Abstract
Drug resistance is a critical factor responsible for the recurrence of non-small cell lung cancer (NSCLC). Previous studies suggest that curcumin acts as a chemosensitizer and radiosensitizer in human malignancies, but the underlying mechanism remains elusive. In the present study, we explored how curcumin regulates the expression of miR-142-5p and sensitizes NSCLC cells to crizotinib. We found that miR-142-5p is significantly downregulated in NSCLC tissue samples and cell lines. Curcumin could increase crizotinib cytotoxicity by epigenetically restoring the expression of miR-142-5p. Furthermore, curcumin treatment suppressed the expression of DNA methylation-related enzymes, including DNMT1, DNMT3A, and DNMT3B, in NSCLC cells. In addition, the upregulation of miR-142-5p expression increased crizotinib cytotoxicity and induced apoptosis in tumor cells in a similar manner to that of curcumin. Strikingly, miR-142-5p overexpression suppressed crizotinib-induced autophagy in A549 and H460 cells. Mechanistically, miR-142-5p inhibited autophagy in lung cancer cells by targeting Ulk1. Overexpression of Ulk1 abrogated the miR-142-5p-induced elevation of crizotinib cytotoxicity in A549 and H460 cells. Collectively, our findings demonstrate that curcumin sensitizes NSCLC cells to crizotinib by inactivating autophagy through the regulation of miR-142-5p and its target Ulk1.
Introduction
Non-small cell lung cancer (NSCLC) includes squamous cell carcinoma, adenocarcinoma, and large cell carcinoma, and accounts for approximately 80% of lung cancers. It is the leading cause of cancer death worldwide [1, 2] and, despite the advances in NSCLC therapy, the 5-year survival rate is still unsatisfactory. Resistance to chemotherapy is one of the most important causes leading to the recurrence of NSCLC. Crizotinib, a small molecular inhibitor of anaplastic lymphoma kinase (ALK), shows superior efficacy on progression-free survival versus chemotherapy in ALK-positive NSCLC cases. Nevertheless, several cycles of treatment with crizotinib may lead to drug resistance and therapy failure [3, 4]. Thus, the mechanism of resistance to crizotinib and its associated chemosensitizers need to be investigated.
Curcumin (diferuloylmethane), a flavoring agent extracted from Curcuma longa rhizomes, exhibits various biological properties such as anti-inflammatory, immunoregulatory, and antitumor activities [5, 6]. Epigenetic modification is a critical mechanism in the tumor-suppressive role of curcumin [7, 8]. Studies have shown that curcumin acts as a DNA hypomethylating agent to restore the expression of multiple tumor suppressors, chemosensitizers, and radiosensitizers [9, 10]. miR-142-5p is considered a tumor suppressive miRNA on multiple cancers, including NSCLC [11, 12]. The expression of miR-142-5p is frequently decreased partly due to epigenetic modification on its promoter regions [13]. However, it remains unknown whether curcumin might restore miR-142-5p expression in NSCLC via epigenetic regulation.
Autophagy is a conserved cellular process that removes misfolded proteins and damaged organelles through the lysosomal pathway, and facilitates essential activity and viability in response to stress [14]. Autophagy plays a critical role in tumor cell growth, metastasis, and drug resistance [15]. Elevation of autophagy can promote crizotinib resistance in lung cancer; thus, targeting autophagy may be a novel strategy to increase drug sensitivity [16].
In the present study, we explored whether curcumin treatment could regulate miR-142-5p expression in NSCLC cells. Moreover, we investigated the role of miR-142-5p in the regulation of crizotinib sensitivity, and its association with autophagy in lung cancer.
Materials and methods
Tissue sample collection
Twenty pairs of human NSCLC tumor tissue and adjacent non-tumor tissue samples were harvested from patients who were subjected to surgical resection at the Second Hospital of Hebei Medical University. The tissue samples were immediately frozen in liquid nitrogen and stored at
Cell culture and drugs
The human NSCLC (A549, H460, H1299, and H1066) and normal lung fibroblast (HLF) cell lines were cultured in DMEM containing 10% fetal bovine serum (FBS) (Gibco, Grand Island, NY, USA), 100 mg/ml streptomycin, and 100 U/ml penicillin. Cells were maintained at 37
Transfection
Transfection of plasmid DNA or miRNAs was performed using Lipofectamine 2000 (ThermoFisher Scientific, Inc., Waltham, MA, USA) according to the manufacturer’s instructions. Cells were transfected with miR-142-5p mimic, miR-142-5p inhibitor, or negative control (NC) (GenePharma, Shanghai, China), and Ulk1 siRNAs or NC siRNA (GenePharma, Shanghai, China).
Quantitative real-time PCR
Total RNA was extracted from human tissue samples or cell lines using TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s instructions. For miRNA detection, RNAs were reversely transcribed to cDNA using a miRNA First-Strand cDNA Synthesis kit (Invitrogen Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s protocol. Quantitative real-time PCR (qPCR) was conducted using the SYBR Green PCR Master Mix (Takara, Japan) with U6 as the internal control. For target gene detection, total RNA was reversely transcribed to cDNA using a First-Strand Synthesis Kit (Invitrogen). qPCR analysis was conducted on an ABI 7700 system with
Cell counting kit-8 (CCK-8) assay
Cell proliferation was examined using the CCK-8 assay (Beyotime, Shanghai, China). Cells from each group were seeded into 96-well plates at a density of 2
Apoptosis assay
The apoptosis of differentially treated A549 and H460 cells was determined using the fluorescein isothiocyanate (FITC)-annexin V/propidium iodide (PI) apoptosis detection kit (KeyGen Biotech. Co. Ltd., Nanjing, China) according to the manufacturer’s instructions. Briefly, cells at a density of 2
Determination of methylation status
The methylation status of the cytosine residues in 5’-cytosine–guanosine (CpG) dinucleotides was detected using bisulfite sequencing. Bisulfite conversion of DNA from A549 and H460 cells was performed using an EpiTect Bisulfite Kit (Qiagen, Hilden, Germany) according to the manufacturer’s protocol. Genomic DNA was isolated and amplified using PCR. The purified products were ligated into the pGEM-T vector (Promega, USA) for sequencing on an ABI DNA sequencer (Applied Biosystems, USA).
