Abstract
BACKGROUND:
Allogeneic hematopoietic stem cell transplantation (ASCT) is the preferred treatment option for patients with several hematologic disorders and immunodeficiency syndromes. Graft-versus-host disease (GVHD) is an immune mediated post-transplant complication which has a major impact on long-term transplant outcomes.
OBJECTIVE:
Current efforts are focused on identification of new markers that serve as potential predictors of GVHD and other post-transplant clinical outcomes.
METHODS:
This study includes donor harvests collected from twenty-three allogeneic donors during period 2008–2009 and respective transplant recipients followed for clinical outcomes till March 2019. Percent CD26+ and CD34+ cells in donor harvest were analyzed using flow cytometry. Percent expression and infused dose of CD26+ and CD34+ cells were evaluated for association with various clinical outcomes.
RESULTS:
Total 23 healthy donors with median age of 28 years (13 males), and transplant recipients with median age of 24 years (17 males) formed the study cohort. The diagnosis included malignant (
CONCLUSIONS:
Our findings suggest a role of CD26 expression on human donor harvest as a potential predictor of acute GVHD. This association warrants further exploration.
Keywords
Graphical abstract
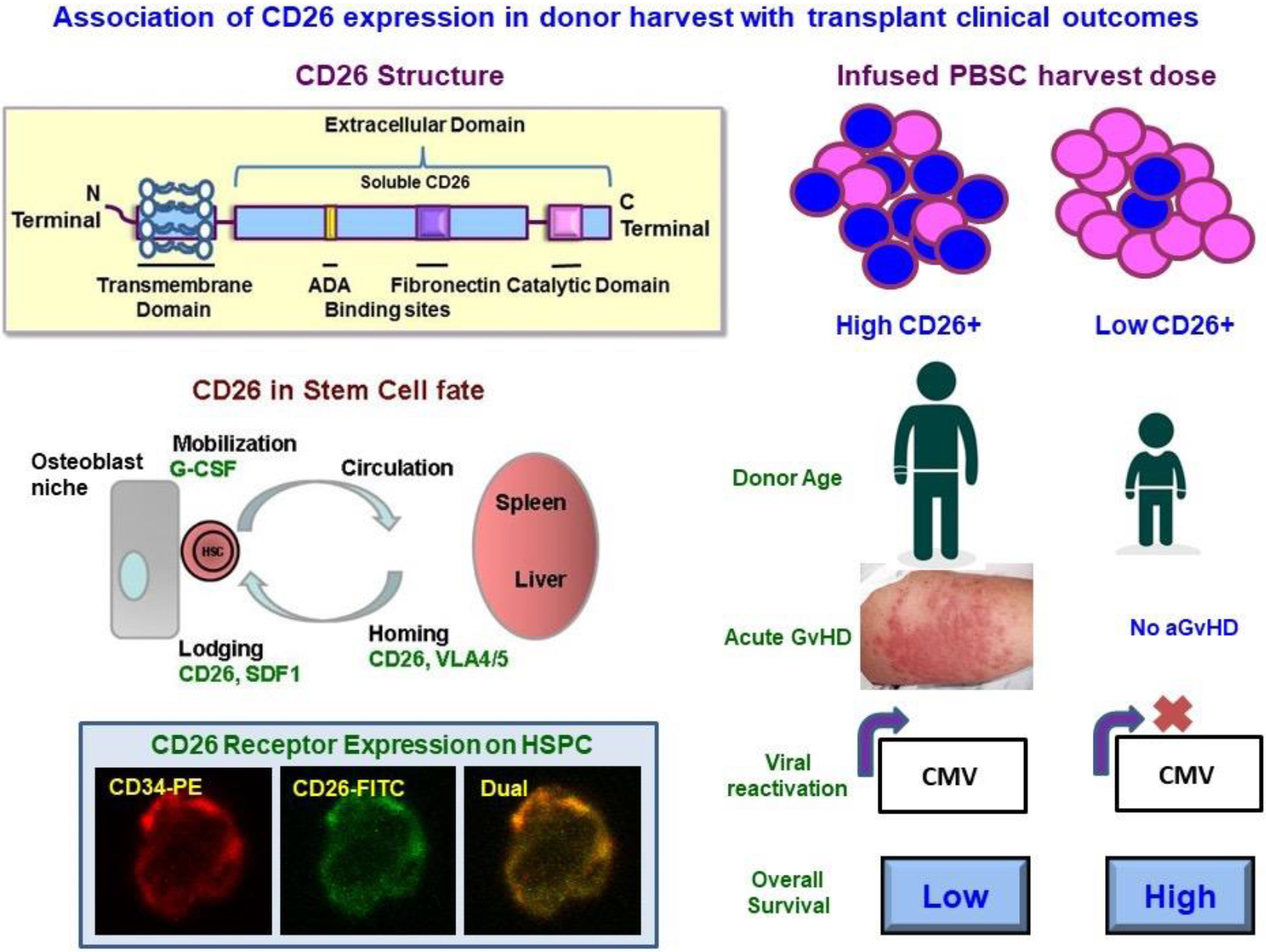
Highlights
Study has identified biomarker immuno-ectoenzyme CD26 for predicting outcome in allogeneic stem cell transplants.
First study correlating CD26 expression on donor stem cell harvest with acute GVHD, and CMV reactivation.
Lower CD26 expression associates with lesser acute GVHD.
These data form the basis of going forward with further studies regarding CD26.
Introduction
Allogeneic stem cell transplantation (ASCT) is the preferred treatment in patients with high-risk hematolymphoid malignancies and few hematological disorders viz. Fanconi’s anemia, aplastic anemia, several hemoglobinopathies, etc. These diseases can be cured by ASCT that brings about donor stem cell-mediated hematopoiesis and immune reconstitution. Among the several factors that affect the long-term success of ASCT (diagnosis, disease status at transplant, age