Abstract
BACKGROUND:
To explore the suppressive effect of Apoptin-loaded oncolytic adenovirus (Ad-VT) on luciferase-labeled human melanoma cells
METHODS:
The stable luciferase-expressing human melanoma cells A375-luc or M14-luc were obtained by transfecting the plasmid pGL4.51 and selection with G418, followed by luciferase activity, genetic stability and bioluminescence intensity assays.
RESULTS:
The growth and migration of A375-luc and M14-luc were significantly inhibited by Ad-VT
CONCLUSION:
Ad-VT reduced the mitochondrial membrane potential in A375-luc or M14-luc cells and induced the mitochondrial release of AIF, ARTS and Cyto-C. Ad-VT induced apoptosis in A375-luc or M14-luc cells via the mitochondrial apoptotic pathway.
Keywords
Introduction
Melanoma is the most lethal skin cancer. In 2014, the National Central Cancer Registry collected data from local cancer registries and showed that there were approximatively 6,761 new cases and 3,637 yearly deaths in China. With the development of molecular biology techniques and immunology, adenovirus-related biological treatment of melanoma has achieved exciting results [1, 2]. In a report of the melanoma-specific human data from the Advanced Therapy Access Program (ATAP), Ad5/3-D24-GMCSF [3, 4], a serotype 5/3 chimeric oncolytic adenovirus expressing granulocyte-macrophage colony-stimulating factor (GM-CSF), showed
An oncolytic adenovirus is a modified adenovirus that can enter and continuously replicate in tumor cells, where it induces their lysis and infect other tumor cells. Oncolytic adenovirus vectors are often used to express therapeutic genes due to their large load [22, 23]. With the continuous replication of oncolytic adenoviruses in tumor cells, such as Ad3-hTERT-CMV-hCD40L in A549 cells [24], SG635-SF in SK-OV3 and HO9010 cells [25], EnAd in DLD-1 cells [26], ONCOS-102 in AB12 cells [27], and ICO15K-FBiTE in HT1080 cells [28], therapeutic genes are expressed to induce a synergistic anti-tumor effect. The dual cancer-specific oncolytic adenovirus Ad-hTERT-E1a-Apoptin (Ad-VT) and Ad-mock have been constructed in our laboratory [29]. Ad-VT contains the human telomerase reverse transcriptase (hTERT), the adenovirus early region 1a (E1a) gene, the CMV promoter and the chicken anemia virus apoptin gene [30, 31]. Ad-mock that lacks hTERT, the E1a and apoptin genes, was used as a blank control. Ad-VT can replicate and express apoptin in the tumor resulting in accelerated tumor cell apoptosis. Research studies using Ad-VT demonstrated its safety in BALB/c mice, Wistar rats, male Hartley guinea pigs and beagles [32]. We also showed that the growth of many tumor cells [33, 34, 35], such as human gastric cancer (SGC7901) cells, human prostate carcinoma (PC-3) cells, human lung cancer (A549) cells and breast cancer (MCF-7) cells, could be significantly suppressed by Ad-VT
Materials and methods
Virus, cell lines, plasmid, reagents and mice
Ad-VT and Ad-mock were previously constructed in our laboratory. The human melanoma cell lines, A375 and M14, were purchased from the Cell Bank, Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China). The MTS cell proliferation kit, Plasmid pGL4.51, ONE-Glo
Establishment of A375-luc or M14-luc cells
A375 or M14 cells were cultured at a density of 2
Detection of A375-luc and M14-luc cells
Stable luciferase-expressing human melanoma cell lines (A375-luc or M14-luc) were detected via their luciferase activity, analysis of their genetic stability and intensity of bioluminescence. To detect luciferase activity, all monoclones that had a good cell growth, were cultured at a density of 5
To analyze the genetic stability of A375 Luc or M14 Luc cell lines, the cells with the highest luciferase activity were subcultured 40 times and their genetic stability determined every 10 generations. The monoclone with the highest luciferase activity and the best genetic stability was named A375-luc or M14-luc.
To evaluate the intensity of bioluminescence
In vitro evaluation of the suppressive effect of Ad-VT on A375-luc or M14-luc cells
The suppressive effects of Ad-VT on A375-luc or M14-luc were evaluated using the MTS cell proliferation assay, FITC-Annexin V apoptosis detection, transwell migration, Matrigel invasion, and the scratch test. To analyze the inhibitory effects of Ad-VT on infected cells, A375-luc or M14-luc cells were seeded in 96-well cell culture plates at 1
For the transwell migration and Matrigel invasion assays, Ad-VT-infected A375-luc or M14-luc cells were seeded in 24 well plates with at a concentration of 5
For the scratch test, Ad-VT-infected A375-luc or M14-luc cells were seeded in 6-well plates at a concentration of 2
Analysis of the inhibitiory pathway in Ad-VT-infected A375-luc or M14-luc cells
The inhibitiory pathway that relied on detecting mitochondrial apoptosis-associated proteins was analyzed using JC-1 staining and Western-blot. To detect changes in the mitochondrial membrane potential in Ad-VT-infected A375-luc or M14-luc, the two cell types were seeded at 2
To detect the levels of mitochondrial apoptosis-related proteins in Ad-VT-infected A375-luc or M14-luc, the two cell types were seeded at 2
Xenograft tumor model in BALB/c nude mice and treatment strategy.
