Abstract
BACKGROUND:
A complicated interplay between radiation doses, tumour microenvironment (TME), and host immune system is linked to the active participation of immune response.
OBJECTIVE:
The effects of single targeted 2 Gy and 8 Gy gamma-ray irradiations on the immune cell population (lymphocytes, B-cells, T-cells, neutrophils, eosinophils, and macrophages) in EMT6 mouse-bearing tumour models was investigated.
METHODS:
The effects of both irradiation doses in early (96 hours) and acute phase (5 to 11 days) post-irradiation on immune parameters were monitored in blood circulation and TME using flow cytometry. Simultaneously, selected cytokines related to immune cells within the TME were measured using multiplex ELISA.
RESULTS:
A temporary reduction in systemic total white blood count (TWBC) resulted from an early phase (96 hours) of gamma-ray irradiation at 2 Gy and 8 Gy compared to sham control group. No difference was obtained in the acute phase. Neutrophils dominated among other immune cells in TME in sham control group. Eosinophils in TME was significantly increased after 8 Gy treatment in acute phase compared to sham control (
CONCLUSION:
The temporary changes in immune cell populations within systemic circulation and TME induced by different doses of gamma-ray irradiation correlated with suppression of several pro-tumorigenic cytokines in mouse-bearing EMT6 tumour models.
Introduction
The incidence of breast cancer among women has increased tremendously in 2020 as compared to 2018, surpassing the other common cancers such as colorectal and lung cancers [1, 2]. Sadly, the actual number of cases might be higher as data provided was based on pre-pandemic COVID-19 (coronavirus disease) era. During the pandemic, the incidence was under-diagnosed, and delayed due to implementation of lockdown, reduced availability of healthcare facilities, and suspension of screening program. Subsequently, leading to increment in advanced stage diagnosis and poor survival rate [3].
Cancer treatments such as surgery and chemotherapy were frequently cancelled or postponed as these treatments potentially cause prolonged immunosuppression and severe infection, especially to elderly patients who were most vulnerable (Centers for Disease Control and Prevention (CDC), 2021). Thus, the used of modified radiotherapy such as hypofractionation radiotherapy as alternative especially in the current situation was justified [4, 5]. Here, higher dose of radiation given in a shorter duration of treatment was suggested for immediate tumour control management [4, 6, 7]. However, more data were required as exposure to different higher doses of radiation may caused chronic toxicity including erythema, epilation, ulceration, fibrosis and telangiectasia [8]. Thus, more preclinical studies were needed to optimise benefits and minimise side effects due to immunosuppression.
The role of immune system in the regulation of tumour control has been elucidated [9, 10]. One of the interesting findings related to immune response within TME was a double-edge sword role of eosinophils infiltration as pro- or anti-tumour agents with inconsistent results in different types of cancers [11, 12, 13]. The effect of radiation-induced immune changes in tumour control produced contradictory outcomes by either promoting immunosuppression leading to poor tumour ablation [14, 15, 16, 17, 18] or radiation related to immunostimulant effect in enhancing recognition for cancer cell death [19, 20]. Active participation of immune reactivity either through bystander or abscopal effects, suggest a complicated interplay between radiation, TME, and host immune system [21, 22]. The capability of radiation in modulating TME is used as a platform in investigating the relationship between different doses of single targeted gamma-ray irradiation with the implication of eosinophils infiltration, cytokine, and subsequent impact on tumour growth.