and gender of patient and donor, degree of human leukocyte antigen (HLA) match or mismatch, and many more) are the infused cell doses [1, 10, 24]. Traditionally, CD34 has been considered as the standard marker for stem cells, however, the correlation between CD34 cell dose and several post-transplant outcomes has not been consistent. Data from our analysis has shown no correlation between CD34 cell dose and acute or chronic GVHD (aGVHD or cGVHD) [10, 24]. In view of these, attempts have been made by us and others to identify other potential markers which may have a correlation with transplant outcomes. CD26/(dipeptidylpeptidase-IV/DPPIV) is an immunoregulatory enzyme which is expressed constitutively on many hematopoietic cell populations (including natural killer cells and activated B and T lymphocytes) endothelial cells, fibroblasts, epithelial cells, and mesenchymal stem cells [14, 19]. The secretory form (which is the extracellular domain of CD26) also carries catalytic activity [7, 22]. CD26 is also crucial for mobilization of hematopoietic stem cells (HSCs) from bone marrow niche and homing towards lymphoid organs. CD26/DPPIV exhibits a complex biology encompassing cell-membrane associated activation of intracellular signal transduction pathways, cell-cell interaction and migration [6, 7, 14, 19]. CD26 plays a key role as a T-cell co-stimulatory molecule. Other known functions of CD26 include cleavage of several neurohormones, and as a receptor for adenosine deaminase [14, 19]. Mice models have shown that inhibition of enzymatic activity of CD26 suppress antibody production [16]. These and several other data point to an important role played by CD26 in immune responses. Previously we had reported for the first time that increased CD26 expression on cells of donor harvest from related donors led to early WBC engraftment in cancer patients [23]. In the current paper, we report long-term outcomes (with a ten-year follow-up) of an expanded cohort of our earlier study. To the best of our knowledge, this is first study to analyze correlation between CD26 expression on donor harvests and CD26+ cell dose with long-term transplant outcomes.
