Abstract
BACKGROUND:
Breast cancer (BC) is one of the most life-threatening cancer types among women. Despite major developments in medical sciences and technologies, the incidence and mortality rates of BC cases are still increasing. One of the reasons for this increase is the absence of an easy to perform early-diagnostic tool. Although there are defined BC biomarkers routinely used for diagnostic and prognostic purposes, none of these biomarkers is useful for early diagnosis. Therefore, early diagnosis of BC remains an important challenge and there is a great need for the early-diagnostic biomarker(s).
OBJECTIVE:
In this study, we aimed to evaluate the diagnostic and prognostic values of glycerol-3-phosphate dehydrogenase (GPD1) and monoacylglycerol lipase (MAGL) proteins as non-invasive serum biomarkers.
METHODS:
GPD1 and MAGL serum levels were determined by ELISA for BC patients (
RESULTS:
The results provided evidence that GPD1 acted as a diagnostic biomarker in distinguishing triple-negative breast cancer (TNBC) patients from other subtypes, and MAGL acted as a diagnostic biomarker in distinguishing healthy individuals from BC patients.
CONCLUSION:
GPD1 and MAGL might be proposed as non-invasive diagnostic biomarkers for BC with high sensitivity and specificity.
Keywords
Introduction
Breast cancer (BC) is a worrisome disease for women around the world [1, 2]. According to the World Health Organization (WHO), 2.1 million women each year suffer from this debilitating disorder [3]. In 2018, WHO estimated that 627.000 women died from BC which accounted for 15% of all cancer deaths among women [3]. To improve BC outcomes and survival rates, better diagnostic and prognostic tools are needed. Those tools should help to screen large women populations and allow early detection of BC. Protein-based biomarkers circulating in serum are among the tools used for the screening of various diseases including BC. Unfortunately, so far, none of the known protein-based serum biomarkers allows the early detection of BC with high specificity and sensitivity [4, 5, 6, 7]. Therefore, the ongoing quest for the discovery of such biomarkers continues.
In a recent comparative proteomics study, several differentially regulated proteins were identified in BC tissue samples [8]. Bioinformatics analysis of the differentially regulated proteins underlined the importance of glycerol-3-phosphate dehydrogenase (GPD1, Uniprot# P21695) and monoacylglycerol lipase (MAGL, Uniprot# Q99685) which play essential roles in triacylglycerol and lipid metabolisms. Our previous findings provided evidence that GPD1 and MAGL were the two tissue-based diagnostic and prognostic biomarker candidates for BC and also held the potential to differentiate the triple-negative breast cancer (TNBC) subtype from the other BC subtypes. Although our findings, overall, are very promising, they may only be useful for monitoring the changes at the tissue level. Therefore, their use in clinics is limited and less practical. In this study, we aimed to carry out a follow-up study to evaluate the possibility of using GPD1 and MAGL as serum-based biomarkers for the diagnosis/prognosis of BC. For this purpose, GPD1 and MAGL serum levels were measured by ELISA, and the diagnostic and prognostic values of these two proteins as non-invasive serum biomarkers were evaluated.
Materials and methods
Ethical approval
Ethical approval for this study was obtained from the Ethics Committee of Kocaeli University with approval ID of KU-GOKAEK-2018/49. Informed consent was obtained from each patient.
Patient characteristics
Patients who were diagnosed with invasive or infiltrating ductal carcinoma at the General Surgery Department of Kocaeli University Medical School and underwent lumpectomy or mastectomy between 2015 and 2017 were evaluated for the study. The patients who received neoadjuvant treatment such as endocrine therapy and/or chemotherapy were excluded from the study group. A total of 100 female BC patients aged between 30–60 years old were selected and included in the study.