Materials and methods
Cell line
EMT6 mouse mammary carcinoma (ATCC, USA) was cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% foetal bovine serum, 100 IU m/L penicillin G, and 100
Study design
The experimental design was summarised in Fig. 1. A total of 36 female BALB/c mice aged between 6 to 8 weeks were divided into 6 groups. Group 1 consisted of 6 healthy uninoculated and untreated mice which served as negative control. Group 2 or sham control were inoculated with EMT6 cells, but not treated with gamma-ray irradiation. Both groups 1 and 2 were euthanised at day 10
The experimental design for mice tumour inoculation, gamma-ray irradiation, and euthanisation during the early and acute post-irradiation phases. Group 1: Mice healthy, uninoculated and untreated, Group 2: Mice inoculated but untreated, Groups 3 and 4: Mice inoculated and treated with 2 Gy and 8 Gy gamma-ray, respectively (early phase), Groups 5 and 6: Mice inoculated and treated with 2 Gy and 8 Gy gamma-ray, respectively (acute phase).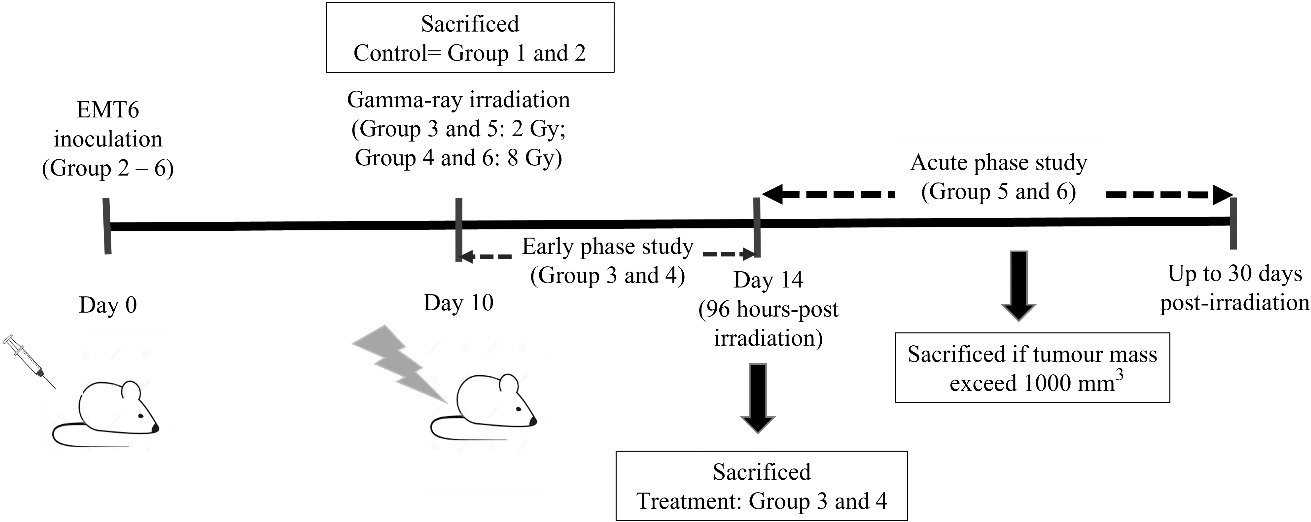
The detailed preparation of mouse-bearing tumour model was previously published by [25]. Briefly, the mice were acclimatised and bred in pathogen-free conditions (Laboratory Control Unit, Faculty of Medicine, Universiti Teknologi MARA). Prior to inoculation, the mice were anaesthetised with ketamine/xylazine at 0.1 mL/mg body weight. Subsequently, inoculation was performed subcutaneously by injecting 10
Gamma-cell irradiation
The tumour was irradiated using a Gamma Cell 220 unit (Nordion, Ottawa, Canada) at Faculty of Science and Technology, Universiti Kebangsaan Malaysia at an operating dose rate of approximately 18.67 Gy/min. Groups 3 to 6 were irradiated on day 10
Mouse euthanisation
Prior to euthanisation, each mouse was anaesthetised and whole blood sample collected into EDTA tubes using cardiac puncture technique. Mice were sacrificed by cervical dislocation. Tumours were excised and used for flow cytometry and protein cytokine analysis. Several mice died during experiment due to unforeseen circumstances were excluded from the study. The final surviving numbers for each Groups 1 to 6 were 6, 4, 6, 5, 5 and 3, respectively.
Blood analysis
Blood analysis was conducted using an automated blood analyser (XN-550, Sysmex Co., Kobe, Japan) at the Centre of Pathology Diagnostics & Research Laboratories, Universiti Teknologi MARA according to the manufacturers protocol. Results were re-confirmed manually with white blood cell (WBC) differential count.