Patients and methods
This study was a single-centre prospective study. Consecutive patients undergoing 1
Donor characteristics, patient demographics, transplant details and clinical outcomes
Donor characteristics, patient demographics, transplant details and clinical outcomes
Cell surface expression of CD34 and CD26 antigens was performed as described previously [23]. Red blood cells were separated and nucleated cells from the stem cell harvest were suspended in FACS (Fluorescence activated cell sorting) buffer (Phosphate buffered saline with 1% Fetal Calf Serum and 0.02% sodium azide) and stained with anti-human CD26 conjugated with Fluorescein-isothiocyanate (FITC), anti-human CD3 conjugated with FITC, anti-human CD34-conjugated with Phycoerythrin (PE) and anti-human CD45–FITC (Becton Dickinson, San Jose, CA) antibodies. Stained cells were acquired using Flow Cytometer (FACS Caliber, Becton Dickinson, San Jose, CA), and analysed with CELLQuest Software (Becton Dickinson). CD34+ cells were defined as described earlier in International Society for Hematotherapy and Graft Engineering (ISHAGE) protocol for CD34 analysis [29]. The gating strategy followed for CD34 and CD26 is shown in Supplementary Figure S1. Infused cell dose of CD26+ cells, CD3+ cells and CD34+ cells was calculated in terms of body weight of transplant recipient.
Post-transplant outcomes
WBC and platelet engraftment
WBC engraftment was considered to have occurred on the first of 3 consecutive days in which the total leukocyte count exceeded 1.0
Graft versus host disease
The diagnosis and grading of aGVHD was done clinically according to Gluksberg criteria [14]. Histological confirmation was done only if clinically necessary. Chronic GVHD was diagnosed clinically with biopsy confirmation obtained only when clinically necessary. Chronic GVHD was classified into limited stage and extensive stage according to the standard modified Seattle criteria. For analysis of aGVHD, patients were divided into 2 groups, those with grade II-IV aGVHD (GVHD-Yes) and those with grade I aGVHD or no GVHD (GVHD-No).
Viral reactivation
Cytomegalovirus (CMV) DNA was monitored by polymerase chain reaction in all patients. Monitoring was started from the beginning of conditioning chemotherapy and continued till day +100 post-transplant (or later, if the patient was on immunosuppression for GVHD). Evaluation of other viruses was done only when clinically indicated.
Scatter plots of percent CD34/CD26 expression and WBC/Platelet engraftment day. This figure depicts scatter plots of percent CD34 expression (Panel A), percent CD26 expression (Panel B) on donor harvest cells. The number and (–) on each panel denote median value of the percent CD34/CD26 expression observed in allogeneic donor harvests (
 ) and solid blue (
) and solid blue ( ) in both the panels depict incidence of GVHD and no incidence of GVHD, respectively reported in transplant recipients at 100
) in both the panels depict incidence of GVHD and no incidence of GVHD, respectively reported in transplant recipients at 100
Event Free Survival (EFS) was measured as the time interval between date of transplant and date of relapse or death. Those patients who had not relapsed or died at their last follow-up were censored on the dates of their last follow-up. Overall Survival (OS) was measured as the time interval between date of transplant and date of death by any cause. Again, patients alive at their last follow-up were censored.
Correlation of cell doses and outcomes
The percentage expression of CD26 and CD34, as well as the infused CD26 and CD34 cell doses were analyzed for correlation with all of the above post-transplant outcomes. Additionally, infused total nucleated cell (TNC) and CD3+ cell doses were also correlated with outcomes.
