a novel marker for predicting bone metastasis in BC. This predictive marker can be easily acquired from CT scans collected in routine clinical care. However, this study has several limitations. First, this study was a single-institutional retrospective study and lacked a mechanistic explanation. Second, the cutoff value of SFI at the T11 slice has not been reported in the literature. Thus, multicentric studies are needed to verify our results. In addition, this was a single T11 slice analysis. SFIs in other CT slices were not assessed. Finally, there is a significant difference in BC incidence, subtype distribution, and cancer survival by ethnicity [37, 38]. The participants in our study were all Chinese. Thus, the results cannot be extrapolated to other ethnic groups.
In this paper, we found that SFI is an independent predictor of bone metastasis in BC patients. Our study indicated that SFI provides additional information on the prevention of skeletal-related events in patients with BC. Further studies are warranted to observe whether early intervention to maintain adipose tissue could improve prognosis in patients with low SFI levels.
Author contributions
Conception: WW, W-J H, and P-P L.
Data collection: SF, M-L Z, and XZ.
Interpretation or analysis of data: WW, W-J H, P-P L, SF, M-L Z, and XZ.
Preparation of the manuscript: WW, W-J H, and P-P L.
Revision for important intellectual content: R-T W and Y-X H.
Supervision: R-T W and Y-X H.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230011.
Footnotes
Acknowledgments
The design of the study was supported by the Harbin Medical University Cancer Hospital [Grant Number JJQN2019-06].