Procurement and storage of the sera
Five mL of blood was collected from each BC patient (
Molecular subtyping of tumour samples
Molecular subtyping of tumour samples was performed at the Pathology Department of Kocaeli University Medical School based on the expression levels of ER, PR, HER2, and Ki-67 proteins. The BC patients were grouped into five different subtypes, namely Luminal A (ER
Preparation of serum pools
Serum pools for each tumour subtype were created using equal volumes of serum samples from each patient of the same subtype. The control serum pool was similarly created using the serum samples of healthy volunteers (
Serum GPD1 and MAGL levels in the BC subtypes and the control group (mean
standard deviation)
Serum GPD1 and MAGL levels in the BC subtypes and the control group (mean
Serum GPD1 and MAGL levels were determined by following the instructions of commercially available ELISA kits (MyBioSource, cat. no. MBS9344172 and MyBioSource, cat. no. MBS167556, respectively). In brief, the positions for the standards, samples, and the blank were assigned to the wells of the ELISA plates and added to each corresponding assigned well. HRP-conjugated reagents were added to each well and the plates were incubated at 37
Statistical analysis
The statistical analyses were carried out using R (Version 3.5.3). All variables were expressed as mean
Evaluation of the diagnostic values of the biomarkers
The diagnostic values of GPD1 and MAGL were determined through sensitivity and specificity metrics, and the Receiver Operating Characteristic (ROC) curve. The optimal cut-off level for biomarker quantification was calculated by the Youden index.