The comparison of mean value for tumour growth volume between gamma-ray irradiated groups compared with sham control (Group 2) during early phase of 2 Gy (Group 3) and 8 Gy (Group 4) post-irradiation. Data were expressed as means values 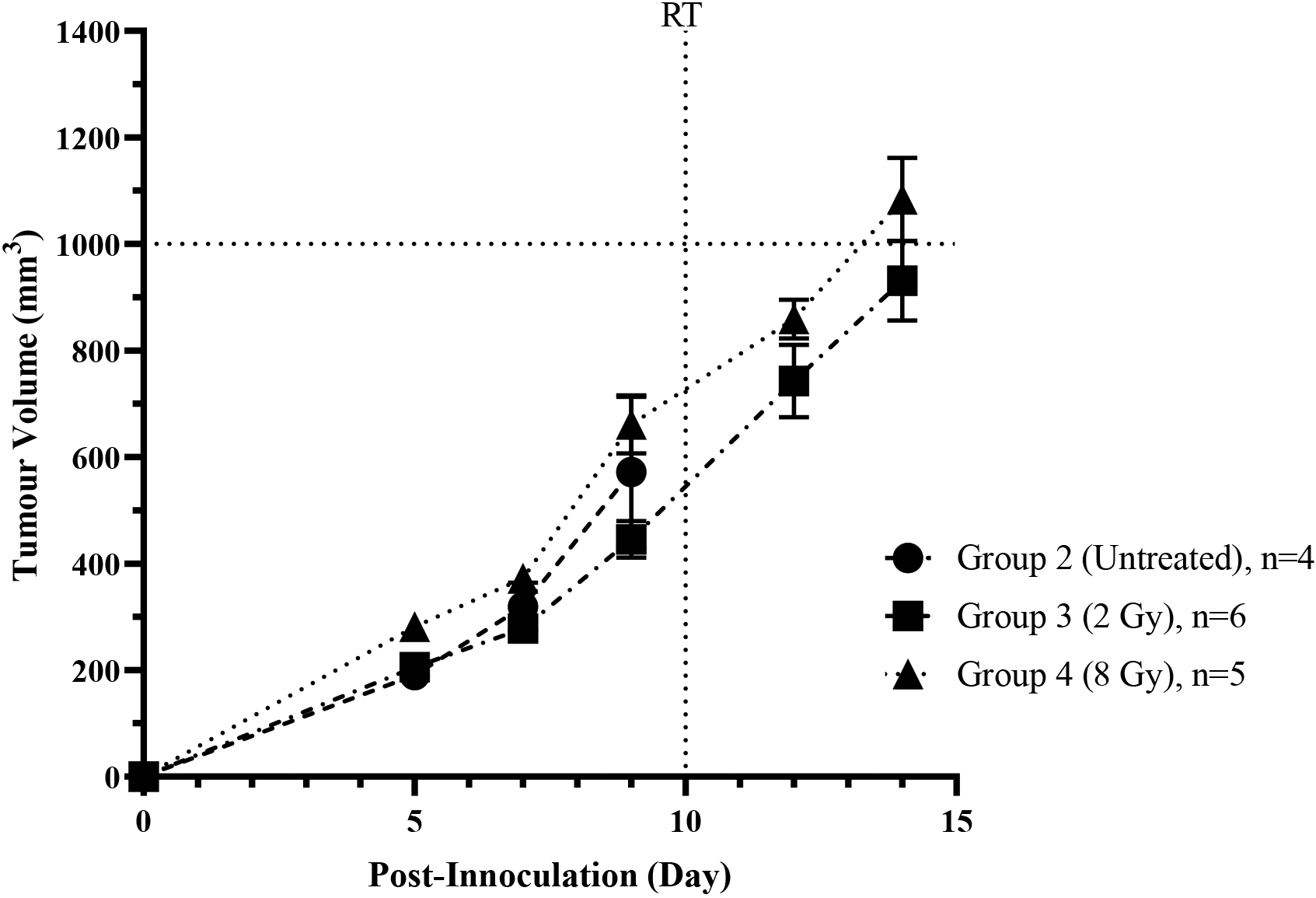
An approximate 0.2 mg tumour section was cut into smaller pieces, crunched with mortar, and filtered using a 70
Multiplex assay
Protein was extracted and estimated using Radioimmunoprecipitation Assay (RIPA, Sigma-Aldrich Solution, Darmstadt, Germany) Buffer and Pierce
Statistical analysis
Statistical analysis was performed using GraphPad Prism (GraphPad Software, San Diego, CA, USA). Changes in blood analysis, immune population, and concentration of cytokines within TME were compared across multiple groups by Kruskal-Wallis and Dunn’s post-tests with
Results
No difference in tumour growth during the early phase post-irradiation in both treatment groups
Gradual increment in tumour volume in 2 Gy and 8 Gy treated-groups 3 and 4, respectively were observed on days 5
No significant difference in the mean life span of post-irrradiation mice in the acute phase in both treatment groups
The mean life span of mice irradiated by 2 Gy gamma-ray (Group 5) was 20
The mean life span of mice in respective Groups 5 and 6, after treatment with a single dose of either 2 Gy or 8 Gy gamma irradiations in the acute phase.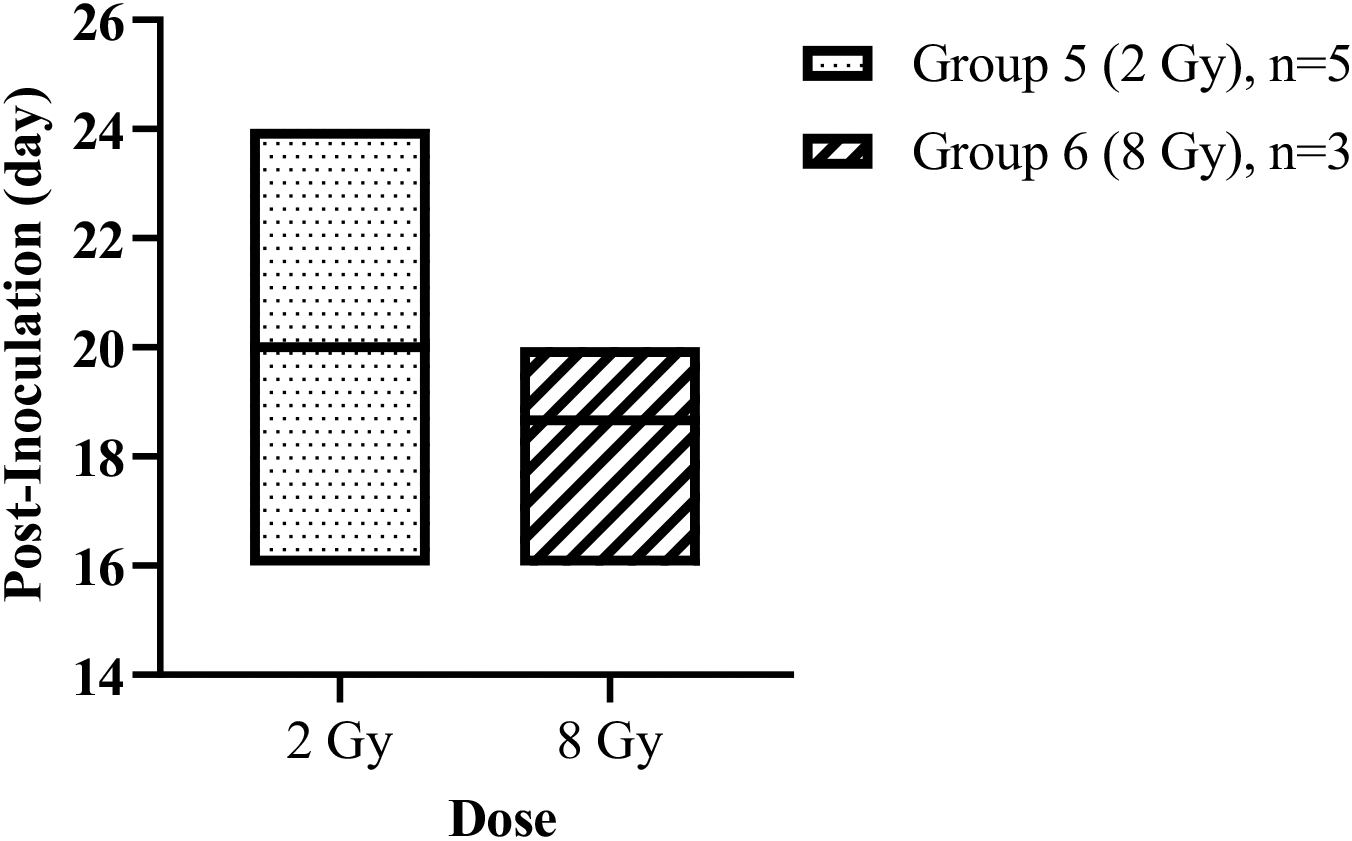
The total white blood cell (TWBC) count, absolute subsets counts, and percentage counts in negative control (Group 1), sham control (Group 2) and mouse-bearing tumour models in early (Groups 3 and 4) and acute (Groups 5 to 6) phases are presented in Table 1. There was no significant difference in white blood cell (WBC) counts and percentages between Groups 1 and 2. Irradiations of 2 Gy (
The total white blood cell count, absolute subsets counts, and percentage counts in negative control, sham control and mouse-bearing tumour models obtained from blood circulation