Six-week-old BALB/c nude mice were purchased from the Experimental Animal Center, Academy of Military Medical Sciences of the PLA (Beijing, China) and were housed under standard pathogen-free conditions. The animal experimental protocols were approved by the Institutional Animal Care and Use Committee of the Chinese Academy of Military Medical Science, Changchun, China (10ZDGG007). All surgeries were performed under sodium pentobarbital anesthesia, and all efforts were made to minimize suffering. The xenograft models were established via subcutaneous injection of A375-luc or M14-luc cells (3
Statistical analysis
The statistical analysis was performed using data from at least three independent experiments using the Statistical Package for the Social Sciences (SPSS) statistical software package (version 15.0; SPSS Inc., Chicago, IL, USA), and the results were obtained using GraphPad Prism version 7.0 (GraphPad Software Inc., La Jolla, CA, USA). Student’s
Results
Construction and identification of A375-luc or M14-luc cells
The optimum concentration of G418 was determined as 200
Identification of A375-luc or M14-luc cells. (A) Luciferase activity of each A375-luc monoclone of A375-luc. Clone 11 that had the highest luciferase activity, was screened with 200 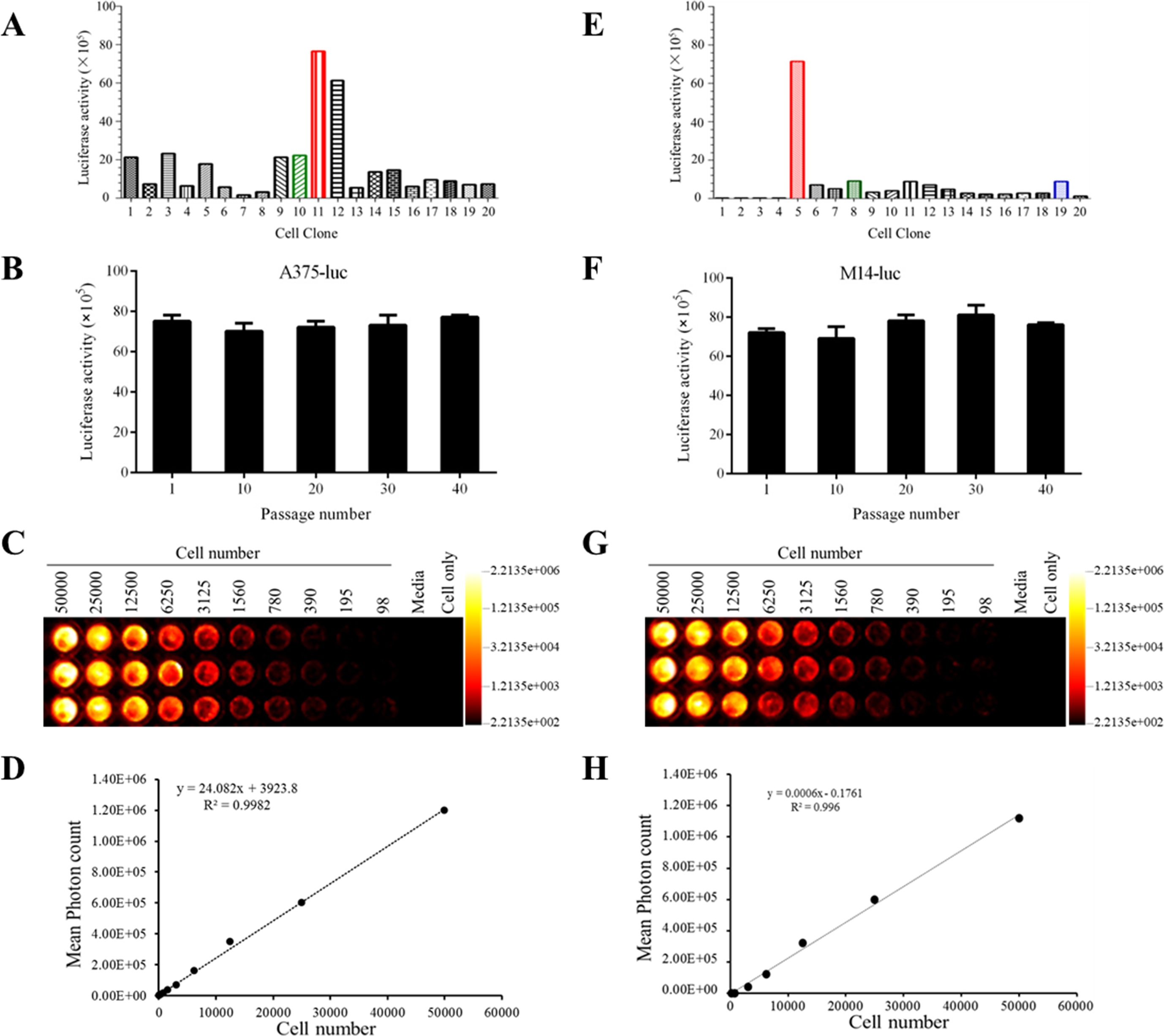
