Abstract
BACKGROUND:
Combination of different cancer markers is often used for predicting tumor growth, for the response to cancer therapy, and for increase in the positive diagnosis ratio in the malignant tumors.
OBJECTIVE:
Evaluation of the diagnostic efficacy of CA 15-3 and CA-62 cancer markers combination for early stages of breast cancer (BC) detection.
METHODS:
This blind study was performed on 2 clinically validated Sets that included serum measurements of CA 15-3 ELISA and CLIA-CA-62 assays in 488 serum samples with TNM classification. A study included 300 BC patients (254 at Stages I and II, 20 with ductal carcinoma in situ (DCIS), and 26 Stages III and IV patients), 47 patients with breast benign diseases, and 141 healthy controls.
RESULTS:
Sensitivity for DCIS & Stage I breast cancer detection was 75% at 100% Specificity (AUC
CONCLUSIONS:
The results obtained in a blind study demonstrate that a combination of CA15-3 with CA-62 yields 75% Sensitivity at 100% Specificity for DCIS and Stage I breast cancer detection, which has a potential to be integrated into existing screening programs.

Introduction
Global cancer statistics confirms that in the last decade female breast cancer has become the most prevalent cancer in women, with more than 2 million cases worldwide corresponding to 11.7 cancer incidence [9]. At the same time the global epidemiological reports [20] indicate that the breast cancer mortality rate has increased significantly in the last decade demonstrating an annual overall increase by 0.23%. Majority of the global breast cancer patients are diagnosed in the USA, Canada, Mexico, South America, Nordic countries, France, Switzerland, Israel, and the Netherlands [26]. In Russia, around 50,000 new BC cases are diagnosed every year, and approximately 30% of them are detected at advanced stages of cancer, which causes high net mortality rate. Cumulative risk of the breast cancer development is equal to 5.4% for a woman’s lifetime, and the incidence rate of breast cancer increases with age from less than 0.5% at 40 years of age to 2.86% at 60 years of age. Some other factors, such as hereditary breast and ovarian syndrome caused by BRCA1 & BRCA2 mutations, age-related changes in the female sex hormones levels up to 10 times increase the risk of breast cancer in this risk group than the baseline [9, 25].
Women with breast cancer diagnosed at Stage I have 98 percent or higher survival rate in the first five years [9, 2]. Obviously that non-invasive ductal carcinoma in situ (Stage 0) and early stages of breast cancer have better outcomes than more advanced stages of cancer. Over the course of the last two decades, a number of achievements have taken place in breast cancer research that significantly improved the chances for patient’s survival. Some significant advances in breast cancer screening methods, breakthroughs in cancer treatments, most recent achievements in molecular biology and molecularly targeted therapies provided better understanding of breast cancer development, enabling the design of the novel highly effective and safe treatment strategies. Breast cancer screening techniques include various tests such as mammography, magnetic resonance imaging, and ultrasound with or without elastography [2]. Currently there are a few ongoing clinical observational or prevention clinical trials for breast cancer such as BRCA-P (BCT 1801/ABCSG 50) or BCT 2001 (Breast MRI Evaluation) aimed to create the new methods to detect, precisely diagnose breast cancer, and reduce the risk of the disease for patients today and in the future.
The development of screening techniques for early detection of breast cancer is challenging due to high prevalence of the disease and the differentiation of benign and malignant breast tumors. The biggest drawback of the overall screening is related to the very high cost of the screening programs, which have significant budget implications, depending on the size of the population and the healthcare system resources involved [22]. Currently the most commonly used screening test for breast cancer in women without any symptoms is mammography, however it allows detecting only some fraction (from 20 to 60%) of Stage I breast cancer overlooking a majority of cases due to calcifications, imprecise aligning, biases, parenchyma density, and misinterpretations [4, 13, 21, 28]. As it turns out, a successful combination of various imaging methods such as mammography & MRI allows providing better detection of early stages of breast cancer (BC) as compared to single method’s use.
