Abstract
Non-muscle invasive bladder cancer (NMIBC) has a high recurrence rate, which places a significant burden on both patients and the healthcare system. Hence, it holds significant importance to predict the recurrence risk following treatment for individuals diagnosed with non-muscle invasive bladder cancer (NMIBC). As new generation technologies continue to emerge, an increasing number of recurrence risk prediction tools are being developed and discovered. This article provides an overview of the primary recurrence risk prediction tools currently available, including the liquid biopsy, tissue biopsy, and risk prediction tables. Each of these tools is described in detail and illustrated with relevant examples. Furthermore, we conduct an analysis of the advantages and disadvantages of these tools. This article aims to enhance the reader’s understanding of the current progress in recurrence prediction tools and encourage their practical utilization in the fields of precision medicine and public health.
Introduction
Bladder cancer ranks as the tenth most prevalent cancer globally, with the sixth highest incidence in men and the tenth highest incidence in women [1]. Non-muscle invasive bladder cancer (NMIBC) constitutes around 75% to 80% of all bladder cancer cases and is typically categorized as early-stage bladder cancer [2, 3].
The frequent recurrence and potential progression to muscle-invasive bladder cancer in patients with non-muscle invasive bladder cancer (NMIBC) necessitate long-term and regular post-treatment follow-up. This places a significant burden on both patients and healthcare systems [4]. Therefore, there is a need to improve the effectiveness of predicting NMIBC recurrence.
NMIBC and Bladder cancer T staging: NMIBC is the malignancy of the bladder confined to the mucosal (TIS/CIS, Ta) and lamina propria (T1) layers of the bladder, with no infiltration of the muscular layer.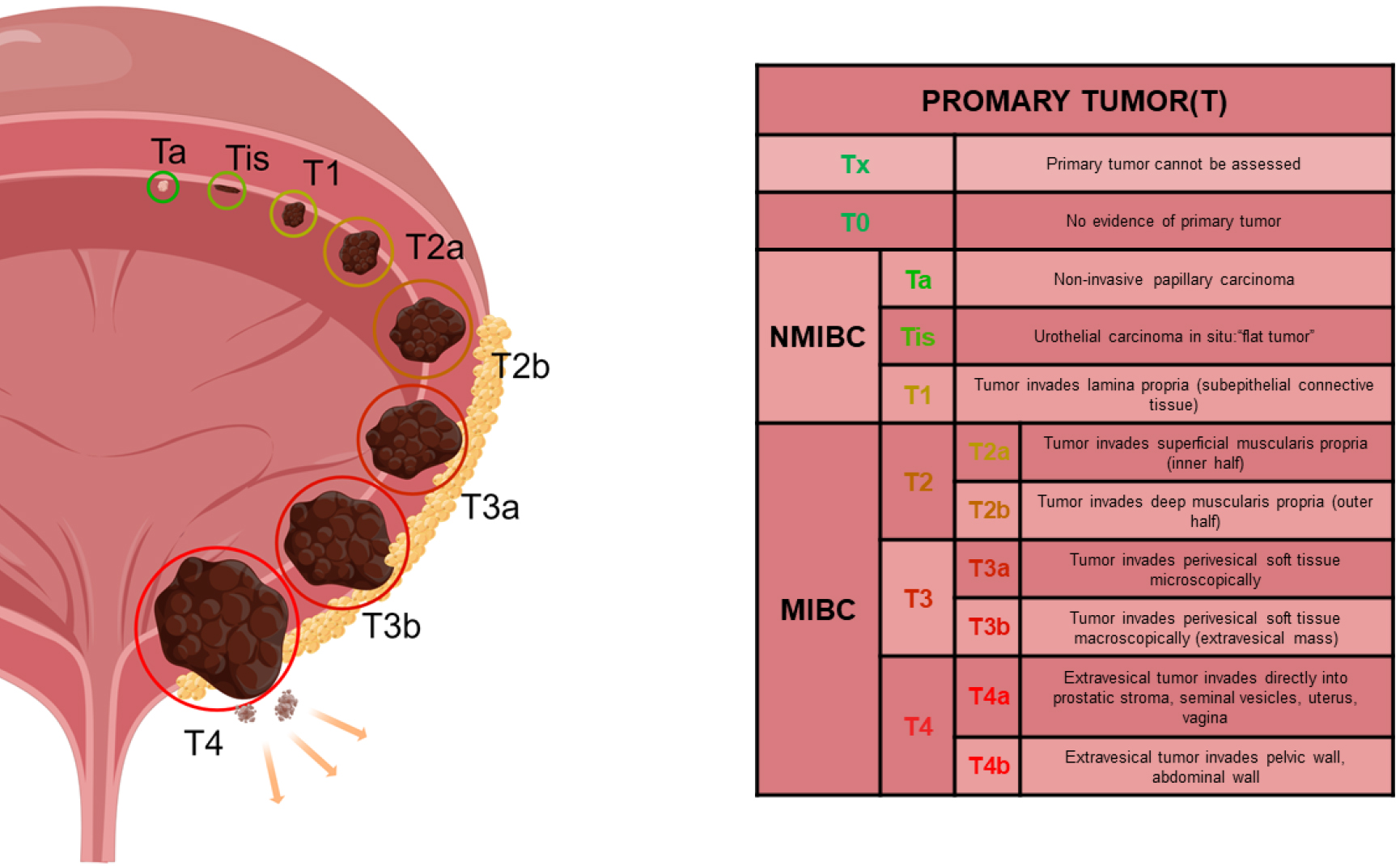
Cystoscopy remains the gold standard for detecting bladder cancer recurrence, offering high sensitivity and specificity [5]. However, cystoscopy is an invasive test that can cause great pain to the patients and may developed complications (infection, bleeding, etc.). Hence, cystoscopy may not be an ideal follow-up test for all patients, and clinicians and researchers have to