The inhibitory effects of Ad-VT on A375-luc or M14-luc cells. (A–B) MTS proliferation assay showing the suppression of the proliferation of A375-luc and M14-luc cells following infection with Ad-VT or Ad-mock at a MOI of 10, 50 and 100. (C–D) Detection of apoptosis rates in Ad-VT-infected A375-luc or M14-luc cells. FITC annexin V flow cytometry was used to detect the apoptosis rate of Ad-VT infected, Ad-mock infected, and control cells at 72 h. (E–F) Transwell migration assay showing the inhibition of the migration of A375-luc or M14-luc cells infected with Ad-VT at MOI of 100 for 72 hours (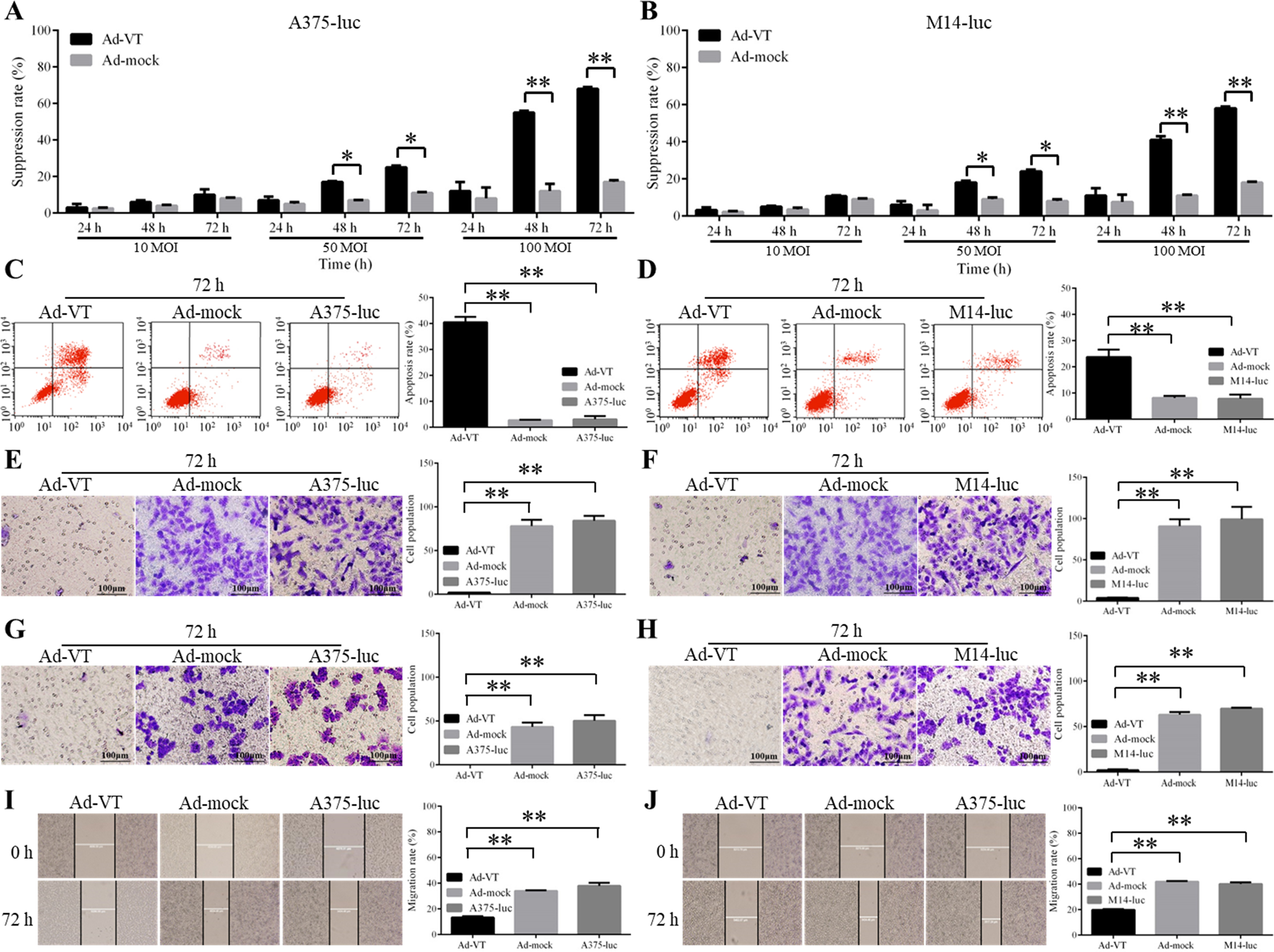
Ad-VT infection of A375 Luc and M14-luc cells inhibited their proliferation (Fig. 2A and B). There was no significant difference in growth suppression between Ad-VT-infected and Ad-Mock-infected cells at a MOI of 10 (
The apoptosis of Ad-VT-infected A375-luc and M14-luc cells at 72 h was evaluated by flow cytometry (Fig. 2C and D). The rate of apoptosis in Ad-VT-infected cells was significantly higher than that in Ad-mock-infected cells and controls at 72 h (
Using the transwell migration assay, we observed that the migration rates of A375-luc and M14-luc cells that were infected with Ad-VT at MOI of 100 for 72 h, were significantly lower than those of other groups (Fig. 2E and F) (
Taken together, the results of the MTS cell proliferation assay, transwell migration, Matrigel invasion and scratch test demonstrate that the proliferation, migration, and invasion of A375-luc or M14-luc cells are inhibited by Ad-VT in a dose- and time-dependent manner.