The total white blood cell count, absolute subsets counts, and percentage counts in negative control, sham control and mouse-bearing tumour models obtained from blood circulation
Data were expressed as mean values
During the early phase, both basophils’ absolute count (
As expected, lymphocytes’ absolute count and eosinophils’ percentage were decreased (
The immune cells within TME were isolated and measured acccording to flow cytometry gating strategy by Ibahim et al. (2022) based on previous studies published by Reichman et al. [28] and Yu et al. [29]. Immune cells within tumour sections in control and radiation-treated groups in the early and acute phases were isolated using the CD45 marker. The process continued with the isolation and measurement of different subpopulations of immune cells, including myeloid neutrophils (CD45
Sham control group has the highest percentage of total immune cells (44.38
Contrary to increment in lymphocytes especially T-cells, (
As expected, higher changes in the differences across study groups compared to systemic circulation was observed in the acute phase. Neutrophils, B-cells, monocytes and natural killer cells in Group 5 remained higher (
Targeted gamma-ray irradiation potentially suppressed pro-tumorigenic cytokines within TME
A total of 25 cytokines-related immune parameters were selected and measured to investigate the effect of single dose irradiation in TME. Except for IL-13, the other 24 cytokines were detected with 15 showing significant changes across the groups (Table 3a), and 9 without changes (Table 3b). IL-17 (
Similarly, in the acute phase of post-irradiation IL-17 (both doses
Discussion
Based on the Mouse Phenome Database (MPD) reports, the healthy 8-week-old female BALB/c mice used in this study have similar total white blood cell count and lymphocytes’ percentage. However, basophils have a twenty-fold greater percentage compared to the standard [30]. Despite identical strain and age, other factors such as environmental, dietary, and handling variables influenced the physiological characteristics, including immunological state of the mice [31].
