Abstract
BACKGROUND:
Nicotine metabolite ratio (NMR) can be used to predict total nicotine clearance. However, it is unknown whether NMR could be used as a marker of lung cancer risk.
OBJECTIVE:
To evaluate the blood metabolites of nicotine relating to the risk of developing lung cancer and investigate the combined effects of NMR and cigarette per day on the risk of lung cancer.
METHODS:
Among the 1,054 eligible subjects from the Korean Cancer Prevention Study-II biobank cohort, those with cotinine values below 0 ng/ml were excluded. Slow and fast metabolizer groups were defined using the median value of the NMR, calculated with the control group data, as the cut-point.
RESULTS:
The multivariable Cox proportional hazard models demonstrated that, the fast metabolizer group had a significantly higher risk of lung cancer than the slow metabolizer group (Adjusted HR: 2.02, 95% CI: 1.32–3.10). Fast metabolizers who smoked more than 15 cigarettes per day had an even higher risk of lung cancer (Adjusted HR: 3.51, 95% CI: 1.96–6.29) than the slow metabolizers who smoked less than 15 cigarettes per day.
CONCLUSIONS:
In summary, the NMR may be an effective marker for estimating tobacco-related disease risks such as lung cancer.
Introduction
Smoking tobacco products is the main cause of high lung cancer mortality and morbidity [1]. A recent study in Korea showed that population attributable risks of smoking for lung cancer deaths were 73.0% in males and 19.8% in females [2]. The number of smoking attributable deaths has continuously increased in Korea since 1985 [3].
The association between lung cancer and smoking is strongly related to the duration of smoking, the number of cigarettes smoked per day, and the inhalation patterns [4]. However, individuals are not equally susceptible to lung cancer for various reasons. In fact, only 11–24% of the smokers develop lung cancer [5]. It has been demonstrated that the differences in individual genetic susceptibility could contribute to the variability of tobacco smoke intake and metabolism of tobacco-specific carcinogens [6].
Consumption of tobacco carcinogens accompanied by intake of nicotine, which is a highly addictive component of tobacco. Nicotine is metabolized by cytochrome P450 (CYP2A6 enzyme) into cotinine and subsequently to trans-3’-hydroxycotinine. Measuring blood cotinine level is meaningful for estimating biological exposure to tobacco smoke as cotinine is a primary breakdown metabolite of nicotine [7]. Also, trans-3’-hydroxycotinine and cotinine have been widely used as the biomarkers for exposure to tobacco smoke [8, 9]. Nicotine metabolite ratio (NMR), the ratio of trans-3’-hydroxycotinine to cotinine, can be used to predict total nicotine clearance [10]. Studies have shown a positive correlation between the NMR level and CYP2A6 metabolizer in a dose-dependent manner [10].
A faster nicotine metabolizer would be more likely to be associated with a persistent smoking behavior for the purpose of maintaining nicotine concentrations in the body when compared with a slow metabolizer [11]. Thus, nicotine metabolism capacity has a direct impact on the degree of exposure to various toxicants and tobacco carcinogens [12, 13]. Increased cigarette consumption ultimately leads to higher potential risk of cigarette-related diseases [14].
To date, several studies have been conducted on the association between smoking status and lung cancer risk [15, 16]. However, whether NMR could be used as a marker of lung cancer risk has not been fully clarified in prospective studies. Thus, the objective of this study was to evaluate the blood metabolites of nicotine concerning the risk of developing lung cancer using data from a population based prospective cohort in Korea. Combined effects of NMR and cigarette per day on the risk of lung cancer among Korean adults were also investigated.
Materials and methods
Study population
This study included individuals of Korean descent in a population-based Korean Cancer Prevention Study-II (KCPS-II) cohort study from 2004 to 2013 [17]. These subjects were from 11 health promotion centers located in Seoul and Gyeonggi Province, Korea. The number of individuals who had provided informed consent for the study was 159,844 [18]. We conducted a case-cohort design within the KCPS-II biobank. Within the 159,844 individuals who had contributed informed consent for the study, we obtained 235 newly diagnosed lung cancer cases, including information on smoking status. Also, we randomly selected sub-cohort (
Endpoints
The primary endpoint of this study was the incident lung cancer which was followed from 2005 to 2018. The number of lung cancer cases was obtained from the Korea Central Cancer Registry based on codes of the International Classification of Diseases 10th revision. Lung cancer codes were C33 and C34.