The inhibition pathway in Ad-VT-infected A375-luc or M14-luc cells. (A–B) JC-1 staining of Ad-VT-infected A375-luc and M14-luc cells at 24, 48 and 72 h (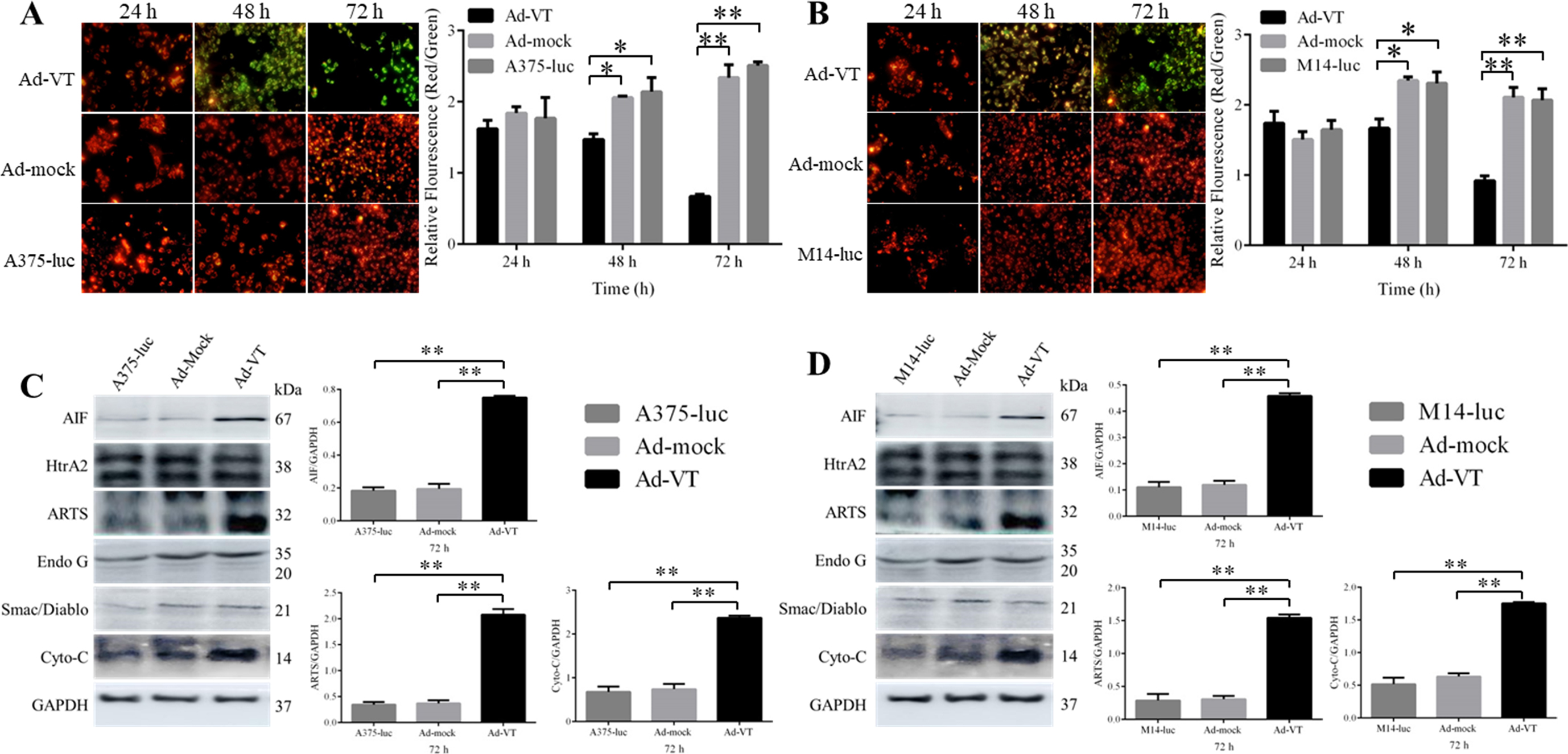
Effects of Ad-VT on BALB/C nude mice A375-luc model. (A) Living imaging of the xenograft tumor BALB/c nude mice model on day 8 day and day 36. (B) Survival rates of xenograft tumor BALB/c nude mice. (C–D) The tumor growth inhibition effect in transplanted nude mice. The tumor volume in the Ad-VT group was significantly smaller than that in Ad-mock and Normal Saline groups (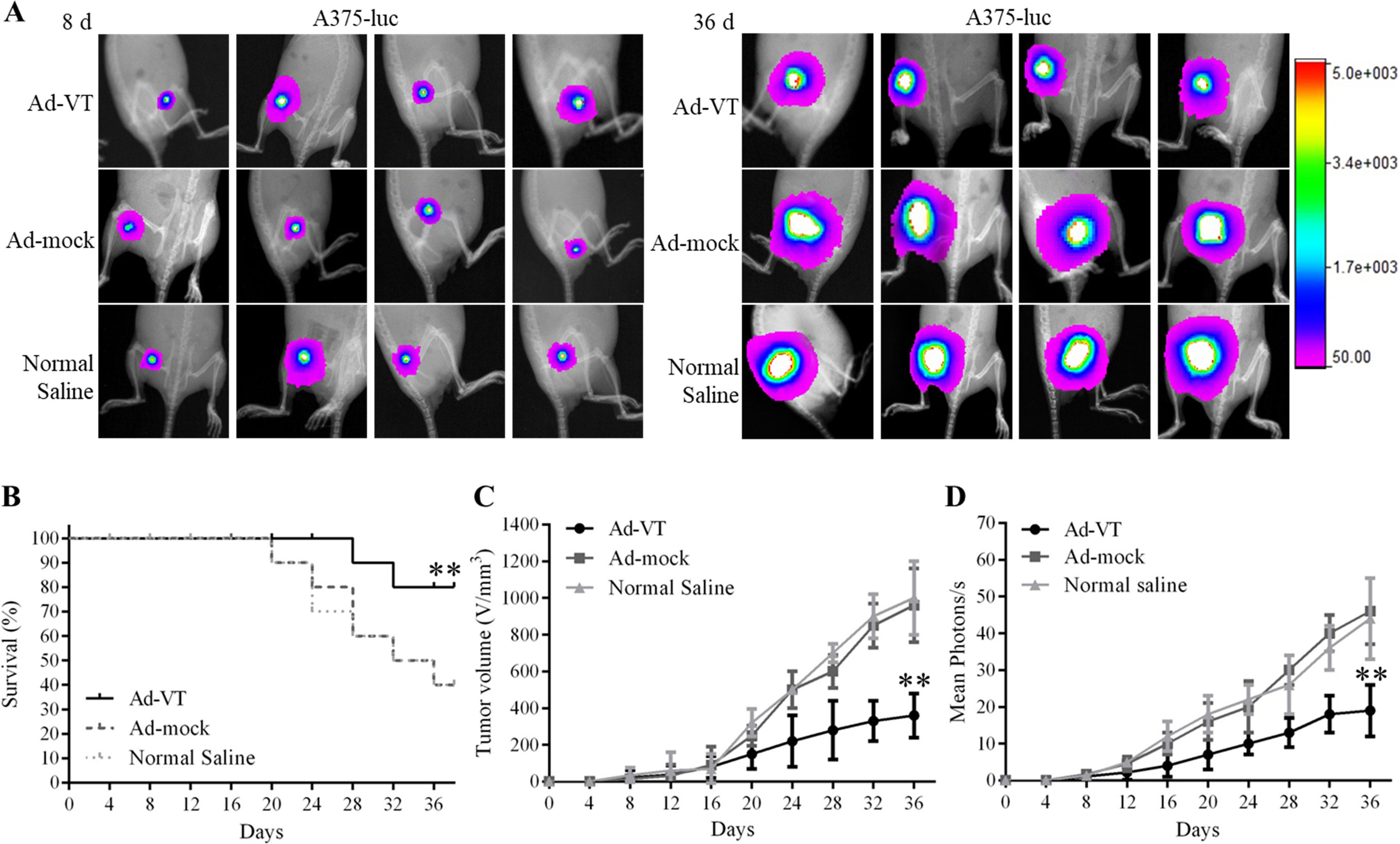
Effects of Ad-VT on BALB/C nude mice M14-luc model. (A) Living imaging of the xenograft tumor BALB/c nude mice model on day 8 and day 36. (B) Survival rates of xenograft tumor BALB/c nude mice. (C–D) Inhibition of tumor in transplanted nude mice. The tumor volume in the Ad-VT group was significantly smaller than that in Ad-mock and Normal Saline groups (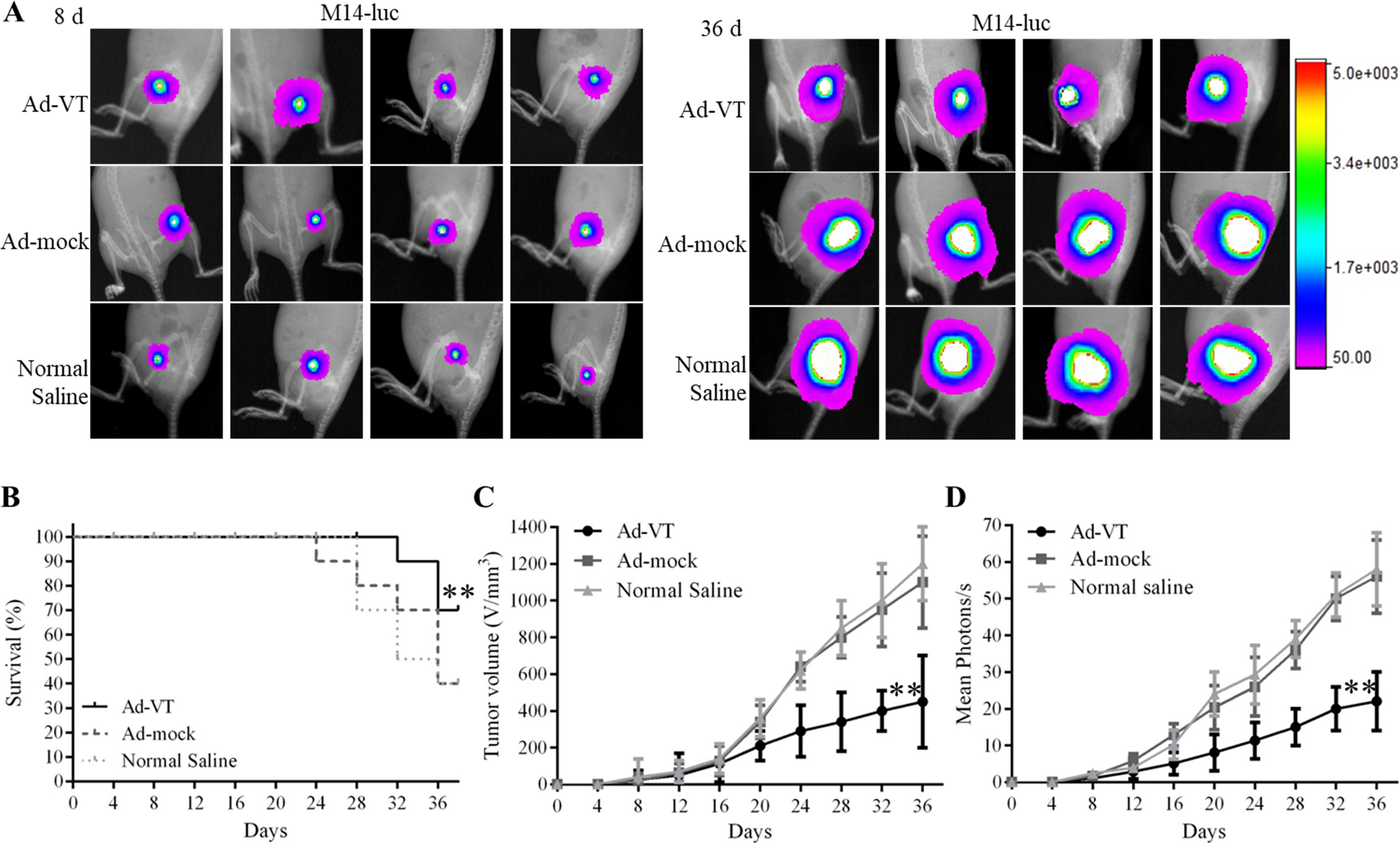
