Abstract
BACKGROUND:
Distinguishing between benign and malignant bile duct strictures has long been a diagnostic challenge in clinical practice.
OBJECTIVE:
This study aimed to discover novel biomarkers in bile to improve the diagnostic accuracy of malignant biliary strictures.
METHODS:
Bile samples were collected from 6 patients with malignant or benign biliary stricture, respectively. Protein profiles of the bile were analyzed with a semi-quantitative human antibody array of 440 proteins. Then the differential expressed proteins were screened by Venn diagram analysis. Following this, the accuracy of these potential biomarkers for discriminating between malignant and non-malignant biliary strictures was validated in a larger (
Results:
Twenty proteins were found differentially expressed in malignant versus benign biliary strictures, 6 of which were identified by Venn diagram analysis to be up-regulated regardless of the location of biliary strictures. Among the 6 biomarkers, bile lipocalin-2, P-cadherin, and adipsin showed better diagnostic utility than that of bile CA19-9. Lasso analysis identified that lipocalin-2, P-cadherin and CA19-9 as a group of makers best distinguished malignant from benign strictures.
CONCLUSIONS:
Lipocalin-2 and P-cadherin measurements in bile could be clinically useful for the detection of malignant biliary strictures.
Introduction
Biliary stricture is an abnormal narrowing of the bile duct caused by benign or malignant conditions that are common in clinical practice. About 30% of bile duct strictures are benign, etiology of which includes iatrogenic injury, primary sclerosing cholangitis (PSC), cholelithiasis, and other less common etiologies. The vast majority of strictures are malignant and usually caused by pancreatobiliary malignant tumors, liver cancer, and in some cases metastatic tumors [1]. Pancreatic cancer is the most common etiology in distal common biliary strictures, and cholangiocarcinoma (CCA) is the major cause for stricture of the mid- and proximal extra-hepatic part of the bile duct [2].
Preoperative diagnosis of biliary strictures is still a major clinical problem that often requires a multi-disciplinary approach, including blood biochemical tests, radiographic imaging endoscopic procedures, and pathological evaluation with ancillary studies [2, 3]. However, these diagnostic methods are not sensitive enough to detect malignant lesions when used alone or in combination. CA19-9 is the most commonly used serum marker and has been reported to be increased in patients with various benign cholangiopathy, such as cholangitis, cholelithiasis and non-neoplastic jaundice [4, 5]. This has led to a low sensitivity, roughly 80%, in the diagnosis of malignant biliary strictures [6]. Furthermore, endoscopic bile duct brush and clamp biopsies as well as related auxiliary techniques, such as fluorescence in situ hybridization (FISH) and Kras mutation detection, have been reported with sensitivities ranging from 10% to 70% [7, 8, 9, 10]. As a result, many patients need to undergo multiple endoscopic examinations to obtain the final diagnosis, which is not only costly but also delays treatment. Therefore, there is an urgent need to develop new biomarkers with higher sensitivity and specificity in order to improve the diagnostic accuracy of malignant biliary strictures.
In this study, we aimed to screen for new biomarkers of malignant biliary strictures using antibody chip technology. Since malignant neoplasm occurs in the bile duct epithelium, we speculated that the bile protein pool more accurately reflects the disease than the blood serum. For this reason, we screened for the differentially expressed proteins in the bile of patients with malignant and benign biliary strictures, and verified the screened proteins in another independent panel of bile and serum samples by ELISA. The specificity and sensitivity of the proteins for diagnosis were evaluated, and lasso analysis was used to select the group of markers with the best diagnostic utility to distinguish malignant from benign biliary strictures.