explore alternative tools and methods to effectively predict the recurrence of NMIBC. Advancements in technology have expanded the scope of recurrence prediction tools beyond the traditional pathological level, such as TNM stage-based risk prediction scales, and conventional tests like cystoscopy. The introduction of novel tools, including liquid biopsies, some of which have obtained US Food and Drug Administration (FDA) approval, presents an opportunity to predict NMIBC recurrence in a more cost, more effective and less invasive manner [6]. For this reason that bladder cancer has the highest lifetime and treatment cost, with 60% of the total expenditure allocated to post-treatment management [7, 8]. This article aims to provide an overview of the prevailing recurrence prediction tools, particularly those that have emerged in recent years. It will examine their advantages and limitations, propose potential solutions to address the existing challenges, and outline potential avenues for future research in this field.
While cystoscopy remains the standard method for detecting bladder cancer recurrence, there is growing interest in the convenience and cost-effectiveness of liquid biopsy compared to the discomfort associated with endoscopy. Liquid biopsy is an emerging technique that involves analyzing various biomarkers present in blood or other body fluids, including circulating tumor cells (CTCs), circulating DNA and RNA (cfDNA and cfRNA), proteins, molecules in exosomes, and tumor-educated platelets. This non-invasive approach holds promise for diagnosing and monitoring diseases such as cancer [9]. Furthermore, considering that bladder cancer comes into direct contact with urine, the collection of urine samples for diagnostic purposes is relatively convenient [10]. Hence, liquid biopsies for bladder cancer, particularly urine biomarker tests, have gained considerable attention. It is noteworthy that certain urine biomarker tests have obtained approval from the FDA [6]. Nevertheless, it is important to acknowledge that liquid biopsies, despite their promise, still face challenges in terms of reliability, sensitivity, and specificity. As a relatively new technique, further research and development are necessary to enhance the accuracy and performance of liquid biopsies for bladder cancer recurrence prediction [9].