Besides imaging methods of diagnostics developed over the past few decades, some immunoassays based on blood serum biomarkers provide a non-invasive promising approach for improving detection and monitoring of breast cancer [30]. For instance, a well-known cancer marker CA 15-3, an O-glycoprotein of the mucin family is commonly used for breast cancer control ma-nagement [2, 28]. Despite its low sensitivity for early stages of breast cancer detection, CA 15-3 cancer antigen is extensively used for cancer treatment monitoring in combination with clinical examination and various imaging methods and for early detection of cancer recurrence [1, 6]. As opposite to the mucin CA 15-3, a marker for epithelial carcinomas CA-62 have demonstrated its diverse clinical potential for early detection of epithelial cancers of the breast, prostate, lung, and colon for screening, cancer treatment monitoring, and cancer recurrence detection [11]. Heavily glycosylated N-glycoprotein CA-62 is a mesenchymal marker that is expressed in large quantities from the onset of cancerogenesis, starting from the abnormal cells production by non-invasive carcinomas in situ, which is the unique quality of this biomarker.
In this paper, we present the results of the clinical blind study to discuss the combined use of two glycoproteins CA 15-3 and CA-62 for Stage I breast cancer detection using an elevated cutoff value to eli-minate false positives among healthy women. Overall, we assessed a combined use of CA 15-3 and CA-62 cancer markers for Stage I breast cancer detection and DCIS, as well as sensitivity (Sen), specificity (Sp), po-sitive predictive value (PPV), negative predictive value (NPV), area under the curve (AUC), and test accuracy for early stages of breast cancer detection.
Baseline characteristics of the patients
Baseline characteristics of the patients
Serum samples
All cancer serum samples were taken before the treatment administration. Tumor staging was performed according to the tumor, lymph node, metastasis (TNM) classification by AJCC 8th Edition [14]. The TNM System for describing the anatomical extent of a neoplasm is based on 3 components: T – the size or direct extent of the primary tumor, N – the degree of cancer spread to regional lymph nodes, and M – the presence of distant metastases. Histopathological classification of the breast tumors was used as the gold standard in breast cancer diagnosis. The entire set of serum samples, including Set 1 and Set 2 was separated by centrifugation (1300 g, 10 minutes) in BD SST tubes with silica clot activator and separating gel, heat inactivated at 56
Total of 488 blind serum samples from 300 histologically confirmed breast cancer patients with known TNM classification, 47 patients with benign breast diseases and 141 healthy individuals were included in this study. The study included 2 validated sets. Blind validated Set 1 included 326 serum samples including healthy controls (
Blood from healthy control subjects (
Ethics approval and consent to participate
The study was approved by the Ethics Committee of the Sechenov First Moscow State Medical University. All patients were given an informed consent to participate in the study. In total 57 patients with histologically-confirmed BC and 69 healthy subjects from the prospective study were included in the analysis. Serum samples were collected at the Sechenov University Hospital after overnight fasting and delivered to the Clinical laboratory. The rest of serum samples were collected as consented donor samples from Federal licensed/re-gistered facility following GMPs. The protocol for the study was approved by the local IRB. The informed consent for archived samples from the bio bank was not required by IRB.
Statistical analysis
The distribution of CA-62 and CA 15-3 levels in sera from healthy, breast benign, and breast cancer patients were tested for normality using the histograms. The determination coefficient (r) was used to evaluate the correlation between cancer antigen’s CA-62 and CA 15-3 serum levels. Since CA-62 values differ by 1000 times from CA 15-3 values, the original measurements for each cancer antigen in different sample cohorts were log transformed (log10) before the analysis for obtaining the same equivalent scales. The ROC curves for the comparison and the distribution of both tumor mar-kers CA-62 and CA 15-3 for different stages of breast cancer and non-cancer samples were plotted using multiple variables graph tool with default settings of the MedCalc statistical software (version 19.7.4, MedCalc Software Ltd, Belgium, EU). A combination of two antigens was presented both in graphical and statistical formats. To plot the ROC curve for the combination of CA-62 and CA 15-3 cancer markers using defined parameters the following technique was developed: a conversion of two independent parameters, such as CA-62 and CA 15-3 values to one arbitrary value corresponding to the principles-based developed testing approach. The method is described in detail in the Results.