Materials and methods
Patient population
Patients with a bile duct stricture from Eastern Hepatobiliary Surgery Hospital between March 2015 and October 2018 were enrolled. All the patients had elevated bilirubin levels due to biliary stricture and met the clinical indications for endoscopic retrograde cholangiopancreatography (ERCP). Protocols for the clinical research were approved by the ethical committees of supporting institutions (Approval number EHBHKY2015-001-18) and conforms with The Code of Ethics of the World Medical Association (Declaration of Helsinki), printed in the British Medical Journal (18 July 1964). Written consent for the use of the samples in biomedical research was signed by all included patients. The final diagnosis of stricture etiology was based on pathology (
Baseline characteristics of included biliary stricture patients at the time of bile extraction
Baseline characteristics of included biliary stricture patients at the time of bile extraction
Patients with malignant biliary stricture and bile extracted at ERCP available were selected retrospectively in two independent cohorts (Derivation panel and Validation panel) and compared to patients with benign cholangiopathy. Data were tested for normal distribution and the Student’s
Baseline characteristics of malignant biliary stricture and benign biliary stricture patients grouped according to the location of stricture in discovery panel
CCA, cholangiocarcinoma; PAAD, pancreatic adenocarcinoma; PSC, primary sclerosing cholangitis; Dbil, direct bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; GGT, gamma-glutamyltransferase; AFP, alpha fetal protein; CA19-9, Carbohydrate antigen 199; CEA, carcino-embryonic antigen; CRP, C-reactive protein; WBC, White blood cells.
The data from the standard biochemical tests were obtained from clinical routine laboratory databases, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyltransferase (GGT), alpha fetal protein (AFP); Carbohydrate antigen 199 (CA19-9); carcino-embryonic antigen (CEA), C-reactive protein (CRP) and white blood count (WBC).
A total of 52 bile samples were extracted during ERCP. Before contrast medium injections, bile was collected upstream from the biliary stricture, then mixed with PBS containing 0.1% Tween 20 and a protease inhibitor cocktail (MedChemExpress) at a ratio of 1:1.5. After centrifuged at 4
Protein antibody assay
Levels of soluble proteins in the bile and serum were measured using a semi-quantitative human cytokine antibody array (G-Series Human Cytokine Antibody Array 440; RayBiotech Inc., Norcross, GA, USA) and preformed according to manufacturers’ instructions. In brief, after blocking, the membranes were incubated with 10-
ELISA
Commercial kits purchased from RayBiotech (Norcross, GA, USA) were used to measure the levels of adipsin, cystatin C, MIF, P-cadherin, lipocalin-2 and MMP10 in the bile and serum samples from 40 malignant or benign biliary stricture patients in accordance with the manufacturer’s instructions. Briefly, standards or samples were added into a 100-well microplate and incubated for 1 hour. Followed by wash buffer, conjugate, substrate solution and stop solution being added according to the manufacturer’s instructions. Finally, microplate reader was used to determine the optical density.
Statistical analysis
Statistical analyses were accomplished using SPSS (version 20, SPSS Inc., Chicago, IL, USA) and R (RStudio, Northern Ave, Boston). Continuous variables were tested for normal distribution using the Kolmogorov-Smirnov test, and analyzed using the Student’s
Diagnostic utility of the putative biomarkers was evaluated using the receiver operator characteristic (ROC) curve, results are shown as area under curve (AUC) with 95% confidence interval (CI). Optimal cut-off values were chosen by ROC analyses according to the Youden’s index, and the sensitivities and specificities of each protein to distinguish malignant from benign bile duct stricture were calculated. Using the R penalized package, lasso analysis was performed to find group of biomarkers with the best diagnostic accuracy and the coefficient estimates of trained models were used to evaluate model performance (AUC) using the bile validation panel.
Results
Discovery putative biomarkers by protein array analysis
Twelve bile samples from the patients with malignant or benign bile duct strictures were collected, and the expression levels of 440 biomarkers were assessed to determine the differential proteome content. A total of 20 proteins (16 up-regulated and 4 down-regulated) were found to be differentially expressed in malignant versus benign (Fig. 1A). Furthermore, subgroups were set up according to the proximal or distal location of the bile duct stricture. Comparison analysis revealed that 21 proteins (15 up-regulated and 5 down-regulated) showed a significant difference between malignant and benign in the distal stricture group, and 19 proteins (14 up-regulated and 5 down-regulated) differed between malignant and benign in the proximal stricture group. A Venn diagram was used to classify the 60 proteins whose altered expression was specific to or common in the comparisons (Fig. 1B). The complete data including Venn diagram sections and protein names are shown in Table 3. From the intersection, 9 proteins showed consistent and significant results in the three comparisons, including CEACAM-1, cystatin C, adipsin, neprilysin, MIF, IL-20, MMP-10, P-cadherin and lipocalin-2 (Fig. 1C), 6 of which were up-regulated. These considered good candidates for further investigation.