Statistical analysis
Descriptive statistics, including mean
Association of CD26 expression in donor harvest with donor age. Scatter plots show association of percent CD26 expression (Panel A) and infused CD26+ cell dose (Panel B) with donor age in years.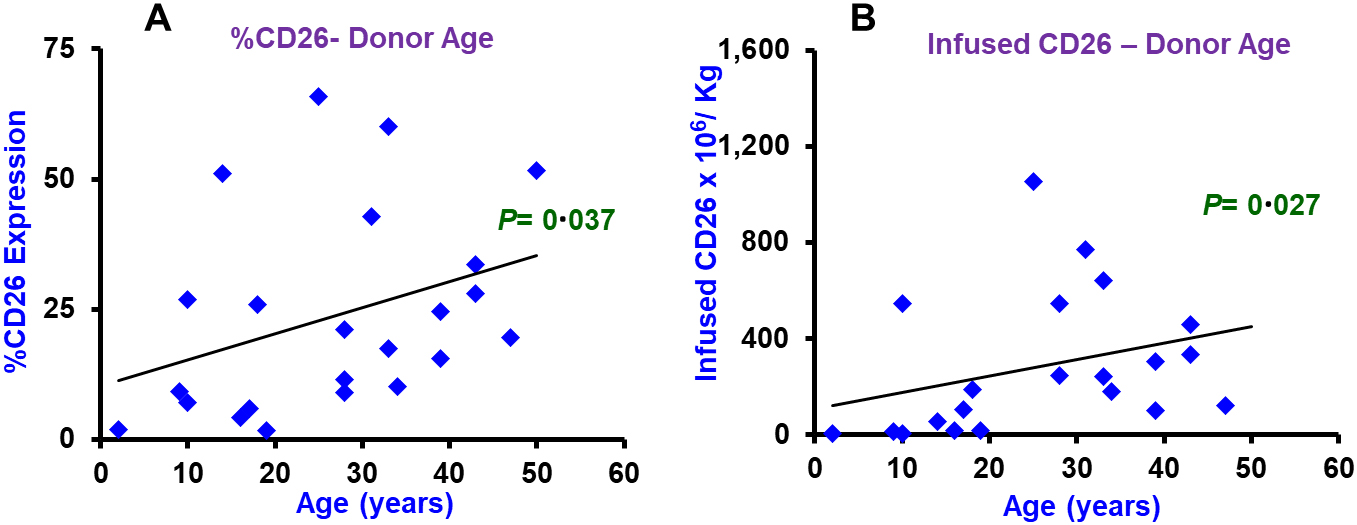
Association of percent CD26 expression and CD34 infused cell dose with post-transplant clinical outcomes. Box-Whisker plots exhibit distribution of percent CD26 expression (Panel A) and infused CD26+ cell dose (Panel B) in patients with (YES, 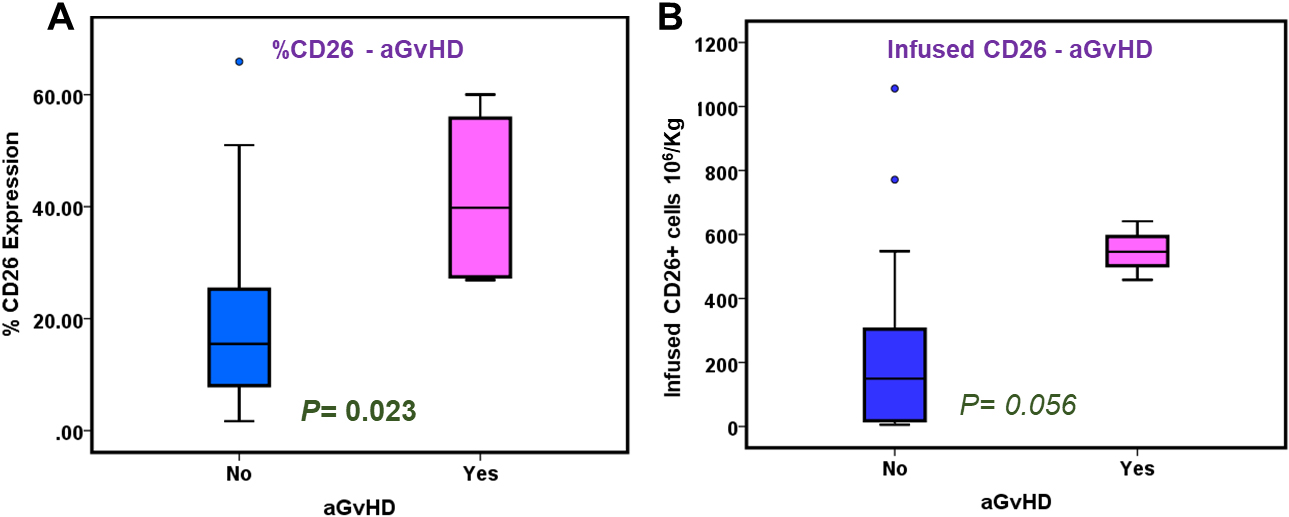
CD34 and CD26 expression on stem cell harvest and infused cell doses
Twenty-three allogeneic donors with median age of 28 (IQR: 17–37) years and 13 (56.52%) males formed the study cohort (Table 1). CD34
Transplant outcomes
WBC engraftment was achieved in all patients at median days of 12 (IQR: 11–13; Fig. 1C). Engraftment of platelets was also achieved with a median of 12 days (IQR: 10–14) (Fig. 1C). Incidence of grade II-IV aGVHD, cGVHD, and CMV reactivation is shown in Table 1. Among 13 patients with malignant disorders, 7 (54%) relapsed. Among the entire cohort of 23 patients, 15 (65%) are long term survivors. Among the 8 patients who died, 7 died of disease, and 1 died due to GVHD.
Correlation of CD26 expression with donor characteristics
Donor age correlated significantly with CD26 percent expression (
Association of CD26 expression on donor harvest and infused cell dose with transplant clinical outcomes
Association of CD26 expression on donor harvest and infused cell dose with transplant clinical outcomes
This table includes CD26 and CD34 percent expression from stem cell harvest of allogeneic donors (PBSC harvest;
Univariate Cox regression analysis