miRNA target prediction
TargetScan (
Fluorescence microscopy
Differentially treated A549 and H460 cells were seeded onto six-well plates, transfected with the GFP-LC3 plasmid, and cultured for 24 h. The accumulation of GFP-LC3 puncta was observed under fluorescence microscopy. The numbers of GFP-LC3 puncta were counted using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Western blot
Cells were washed with cold PBS and lysed in lysis buffer (Beyotime, Shanghai, China). Protein samples (20
Statistical analysis
Data are presented as mean
Results
Downregulation of miR-142-5p in NSCLC tissues and cell lines
To explore the clinical relevance of miR-142-5p in NSCLC, we examined its expression pattern in twenty pairs of tumors versus adjacent non-tumor tissues. qPCR data revealed that the expression of miR-142-5p in tumor tissues was significantly lower than that in non-tumor tissues (Fig. 1A). Consistently, the expression of miR-142-5p was much lower in human NSCLC cell lines (A549, H460, H1299, and H1066) than in normal lung fibroblast HLF cells (Fig. 1B). A549 and H460 cells were chosen for subsequent experiments because they showed the lowest miR-142-5p expression.
Downregulation of miR-142-5p expression in NSCLC tissues and cells. qPCR results showing miR-142-5p expression in tumor and non-tumor tissues (A), and in human NSCLC cell lines, including A549, H460, H1299, and H1066, and the normal lung fibroblast cell line HLF (B). 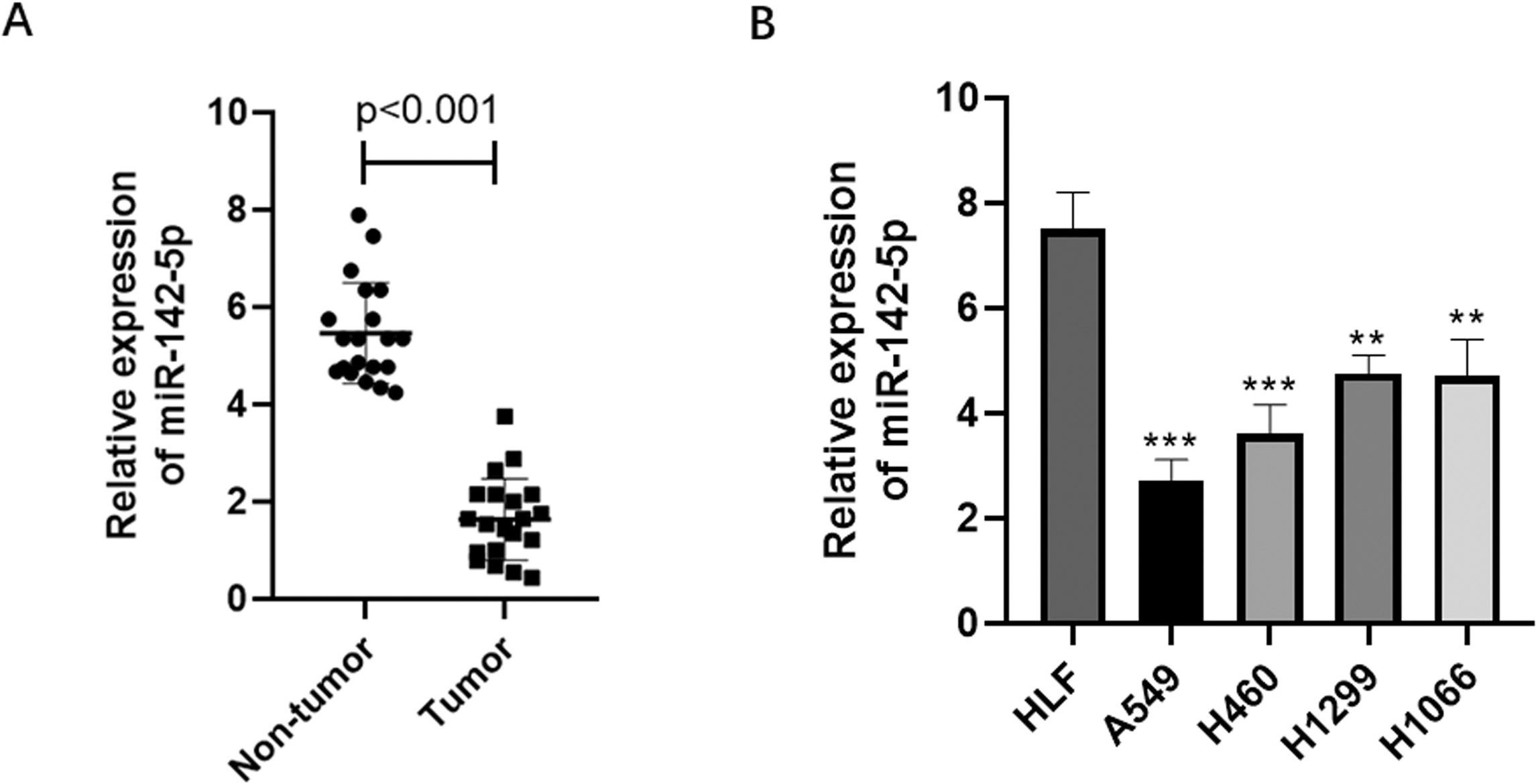
Curcumin restores miR-142-5p expression in NSCLC cells. A549 and H460 cells exposed to crizotinib (A) or curcumin (B) alone or in combination (C); tumor cell viability in each group was evaluated using the CCK-8 assay. (D) qPCR analysis of miR-142-5p expression in A549 and H460 cells exposed to curcumin. 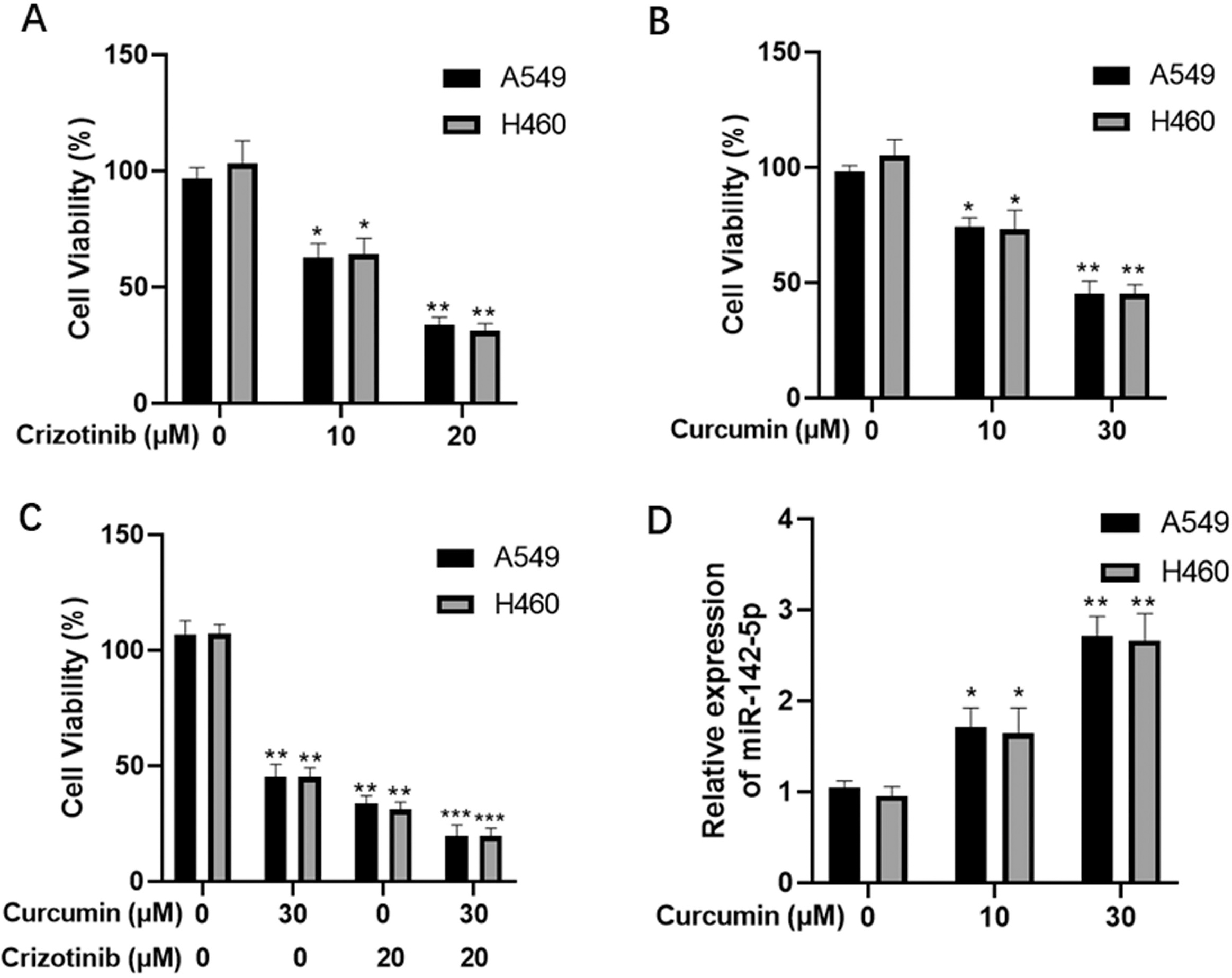