Differentially expressed bile proteins in malignant group against benign group showed in Venn diagram
Differentially expressed bile proteins in malignant group against benign group showed in Venn diagram
A. Heatmap of the 20 differentially expressed proteins between malignant versus benign. B. Venn diagram of the differentially expressed proteins in three comparisons between malignant stricture versus benign stricture, distal malignant stricture versus distal benign stricture, and perihilar malignant stricture versus perihilar benign stricture. The protein details are provided in Supplementary Table 2. C. Relative expression of indicated proteins in bile samples of derivation panel (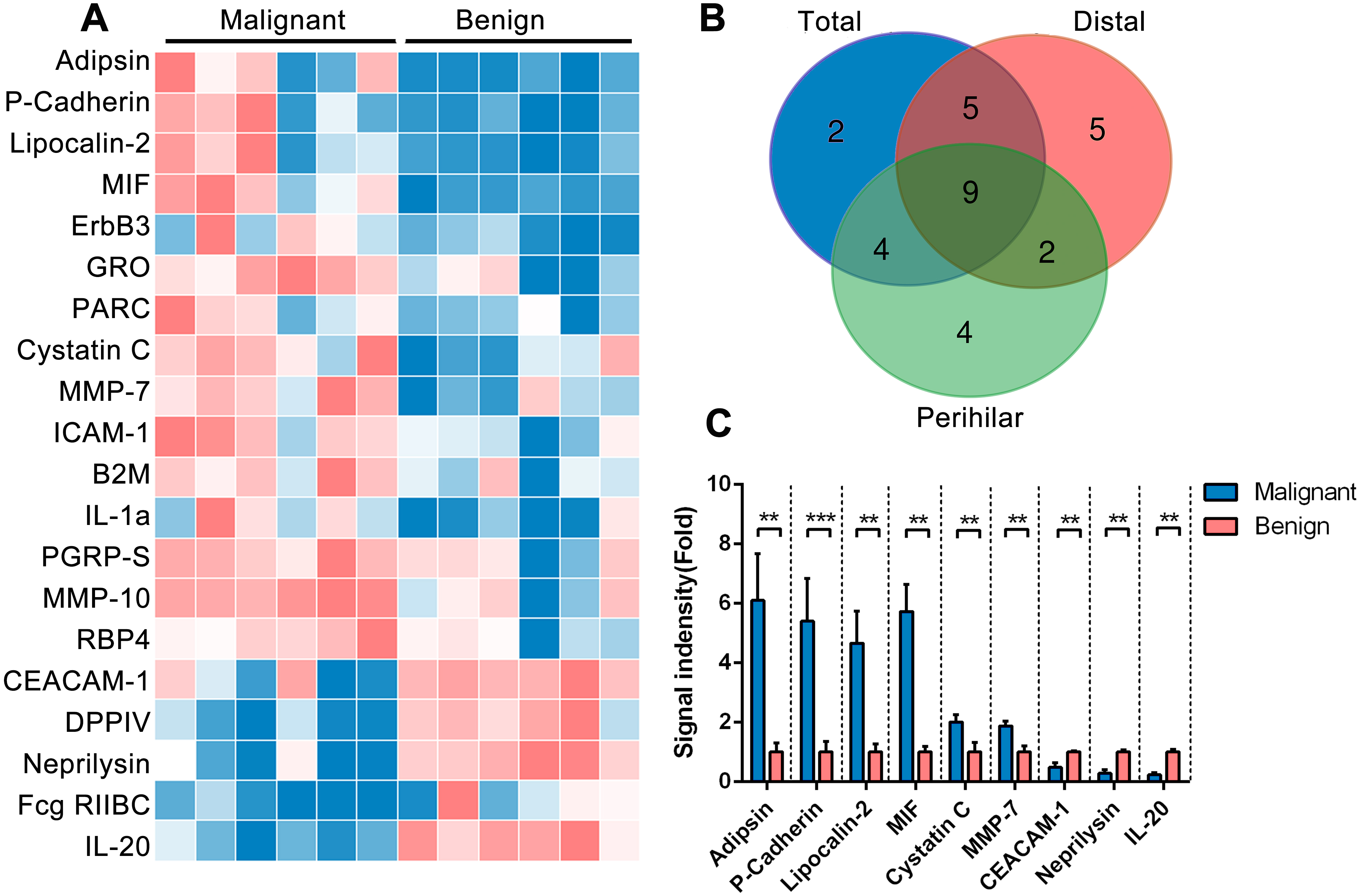
We further tested the level of the 6 selected proteins in bile and serum samples from an independent cohort, including 14 cholangiocarcinoma, 4 pancreatic ductal adenocarcinoma, 2 ampullary adenocarcinoma, 6 PSC and 14 cholelithiasis patients. We also measured the level of CA19-9 in the bile samples for comparison. As expected, the levels of all six proteins, along with CA19-9, were significantly increased in the bile of the malignant patients (Fig. 2) and 4 of the 6 proteins, including CYSTATINC, MIF, P-cadherin and lipocalin-2, showed significantly enhanced levels in the serum of the malignant patients. However, no difference was observed in serum for adipsin and the level of MMP-10 was undetectable in serum.