Evaluation of the prognostic values of the biomarkers
The prognostic values of GPD1 and MAGL were determined by using BC RNA-Seq dataset retrieved from The Cancer Genome Atlas (TGCA) and survival analysis via the SurvExpress validation tool [10]. In SurvExpress, the samples were partitioned into low- and high-risk groups according to their prognostic index. The prognostic capabilities of the potential biomarkers were characterized through Kaplan-Meier plots using the overall survival times of patients, the log-rank test, and the hazard ratio (HR).
Results
Serum GPD1 levels discriminated TNBC patients from the other BC subtypes and the control
Serum GPD1 levels for each BC subtype and the control were initially analyzed in serum pools by ELISA. Serum GPD1 levels for the TNBC, LumB-like HER2
Serum expression levels of GPD1 and MAGL determined by ELISA (A) Comparative analysis of the GPD1 serum expression levels among the BC subtypes and control group (B) Comparative analysis of the MAGL serum expression levels among the BC subtypes and control group.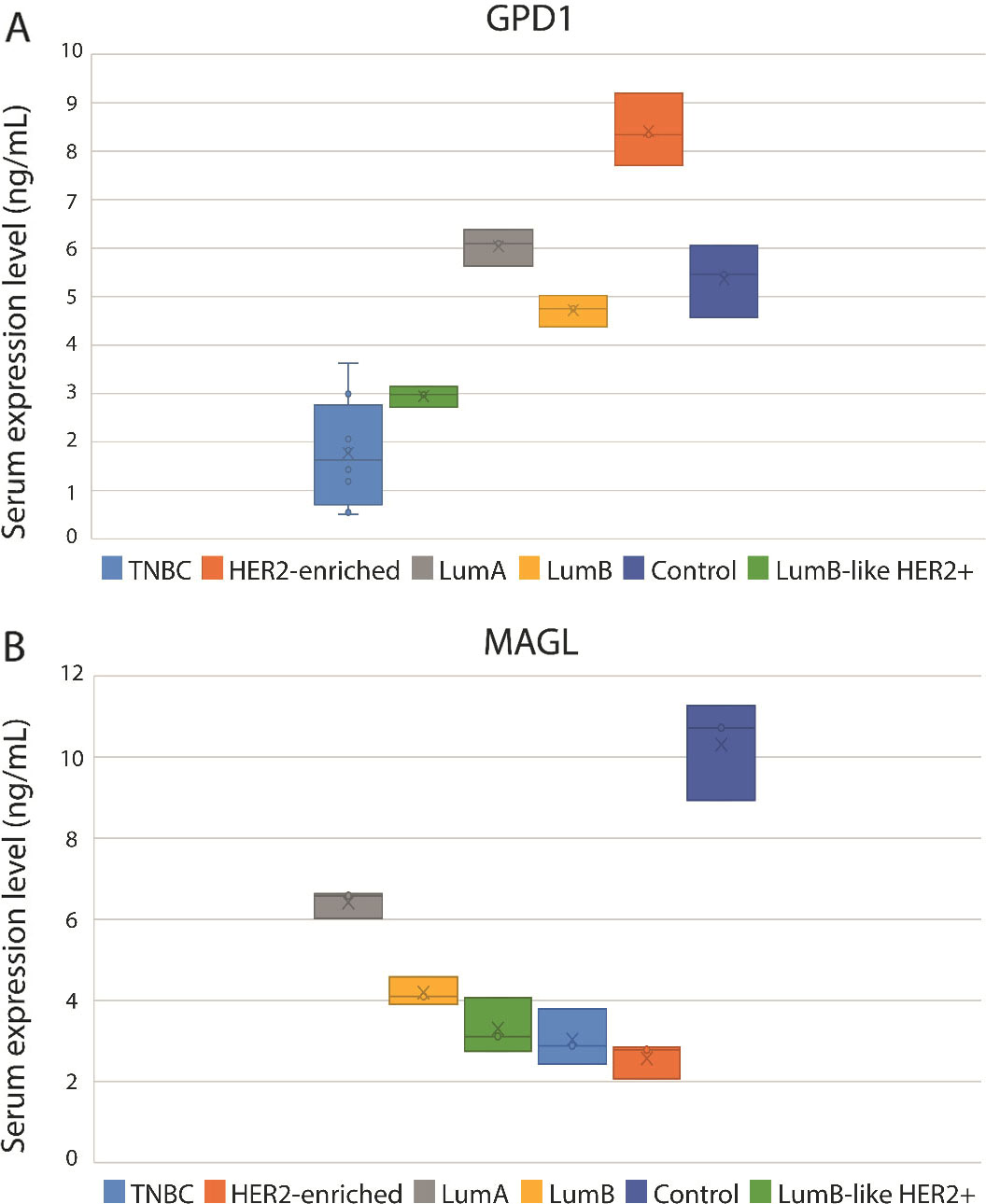
To further investigate serum GPD1 levels in the individual TNBC serum samples (
Serum MAGL levels for each BC subtype and the control were determined in serum pools by ELISA. The serum MAGL levels in all BC subtypes were significantly lower than in the control pool (Fig. 1B; Table 1). The lowest MAGL level was detected in the HER2-enriched serum pool while the control pool had the highest MAGL level. Like GPD1, serum MAGL levels in each TNBC sample were determined for