Molecules in liquid biopsies
Currently, various commercial liquid biopsy kits are available for clinical use in bladder cancer, focusing on the detection of specific molecules in body fluids. Some notable examples include AssureMDx, CxBladder series (including Cxbladder Monitor), NMP22 series (including NMP22 ELISA and NMP22 bladderchek), and BTA series (including BTA TRAK and BTA stat). These commercially available test kits encompass a wide range of molecular liquid biopsy assays, providing comprehensive coverage for various aspects of bladder cancer detection and monitoring. AssureMDx is a liquid biopsy test that focuses on the detection of DNA mutations and methylation patterns. In this test, DNA is extracted from urine samples collected prior to cystoscopy. The extracted DNA is then analyzed for specific mutations in genes such as FGFR3, TERT, and HRAS, as well as for methylation patterns in genes including OTX1, ONECUT2, and TWIST1 [11], Cxbladder is a liquid biopsy test that utilizes the quantitative detection of mRNA. In this test, mRNA is extracted from urine samples collected before cystoscopy. The extracted mRNA is then analyzed using a technique called RT-qPCR (Reverse Transcription-quantitative Polymerase Chain Reaction). The test focuses on measuring the expression levels of specific genes, including IGFBP5, HOXA13, MDK, CDK1, and CXCR2 [12]. The NMP22 series and BTA series are liquid biopsy tests that utilize well-established protein assays. These tests use immunoassays to detect specific proteins, namely the NMP22 protein and factor H-related protein, in urine samples collected prior to cystoscopy. Importantly, both of these tests have received approval from the FDA [13, 14]. They have also developed a more convenient version for clinical use: NMP22 bladderchek and BTA stat. The NMP22 bladderchek, for example, is a modification of the NMP22 Elisa. In other words the NMP22 bladderchek is an improvement on the “Point of care” (POC) version, which uses dipstick immunoassay technology to make the NMP22 test faster and more convenient. The bladderchek Test is a single-step immunoassay that requires five drops of urine and can complete within five minutes, so does BTA stat.
The foundation of these commercial test kits lies in an expanding body of fundamental research on biomarkers for liquid biopsies. This research encompasses traditional biomolecules such as DNA, RNA, and proteins, as well as the emerging field of non-coding RNA and exosomes. As a result of these findings, the availability of biomarkers for liquid biopsies has significantly increased. Additionally, numerous clinical cohort studies utilizing commercial kits have enhanced the reliability of their clinical application. Consequently, these biomarkers hold greater significance in guiding clinical practices [15, 16, 17].
The discovery of circulating cell-free nucleic acids in the bloodstream was first reported by Mandel and Metais in 1948 [18], Circulating cell-free nucleic acids consist of cfDNA and cfRNA. Within the cfRNA fraction, there are circulating coding RNAs as well as various circulating non-coding RNAs. Circulating cell-free nucleic acids are derived from cellular necrosis or apoptosis processes. Specifically, in the case of cfDNA, it has been observed that around 70% of patients with low-grade NMIBC exhibit mutations in the FGFR3 gene [19]. As a result, researchers have developed various approaches to monitor and predict the recurrence of bladder cancer through the analysis of FGFR3. Tahlita Zuiverloon et al. conducted a cohort study focusing on the FGFR3 single gene mutation in non-muscle invasive bladder cancer, while Christensen et al. developed a cohort study investigating the potential of hotspot FGFR3 and PIK3CA mutations as molecular liquid biopsy biomarkers [20, 21]. However, there has been comparatively less research on circulating cell-free DNA (cfDNA) compared to circulating cell-free RNA (cfRNA). This is primarily due to the fact that cfRNA exhibits a greater diversity of molecular characteristics and potential applications than cfDNA. Besides mRNA, there has been extensive research on the emerging field of noncoding RNA, including microRNA (miRNA), circular RNA (circRNA), long noncoding RNA (lncRNA), and others, in the context of liquid biopsies for predicting NMIBC recurrence [22, 23, 24, 25].
Protein detection has emerged as one of the earliest and more mature biomarkers in liquid biopsies. Compared to RNA and DNA analysis, protein detection is relatively easier and more established [26]. The research on liquid biopsies of proteins started earlier and had been very inclusive so far, such as the NMP22 protein we mentioned previously. In addition, Rosser et al. utilized the ELISA to conduct a comprehensive evaluation of ten protein biomarkers in urine (ANG, APOE, CA9, IL8, MMP9, MMP10, SDC1, SERPINA1, SERPINE1, and VEGFA), as an approach to monitor and predict the recurrence of NMIBC [27].