A–G. Level of indicated proteins in bile samples of patients in validation panel (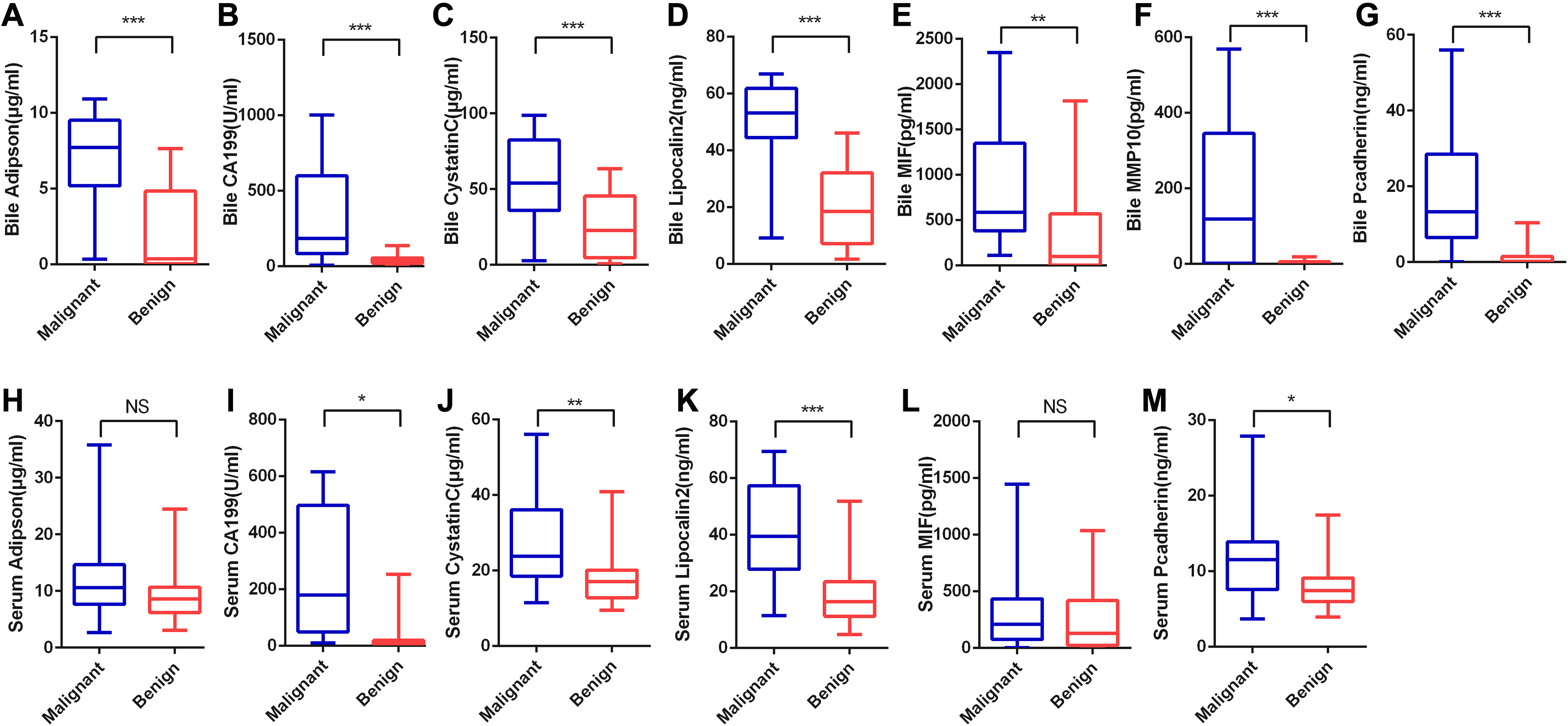
A–G. The ROC curve analysis for the indicated biomarkers in bile, the area under curve (AUC) was shown. H. The ROC curve analysis for the lasso score calculated using the Regression model, in which bile Lipocalin-2, P-cadherin and CA19-9 were included.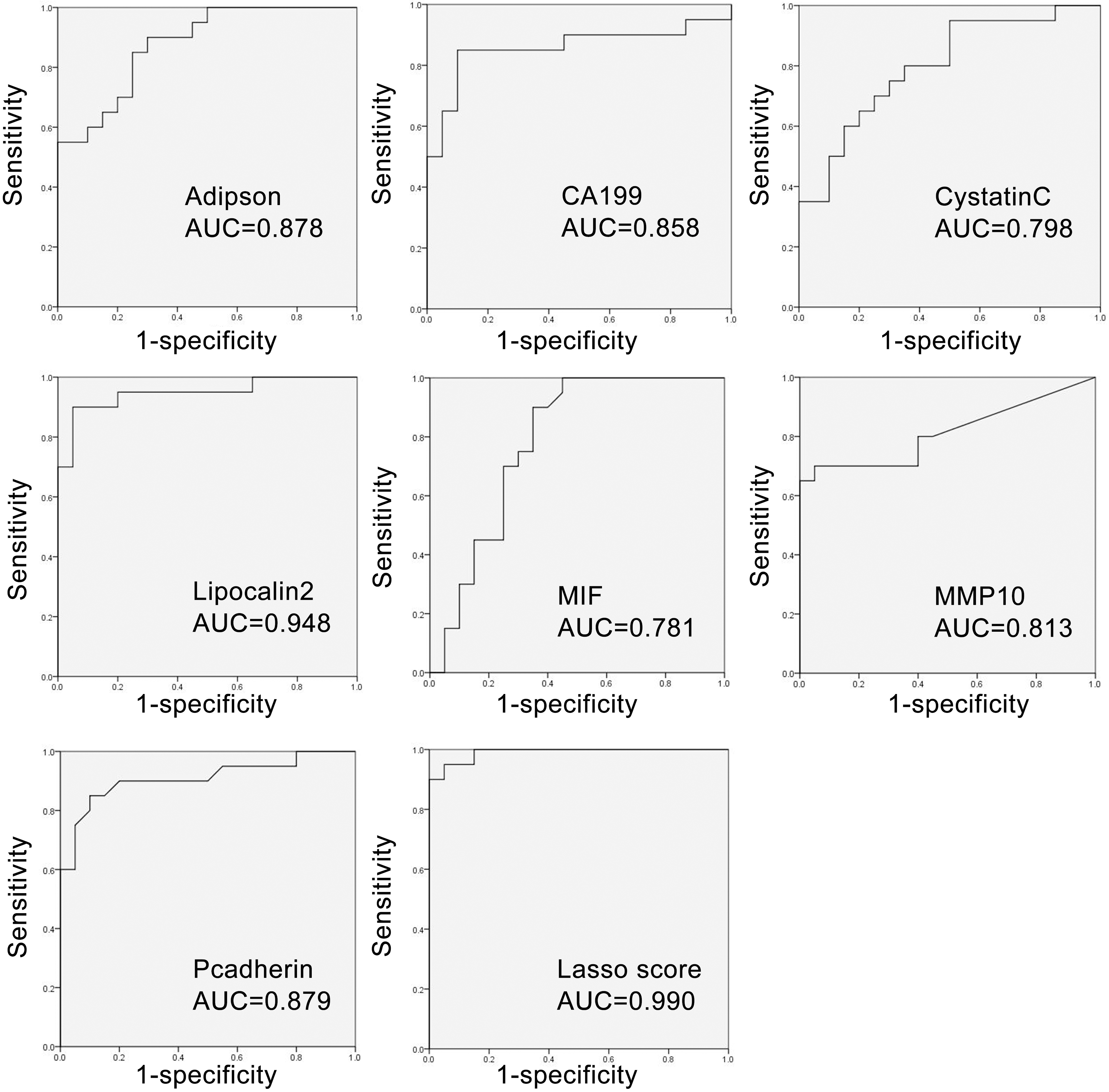
ROC curves were generated for the seven ELISA-tested biomarkers to ascertain their capacity in distinguishing malignant from benign strictures. As shown in Table 4 and Fig. 3, the AUC values range from 0.781 to 0.948, all with
Evaluation of putative biomarkers in bile to distinguish malignant and benign stricture by ROC-AUC analyses in validation panel
Evaluation of putative biomarkers in bile to distinguish malignant and benign stricture by ROC-AUC analyses in validation panel
ROC-AUC analysis was performed for each protein target showing significantly different levels in bile stricture patients in validation panel. The optimal cut-off was determined according to Youden’s index. For cases with biliary brushings (