Epigenetic regulation of miR-142-5p expression in NSCLC cells. qPCR analysis of DNMT1 (A), DNMT3A (B), and DNMT3B (C) mRNA levels in A549, H460, and HLF cells. (D) Western blot analysis of DNMT1, DNMT3A, and DNMT3B protein levels in A549, H460, and HLF cells. (E) Methylation levels in the miR-142 promoter region of A549, H460, and HLF cells. (F) qPCR analysis of miR-142-5p expression in 5-AZA-dC-treated A549 and H460 cells. 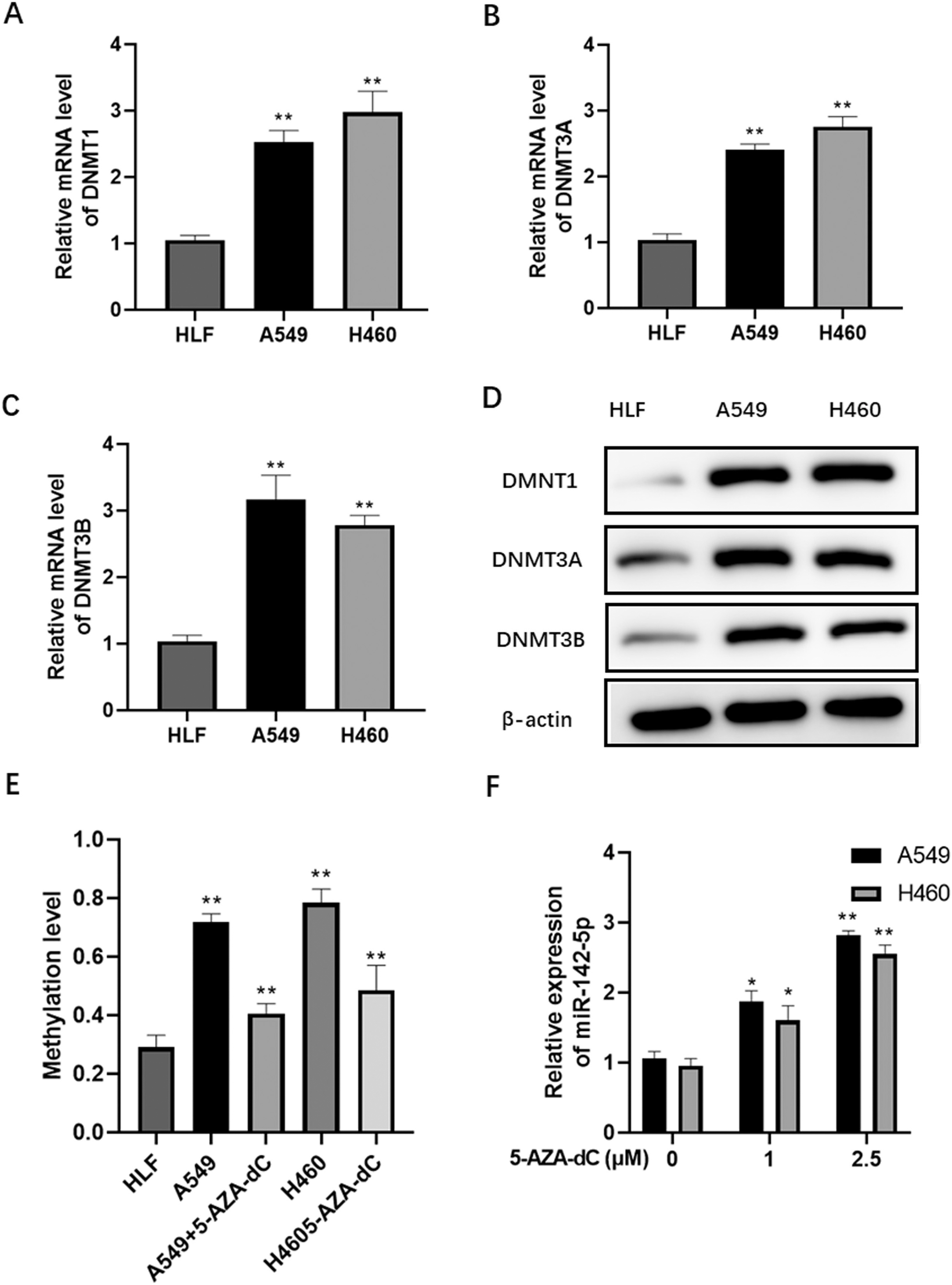
Curcumin modulates DNA methylation in NSCLC cells. qPCR analysis of DNMT1, DNMT3A, and DNMT3B mRNA levels in curcumin-treated A549 (A) and H460 (B) cells. Western blot analysis of DNMT1, DNMT3A, and DNMT3B protein levels in curcumin-treated A549 (C and E) and H460 (D and F) cells. Methylation levels in the miR-142 promoter region of curcumin-treated A549 (G) and H460 (H) cells. 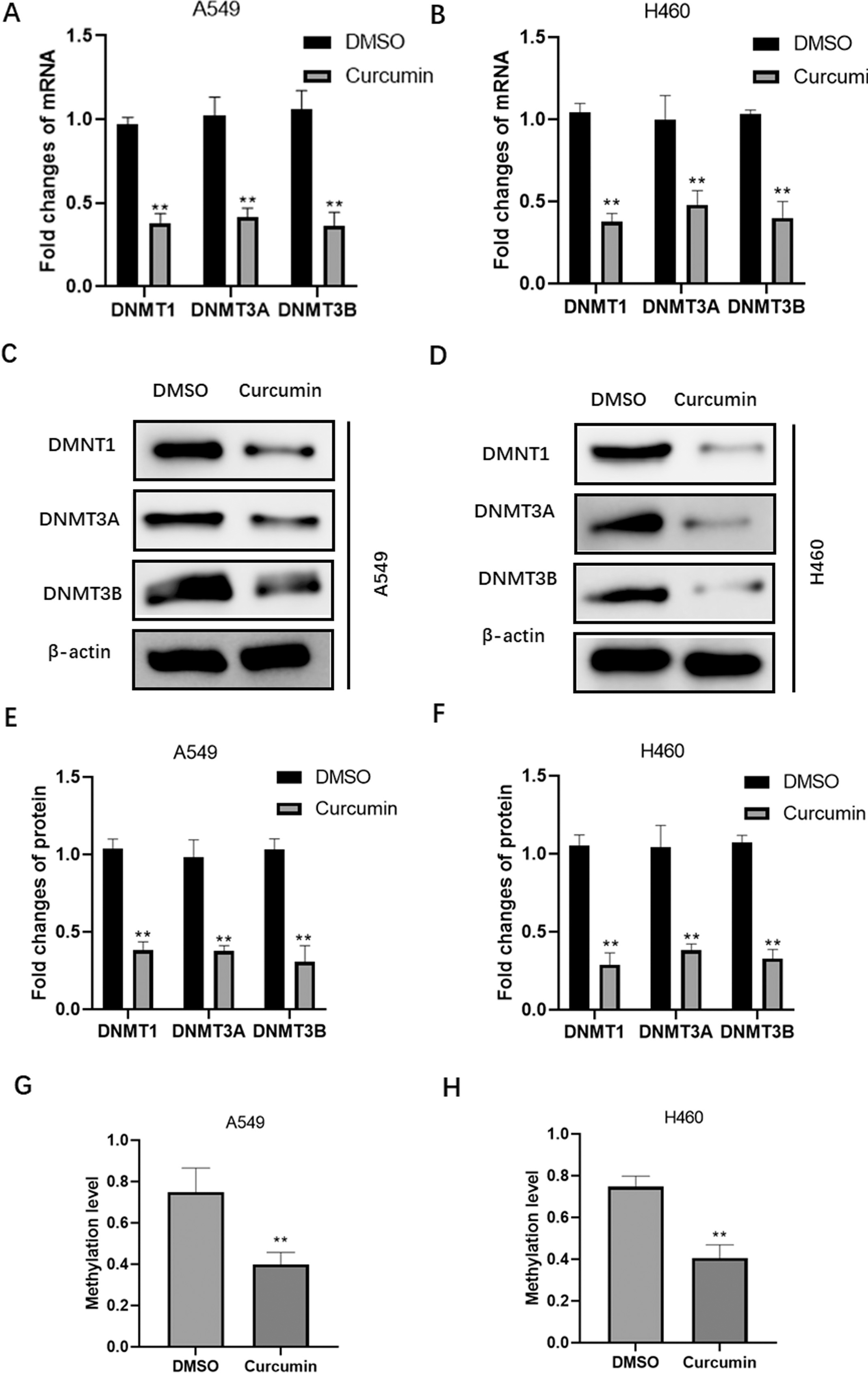
To evaluate the cytotoxic effects of curcumin on human NSCLC, A549 and H460 cells were exposed to crizotinib and curcumin alone or in combination. After 48 h treatment, the CCK-8 assay was performed to