The immunohistochemical detection of proteins related to the mitochondrial apoptotic pathway. (A–B) Expression levels of proteins related to the mitochondrial apoptotic pathway (AIF, ARTS Cyto-C) were detected by immunocytochemistry (800 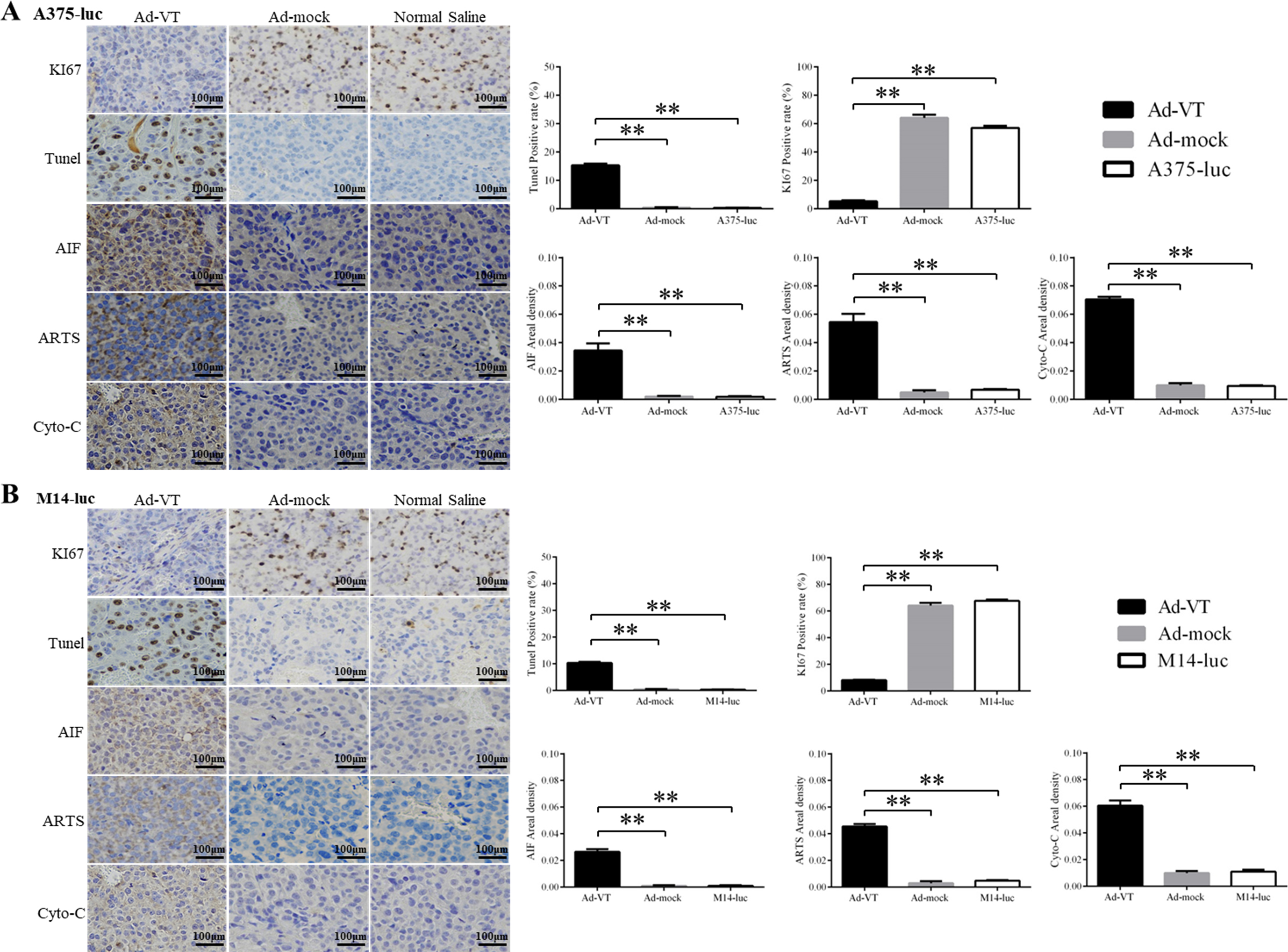
The results of the inhibition pathway analysis in A375-luc or M14-luc cells are shown in Fig. 3A–D. The fluorescence of A375-luc and M14-luc cells that were stained with JC-1 changed from red to green within 24 and 72 hours after infection with Ad-VT at a MOI of 100 (Fig. 3A and B). The relative fluorescence (Red/Green) of Ad-VT-infected cells was significantly higher than that of Ad-mock-infected cells and controls (
The expression levels of AIF, ARTS, and Cyto-C, in Ad-VT-infected A375-luc and M14-luc cells were significantly higher than those in Ad-Mock-infected cells at 72 h (Fig. 3C and D) (