cross-comparisons. Serum MAGL levels of two individuals (P2 and P6) were similar to the level detected in the control group, whereas all the other nine individuals had lower MAGL levels than the control pool. It was possible to state that control samples had much higher MAGL levels than all BC subtypes. The difference in serum MAGL levels between each BC subtype and the control were statistically significant (TNBC (
GPD1 and MAGL mRNA expression levels were determined using the BC RNA-Seq dataset retrieved from The Cancer Genome Atlas (A) Comparative analysis of the GPD1 mRNA expression levels among the BC subtypes and control group (B) Comparative analysis of the MAGL mRNA expression levels among the BC subtypes and control group.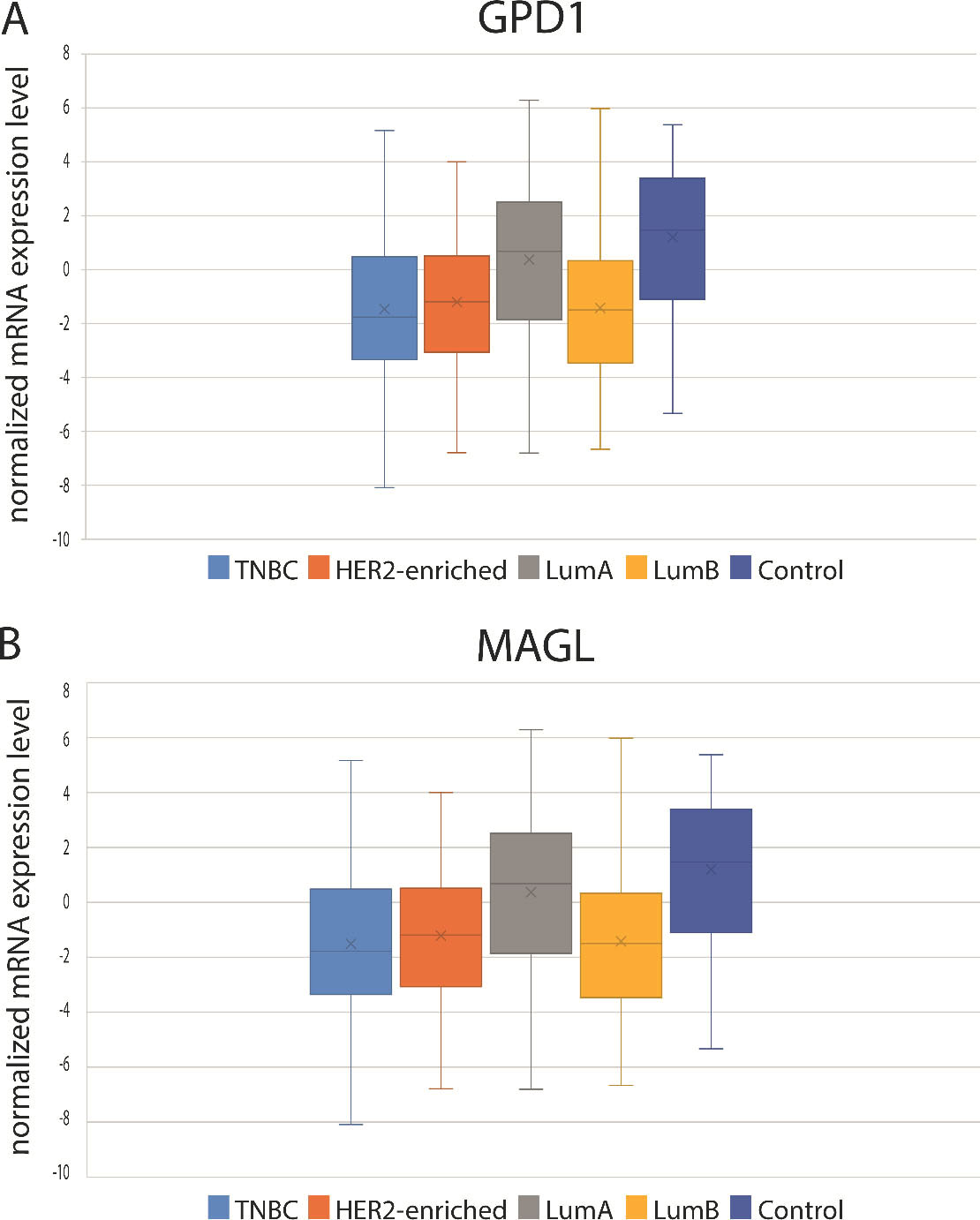
Using the mRNA expression data retrieved from TCGA (BRCA dataset with 155 TNBC, 74 HER2, 507 LumA, 191 LumB, and 34 control samples), the GPD1 mRNA expression levels of each subtype were evaluated (Fig. 2A). A profile similar to the serum GPD1 levels was observed in GPD1 at mRNA expression levels for BC patients. However, when pairwise comparisons were performed between the subtypes using the mRNA levels, the differences were significant for only TNBC vs. LumA (
Serum MAGL levels rather than mRNA levels were more predictive in discriminating TNBC patients
Using the mRNA expression data retrieved from TCGA, the mRNA expression levels of MAGL in each subtype were also evaluated (Fig. 2B). mRNA expressions were the lowest in the HER2-enriched subtype and highest in the control group. But the differences were not statistically significant.