Generally, uncontrolled growth of either cells or tissues or tumour resulted from failure of innate and adaptive immune responses leading to various homeostasis abnormalities. On day 5
The percentage of immune cells presence within TME in response to 2 Gy and 8 Gy gamma-ray irradiations duing early and acute phases
The percentage of immune cells presence within TME in response to 2 Gy and 8 Gy gamma-ray irradiations duing early and acute phases
Data were expressed as means values
Targeted gamma-ray irradiation of 2 Gy and 8 Gy induce changes to cytokines within TME in mouse-bearing tumour model
Data expressed as means values
Targeted gamma-ray irradiation of 2 Gy and 8 Gy have no effects on following cytokines within TME in mouse-bearing tumour model
Data expressed as means values
count decreased without significant difference in comparison to healthy mice. Tumour progression also altered immune cell percentage with significant elevation in eosinophil’s percentage. Tumour development might be decelerated or accelerated depending on the percentage of eosinophils present [32]. Despite several investigations, the involvement of eosinophils in cancer remains unclear due to different contradictory tumorigenic activities obtained from various cells [11, 33, 34].
Alteration within microenvironment’s residence was expected as the tumour was directly exposed to gamma-ray irradiation. Similarities in irradiation-induced immunosuppression has been established [35, 36, 37]. But presently, no significant difference was obtained in tumour development between irradiation-treated and sham control groups. A declined in the number of systemic TWBC compared to untreated group in early response to both irradiation doses indicated that irradiation as the caused for systemic effect. The indication is concluded based on our experimental setting where single targeted irradiation was exposed specifically to tumour site at hind leg, as well as the presented impact on TWBC in the systemic circulation [38].