Tumor growth suppression and survival rate in BALB/c nude mice model
The proliferation of A375-luc or M14-luc cells can be significantly suppressed by Ad-VT in tumor bearing nude mice. The results of the tumor growth suppression experiment are shown in Figs 4A–D and 5A–D. Tumor growth in the Ad-VT group was significantly lower than that in the Ad-mock and Normal Saline groups (
These results indicate that we have successfully established a tumor model of A375 Luc or M14 Luc in BALB/c nude mice and demonstrated that Ad-VT inhibits tumor growth and improve survival rate
Immunohistochemical studies of the tumors showed that Ad-VT significantly inhibits the proliferation of A375-luc and M14-luc cells and causes significant changes in proteins related to mitochondria (Fig. 6A and B). The positive rates for Ki67 staining in tumors grown by the Ad-VT group were significantly lower than those in the Ad-Mock and Normal Saline groups (
Discussion
Melanoma is a type of skin cancer that originates from the abnormal proliferation of melanocytes in the neural crest located in the epidermal layer of the skin [36]. It is highly malignant and invasive, and prone to blood and lymph node metastasis. Once metastasis occurs, the mortality is extremely high and approximatively 80% of skin cancer patients die of melanoma [37]. The treatment of melanoma patients involves surgery, radiation, chemical, and molecular targeted therapies; however, after chemotherapy, there are often adverse reactions. Although melanoma diagnosis technology is well advanced, late detection of the disease in some patients with recurrence and metastasis results in poor treatment. In recent years, with the development of biomedical and genetic engineering technologies, many new therapeutic programs have been developed, including gene-targeted therapy and oncolytic virotherapy [38].