Laboratory measurements
Blood samples were drawn from KCPS-II participants after 8 hours of fasting and stored at a temperature of
The limit of detection was 0.073 ng/ml for cotinine and 0.092 ng/ml for trans-3’-hydroxycotinine. Serum NMR was defined as the ratio of serum trans-3’-hydroxycotinine to serum cotinine. Samples with cotinine values below 0 ng/ml were excluded as those with such values were not likely to be exposed to nicotine. Samples with cotinine levels above 10 ng/ml and trans-3’-hydroxycotinine below the limit of detection were imputed as a value of 0.065 (LOD/sqrt
Statistical analyses
Baseline characteristics of smokers with cotinine-verified smoking status
Baseline characteristics of smokers with cotinine-verified smoking status
BMI, body mass index; SBP, systolic blood pressure; FBS, fasting blood sugar; LDL cholesterol, low-density lipoprotein cholesterol; NMR, nicotine metabolite ratio. Analyzed using
Smoking amount of smokers by age group
Estimated hazard ratios (HRs) and their 95% confidence intervals (CIs) of lung cancer risk were calculated using the Cox proportional hazards models. All statistical tests were two-sided and a
The analysis of 509 subjects showed the median follow-up time of 9.1 years. Subjects were divided into fast metabolizers and slow metabolizers based on the median NMR of the control subjects (NMR cut-off: 0.09). Table 1 shows the baseline characteristics of the subjects by the NMR status. Fast metabolizers were more likely to be males, showed higher lung cancer incidence, and were associated with a higher level of cigarettes per day. There was no statistically significant difference in age or BMI between the two groups. Mean serum concentrations of trans-3’-hydroxycotinine and cotinine at baseline are shown in Table 1. The mean NMR was 0.02 for the fast metabolizer group and 0.24 for the control group (Table 1). There were significant differences in levels of biomarkers for tobacco smoke exposure between the two groups (
The distribution of blood cotinine and trans-3’-hydroxycotinine and NMR for the study sample are shown in Fig. 1. The number of participants having higher concentrations of serum cotinine and trans-3’-hydroxycotinine was decreased. Table 2 shows the number of cigarette per day by the age group at baseline. In all age groups, fast metabolizers smoked more cigarettes per day than slow metabolizers.
In multivariable Cox proportional hazard models after adjusting for age, sex, BMI, and alcohol consumption, the fast metabolizer group was significantly associated with a higher risk of lung cancer than the slow metabolizer group (HR: 2.02, 95% CI: 1.32–3.10) (Table 3).