Diagnostic characteristics of CA 15-3 ELISA, CLIA-CA-62 assay, and their combination were evaluated based on sensitivity and specificity, test accuracy, PPV, NPV, and the ROC-curves AUC for different IVD me-thods were compared. The level of significance was set at
The sandwich CA 15-3 ELISA immunoassay
CA 15-3 solid-phase one-step sandwich ELISA (Federal Service for Surveillance in Healthcare of Russia (FSSH) – approved in vitro diagnostic (IVD) medical device from Vector-BEST, Novosibirsk, Russia, Serial No # T-8472) is designed for the quantitative measurement of the cancer antigen CA 15-3 bound between two types of monoclonal antibodies specific to different epitopes of CA 15-3 cancer antigen in human serum. Capture CA 15-3 specific monoclonal antibodies (Mabs) were immobilized in the wells of the microplate provided in the kit. CA 15-3 calibration standards, serum samples. Positive control, and a working solution of the secondary anti-CA 15-3-HRP Mab are then added to the wells that react with the antibodies to form the immune complex of the HRP-enzyme with Mab-CA-15-3 that generates measurable optical signal to be recorded by ELISA colorimetric reader. A linear calibration curve CA 15-3 is plotted for the quantitative measurement of the analyte concentration in unknown specimens. The intensity of the optical density (O.D) is directly proportional to the concentration of CA 15-3 antigen present in the analyzed specimens. Time-to-result: 3.5 Hrs. Sensitivity of the assay: 0.5 U/ml, a diagnostic range: 10–250 U/ml, a detection method: colorimetric. The cut-off value (30 U/ml) was recommended by the manufacturer based on CA 15-3 levels in sera from healthy females (
Human CLIA-CA-62 immunoassay
The Human CLIA-CA-62 assay (FSSH – approved IVD medical device from JVS Diagnostics LLC, Moscow, Russia, Serial No # CLIA-CA-62-200221) is designed for the quantitative measurement of the carcinoma-specific antigen CA-62 in human blood serum for the detection of various epithelial carcinomas in the adult population, regardless of their gender and race. A set of reagents is based on a competitive solid-phase chemiluminescent immunoassay, where the competition for active binding sites of specific anti-CA-62 Mab, captured on the wells of the provided microplate carried out between the cancer antigen present in the analyzed sample and the luminescent CA-62 conjugate. Serum samples, CA-62 standards, Positive control, and CA-62 conjugate solution are then added simultaneously to the wells that react with the
Descriptive statistics of serum CA 15-3 and CA-62 values in healthy, breast benign and breast cancer patients (median, mean, SD, 95 percentile)
Descriptive statistics of serum CA 15-3 and CA-62 values in healthy, breast benign and breast cancer patients (median, mean, SD, 95 percentile)
capture antibodies to form the
Measurement of CA 15-3 and CA-62 serum levels in breast cancer, breast benign patients, and healthy controls