Univariate Cox regression was performed to detect the coefficients which relate to hazard. Hazard ratio (HR) of less than 1 indicate that the predictor is protective (i.e., associated with improved survival). While Hazard ratio of greater than 1 signifies that the predictor is associated with increased risk (or decreased survival).
CD26, CD34 and CD3 expression and infused cell doses were correlated with transplant outcomes viz. WBC/ platelet engraftment day, aGVHD, cGVHD, viral reactivation, EFS and OS.
Engraftment
CD26 percent expression (
Graft-versus-host disease
CD26 percent expression (
Viral reactivation
Infused CD26+ cell dose correlated significantly with viral (CMV) reactivation (
Survival
Median Survival time was 92 months (95% C.I. 87.5, 96.15) months. More than 50% patients were survived till 96 months. Median Event free survival time was 28 (95% C.I. 16, 41) months. 3-year OS and EFS probability was 64.7% and 42.7%, respectively. ROC identified an optimal cut-off of 20.4% for CD26 expression and 217
Association of percent CD26 expression and CD26 infused cell dose with post-transplant overall and event-free survival. ROC curves demonstrated area under curve for % CD26 expression (Panel A) and infused CD26 cell dose (Panel D) in donor harvest. Overall survival Kaplan Meier curves of donor harvest % CD26 expression (Panel B) and infused CD26 cell dose (Panel E) and Event free survival Kaplan Meier curves of donor harvest % CD26 expression (Panel C) and infused CD26 cell dose (Panel F). Values mentioned at right bottom of each panel indicate statistical significance by Log Rank test. 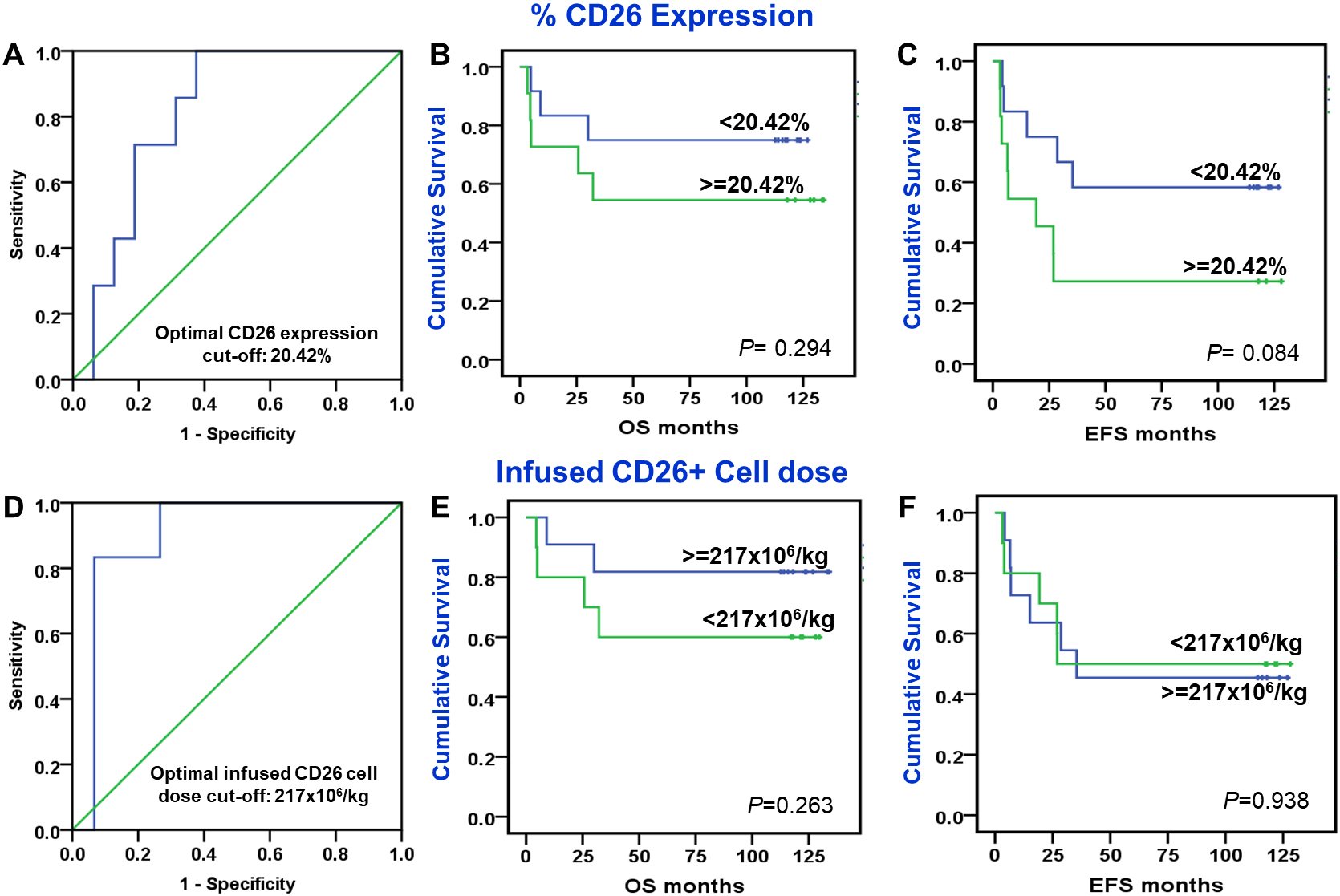
Association of CD26 expression was also evaluated with that of CD34, CD3 expression and TNC. Spearman’s correlation applied to evaluate the association of CD26 percent expression (