Decreased serum GPD1 level may be used as a potential diagnostic biomarker for TNBC
To assess the value of serum GPD1 levels as a diagnostic biomarker in differentiating the TNBC subtype from the other subtypes, a ROC curve was created and the area under the curve (AUC) metric was determined (Fig. 3A). The ROC curve indicated an AUC that corresponded to test the accuracy of 0.948 (95% CI: 0.928–0.968,
Evaluation of the diagnostic value of GPD1 as a biomarker in distinguishing TNBC patients from other BC subtypes (A) To determine the diagnostic value of GPD1, the Receiver Operating Characteristic (ROC) curve, and the area under the curve (AUC) metric was employed. The ROC analysis showed that sensitivity and specificity were 95.0% and 77.5%, respectively and the AUC was 0.948 (95% CI: 0.928–0.968, 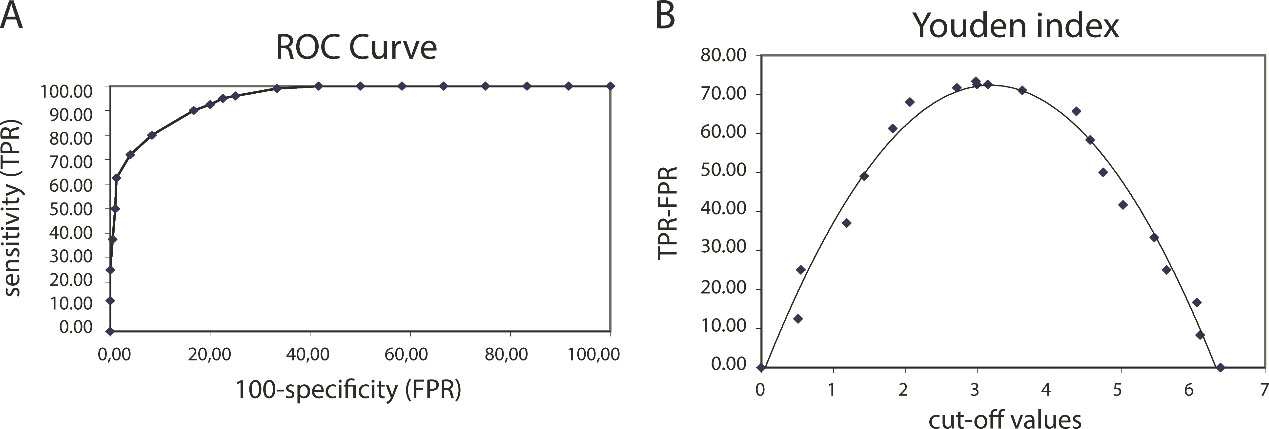
Evaluation of the diagnostic value of MAGL as a biomarker in distinguishing healthy subjects from BC patients from any subtypes (A) To determine the diagnostic value of MAGL, the Receiver Operating Characteristic (ROC) curve and the area under the curve (AUC) metric was employed. The ROC analysis showed that the sensitivity and specificity were 100% and 98.5%, respectively and AUC was 0.980 (95% CI: 0.968–0.992, 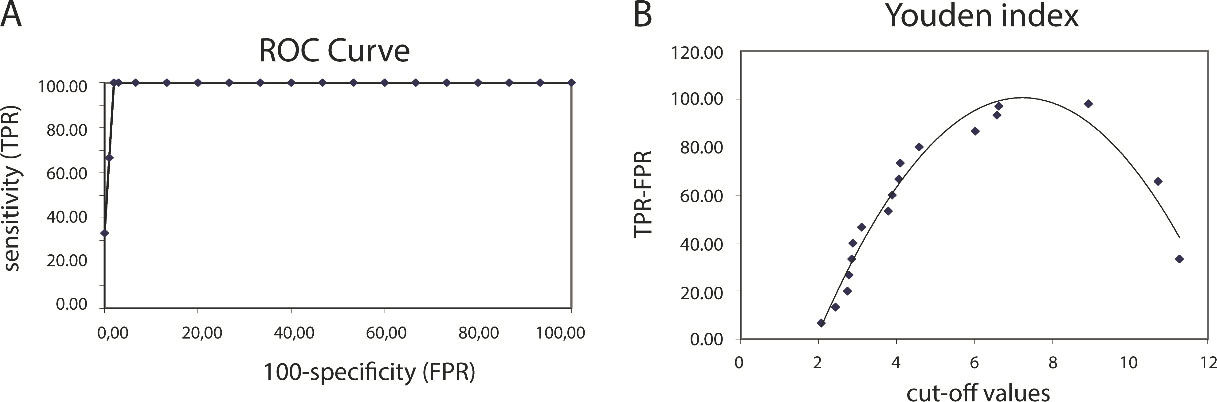
Evaluation of the prognostic value of GPD1 and MAGL. Patients’ outcomes were analysed through Kaplan-Meier survival analysis using mRNA expression data retrieved from TCGA. (A) Box blot graph representing low- and high-risk groups for GPD1 in BC patients. Using the mean expression level of GPD1 mRNA, the samples were partitioned into two risk groups. (B) Box blot graph