Discovered in 1983, exosomes are crucial constituents of molecular liquid biopsies. Exosomes are small extracellular vesicles with sizes ranging from 30 to 150 nm and represent a subtype of extracellular vesicles (EVs) that are released by various cell types. Exosomes play a significant role in facilitating intercellular communication [28, 29]. Individual tumor cells have the ability to communicate with normal cells through exosomes, facilitating tumor cell growth and survival. Studies have demonstrated that this communication is mediated, in part, by abnormal alterations in molecules contained within exosomes, including non-coding RNAs and proteins. These molecular changes contribute to the functional effects exerted by exosomes in promoting tumor progression [30]. Exosomes possess a unique lipid bilayer structure that safeguards their contents from enzymatic degradation, ensuring their stability. They are also enriched with specific cellular components, allowing for efficient long-distance transport and accessibility. Hence, there is growing interest in investigating the prognostic implications of exosomes in the field of bladder cancer. However, research in this area is relatively recent and has primarily focused on analyzing proteins and miRNAs. Exploring the role of other non-coding RNAs and novel molecules within exosomes may yield valuable insights into their prognostic value for bladder cancer [31]. Furthermore, the development of commercially available products for exosome analysis is still in its early stages and requires further refinement and validation to achieve maturity. It is important to mention that a study published in the journal Cell has provided evidence indicating the absence of double-stranded DNA and DNA-binding histones in exosomes and other small extracellular vesicles. Autophagy and multivesicular endosome-related pathways have been identified as the primary mechanisms responsible for the secretion of extracellular DNA, rather than the exosome-dependent pathway. However, subsequent research using imaging flow cytometry has confirmed the presence of double-stranded DNA in exosomes [31, 32, 33]. The conflicting findings highlight that exosomes are worth further researching as a potential liquid biopsy biomarker.
An interesting observation in the field of molecular liquid biopsies for bladder cancer is the predominant use of urine specimens rather than blood. Unlike other types of cancer, bladder cancer has the unique advantage of direct contact with urine, making urine samples more easily accessible for diagnostic purposes. The utilization of urine specimens in liquid biopsies for bladder cancer highlights the practicality and convenience of this non-invasive approach in clinical settings. The preference for urine specimens in liquid biopsies for bladder cancer can be attributed to two factors. Firstly, bladder cancer directly interacts with urine, making it a more relevant source of biomarkers. Additionally, compared to blood, urine tends to have fewer impurities. It is worth noting that the study of biomarkers in liquid biopsies is evolving from single variables to multiple variables and from single types to mixed types. However, it is important to acknowledge that while many biomarkers in urine originate from direct contact with the tumor, some of these biomarkers may also result from the filtration of blood by the kidneys.
Both UroVysion and uCyt+ have received FDA approval, and these two kits offer different assays for molecular liquid biopsies and circulating tumor cells liquid biopsies, respectively. Both UroVysion and uCyt+ tests are focused on the detection of exfoliated cells in urine rather than circulating tumor cells. UroVysion utilizes a multi-targeted FISH (fluorescence in situ hybridization) technique to analyze chromosomal alterations in exfoliated cells from the urinary bladder, enabling the identification of bladder cancer cells [34]. In contrast, the uCyt+ test relies on three BC antigens (M344, LDQ10 and 19A11) by Immunofluorescence technology to detect urothelial cells in voided urine [35]. Numerous studies have demonstrated the efficacy of both UroVysion and uCyt+ in predicting the recurrence of NMIBC [36, 37, 38, 39].