The entire set of serum samples was analyzed using in vitro diagnostic FSSH – approved IVD medical devices, such as sandwich CA 15-3 ELISA assay and a competitive chemiluminescent immunoassay CLIA-CA-62 for CA-62 glycoprotein concentration measurement. Table 2 shows the descriptive statistics of CA 15-3 and CA-62 values in the serum of various stages of breast cancer, benign breast diseases, and healthy controls. CA 15-3 values correlated with the literature data [12, 14] for the early and advanced stages of breast cancer. Measuring serum levels of cancer markers CA-62 and CA 15-3 in 488 serum samples (300 samples from patients with histopathological confirmation of breast cancer, 47 samples from breast benign patients and 141 samples from apparently healthy women) have established that CA-62 antigen was found to be elevated (12286 U/ml) in 279/300 (93%) breast cancer patient’s samples (111/300 (37%) elevated serum level for CA 15-3 marker) as compared to its level in healthy women (2679 U/ml) and women with breast benign diseases (4465 U/ml). As opposed to CA 15-3, serum values for CA-62 glycoprotein have demonstrated the highest mean value for DCIS (Stage 0) (12223 U/ml) and Stage I (11856 U/ml) of breast cancer, that were decreasing with more advanced stages: Stage II (10092 U/ml), and Stages III and IV (9850 U/ml). Using recommended cutoff values for CA 15-3 ELISA and the CLIA-CA-62 assay the sensitivity was calculated for all stages of breast cancer, including Stage 0 (DCIS). The results obtained are presented in Table 3. It is worth mentioning that whereas the sensitivity of CA 15-3 is lower for early stages (20%–37%) as compared to advanced stages of breast cancer (46%), a sensitivity for the marker for epithelial carcinomas CA-62 is very high in all stages, with its maximum at pre-invasive DCIS (Sen
Sensitivity of CA 15-3 ELISA and CLIA-CA-62 assays for different stages of breast cancer using recommended cutoff values
Sensitivity of CA 15-3 ELISA and CLIA-CA-62 assays for different stages of breast cancer using recommended cutoff values
Despite the CA-62 marker’s close mean and median values, the difference in the sensitivity of CA-62 for various stages is significant. The
Combined ROC curves of CA 15-3 (a) and CA-62 (b) cancer markers for each stage.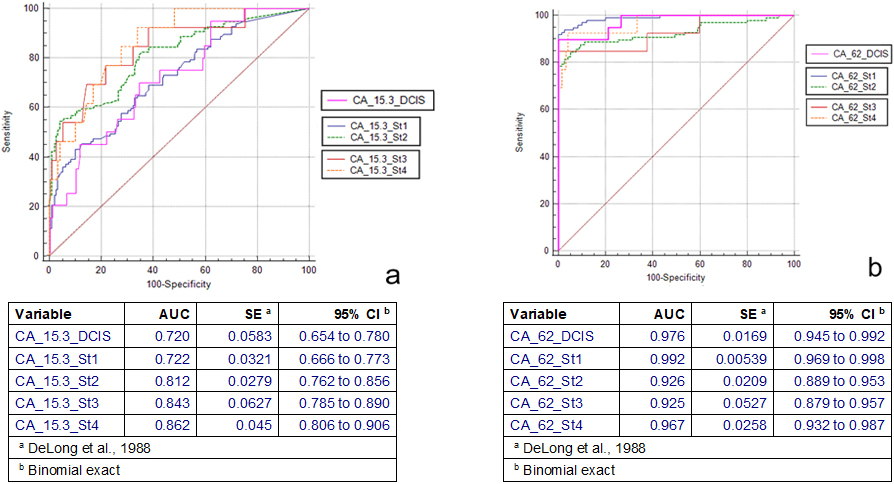
The comparison of ROC curves for both tumor mar-kers CA-62 and CA 15-3 for different stages of breast cancer and non-cancer samples (breast benign and healthy control subjects) were plotted using graph tool with default settings of the MedCalc statistical software. To classify a sample as positive or negative, originally were used recommended cutoff values for single cancer antigens and later was determined a new combination of cutoff values for each antigen that resulted in 100% specificity. Figure 1 shows the CA 15-3 (a) and CA-62 (b) ROC curves for Stage 0 (DCIS), Stage I, Stage II, and Stages III and IV. The overall performance of the CA 15-3 ELISA was lower (AUC