Association of percent CD26 expression and CD26 infused cell dose with CD34 infused cell dose. The figure exhibits association between percent CD26 expression (left panel) and infused CD26 cell dose (right panel) with a known marker of post-transplantation recovery – infused CD34 cell dose in nucleated cells of stem cell harvest (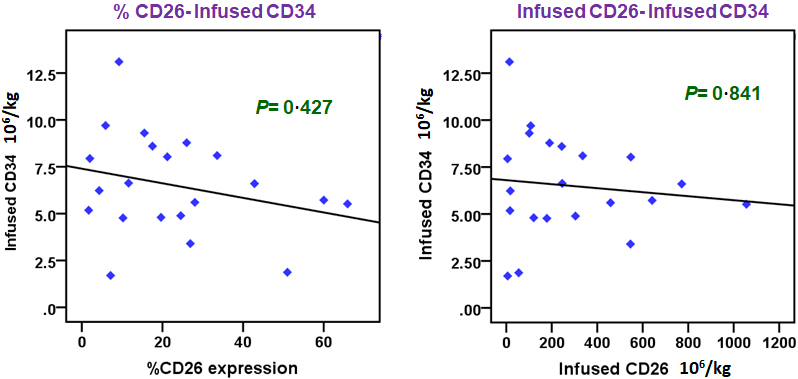
Variable reports on correlation of CD34 expression and/or infused CD34+ cell dose and transplant outcome have led to search of novel markers for predicting post-transplant outcomes [1, 2, 3, 13, 14, 15, 16, 17]. CD26 is expressed on several hematopoietic cells (and also other cells) as well as exist in soluble form in the blood. All functions of CD26 are not known. However, some of its important functions include – acting as a co-stimulatory molecule for T-cell activation, cleaving several cytokines and neuropeptides, and as receptor for adenosine deaminase [14, 19]. Thus, it has many significant functions in the immune system. Simeoni et al. [28] demonstrated that CD26-knock-out and -deficient animal models, transgenic mice with human CD26 displayed impairment in thymocyte proliferation to mitogens and CD4
In the field of allogeneic stem cell transplant, there is sparse literature regarding the role of CD26. The fact that CD26/DPP-IV regulates CXCL12 (stromal cell derived factor 1 or SDF1) led to the hypothesis that CD26 inhibition may have an impact on engraftment. However, studies have yielded conflicting results [4, 26]. Subsequently, following the identification of role of CD26+T cells in several auto-immune disorders, their role in pathology and prevention of GVHD has been studied. It has been shown in human-derived xenograft models of GVHD that there is an infiltration of CD26+ T cells in target organs affected by acute GVHD [12]. The same study also reports a significant improvement of GVHD with anti CD26 monoclonal antibody [12]. However, there is no correlation in literature about the CD26+ cells in donor stem cell harvest and transplant outcomes.