representing low- and high-risk groups for MAGL in BC patients. Using the mean expression level of MAGL mRNA, the samples were partitioned into two risk groups. (C) Kaplan-Meier survival curves of BC patients for GPD1. The group with higher GPD1 gene expression levels received a poor prognosis compared to the low-risk group with lower GPD1 levels (log-rank 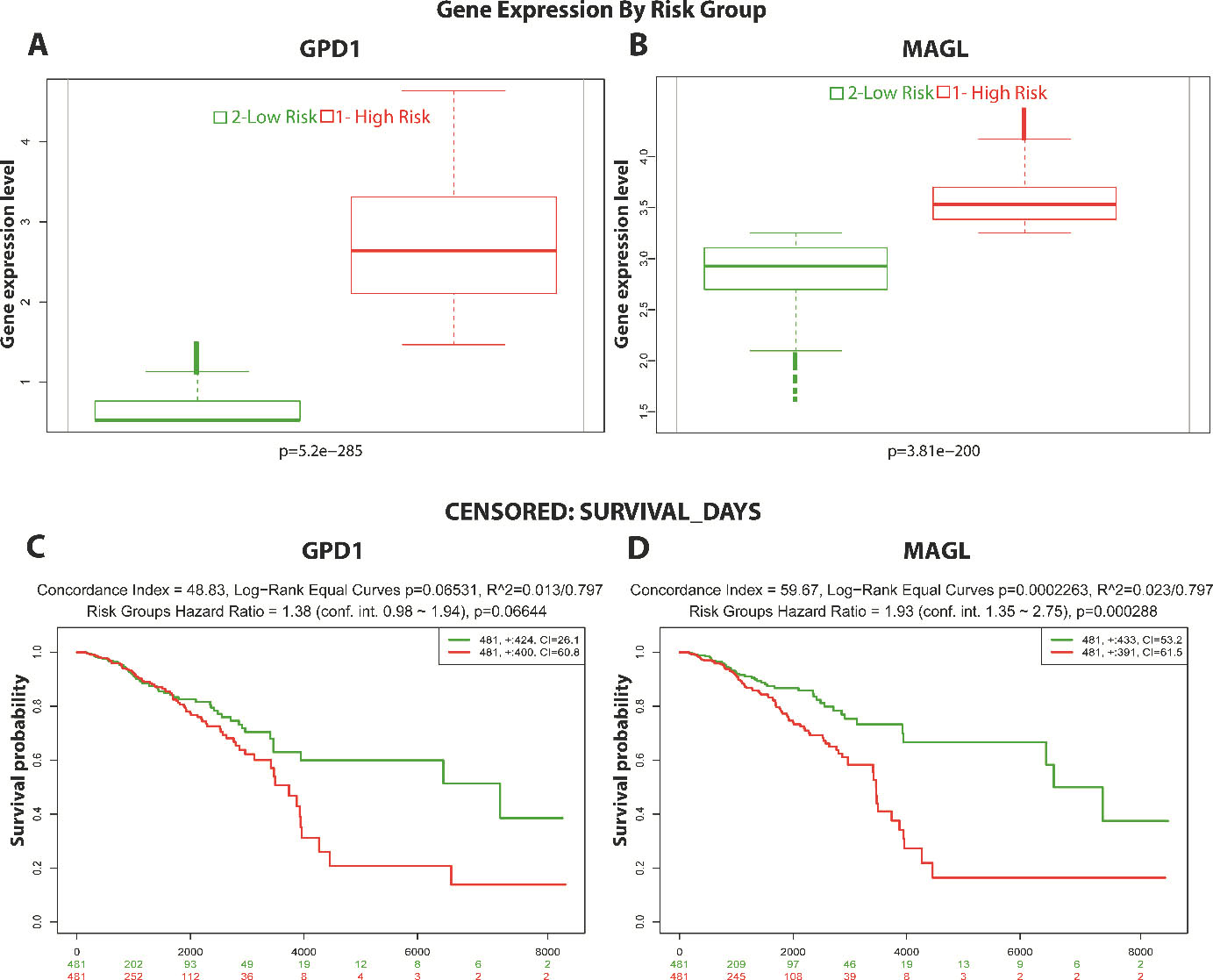
To assess the value of the serum MAGL levels as a diagnostic biomarker in differentiating healthy individuals from the BC patients, a ROC curve was created and the AUC was analysed (Fig. 4A). The ROC analysis indicated an AUC that corresponded to test the accuracy of 0.980 (95% CI: 0.968–0.992,
Elevated GPD1 and MAGL mRNA levels were correlated with the poor prognosis of BC
To further explore the prognostic performances of GPD1 and MAGL in BC, the outcomes of the patients were analysed through the Kaplan-Meier survival estimator (Fig. 5). GPD1 and MAGL mRNA data retrieved from TCGA was used to partition the samples into two risk groups (Fig. 5A and B). The group with higher GPD1 gene expression levels received a poor prognosis compared to the low-risk group with lower GPD1 levels (log-rank
Discussion
BC is a disease with serious consequences and may result in death. The only way to escape from this disorder is to diagnose it at an early stage so that necessary precautions can be taken to prevent its spread. However, early detection of BC is not an easy task since it requires a physical examination of the person. In most cases, highly sophisticated instrumentation like the mammogram and MRI is also required. In cases where a suspicious lump is detected in the