Hazard ratios of lung cancer by NMR level in those with cotinine-verified smoking status
Hazard ratios of lung cancer by NMR level in those with cotinine-verified smoking status
NMR, nicotine metabolite ratio; ref, reference; CI, confidence interval. HR is based on proportional hazards regression analysis. Nicotine metabolite ratio
Distribution of biomarkers for cigarette smoking exposure level. (a) Distribution of cotinine level. (b) Distribution of Trans-3’-hydroxycotinine level. (c) Distribution of NMR level.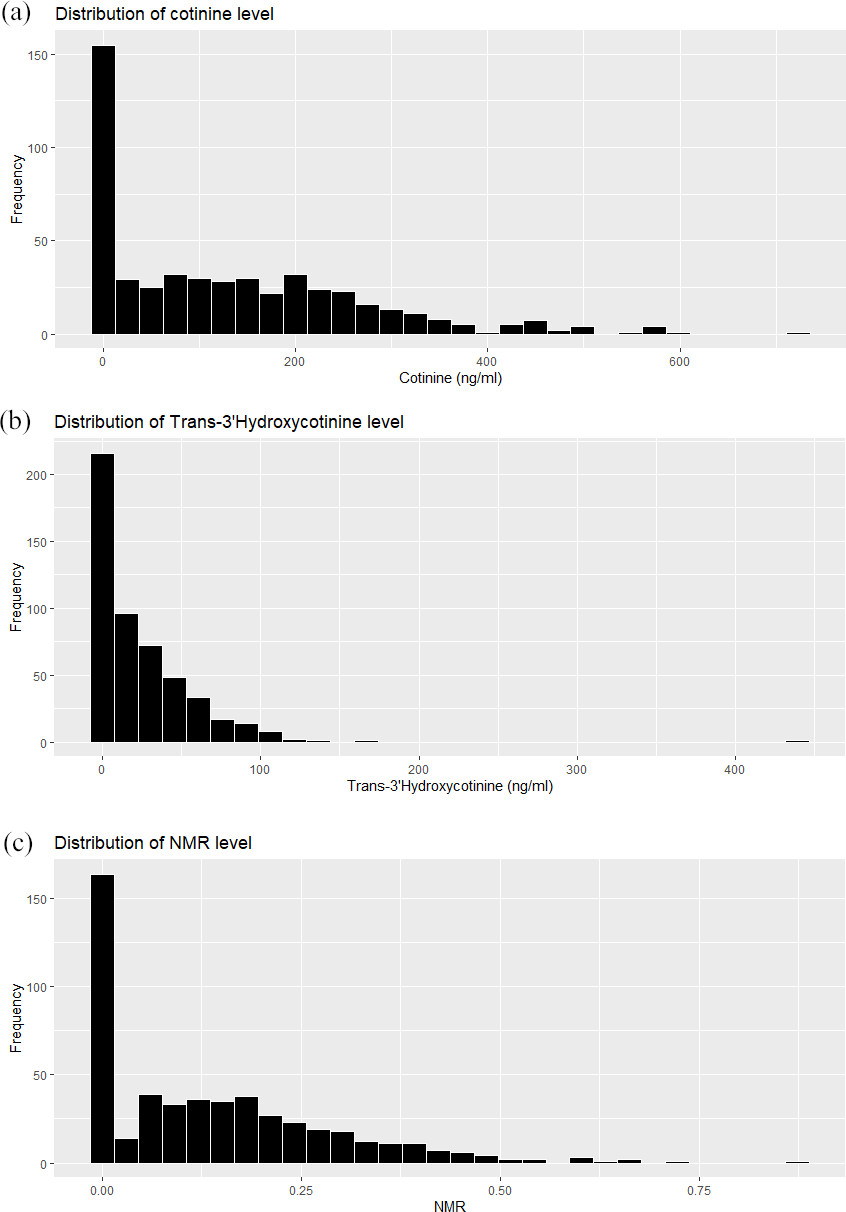
Hazard ratios of lung cancer by Cotinine, Trans-3’-hydroxycotinine, and NMR levels
P, percentiles; HC, trans-3’-hydroxycotinine; NMR, nicotine metabolite ratio; ref, reference. HR is based on proportional hazards regression analysis. Nicotine metabolite ratio
After repeating the main analysis by defining the NMR status into four groups, it was found that the NMR status was associated with lung cancer risk (fast metabolizers [100th percentile group] vs. slow metabolizers [50th percentile group as reference], adjusted HR: 2.78, 95% CI: 1.58–4.90) (Table 4). The associations between the biomarkers for exposure to tobacco smoke and lung cancer risk are shown in Table 4. In the adjusted Cox proportional hazard models, the highest cotinine 100th percentile group and the 90th percentile group were significantly associated with a higher risk of lung cancer when compared to those in the lowest 50th percentile group (100th percentile group, HR: 3.65, 95% CI: 2.06–6.45; 90th percentile group, HR: 2.26, 95% CI: 1.30–3.92). Moreover, compared with the lowest 50th percentile group of trans-3’-hydroxycotinine, adjusted HRs for developing lung cancer in the 90th percentile group and the 100th percentile group were 2.39 (95% CI: 1.36–4.19) and 3.29 (95% CI: 1.91–5.66), respectively (Table 4).
The cumulative hazard of lung cancer in the fast metabolizer group (NMR
Figure 3 illustrates the combined effects of NMR and the number of cigarettes smoked per day with the risk of lung cancer. We divided the participants into two groups by the median number of cigarettes smoked per day (CPD cut-point: 15). We found an additive model when we examined the modifying effect of smoking amount on the association between NMR levels and lung cancer risk. After adjusting for age, sex, BMI, and alcohol consumption, HRs (95% CI) for the risk of lung cancer among fast metabolizers who smoked less than 15 cigarettes per day were 2.28 (1.21–4.30) compared with slow metabolizers who smoked less than 15 cigarettes per day. The HR was increased to 3.51 when the fast metabolizers smoked more than 15 cigarettes per day.