Comparison of ROC curves
Comparison of ROC curves
1. Standard Error, DeLong et al., 1988. 2. 95% Confidence Interval, Binomial exact.
The diagnostic characteristics of the biomarkers
To plot the ROC curve for the combination of CA-62 and CA 15-3 cancer markers using defined parameters was implemented the following mathematical technique: a conversion of two independent parameters, such as CA-62 and CA 15-3 values, to one arbitrary value corresponding to the principles-based developed testing approach in accordance with the following ma-thematical transformations:
Samples with 10 Samples with CA 15-3
Samples with CA 15-3
Therefore, samples were considered as “positive” if: CA15-3
ROC-curve for Stage I with DCIS using a combination of CA 15-3 and CA-62.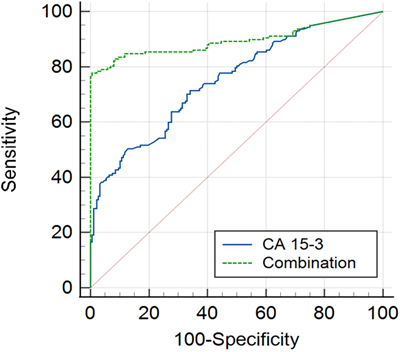
A correlation between CA 15-3 and CA-62 cancer markers. A 100% Specificity cutoff value for CA 15-3 and CA-62 required a log scale on the x-values.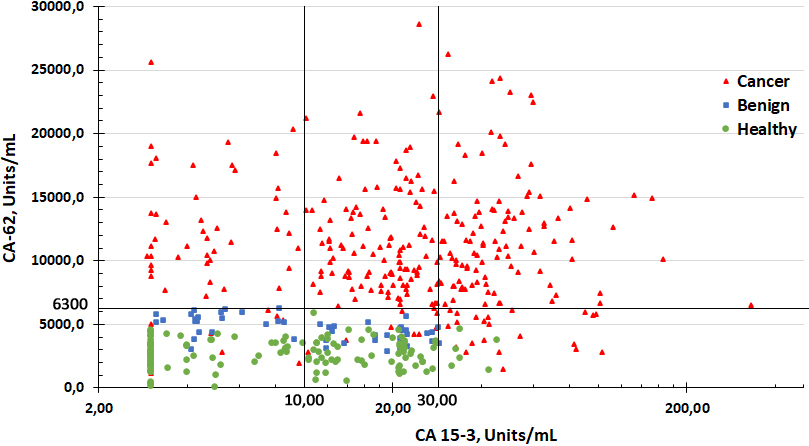
Given the incidence rate of breast cancer and the limited sensitivity (60–90%) of the mammography commonly used for breast cancer detection caused by many possible influences, such screening approach produces a large number of “false positive” results for women with “suspicious mammograms”, which are incorrectly categorized as positive for the presence of disease. Pearson correlation analysis demonstrated low correlation coefficient between CA 15-3 and CA-62 values for the entire set of samples (
We have determined the optimal combination for two biomarkers (Ca 15-3 & CA-62), which considers samples as “positive” if 10
Discussion
In this paper we present the results of the blind study to discuss a combined use of two glycoproteins CA 15-3 and CA-62 for Stage I breast cancer detection using a very high cutoff value for both markers to eliminate false positives among healthy women. A combination of the IVD chemiluminescent CLIA-CA-62 and colorimetric CA 15-3 ELISA assays appears to offer prospective cost-effective solution for novel breast cancer primary screening strategy prior mammography.
Breast cancer is a silent killer, with very minor symptoms that are associated with yet invasive late-stages of the disease. The incidence rate of breast cancer is around 90 per 100,000 women in Western Europe and North America and around 57 per 100,000 women in Eastern Europe. With such frequency a successful screening strategy must provide a sensitivity of at least 75% at close to a 100% specificity to significantly decrease a probability of detecting false positive results. A number of screening approaches using cancer biomar-kers and their panels were used in the past such as a screening study in the UK Collaborative Trial [15, 24].