Circulating tumor cells (CTCs)
Circulating tumor cells (CTCs) have become a crucial element in liquid biopsies in recent years. Although researchers observed CTCs under the microscope as early as the 1860s [40]. However, the low abundance and short half-life of circulating tumor cells (CTCs), with a lifespan of only 1–2.4 hours, present significant challenges in their isolation and subsequent analysis from body fluids. Due to their rarity, isolating a sufficient number of CTCs for comprehensive characterization and analysis has been technically demanding [41]. The progress in CTC isolation techniques in the 21st century has significantly advanced research on CTCs and paved the way for their clinical applications. The CellSearch system, based on EpCAM (epithelial cell adhesion molecule), is the only FDA-approved platform for capturing and counting CTCs in a clinical setting. Unlike molecular liquid biopsies, CTCs derived from primary or metastatic tumors are directly associated with metastatic recurrence. CTCs carry complete biological information and have the potential to guide precise drug selection and treatment decisions, offering advantages over molecular liquid biopsies [42, 43]. Given that some patients with NMIBC may already have micrometastases, it is noteworthy that studies have demonstrated the presence of CTCs in the blood of NMIBC patients even after treatment. This suggests that CTCs hold promise as a valuable marker for predicting bladder cancer recurrence [44, 45, 46]. Furthermore, similar to liquid biopsies for other types of cancer, CTC-based liquid biopsies in NMIBC primarily focus on the detection of CTCs and the quantification of their numbers as indicators of recurrence. The presence of CTCs in the bloodstream suggests the potential for metastatic spread and recurrence of the disease. One notable study conducted by Michael Rink et al. demonstrated the significance of preoperative CTCs as a robust predictor of disease recurrence in NMIBC [47]. However, it has been suggested in some articles that relying solely on CTC counts may not be sufficient to accurately reflect tumor prognosis. Additional cytomics studies, such as analyzing mutations, transcriptomes, and other cellular characteristics, are needed to provide a more comprehensive understanding of the tumor and its behavior [48]. Certainly, the sequencing technology for CTCs requires further development, especially in the realm of single-cell analysis. CTCs liquid biopsies have made significant progress in terms of isolation and detection of the recurrent bladder cancer. However, a number of issues remain unresolved. One major challenge is the lack of a standardized method for detecting CTCs beyond the CellSearch system, which is currently the only FDA-approved platform for clinical use.
Risk tables
Recurrence risk scales, such as the EORTC model developed by the European Organisation for Research and Treatment of Cancer and the CUETO model developed by the Club Urológico Español de Tratamiento Oncológico, are widely accepted in clinical guidelines for assessing the risk of recurrence in bladder cancer [49, 50].
The EORTC and CUETO models are both recurrence risk scales used in bladder cancer. They consider similar factors such as the number of tumors, previous recurrence, G classification, and the presence of carcinoma in situ (CIS). However, there are some differences between the two models. The EORTC model places emphasis on tumor diameter and T stage, while the CUETO model also considers gender, age, and previous recurrences as additional factors in assessing the risk of recurrence. But more importantly, the application of the two models are different, as both models have limitations in the selection of patients at the beginning of their design which lead to the two models’ limitations as well: for the EORTC model, few patients in the study received BCG and did not undergo a second transurethral resection of bladder tumor (TURBT); for the CUETO model, no second TURBT was performed and BCG was not used as maintenance therapy. As a result, the European Association of Urology (EAU) has recommended using the CUETO risk model for patients who have previously received BCG and the EORTC risk model for those who have undergone TURBT. These recommendations take into account the specific treatment history of the patients to guide the selection and application of the appropriate risk model [51]. Further, for patients with NMIBC, both models can predict disease recurrence to some extent [52, 53, 54, 55]. And many studies have indicated that the EORTC risk model exhibits superior predictive ability compared to the CUETO risk model in terms of disease recurrence in patients with NMIBC [52, 56, 57]. However, many institutions and their guidelines have varying perspectives on the use and endorsement of risk prediction models. The American Urological Association (AUA) has chosen not to endorse the two model in its guidelines, suggesting that it may be too limiting and may not encompass all relevant variables for predicting recurrence. The Canadian Urological Association (CUA) has identified additional factors such as lymph vascular invasion (LVI), prostatic urethral carcinoma in situ, and variant histology, which they consider important variables that should be considered alongside existing risk prediction models to improve the accuracy of recurrence prediction in NMIBC [2]. Lastly, it is important to acknowledge that the two recurrence risk models are primarily based on clinical information. While these models offer accessibility and cost-effectiveness compared to other methods like biopsies, their predictive accuracy is not considered optimal. Numerous studies have highlighted the limitations of these models in accurately predicting recurrence in NMIBC [57, 58].