Cumulative hazard curve of lung cancer risk: there is a significant difference in risk between the slow metabolizer group and the fast metabolizer group (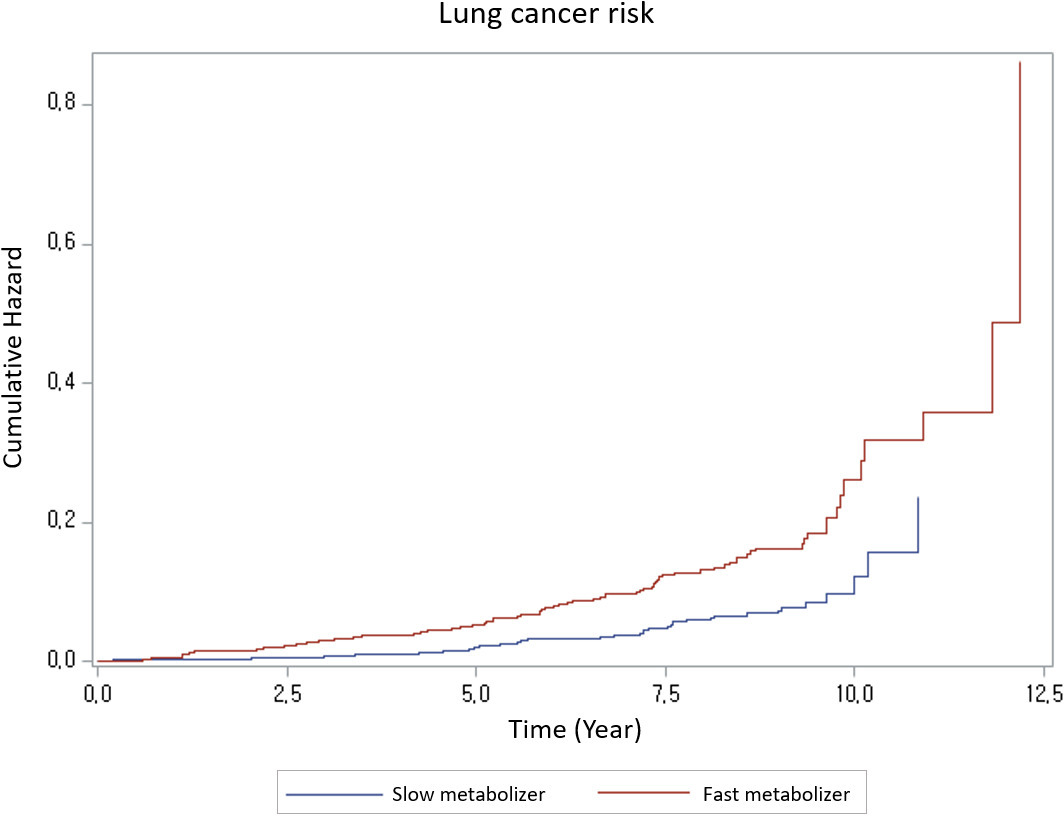
Hazard ratios (HRs) for lung cancer by NMR and cigarettes per day. 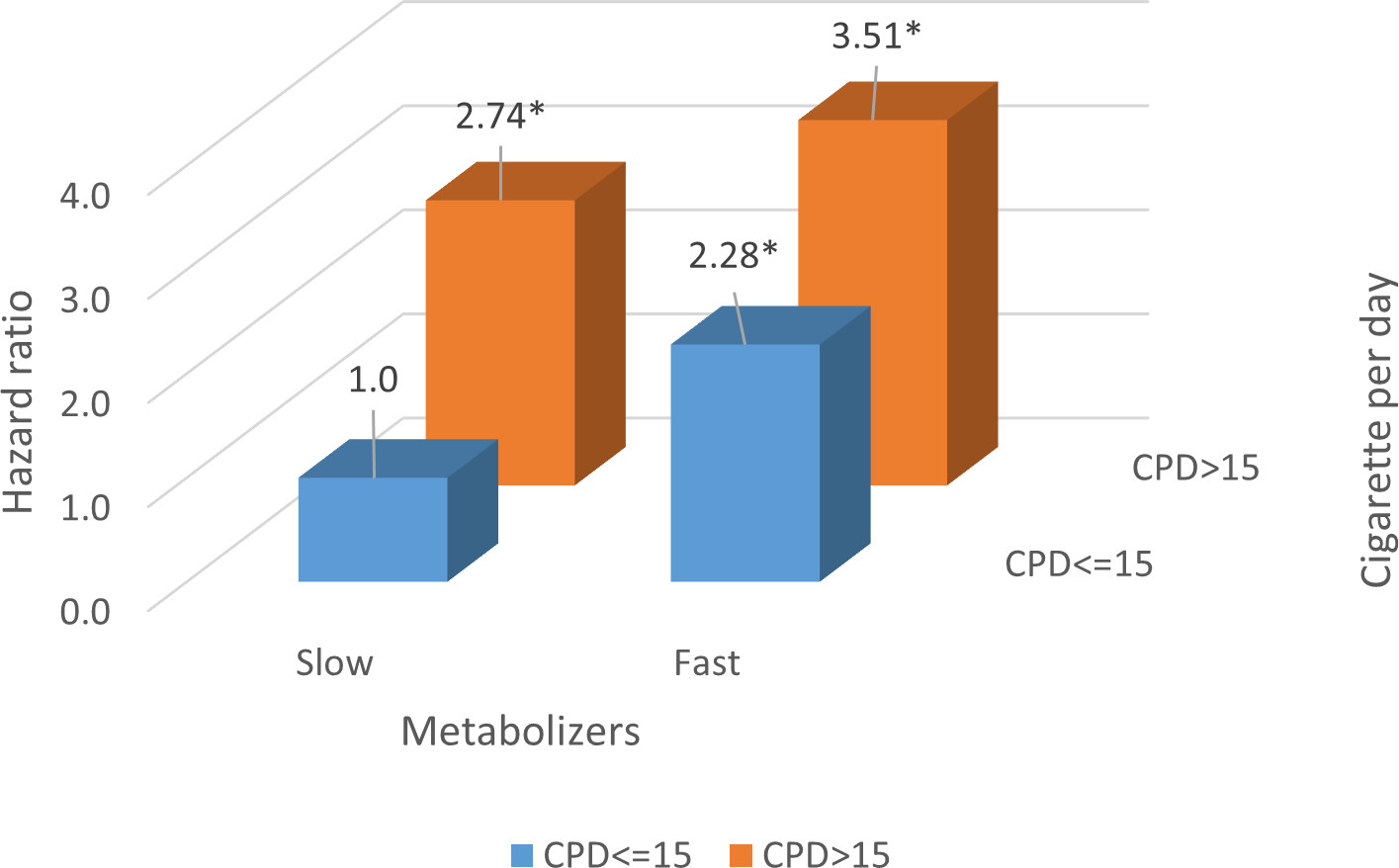
The aim of this study was to evaluate the association between the blood NMR and the risk of lung cancer. No previous study has examined whether serum NMR as a biomarker could directly estimate the risk of lung cancer in a general population cohort. In this study of 509 individuals from a population-based cohort, we found a significant association between serum NMR and lung cancer incidence. Those with high levels of NMR were associated with an increased risk for lung cancer. This study showed that the fast metabolizers have an approximately two-fold higher risk of developing lung cancer than the slow metabolizers even after adjusting for other variables. In addition, fast metabolizers who smoked a higher number of cigarettes per day showed the highest risk for lung cancer.
In this study of the hazard ratio model, we did not adjust the number of cigarettes per day and smoking years since cotinine, Trans-3’-hydroxycotinine, and NMR are already identified as the indicators of active smoking and markers for secondhand smoke exposure. Also, the previous paper adjusted only the fundamental factors to predict the performance of cotinine level for lung cancer risk [13]. Furthermore, we have tried additional analyses to address the issue regarding those factors, and the results showed consistency of positive association in the model including the number of cigarettes per day (Supplementary Table 1).