Currently the mammographic screening is the most common approach to breast cancer detection worldwide and it is the only one test clinically proven to decrease mortality rates [2]. Besides the obvious advantage of the mammography which allows detecting the malignant tumors less than 5 mm (26.9 % of BC cases) including some carcinomas in situ (DCIS), it has some significant drawbacks. For example, the mammographic screening of women is producing a significant amount (3/4) of “suspicious mammograms” (2.2%; 2200 per 100,000 women) that are not associated with breast cancer and considered as false positive results, decreasing a diagnostics efficacy of this screening strategy [20]. As it was demonstrated by authors [4, 23] based on the results of the multicenter clinical trials, about 40% of BC cases are “missed” early-stage as a result of the detection and misinterpretation errors (dense parenchyma, calcifications, distortions, poor aligning etc). Therefore, the mammography detects only 63–90% of breast cancers in patients at screening that gives a probability of 10 to 37% that the breast cancer will not be detected. According to the WHO screening recommendations [5] the overpriced economic costs to the health care system in regards to screening programs are related to a large number of false positive results that causes over-diagnosis, over-treatment, false positives, and strain on health systems. The quality of the screening test is described in terms of Sensitivity and Specificity. It is desirable that the screening test has a high PPV. On another hand, high NPV, a posteriori probability given a negative test result, reflects, first of all, the probability for someone to truly be healthy, in case of a negative test result, but not a reduction in costs. This leads to late cancer detection, which can be offset by regular screening with high sensitivity and specificity.
Therefore, there is a need in a new cost-effective approach that will result in a high positive predictive value (PPV) and a minimum rate of false positive results. It could be an improvement of the current mammographic approach using an additional instrumental method for visual detection of the vascularization associated with malignant tumors or a combination of the mammography with sensitive in-vitro diagnostic methods.
The results reported in this paper strongly suggest that the CLIA-CA-62 chemiluminescent assay is able to detect a significant increase in the CA-62 serum level of breast cancer patients in the DCIS, Stage I, Stage II, and Stages III and IV as compared to healthy women and women with benign breast lesions. The established overall sensitivity of CA 15-3 assay for breast cancer detection is in the range from 20 to 50%, which correlates with our findings (Sen
Unique quality of the marker for epithelial carcinomas CA-62 consists in its significant expression from the onset of cancerogenesis with its maximum production at non-invasive carcinoma in situ DCIS and Stage I breast cancer (Sen
Comparison of different breast cancer diagnostic methods allows making a suggestion that only a combination of several methods is superior to the single use of either method for Stage I breast cancer detection providing a significant improvement in the DCIS and Stage I detection having high survival rate.
Our results validate that a combination of CA 15-3 with CA-62 biomarker could be potentially useful for precise differential diagnosis in breast cancer (DDx). A combination of two different N- and O-glycoproteins could significantly improve overall diagnostic results due to their specific features allowing successful diffe-rentiating benign from malignant breast tumors. Clinical value of CA 15-3 as a biomarker in breast cancer is very significant for management of BC patients, such as treatment monitoring and early detection of cancer recurrence [16].
CA 15-3 is a secretory heavily O-glycosylated protein with high molecular weight that belongs to the MUCIN family, which secrets into the blood stream from the polysaccharide surface of the epithelial cells [16]. In cancer cells some structural changes of CA 15-3 occurred due to abnormal modifications in the glycosylation pattern to less glycosylated and more mobile form, inducing metabolic re-programming associated with tumor cell proliferative activity, resistance to hypoxia and stimulation of angiogenesis. Hyper expression of the mucin CA 15-3 occurs as the result of the Tumor necrosis factor (TNF) and Interleukin IL-8 production [25]. Increased expression of the mucin (CA 15-3) causes a suppression of the Tall-like receptors signaling and a decrease of the IL-8 expression. Therefore, understanding of the mucin expression role in cancer cells survival has an important impact on the development of the novel therapeutic, screening, and cancer treatment monitoring approaches for the successful treatment of breast cancer patients. A majority of other cancer markers including CA 15-3 represent heavy weight O-mucins (up to 800 kDa) that can get released into the blood only after tumor cells lysis. In this case, tumor cell death releases into the blood all the accumulated tumor-specific, tumor-associated and genetic markers which levels are directly proportional to the tumor growth.