Evaluation of putative biomarkers in serum to distinguish malignant and benign stricture by ROC-AUC analyses in validation panel
ROC-AUC analysis was performed for each protein target showing significantly different levels in bile from bile duct stricture patients in validation panel. The optimal cut-off was determined according to Youden’s index. AUC, area under the curve; CI, confidence interval; ROC-AUC, area under the curve of the receiver operating characteristics curve.
A–G. The ROC curve analysis for the indicated biomarkers in serum, the area under curve (AUC) was shown.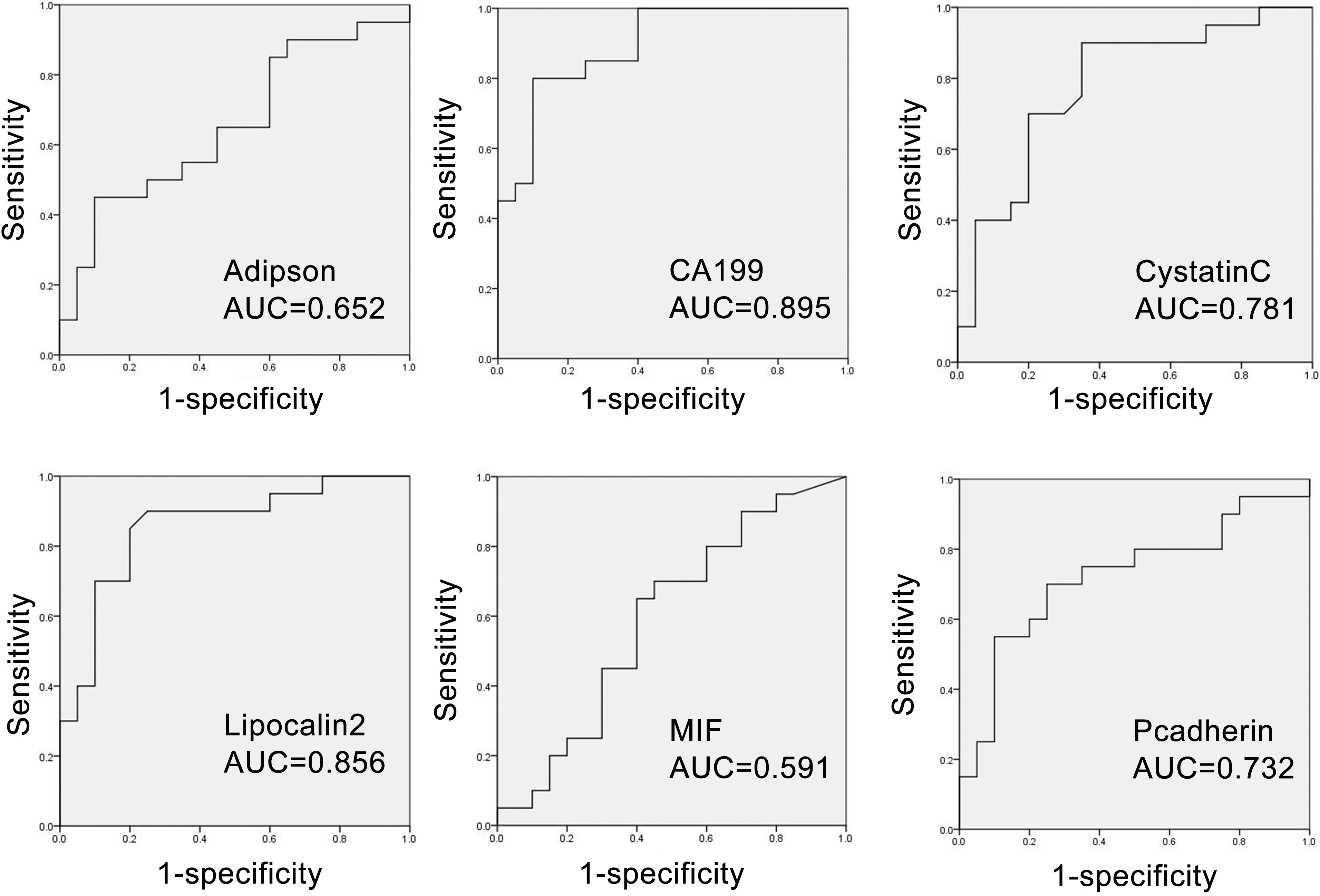
In serum samples, ROC-AUC analysis showed that CA19-9, CYSTATINC, lipocalin-2, and P-cadherin had the capacity to discriminant malignant from benign strictures, with a
Distinguishing malignant bile duct strictures from benign has long been a challenge in clinical practice. Although the specificity of diagnosis is high using bile duct brushings and forceps biopsies during ERCP, the sensitivity remains low. In this study, we used a high-throughput protein microarray platform to discover differentially expressed