evaluate the viability of tumor cells. We found that both crizotinib and curcumin significantly suppressed tumor cell viability in a dose-dependent manner (crizotinib IC50 values: 15.3
miR-142-5p sensitizes NSCLC cells to crizotinib. (A) qPCR analysis of miR-142-5p expression after transfection of miR-142-5p mimic into A549 and H460 cells. Cell survival analysis in A549 (B) and H460 (C) cells treated with crizotinib alone or in combination with curcumin or miR-142-5p. (D and E) Flow cytometric analysis of the apoptosis rate in A549 and H460 cells treated with crizotinib alone or in combination with curcumin or miR-142-5p. 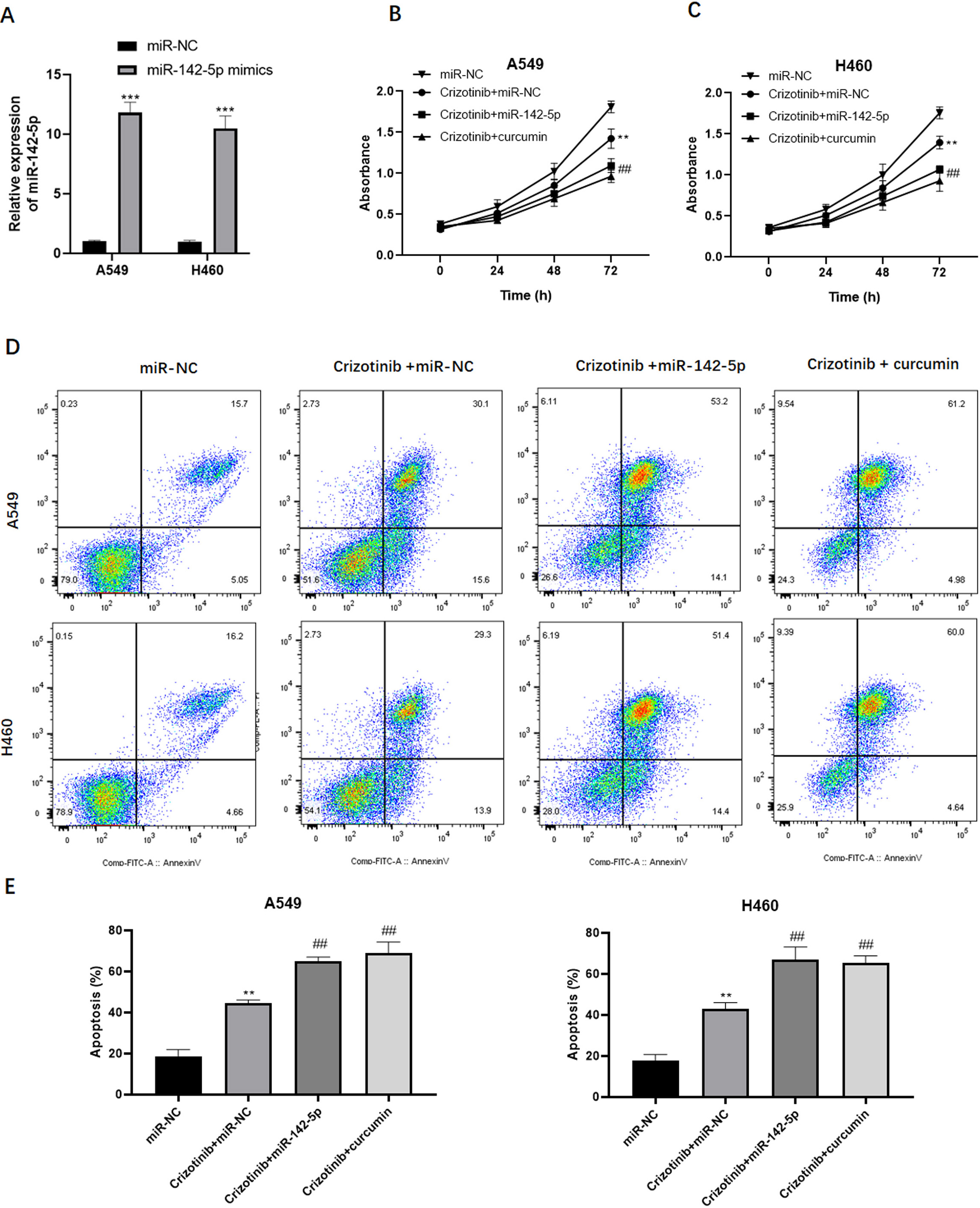
miR-142-5p targets Ulk1 and regulates autophagy in NSCLC cells. (A and B) Representative images and quantitation of GFP-LC3 punctum formation in A549 cells using a fluorescence microscope. (C) Western blot analysis of the expression of proteins associated with autophagy, including LC3-II, beclin-1, and p62, in