Early immunomodulatory effects are dose-dependent [39, 40], since less alteration in the systemic immune population observed with considerably lower dosage of 2 Gy compared to 8 Gy gamma-ray irradiation. In this study, systemic modulation was presented in certain immune cells in either their absolute number or/and percentages. It can be seen in this study for both the percentages and absolute numbers of systemic eosinophils and basophils irradiated with 8 Gy gamma-ray were significantly reduced, however, only the absolute numbers were reduced for neutrophils and lymphocytes. Since eosinophil changes are low, several human studies reported the changes in eosinophil and neutrophil as eosinophil to lymphocyte ratio (ELR) or neutrophil to lymphocyte ratio (NLR) and have been used as a prognosis marker for inflammation event [41, 42, 43, 44]. However, in this study, the ratio of ELR obtained in normal mice is too large (1:1132) while the other radiation-treated groups have a 0 value that is unable to get the ratio. Changes in eosinophils absolute count appeared to vary depending on species and site of irradiation exposure [11, 45].
Lymphoid system’s vulnerability to ionising radiation is well known to cause lymphopenia [46, 47]. However, our study suggested basophils as more likely to be impacted. The effects of irradiation on basophils in both human and mouse models were unclear [48]. Monocytes were the most radioresistance, while lymphocytes, neutrophils, and basophils were radiosensitive [49, 50]. Accordingly, an increment in the percentage of monocytes by 10
The main effect of high-energy gamma-ray irradiation on TME was the destruction of tumour endothelial cells involved in triggering an immune response. TME consisted of various cells, including blood vessels, connective tissues, immune cells, and epithelial cells that emitted a variety of signalling molecules, including cytokines which governed the progression and growth of cancer. Certain cytokines, chemokines and growth factors influenced the immune response within TME [55, 56, 57, 58]. Various factors, including dosages and fractions of radiation treatments, potentially altered these mediators. Thus, the impact would affect either the activation or suppression of immune cells involved in either pro- or anti-tumorigenic activities [59].
Our experimental design enabled the isolation and measurement of the most common immune cells infiltrating TME. In contrast to eosinophils, the involvement of neutrophils, macrophages, myeloid-derived suppressor cells (MDSCs), natural killer cells, and both B- and T-lymphocytes in cancer progression and suppression have been extensively addressed [14, 60, 61]. Our findings were consistent with prior studies, which reported neutrophils and macrophages as the most prevalent immune cells penetrating tumours [62, 63]. Tumour-associated neutrophils (TANs), a term for neutrophils invading TME, appeared to have both pro- and antitumour effects [62, 64].
One of neutrophils’ anticancer effects was releasing mediators involved in the recruitment and activation of other immune cells under innate or adaptive immune systems [10, 65]. In contrast, pro-inflammatory mediators, including IL-6 and IL-1 were also secreted by TANs. In our study, the early response to single targeted irradiation resulted in a substantial drop in IL-6 level compared to sham control. Notably, a decreased in TANs was parallel with a reduction in IL-6, but not IL-1 in the irradiation-treated groups, thus suggesting pro-tumour stimulators were suppressed in early response to irradiation.
The released of IL-6 was also contributed by cancer-associated fibroblasts (CAFs) which was crucial for cancer progression [66, 67, 68]. An increased in CAFs activity accelerated tumour proliferation, angiogenesis, metastasis, and survival, as well as poor chemotherapy response [67]. On the other hand, suppressing IL-6 expression was associated with CAFs inhibiting the spreading of certain cancers [68]. Although the existence of CAFs was not determined in this study, the lower level of IL-6 in irradiation groups denoted the destruction of CAFs and consequently suggesting an anti-tumour impact. The released of IL-6 from CAFs interfered with the action of immune surveillance, such as hindering tumour infiltrating lymphocytes (TILs) and increasing tumour survival [66].
Interestingly, decreased in TANs and IL-6 occurred concurrently with elevation of TILs population within TME. Growing evidence suggested certain TILs subtypes reflected different prognostic significance in various cancers [69, 70, 71]. TILs mainly composed of T-cells consisting of different subsets, including regulatory T cells (Tregs) or CD4
Another result from single targeted irradiation of 2 Gy in parallel with reduction in TANs was a decreased in macrophages and dendritic cells’ populations. As with TANs, there are two subtypes of tumour-associated macrophages (TAM) responsible for antitumour and pro-tumour effects: type 1 macrophage (M1) and type-2 macrophage (M2) [76, 77]. M2 was associated with the expression of numerous pro-tumour mediators, including VEGF and IL-6 [78]. The inhibition of both cytokines suggested the treatment given induced suppression of M2 rather than M1. In addition, the rise of TNF i.e., a cytokine released by M1 [79], provided additional evidence for the notion that targeting gamma-ray irradiation at 2 Gy induced repression of pro-tumour activity.