The association between the high levels of NMR and an increased risk of lung cancer could be explained by the smoking intensity owing to the nicotine dependence. The high intensity of smoking may induce sequential carcinogenic effect on people and can lead to the development of lung cancer. In many studies, it has been hypothesized that nicotine metabolism can increase nicotine dependence [20, 21, 22]. In accordance with the previous studies, the results of our study showed a positive relationship between nicotine dependence and smoking frequency [20, 21, 22]. Also, the number of cigarettes smoked per day was highly correlated with NMR in a dose-dependent manner.
The current study also found that in Korean adults with exposure to nicotine either directly or indirectly by smoking, an increased risk of lung cancer was associated with high levels of cotinine, trans-3’-hydroxycotinine, and NMR. This is consistent with a previous U.S. study that found that the fast nicotine metabolizers were associated with an increased risk for tobacco-related disease, and also were heavier smokers with higher exposure to numerous toxicants than the slow metabolizers [14]. Moreover, a significant positive association between CYP2A6 activity and the risk of smoking-related lung cancer has been observed in a multiethnic cohort study [9]. Also, the results of the present study were in close agreement with the preceding studies demonstrating that the odds ratio for lung cancer increased linearly with increasing cotinine levels [13]. Among the subjects with cotinine detected, the association between low levels of cotinine and an increased risk of lung cancer was weaker than that for individuals with high levels of cotinine. Increased levels of cotinine and trans-3’-hydroxycotinine were also associated with an increased risk of lung cancer.