A549 cells treated with crizotinib alone or in combination with curcumin or miR-142-5p. (D) Bioinformatic analysis of the binding sites between miR-142-5p and Ulk1. (E) Western blot analysis of Ulk expression in A549, H460, and HLF cells. (E) Western blot analysis of Ulk expression in A549 and H460 cells transfected with miR-142-5p mimic. 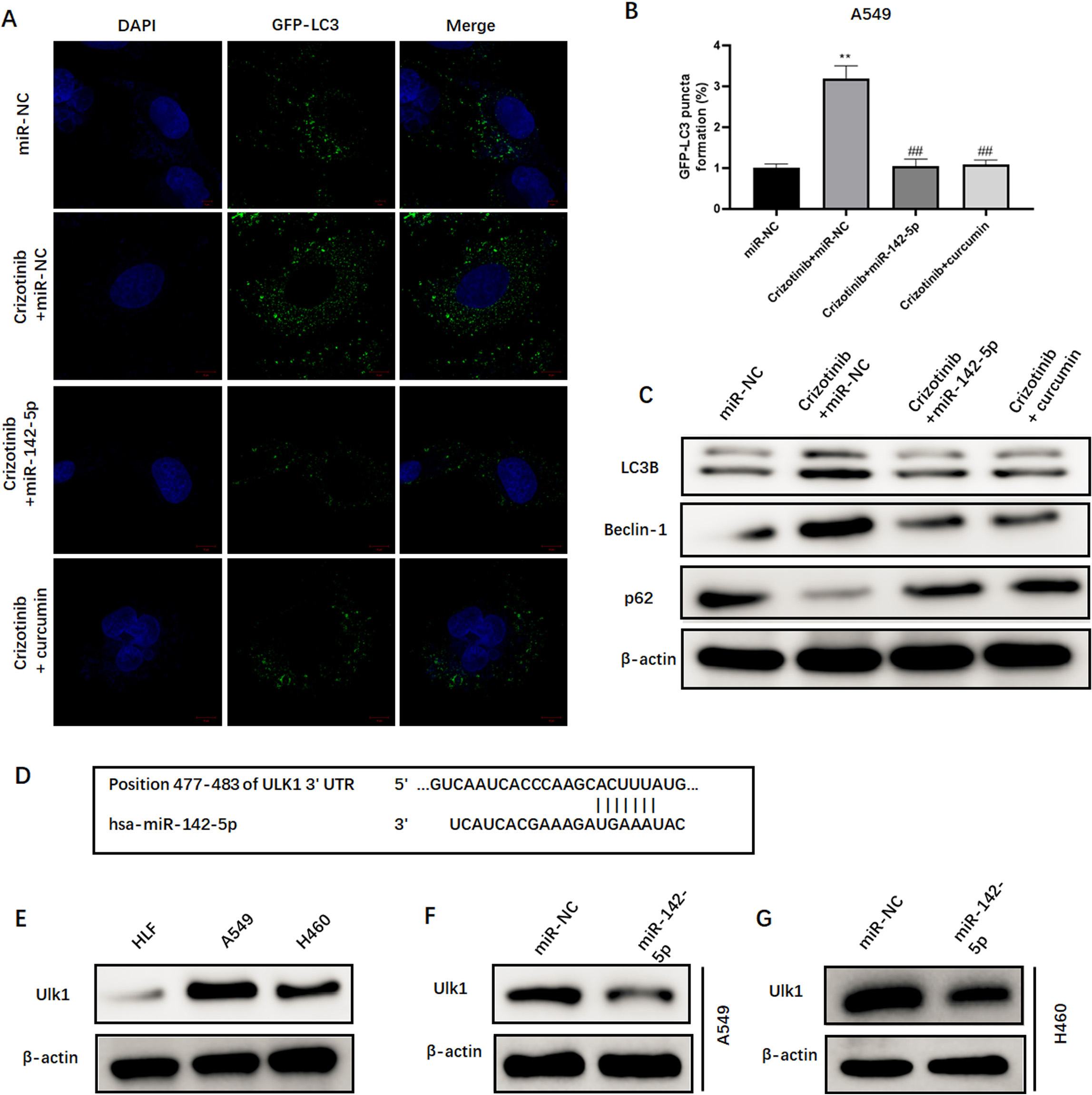
Next, we examined whether curcumin could modulate the methylation status in NSCLC cells. Data from qPCR and western blot assays confirmed that curcumin treatment substantially suppresses both the mRNA (Fig. 4A and B) and protein (Fig. 4C–F) levels of DNA methylation-related enzymes, including DNMT1, DNMT3A, and DNMT3B, in A549 and H460 cells. In addition, curcumin treatment inhibited CpG dinucleotide methylation in the miR-142 promoter region of A549 and H460 cells (Fig. 4G and H), suggesting that curcumin restores miR-142-5p expression by suppressing the methylation levels in tumor cells.