An interesting finding in the acute response to single targeted 8 Gy was an increment of eosinophils within TME, known as tumour-associated tissue eosinophilia (TATE). This discovery was consistent with a previous study by Ibahim et al. [25] that reporting an increment of eosinophil-related gene expression, CCL11 or eotaxin after exposure to 7 Gy gamma-ray irradiation. Nine of the selected cytokines exhibited substantial alterations in response to both gamma-ray irradiation dosages of 2 Gy and 8 Gy was presented Table 3a. Three cytokines were associated with response to 2 Gy irradiation, but only RANTES was associated with response to 8 Gy irradiation. RANTES/CCL5, CCL11/eotaxin, VEGF, GM-CSF, IL-1, IL-6, IL-7, IL-15, IL-17, and TNF were considerably altered in response to 8 Gy irradiation and corresponded with TATE. These chemokines were also responsible for anti-angiogenesis, thus collectively serving as an anti-tumorigenic factor because increment in CCL11 expression favourable to the survival of cancer patients by metastasis inhibition [80, 81].
Damage to tumour vascularization was another well-documented consequence of irradiation on TME and has been extensively [82]. Deficiencies in blood flow and oxygen supply led to a hypoxic microenvironment within the tumour. Thus, hypoxic situation stimulated a rise in the expression of VEGF by tumour tissue. As an important player in tumour angiogenesis, VEGF helps delivered nutrients and oxygen necessary for tumour growth and development [83, 84]. Astonishingly, we found decrement in VEGF in radiation-treated groups slowing tumour angiogenesis. A decreased in VEGF expression resulted from irradiation exposure suggested an anti-tumorigenic activity. This was confirmed by metanalysis study demonstrating a connection between poor prognosis with high expression of VEGF among ovarian cancer patients [85]. Apart from that, VEGF can dampen an immune response by decreasing the maturation of anti-tumorigenic immune cells such as dendritic cells and increasing pro-tumorigenic cell types, such as T-regulatory cells, MDSC, and TAMs [86].
The role of different subtypes of IL-17 in both preclinical and clinical breast cancer experiments was discussed Fabre et al. [87]. In summary, IL-17 was suggested as the new target in cancer therapy as it promoted tumour progression [85, 88], angiogenesis, and resistance toward anti-tumour immunity, despite demonstrating anti-tumourigenic effect [87, 89]. The correlation between IL-17 with TNF-a in tumour progression and systemic inflammation were discussed. Depending on the IL-17 subtypes, they either served as pro-tumour cytokines leading to the production of other pro-tumorigenic cytokines [55, 88, 90] and/or anti-tumour cytokines inducing immune recruitment [88, 91, 92]. However, the suppressive effects on other pro-tumorigenic cytokines i.e., IL-6, GM-CSF, and VEGF obtained in our initial study suggested IL-17 acted synergistic with TNF-a cytokine post-radiation therapy and exhibited an anti-tumorigenic bias activity.
Different doses of single targeted gamma-ray irradiation were proposed to cause temporary changes in the immune population within TME, which differ in immune population in the systemic blood circulation and mainly correlated with suppression of pro-tumorigenic cytokines. Further investigation and a more extended experimental period is needed to understand the detailed mechanism.
Footnotes
Acknowledgments
The study was funded by a Fundamental Research Grant Scheme, Ministry of Education Malaysia (FRGS/1/2016/SKK08/UITM/03/2).
Conflict of interest
The authors have no conflicts of interest to disclose.
Author contributions
Funding acquisition: MJI.
Conception: NH, MJI.
Methodology: NH, NFRS, MJI.
Interpretation or analysis of data: NH, NAHH, MKAK, MJI.
Preparation of the manuscript: NH, NAHH, MKAK, SBSAF, EO, MJI.
Revision for important intellectual content: NH, NAHH, MKAK, SBSAF, EO, MJI.
Supervision: NAHH, EO, MJI.