Oncolytic adenoviruses are a type of selective tumor-killing viruses that have many advantages compared with other types of tumor therapies, including a multi-path killing mechanism and a broad anti-tumor spectrum, safe and lower toxic side effects, and a low cost compared with cell immunotherapy such as CAR-T Because of the outstanding performance, adenoviruses have attracted a significant attention in the field of oncolytic virotherapy and tumors’ targeting strategies using adenoviruses as vectorshave been shown to have great applications’prospect. In previous studies, we used the CMV promoter, apoptin, the hTERT promoter and E1a, to generate a tumor-specific replicative recombinant adenovirus [39, 40]. Apoptin has a broad range of antitumor properties that can specifically induce apoptosis of tumor cells without affecting normal cells [41]. Most normal human cells lack telomerase activity; however, hTERT expression and telomerase activation have been observed in most human malignances [42]. Therefore, Ad-VT can replicate and continuously express apoptin in tumor cells, but not in normal cells.
In this study, we investigated the effects of Ad-VT on the tumorigenesis and tumor progression of human melanoma cells tumorigenesis and progression, and we plan to establish a visualized human melanoma bearing nude mouse model using living imaging technology, one of the most common techniques. At present, luciferase that has the advantages of luminescence without excitation, strong penetrability, high sensitivity, and specificificity of interfence excitation, is one of the most commonly used marker gene in living imaging technology that is widely used in anti-tumor research. Tumorigenesis, tumor progression and metastasis in luciferase-labeled tumor bearing nude mouse model can be detected using living imaging technology. The bioluminescence intensity in the tumor area is detected using living imaging technology in orthotopic, metastatic and xenograft luciferase-labeled tumor models, where it can directly reflect the change in tumors’ volume. Tumor metastasis can be traced according to the location of bioluminescent regions [43, 44]. In our model, with the increase of tumor volume, the intensity of bioluminescence increased gradually and according to the bioluminescence localization, no metastasis occurred during the observation period.
In the present study, we used the plasmid pGL4.51 (Promega), which expresses the luciferase type 2 gene at a high level, to establish A375-luc and M14-luc stable luciferase-expressing human melanoma cell lines, that were used to visualize tumor growth in our
The inhibition pathway in Ad-VT-infected A375-luc and M14 cells was analyzed using FITC-Annexin V apoptosis flow assays, JC-1 staining and Western-blot. These results showed that after infection with Ad-VT, the apoptosis levels of A375-luc and M14-luc cells were significantly increased, reaching 40.54% and 23.7%, respectively. The Western-blot results demonstrated that the mitochondrial membrane potential and apoptotic-related protein expression levels in Ad-VT-infected A375-luc or M14-luc cells were significantly higher than those of the control group (
In conclusion, the oncolytic adenovirus Ad-VT has a significant inhibitory effect on A375-luc and M14-luc stably expressing luciferase cells on vitro.
Footnotes
Acknowledgments
This work was supported by the National Key Research and Development Program of China [grant number 2016YFC1200901], the Major Technological Program of Changchun City [grant number 16ss11], the National Science and Technology Major Project (Major New Drugs Innovation and Development) [grant number 2018ZX09301053-004-001] and Jilin Province Science and Technology Development Plan Project [grant number 20160209015YY].
Conflict of interest
The authors have no conflicts of interest to declare.
Author contributions
Conception: Xiao Li and Rihua Jiang.
Interpretation or data analysis: Min Li, Yilong Zhu, Bing Bai, Wei Yao, Yiquan Li and Shanzhi Li.
Manuscript preparation: Yilong Zhu and Min Li.
Revision for important intellectual content: Min Li and Jinbo Fang.
Supervision: Ningyi Jin and Rihua Jiang.