Ulk1 abolishes the sensitizing effect of miR-142-5p on NSCLC cells. Western blot analysis of Ulk1, LC3-II, beclin-1, and p62 expression in A549 (A) and H460 (B) cells after overexpression of Ulk1. Cell survival analysis in A549 (C) and H460 (D) cells treated with miR-142-5p alone or in combination with Ulk1 in the presence of crizotinib. Flow cytometric analysis of the apoptosis rate in A549 (E) and H460 (F) cells treated with miR-142-5p alone or in combination with Ulk1 in the presence of crizotinib. 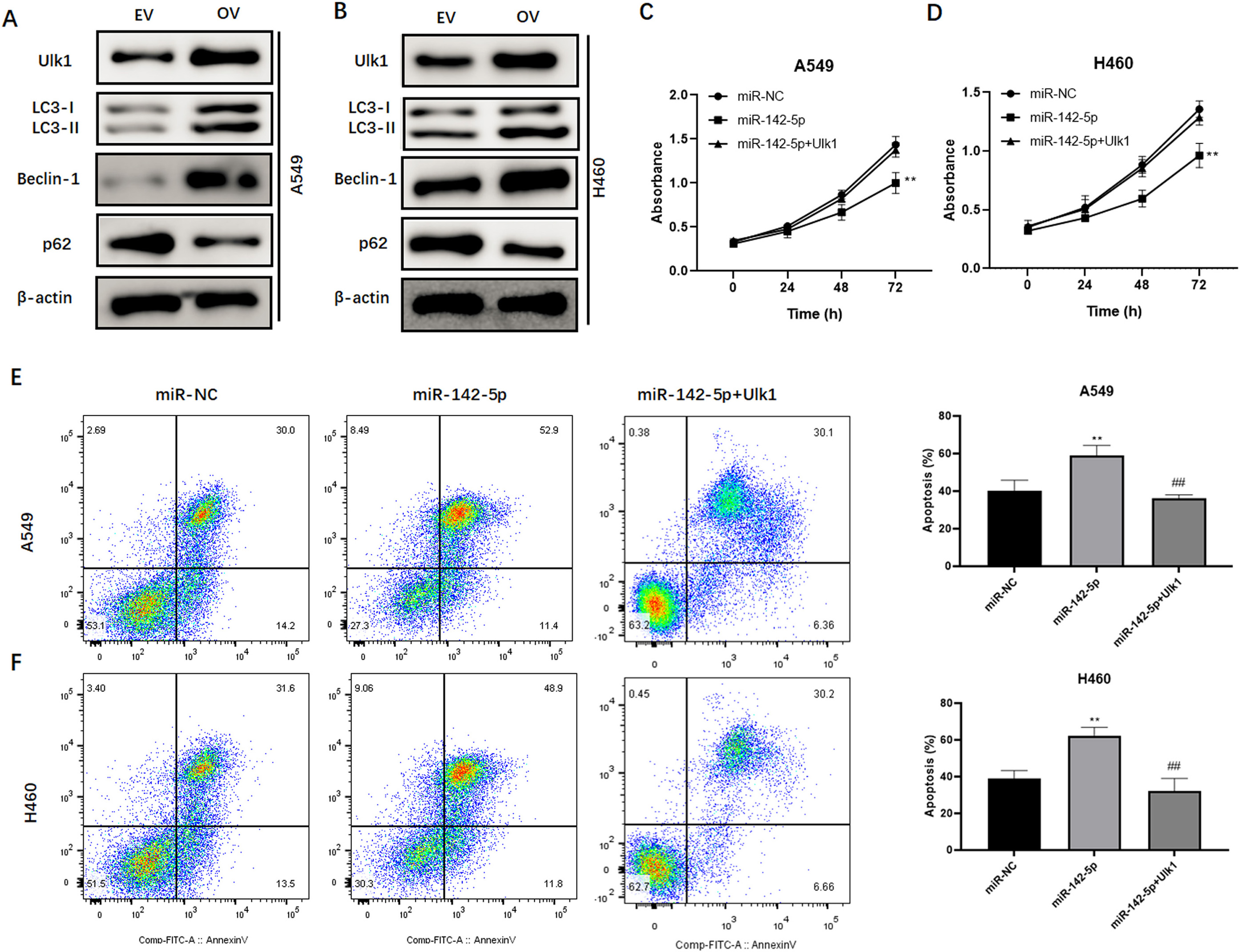
Furthermore, we explored the underlying mechanisms whereby curcumin increases crizotinib cytotoxicity in NSCLC cells. Given that miR-142-5p expression is affected by curcumin treatment, we examined the functional role of miR-142-5p in the sensitivity of tumor cells to crizotinib. Initially, we transfected miR-142-5p mimic into A549 and H460 cells (Fig. 5A). Consequently, treatment with crizotinib exhibited an obvious tumor suppressive effect. Interestingly, the overexpression of miR-142-5p substantially inhibited the growth of A549 and H460 cells exposed to crizotinib (Fig. 5B and C). In addition, flow cytometric analysis revealed that the upregulation of miR-142-5p expression potentiated the pro-apoptotic effect of crizotinib in tumor cells (Fig. 5D and E). Collectively, these findings suggested that miR-142-5p may enhance the response to crizotinib in NSCLC cells.
miR-142-5p decreases autophagy by targeting Ulk1 in NSCLC cells
Next, we explored the underlying mechanism where-by miR-142-5p increases drug sensitivity in NSCLC cells. We found that treatment with crizotinib induces significant GFP-LC3 punctum formation (Fig. 6A and B). However, both the upregulation of miR-142-5p expression and curcumin treatment suppressed crizotinib-induced punctum formation (Fig. 6A and B). Then, proteins associated with autophagy were examined using western blot. Both increased miR-142-5p expression and curcumin treatment significantly suppressed crizotinib-induced upregulation of LC3-II and beclin-1, and promoted crizotinib-induced degradation of p62 in tumor cells (Fig. 6C). Furthermore, bioinformatic analysis predicted that Ulk1, which is an important gene involved in autophagy, may be a potential target of miR-142-5p. Subsequently, we observed that the protein levels of Ulk1 were significantly elevated in A549 and H460 cells (Fig. 6D). However, the overexpression of miR-142-5p remarkably decreased Ulk1 expression (Fig. 6E and F). Collectively, these data suggested that miR-142-5p targets Ulk1 and regulates autophagy in NSCLC cells.