breast, then a biopsy sample is to be taken for a definite diagnosis. Unfortunately, the presently used screening methods for the diagnosis of BC are cumbersome and not practical [11, 12]. A more practical way of screening BC is to use serum biomarkers. However, the currently available serum biomarkers are not that helpful for the diagnosis of BC since they have low specificity [13]. For example, one of the most commonly used biomarkers, CEA, has a sensitivity of 95% with 18% specificity [14]. Similarly, CA 15-3, the soluble form of MUC-1 protein and widely used serum marker for BC, has a sensitivity of 95% with 32% specificity [14].
It has been long known that cancer cells can reprogram their central carbon metabolism and maintain an elevated level of glycolysis to meet their high energy needs [15, 16]. In addition to the changes in carbon metabolism, another aspect of metabolic reprogramming in cancer cells is aberrant lipogenesis [17]. The dysregulation of core enzymes in lipid metabolism and their contribution to cancer cell migration, invasion, and metastasis has been well established in the literature [18, 19]. The genes involved in cellular fatty acid uptake and de novo lipogenesis are amplified in metastatic tumours and their expressions have been associated with the aggressive properties of malignant cancers [20, 21]. In our previous work, we identified two differentially regulated proteins, namely GPD1 and MAGL, and established an association between lipid metabolism and BC. These two proteins were down-regulated in tumour tissues of BC subtypes in comparison to the control group, and the lowest tissue expression was in the TNBC subtype.