Furthermore, this study showed that the cotinine level or Trans-3’-hydroxycotinine level better estimated the lung cancer risk than NMR. Since the serum Trans-3’-hydroxycotinine is a further metabolite of serum cotinine, Trans-3’-hydroxycotinine levels generally were two to four times lower than serum cotinine levels [23]. As the subjects of our study were chosen from the general population regardless of their smoking status, some subjects would have a much lower level of serum Trans-3’-hydroxycotinine that would be difficult to detect. Therefore, in the process of calculating NMR, 0 value of NMR has been founded more than that of cotinine and Trans-3’-hydroxycotinine. This could be diminishing the NMR performance of quantitative assessment of exposure to smoke with lung cancer than serum cotinine and Trans-3’-hydroxycotinine. Thus, further study would be needed with a larger study sample size that would improve the performance of NMR associated with lung cancer.
Recent studies have found a range of blood NMR cut-points of 0.2–0.3 in current smokers [22, 24, 25, 26]. Interestingly, however, our results showed that a low level of NMR cut-point was 0.09. In addition, there was no significant association between the NMR cut point, which has been derived from previous studies, and lung cancer risk in our study. This result could be explained by the fact that our study included not only active smokers, but also secondhand smokers as measured by the cotinine levels. Lung cancer risk among non-smokers is increasing as secondhand smokers are exposed to nicotine and toxins from smokers [27]. Therefore, we think our study with detectable cotinine levels at the population level is very meaningful.
Finally, this study has several limitations. First, we only had information on biomarkers for exposure to tobacco smoke, which limited the applicability of our results to an evaluation of genetic polymorphisms of the CYP2A6 gene. Plasma is a very reliable and reproducible biological source to measure NMR which can be used as a surrogate of CYP2A6 activity [28, 29]. However, future studies with genetic data are needed to verify the results of our study. Second, this study lacked specific information regarding the degree of exposure to passive smoking in participants. Therefore, further assessment is needed to identify trends in nicotine exposure among non-smokers. Third, this study has a limitation stemming from its small sample size.
Nevertheless, this study also has a few strengths. First, this is the first cohort study to investigate the comprehensive association between biomarkers for exposure to tobacco smoke, such as cotinine, trans-3’-hydroxycotinine, and NMR, and lung cancer. In addition, this study assessed the health risk from smoking in a general population of Korean adults. While the earlier experimental studies obtained the NMR cut-point only in active current smokers to find the effect of smoking cessation [30, 31], we defined the NMR cut-point for a general population who were exposed to smoking in any way possible. Therefore, we believe that the results of the current study are more applicable to the general population.
Conclusion
Based on a Korean cohort who is exposed to nicotine, the results of this study suggest that fast nicotine metabolizers might have a higher risk for lung cancer. In addition, the highest risk of lung cancer was observed in individuals with a fast metabolism who smoked a high number of cigarettes per day. In this respect, the individual NMR might be highly applicable to developing individual pharmacotherapies to treat tobacco dependence. If these results are validated in more studies, it will contribute to an in-depth understanding of the association between nicotine metabolism and lung cancer in the general population, thus having important implications for public health.
Author contributions
Conception: Guen Hui Kim, Sun Ha Jee and Keum Ji Jung.
Interpretation or analysis of data: Guen Hui Kim and Keum Ji Jung.
Preparation of the manuscript: Guen Hui Kim, Sun Ha Jee and Keum Ji Jung.
Revision for important intellectual content: Guen Hui Kim, Joyce Mary Kim, Sun Ha Jee and Keum Ji Jung.
Supervision: Keum Ji Jung.
Supplementary data
The supplementary files are available to download from https://dx.doi.org/10.3233-CBM220023.
sj-docx-1-cbm-10.3233_CBM-220023.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-220023.docx
Footnotes
Acknowledgments
This study was funded by the Research Program funded by the Korea Disease Control and Prevention Agency [fund code 2019-E6704-00] and the National R&D Program for Cancer Control through the National Cancer Center(NCC) funded by the Ministry of Health & Welfare, Republic of Korea (HA21C0142).