Overexpression of Ulk1 abolishes the effect of miR-142-5p on the sensitivity of NSCLC cells to crizotinib
To verify that miR-142-5p increases crizotinib cytotoxicity by suppressing Ulk1-mediated autophagy, we transfected a plasmid containing recombinant Ulk1 into NSCLC cells, and examined cellular behaviors in response to crizotinib. Western blotting showed that the expression of Ulk1 was efficiently upregulated in A549 and H460 cells (Fig. 7A and B), which was accompanied by elevated levels of autophagy-associated LC3-II and beclin-1, and decreased levels of p62 (Fig. 7A and B). While the overexpression of miR-142-5p increased the cytotoxic effect of crizotinib on tumor cells, this effect was abolished by co-overexpression of Ulk1 (Fig. 7C and D). Consistently, the potentiating effect of miR-142-5p on crizotinib-induced apoptosis was reversed upon Ulk1 upregulation in A549 and H460 cells (Fig. 7E and F). Collectively, the results showed that the upregulation of Ulk1 can abrogate the sensitization of NSCLC cells to crizotinib induced by miR-142-5p.
Discussion
Many studies have demonstrated that curcumin has promising therapeutic values in multiple types of cancer. Additionally, epigenetic modification has been proposed to be the underlying mechanism whereby curcumin exerts its effects on tumor pathogenesis [17]. In the present study, we report for the first time that curcumin increases crizotinib cytotoxicity through the epigenetic regulation of miR-142-5p, providing additional experimental data regarding drug resistance in NSCLC.
miRNAs play critical roles in NSCLC progression by manipulating cell proliferation, apoptosis, and drug resistance [18, 19, 20]. Identifying the miRNAs and associated targets that are important for tumor initiation and progression may provide valuable therapeutic strategies [21]. Various studies have demonstrated that miR-142-5p plays a diverse role in human pathophysiological processes, including immune tolerance, inflammation, and viral infection [22, 23, 24]. Specifically, miR-142-5p was reported to inhibit tumor growth and metastasis by targeting Yin Yang 1 in NSCLC cells [25]. Recently, the overexpression of miR-142-5p was shown to reverse resistance to gefitinib by suppressing HOXD8 in lung cancer [26]. Here, we found that miR-142-5p was downregulated in NSCLC clinical samples and cell lines. In addition, many miRNAs are involved in the antitumor activity of curcumin. In prostate cancer, curcumin increases radiosensitivity by activating miR-143 and miR-143 [27]. In NSCLC, the pharmacological effects of curcumin were shown to be mediated through the regulation of several miRNAs, including miR-21, miR-192-5p, and miR-215 [28]. Here, both curcumin and crizotinib remarkably suppressed cell viability in NSCLC cells. In addition, combined treatment potentiated the cytotoxicity of crizotinib in tumor cells. Strikingly, curcumin substantially restored miR-142-5p expression and suppressed the expression of DNMT1, DNMT3A and DNMT3B, which act as DNA methylation-related enzymes [29]. These findings suggested that curcumin sensitizes tumor cells to crizotinib by epigenetically activating miR-142-5p expression in NSCLC.
Previous studies have suggested that miR-142-5p may function as an oncogenic miRNA or a tumor suppressor depending on the specific type of cancer [11, 12]. In our study, ectopic expression of miR-142-5p could potentiate crizotinib-induced inhibition of cell growth and induction of apoptosis in NSCLC cells, suggesting that miR-142-5p might function as a tumor suppressor and that its downregulation may contribute to the resistance to crizotinib in NSCLC.
Autophagy is a conserved cellular process that degrades intracellular misfolded proteins and damaged organelles, which is dependent on the lysosomal pathway and activated upon external and internal stimuli. The induction of autophagy contributes to drug resistance in NSCLC cells [16, 30]. A recent study found that crizotinib treatment induces higher autophagic response in anaplastic large cell lymphoma; chloroquine-mediated inhibition of autophagy has a tumor suppressive role against the chemoresistance phenotype [31]. Interestingly, we found that both miR-142-5p and curcumin attenuated crizotinib-induced autophagy in NSCLC cells. Moreover, we identified Ulk1 as a target of miR-142-5p, which is a regulator of autophagy [32]. Furthermore, we found that the overexpression of Ulk1 potentiated the autophagic response and abrogated the sensitizing role of miR-142-5p in crizotinib-treated NSCLC cells.
Conclusion
In conclusion, our study suggests that curcumin enhances lung cancer cell sensitivity to crizotinib treatment via the epigenetic activation of miR-142-5p expression. Furthermore, our findings reveal that miR-142-5p increases drug sensitivity by targeting Ulk1 and suppressing autophagy.
Author contributions
Conception: Yu-Zheng He and Shan-Ling Yu.
Interpretation or data analysis: Xiao-Ning Li, Xian-Hua Bai and Hai-Tao Li.
Manuscript preparation: Yan-Chao Liu, Bao-Lei Lv, and Xiu-Min Zhao.
Revision for important intellectual content: Dong Wei and He-Lin Zhang.
Supervision: Fan-Nian Li and Shuai Li.
Disclosure statement
The authors declare that they have no competing financial interests.
Funding
The present study was supported by the Humanities and Social Science Research Project of Hebei Education Department (Grant no. QN2020112) and the Natural Science Foundation of Hebei Province (Grant no. H2021206067).
Footnotes
Acknowledgments
We appreciate the assistance of Large-scale Data Analysis Center of Cancer Precision Medicine-LinkDoc for clinical and pathological data collection.