GPD1 is a member of the NAD
MAGL is an enzyme responsible for the breakdown of monoacylglycerols to free fatty acids (FFA) and is considered to be the key enzyme in the regulation of the FFAs network [26]. Its expression was stated to be regulated in numerous aggressive tumours such as colorectal cancer, neuroblastoma, and nasopharyngeal carcinoma [27, 28, 29]. It is reported in the literature that cancer cells use de novo fatty acid synthesis rather than hydrolysis of neutral lipid stores [21, 30, 31, 32, 33]. However, there is also a compelling study stating that cancer cells use MAGL-dependent hydrolysis of monoacylglycerols as the major source of FFAs in aggressive malignant cells [34]. The current knowledge about the function of MAGL is not sufficient to explain this contradiction.
In this study, a comparative analysis of the serum GPD1 and MAGL levels was performed among the BC subtypes. The results indicated significantly low serum concentrations of both proteins in the TNBC subtype compared with the other BC subtypes. These results were consistent with our previous tissue-based mass spectrometry results [8]. GPD1 serum expression levels less than 3.18 ng/mL could be used to diagnose TNBC with a sensitivity of 95% and a specificity of 77.5%. The ELISA results suggested that serum GPD1 levels in BC patients had a higher diagnostic value than those used in clinics today. These results are also consistent with transcriptome data from TCGA. However, monitoring serum GPD1 levels was more informative than mRNA levels in distinguishing TNBC patients. The prognostic value of GPD1 in BC was determined via Kaplan-Meier survival analysis using mRNA expression data. Patients with higher GPD1 gene expression levels had a worse prognosis than the low-risk group with lower GPD1 levels. Although GPD1 had a high value as a diagnostic biomarker for BC, its prognostic value was not the same.
Comparative analysis of serum MAGL levels among BC subtypes and healthy subjects revealed significantly higher MAGL levels in controls. When serum levels were compared with the MAGL mRNA levels, serum MAGL levels were found to be more informative in distinguishing healthy subjects from BC patients. Analysis of the ROC revealed that subjects with serum MAGL levels higher than
Conclusion
The quest to find a biomarker for early detection of BC continues. The biomarkers in use today are either not specific or do not allow early detection of BC. The classical radiological scans and the other clinical approaches do not appear to be powerful and convenient enough to allow early detection. In the late few years, novel biomarker candidates have emerged but they also suffered from sensitivity and specificity issues [35, 36]. Gene/protein signatures are proposed as biomarker panels but they stay short due to their high cost and availability. The work presented here analysed the serum expression profiles of GPD1 and MAGL proteins in BC subtypes compared to healthy controls. Statistical analysis of the ELISA results provided strong evidence that GPD1 acts as a diagnostic biomarker in distinguishing TNBC patients from other subtypes (sensitivity of 95% and specificity of 77.5%). Also, MAGL displayed value as a diagnostic biomarker in distinguishing healthy individuals from BC patients (sensitivity of 100% and specificity of 98.5%). The experimental findings directed us to evaluate the prognostic values of these two proteins at the transcriptome level by retrieving data from the TCGA database. The statistical analysis of transcriptomics data failed to support the evidence that GPD1 and MAGL hold prognostic values. However, our experimental data suggested that GPD1 and MAGL might be used as non-invasive biomarkers with high sensitivity and specificity for the diagnosis of BC.
Footnotes
Acknowledgments
This study was carried out in the Medical Biology Proteomics laboratory at Kocaeli University Medical School and supported by Kocaeli University, Scientific Research Unit under the grant number 2018/123. The funding body had no role in the design or execution of the study.
Author contributions
Conception: Murat Kasap, Kubra Karaosmanoglu yoneten.
Interpretation or analysis of data: Kubra Karaosmanoglu Yoneten, Kazim Yalcin Arga, Murat Kasap.
Preparation of the manuscript: Kubra Karaosmanoglu Yoneten, Murat Kasap.
Revision for important intellectual content: Gurler Akpinar, Kazim Yalcin Arga.
Supervision: Murat Kasap, Nihat Zafer Utkan.