proteins in the bile of patients with malignant and benign bile duct strictures. Six biomarkers (adipsin, cystatin C, MIF, P-cadherin, lipocalin-2 and MMP10) were identified, among which the best single discriminator was lipocalin-2. Further analysis demonstrated that bile lipocalin-2, P-cadherin and CA19-9 as a group of markers best distinguished malignant from benign bile duct stricture, resulting in a sensitivity of 95% and specificity of 95%. In comparison, pathological evaluation of biliary brushings and biliary biopsies showed 100% specificity, but lower sensitivity (35.29% and 46.67%, respectively).
Bile is the immediate substance for malignant tumors involving the bile duct, tumor-related proteins may become detectable in bile rather than serum, especially during earlier stages of disease [11]. Thus the molecular diagnosis of malignant biliary stricture using biomarkers in bile has been the subject of intensive investigation. To date, various biomarkers have been identified in bile, such as ApoA-I, actinin-1, S100A9, CEAM6 and MCM5 [12, 13, 14, 15, 16], but only a few of these markers have been incorporated into routine clinical practice. This is probably due to the inconsistencies in the criteria for patient recruitment and the biological variability of single biomarkers, which results in conflicting conclusions. In the present study, the patients with malignant or benign biliary stricture showed comparable serum levels of CRP and WBCs and this suggested that the included patients had no significant differences in their inflammatory states. Thus, this eliminated the influence of conflicting factors such as cholangitis. However, different locations of stricture may lead to varying degrees of biliary obstruction and result in an altered composition of bile. We took this into consideration and performed subgroup analysis. By taking the intersection of three comparisons, we identified putative biomarkers which were differentially expressed regardless of the stricture site. All the selected biomarkers showed considerable diagnostic performance in the validation panel, and three of the six biomarkers exhibited a higher AUC than CA19-9. The following summarizes possible connections with malignant disease and the roles of these three biomarkers.