Tissue biopsy
Bladder tumor tissue biopsy remains the gold standard for diagnosing bladder cancer recurrence and is widely utilized in clinical practice. For NMIBC recurrence, tissue biopsy can provide valuable prognostic information based on pathological features such as tumour stage and grade, and these pathological elements are often considered in recurrence risk prediction models. In comparison to liquid biopsies, tissue biopsies have limitations when it comes to evaluating cytological components. Biomarkers in tissues are often challenging to track dynamically, impeding their use in monitoring tumor recurrence or progression over time. But the tissue biopsy has some notable advantages: detecting tumor tissue directly and the large sample size, which allow for more accurate information. These characteristics also make tissue biopsies compatible with high-throughput sequencing such as DNA-, mRNA-, LncRNA-, or miRNA-seq [59, 60, 61]. Sequencing allow for the analysis of genetic mutations, differential gene expression, and other molecular factors that play a role in tumor progression and clinical outcomes. As second-generation high-throughput sequencing became more popular and the gradual application of third- and fourth-generation high-throughput sequencing [62], high-throughput sequencing is becoming more affordable. As sequencing costs decrease and additional prognostic biomarkers are validated, high-throughput sequencing, particularly transcriptome sequencing, is poised to become a prominent force in cancer research and clinical applications. These sequencing results can help clinicians assess patients’ prognoses from multiple molecular dimensions. The cytological analysis of tumour tissue biopsies is similar to that of liquid biopsies. They both analyse the genomics of tumour cells, the proteomics and even many well-known molecules present in liquid biopsies are still valuable in tissue biopsies. Compared to liquid biopsies, immunohistochemistry is more frequently used for analysis in tissue biopsy due to the larger sample size and easier accessibility [63]. Biomarkers for tissue biopsy can be categorized into several main groups. The first category includes cell cycle markers such as p53, Ki67, p21, Cadherins, Survivin, and FGFR3, which provide insights into the proliferation and cell cycle control of tumour cells. Another important category encompasses immune and inflammation-related markers, including COX-2, CD103, and other tumour antigens that play a role in mediating the host’s immune response [64, 65, 66, 67, 68, 69, 70, 71, 72]. Combining tissue biopsy biomarkers with clinical decision-making tools, such as recurrence risk prediction tables, has shown promising results in research studies [73, 74]. However, the combination makes the clinical decision-making tools less practical as it makes the clinical information decision tools more complex while improving risk stratification for recurrence and progression of NMIBC.