Previously, we had reported the correlation between CD26 expression on donor harvest and engraftment [23]. However, it was also important to address the correlation between CD26 expression and other ASCT outcomes. With a long-term follow-up, we found a strong association between CD26 percent expression on donor harvest and infused CD26 cell dose with acute GVHD. However, we did not find an association between chronic GVHD, and event-free and overall survival. Our finding of correlation between CD26 expression and donor age could provide a biological basis for clinical observation of increased risk of GVHD seen with older donors [25, 32].
Given the potential role of CD26+ T cells in GVHD, several groups have studied inhibition of CD26 for prevention and treatment of GVHD. Bacigalupo et al. [2] studied the role of anti CD26 monoclonal antibody begelomab for the treatment of steroid refractory GVHD and found an impressive response rate of about 60%. More recently, sitagliptin, a DPP-IV inhibitor has been shown in a clinical trial to very effectively prevent acute GVHD [8]. Importantly, experimental [12] and human studies [8] targeting CD26 for prevention of GVHD suggest that the graft-versus-leukemia effect remains preserved making this an even more attractive option for prevention and treatment of acute GVHD. The predictive and prognostic values of CD26 and their comparisons with some other known predictive and prognostic biomarkers (like ST2 and Reg3a) [11] remains to be studied.
Infused CD34
Since circulatory CD26 (sCD26) carries enzymatic activity [23], it would also be important to correlate circulating levels of CD26 with the risk of developing GVHD. We could not evaluate sCD26 in the current long-term follow-up study, however, in our new ongoing prospective study we have been finding leads that sCD26 correlates negatively with the risk of GVHD occurrence (data not shown).