Lipocalin-2 has been reported to be a key element in the physiopathology of the neoplastic process [17]. However, the information on the direct relations between lipocalin-2 and pancreatobiliary malignant tumors is contradictory. The ability of lipocalin-2 to protect MMP9 from proteolytic degradation by forming the lipocalin-2/MMP9 complex would trigger an enhancement of the enzymatic activity of MMP9 and support enhanced tumoral invasiveness and diffusion [18]. On the other hand, lipocalin-2 can inhibit focal adhesion kinase (FAK) phosphorylation and reduce angiogenic capacity by diminishing the production of VEGF in cancer of the pancreas, thus blocking cell adhesion and invasiveness [19]. Although the role of lipocalin-2 in tumorigenesis is still uncertain, the level of lipocalin-2 in serum or urine has previously been proposed as a prognostic or diagnostic marker for different types of human tumors. A recent meta-analysis reported that lipocalin-2 determination in plasma and urine could be useful in the prognosis of colorectal and breast cancer, but its prognostic and diagnostic accuracy remains uncertain for other human tumors [20]. Abigail et al. first reported the raised level of lipocalin-2 in the bile of patients with malignant biliary obstruction, the AUC was 0.76, with 94% sensitivity and 55% specificity for distinguishing malignant from benign causes [21]. Kun et al. also revealed lipocalin-2 as a promising target for CCA treatment and bile lipocalin-2 levels as a potential diagnostic marker for CCA [22]. In corroboration with their findings, our results confirmed that the concentration of lipocalin-2 in bile was increased in the presence of a malignant tumor. Furthermore, the diagnostic performance of bile lipocalin-2 was compared in parallel with bile CA19-9 in the same cohort, and lipocalin-2 showed better diagnostic utility in both sensitivity (90% vs 85%) and specificity (95% vs 90%). In serum, the level of lipocalin-2 was also significantly increased in the malignant versus the non-malignant resulting in a lower, but still considerable diagnostic accuracy (AUC: 0.856).
As a classical cell-to-cell adhesion molecule, P-cadherin is highly expressed in undifferentiated cells in normal adult epithelial tissues, as well as in poorly differentiated carcinomas [23]. However, in the malignant setting the behavior of P-cadherin is contradictory and dependent on the cellular context. In some malignant tumors, such as kidney and oral squamous cell cancer, the absence of P-cadherin is associated with a more aggressive cancer cell phenotype. However, the overexpression of this molecule accelerate the growth of tumor in breast, ovarian, prostate, pancreas and colon neoplasms [24]. Recent meta-analysis demonstrated that P-cadherin overexpression can be used as a prognostic indicator in breast cancer [25]. Our study was the first to report P-cadherin in bile as a new and independent biomarker for differentiating between malignant and benign biliary strictures We also showed its superior diagnostic sensitivity for malignant tumors when compared to CA19-9 (0.879 vs 0.858).
Adipsin is highly and specifically expressed in adipocytes. The research on this molecule is focused in the field of metabolism as it was elucidated that adipsin can preserve beta cells in diabetic mice and protect from type 2 diabetes in humans [26, 27]. A recent study reported the association between adipsin and breast cancer, in which the adipsin secreted by adipose-derived stem cells enhanced human breast cancer growth and cancer stem cell-like properties [28]. In our study, we discovered the differential expression of adipsin in bile between malignant and benign biliary stricture, suggesting that adipsin could be a valued diagnostic marker for the malignant tumors in bile ducts, with an AUC of 0.878 and a sensitivity and specificity of 90% and 70%, respectively.
Our study comprehensively examined bile proteins as biomarkers that could be used to distinguish malignant from benign biliary strictures. The data in this study provides evidence that bile lipocalin-2, P-cadherin, and adipsin may be useful as single biomarkers, better than CA19-9. Lipocalin-2, P-cadherin and bile CA19-9, as a group, could be used collectively to improve the sensitivity and specificity of the diagnosis of biliary malignant tumors. The main limitation of this study is that the number of cases was relatively modest, and so we cannot ascertain whether the raised level of biomarkers in the bile is a common change under tumor burden or a specific change related to the kinds of included malignant diseases. Larger studies are needed to validate our findings and to assess whether the use of these biomarkers could improve patient management for a better clinical outcome.
Author contributions
Conception: W.H.Q, B.H.
Interpretation or analysis of data: W.H.Q, J.T.L, S.P.W, Z.S.Y.
Preparation of the manuscript: W.H.Q, J.T.L.
Revision for important intellectual content: W.H.Q, B.H.
Supervision: B.H.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210095.
sj-docx-1-cbm-10.3233_CBM-210095.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-210095.docx
Footnotes
Acknowledgments
This study was partially supported by funding from National Natural Science Foundation of China (82003005), Shanghai Committee of Science and Technology (20YF1459100).