Currently, there is improved understanding of tumor heterogeneity, acknowledging the significance of a specific cell population known as “tumor stem cells” in tumor recurrence and metastasis. These cells, commonly referred to as “tumor-initiating cells,” have been identified as crucial players in driving tumor progression [75]. With the gradual maturation of single-cell techniques, the role of single-cell technology in guiding tumor prognosis is beginning to initiate. Unlike single-cell technologies applied to CTCs, which focus mainly on cell capture and low-abundance single-cell sequencing, single-cell technologies for tissue samples focus more on tumor cell heterogeneity [76, 77]. The development of high-throughput single-cell sequencing platforms like 10
Different components in liquid biopsies and tissue biopsies.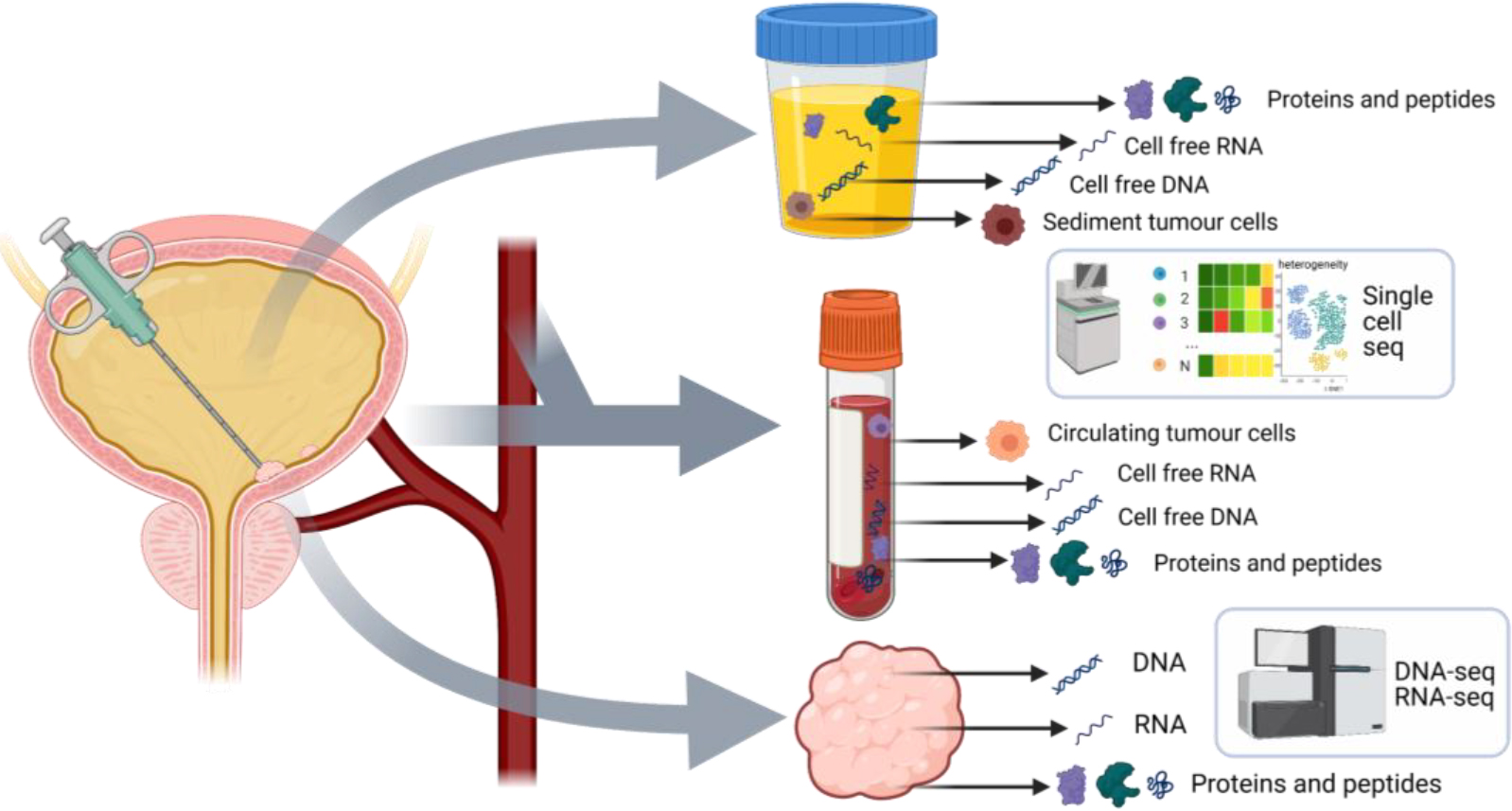
While recurrence risk scoring models and biopsies provide valuable information for assessing NMIBC, there are many factors associated with recurrence that are challenging to quantify or often overlooked. These factors include a patient’s history of smoking, exposure to benzene chemicals, or the presence of chronic kidney disease, among others. When dealing with patients who have poor lifestyle habits, such as smoking, or those at risk of occupational exposure, such as in the chemical or rubber industry, it is crucial to recognize that these individuals have a higher likelihood of NMIBC recurrence. Consequently, it is recommended to increase the frequency of regular check-ups for these patients. Moreover, it is of utmost importance to advise and encourage patients to steer clear of these risk factors as soon as possible to minimize the risk of recurrence [81, 82, 83].
Discussion
The high recurrence rate observed in NMIBC poses a significant challenge. Currently, one of the primary strategies to address this issue is through regular cystoscopy for patients. While effective in detecting recurrence, regular cystoscopy can be uncomfortable and may deter patients from undergoing frequent examinations. As a result, the EORTC and CUETO models have gained popularity as alternative tools for predicting recurrence in NMIBC. The greatest features of these clinical information decision tools are their ease of use and low cost. However, one limitation of these models is their inability to provide real-time monitoring of the disease’s progression. Additionally, the accuracy of these models may be compromised due to the diverse treatment options available for NMIBC. Compared to those clinical information decision tools, tissue biopsy still has the problem of not being able to follow the disease dynamically. Tissue biopsy is a well-established method for predicting the recurrence of NMIBC. Unlike clinical information decision tools, tissue biopsy provides genomic and proteomic testing, which offers more personalized insights for individuals. However, similar to clinical information decision tools, tissue biopsy has limitations in its ability to dynamically monitor the disease. Nevertheless, it remains valuable in guiding individualized treatment decisions and reducing the likelihood of recurrence. With rapid technological advancements, liquid biopsies are emerging as a promising approach to improve dynamic disease monitoring and enhance recurrence prediction in NMIBC. Liquid biopsies, particularly when utilizing urine as the specimen, offer the advantage of being no invasive and reducing patient discomfort. This minimally invasive nature makes liquid biopsy an attractive option for improved follow-up and recurrence prediction in NMIBC.