CD26 (DPP4) has been shown to be a therapeutic target in autoimmune diabetes (Type 1 Diabetes Mellitus, T1DM) patients [27]. Inhibitors of CD26 have demonstrated an increase in T-regulatory (Treg) cells and a decrease in Th1 cell response [20]. Preclinical trials with CD26 inhibitor therapy have led to reversal or delay in the onset of diabetes in streptozotocin-induced NOD-SCID mice model of autoimmune T1DM [13, 29]. In mouse models of aGVHD, down-regulation of CD26 prevented GVHD but retained graft-versus-tumor effect. Further a two-stage, phase 2, a non-randomized clinical trial conducted to test the therapeutic effect of CD26 inhibitor – sitagliptin along with immunosuppressors tacrolimus and sirolimus resulted in considerable reduction in the incidence of grade II to IV acute GVHD by day 100 post-stem cell transplantation with filgrastim-mobilized blood cells from HLA-matched related or unrelated donors [8]. These studies have focused on exploring CD26 as a therapeutic target to improve stem cell transplantation clinical outcome. As against, our study is the first study which shows CD26 surface receptor on stem cell harvest as a predictive biomarker for risk of developing aGVHD post-transplantation.
Our study has limitations of small sample size, heterogenous diagnosis, and different conditioning regimens. Additionally, the stem cell source included marrow as well as peripheral blood stem cells. Also, we could not perform a multivariate analysis because of the small sample size. Nonetheless, our study does provide an important insight into the potential role of an easily measurable surface protein CD26 as a predictive biomarker of acute GVHD.
Summarizing, our study forms the first study demonstrating the association of CD26 expression and/or infused CD26+ cell dose with engraftment and acute GVHD. It adds to the existing body of literature in suggesting that CD26 may be a potential target for prophylaxis and treatment of GVHD. Further studies are warranted to explore the association of CD26 with GVHD. Additionally, studies are also warranted to address whether CD26 expression could potentially be used to individualize GVHD prophylaxis.
Funding
This work was supported by a grant from the Indian Co-operative Oncology Network-Academic Research Organization, Mumbai, India. (Project # 266, grant to KP).
Author information
Sachin Punatar and Shruti Kandekar contributed equally to this work.
Author contributions
Conception: Jyoti Kode, Sachin Punatar, Kumar Prabhash and Navin Khattry.
Interpretation or analysis of data: Jyoti Kode, Navin Khattry, Sachin Punatar, Anant Gokarn, Kumar Prabhsh, Ashish Bakshi, Pallavi Rane, Libin Mathew, Shubhada Chiplunkar.
Preparation of the manuscript: Jyoti Kode, Sachin Punatar, Shruti Kandekar and Navin Khattry.
Revision for important intellectual content: Kumar Prabhash, Shubhada Chiplunkar.
Supervision: Jyoti Kode, Sachin Punatar, Navin Khattry.
Final approval of the manuscript: Jyoti Kode, Sachin Punatar and all coauthors.
Research involving human participants
Informed consent for participation in this study was obtained in accordance with guidelines by Institutional Ethics Committee (IEC-I, Tata Memorial Centre, Mumbai), with approval from the IRB (Study # 266; IEC approval dated 16/June/2006. The experiments described here were performed in ACTREC, Tata Memorial Centre, Navi Mumbai, in accordance with IEC regulations.
Consent to participate
Written informed consent was obtained from the donors and patients selected for stem cell transplant procedure.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210137.
sj-docx-1-cbm-10.3233_CBM-210137.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-210137.docx
Footnotes
Acknowledgments
Study was funded by Indian Co-operative Oncology Network-Academic Research Organization, Mumbai, India. The authors thank all the patients, their families, healthy donors and the support groups who cared for participants for their contributions to this study. Authors wish to acknowledge the support provided by Transfusion department and flow cytometry facility of the institute.
Conflict of interest
The authors declare that they have no conflict of interest.