As opposed to mucins, a marker for epithelial carcinomas CA-62 belongs to a family of membrane N-glycoproteins that bind alpha-fetoprotein (AFP) using a special combination of oligosaccharides located on the surface of the antigen and function by receptor-mediated endocytosis [11]. N-glycoprotein CA-62 is a mesenchymal marker produced as a result of the malignant cells transformation by epithelial-mesenchymal transition (EMT) in which epithelial cells lose their characteristics (cell to cell junctions, their polarity, some epithelial markers) and acquire mobile cell shape and mesenchymal features. As a result, various epi-thelial cells possessing different morphological and phylogenetic classifications are transformed into the same pluripotent stem cells. Heavily branched N-glycoprotein is expressed in large quantities on the cell membrane of such degenerated mesenchymal stem cells from the onset of cancerogenesis, and is getting released into the blood stream from the cell membrane on the very early stages of cancer, including carcinomas in situ and detected by the CLIA-CA-62 chemiluminescent assay.
Previous studies have shown that malignant epithelial cells demonstrate significant expression of glycoprotein antigens CA-62 in all studied localizations of epithelial carcinomas (such as breast, prostate, lung, uterus, stomach, kidney, colon, and ovaries) [11]. At the same time, healthy controls do not demonstrate increased expression of CA-62 markers. However some breast benign specimens have demonstrated a slight increase in CA-62 level that might indicate a transitional stage of the tumor becoming malignant, which was actually confirmed lately for some benign patients. Patients with significant elevation in serum CA-62 level might have another type of carcinoma, which does not make it false positive for breast cancer detection, but rather beneficial for simultaneous detection of some other existing primary cancer.
Proposed screening algorithm for target population over 30 years old using a combination CA-62 and CA 15-3.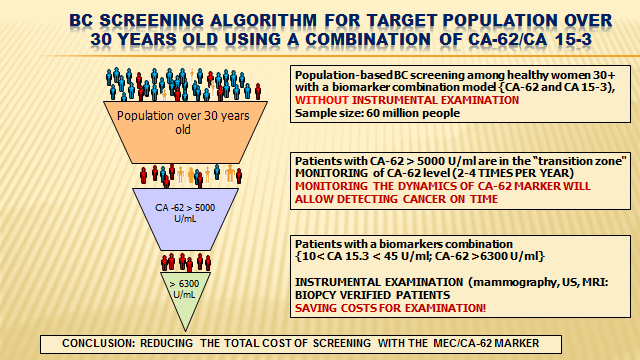
Taken together the results of the biomarkers (CA 15-3 and CA-62) combination may possibly be considered as indicator for early stages of BC and demonstrate their potential implementation into breast cancer screening programs. A proposed screening algorithm is presented in Fig. 4. Briefly, patients with CA-62
Thus, the results obtained demonstrate that a combined use of serum CA 15-3 and CA-62 values provides high level of specificity (100%) and sensitivity (75%) suitable for the clinical detection of early stages of breast cancer, which could provide an effective tool for prior mammography breast cancer screening within generally healthy women above 35 years of age. This approach could also increase the diagnostic sensitivity in DCIS and Stage I breast cancer detection.
The indisputable advantage of using CA-62 cancer antigen for a biomarker’s combination consists in its very high sensitivity (Sen
In the context of this blind study, a biomarker’s combination approach could prospectively be integrated into existing screening programs. The results obtained in the study confirm that by combining serum values of CA 15-3 and CA-62 it is possible to obtain a useful non-invasive screening test that will detect two-thirds of Stage I breast cancer without any false positives among healthy women. Such algorithm could supplement existing mammography screening as well as other diagnostic imaging methods. For the implementation of such biomarkers combination in the screening of BC our findings need to be confirmed in a clinical approbation.
The evidence from this study presents remarkable results in regards to the biomarker’s combination for Stage 0 and Stage I breast cancer detection (Sen
Footnotes
Acknowledgments
This work was financed by the Ministry of Science and Higher Education of the Russian Federation within the framework of state support for the creation and development of World-Class Research Center “Digital biodesign and personalized healthcare” No 075-15-2020-926.
Author contributions
Conception: Tcherkassova J.
Interpretation or analysis of data: Tcherkassova J, Ragoulin V.
Preparation of the manuscript: Prostyakova A.
Revision for important intellectual content: Tcherkassova J, Prostyakova A, Tsurkan S, Boroda A and Sekacheva M.
Supervision: Tsurkan S, Sekacheva M.