The application of liquid biopsy in clinical practice still faces several challenges. Firstly, the abundance of different biomarkers makes it difficult to conduct large-scale prospective and retrospective studies across multiple centers, hindering the validation and standardization of the technique. Secondly, the requirement for specialized laboratory equipment and expertise increases the cost of liquid biopsies, making them less accessible to patients. In our clinical practice, many patients have consulted about the value of the technique, but finally, most of them step back after knowing the cost. Consequently, the practical implementation of liquid biopsies is limited, let alone the more expensive single-cell technology. In addition, as the result of the liquid biopsies’ high cost, there is always a significant difference in income between patients who undergo liquid biopsy and those who do not, and the significant differences in living conditions due to different income may lead to serious statistical bias, making the predictive power of liquid biopsy overestimated. Apart from the price, there are some other concerns about liquid biopsy, such as whether the convenience of liquid biopsy may encourage doctors to perform non-invasive tests that are unnecessary, further increase the cost of treatment. In summary, the three main types of NMIBC recurrence risk prediction tools discussed above have their limitations due to the diverse treatment methods for NMIBC. However, it is evident that these tools are continuously evolving to enhance their ability to predict recurrence risk and alleviate patient discomfort.
In the foreseeable future, establishing a global consensus on the predictive power of specific models or molecules for NMIBC recurrence is crucial to provide patients with cost-effective and accurate prediction tools. While the identified mutual recognition may not be the ultimate solution, it would encourage further clinical studies and accelerate the commercialization of these models or molecule detection methods, leading to cost reduction and benefiting national health insurance systems. This iterative process of searching for improved NMIBC recurrence risk prediction tools will continue, just like a circle. It is only a matter of time for more accurate, more practical and affordable recurrence risk prediction tools are introduced into various guidelines.
Method
A literature review was performed using the PubMed database to identify relevant studies on the prediction of recurrence in non-muscle invasive bladder cancer (NMIBC). The search was conducted from January 2000 to December 2021, and keywords such as bladder cancer, NMIBC, liquid biopsy, recurrence, biomarkers, prognosis, and prediction were used to retrieve relevant articles. The purpose of this review was to gather evidence on the various approaches and biomarkers used in predicting NMIBC recurrence, as well as to assess their prognostic value and potential for clinical application. The findings of this review will contribute to the understanding and advancement of recurrence prediction in NMIBC. Using a database of search terms
To ensure the quality of the literature, papers presenting data in the form of letters, editorials, study protocols, case reports, short communications and articles not published in English were excluded. Colleagues examined the literature of all included papers for additional studies of interest. On this basis, 11 articles published before 2000 and 6 article published after 2021 and 2022 were included.
Three independent researchers (two urology researchers, one clinical laboratory researcher) assessed each of these articles for quality and thematic suitability. If two researchers provided negative comments on a particular study, it was excluded from the final selection.
Funding
This work was supported by the National Natural Science Foundation of China (no. 82172564) and the Science and Technology Research Project of Henan Province (no. 212102310116).
Author contributions
Conception: Keqiang Li, Zhenlin Huang.
Interpretation or analysis of data: Guoqing Xie, Haofan Wu, Keqiang Li, Zhenlin Huang.
Preparation of the manuscript: Aravind Raveendran, Yu Zhang.
Revision for important intellectual content: Keqiang Li, Zhenlin Huang, Zhankui Jia.
Supervision: Jinjian Yang, Zhankui Jia.
Footnotes
Acknowledgments
We thank Professor Jun Wang, Professor Songchao Li and Professor Lin Sun for assessing the quality and thematic suitability of the articles we selected.
All figures are created by Figdraw and Biorender, we thank for their brilliant work.
