Abstract
BACKGROUND:
Resistance to PD-1 blocking agents is not uncommon, limiting their wide clinical success. Certain tumor-infiltrating immune cells (e.g., TILs/CTLs) have emerged as biomarkers of response, and absence of such immune cells contributes to resistance.
OBJECTIVE:
We deconvoluted the dynamic immune microenvironment in a mouse model of oral carcinogenesis for augmenting the resistance to PD-1 blocking agents by combination.
METHODS:
Bioinformatics methods and routine biological experiments were adopted such as morphological analysis and ELISA in the 4NQO-treated mice model.
RESULTS:
Our findings revealed that dysplastic tongue tissues from 4NQO-treated mice were characterized by an immunosuppressive tumor microenvironment. Tongue tissues from mice treated with 4NQO for 12 weeks had higher levels of Th2 cells and Tregs compared to tissues taken from control mice or mice treated with 4NQO for 28 weeks; these results suggested a potential therapeutic benefit of anti-PD-1 in the oral cancer. The IL-17 pathway was significantly upregulated during progression from normal mucosa to hyperplasia and tumor formation in mice. Inhibition of IL-17
CONCLUSIONS:
Our data suggested a strong rationale of IL-17
Keywords
Abbreviations
Introduction
Cancer immunotherapies targeting immune checkpoint molecules, such as the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), programmed cell death protein 1 (PD-1), and programmed death-ligand 1 (PD-L1), have emerged as a promising therapeutic approach in several advanced cancers [1]. The blockade of immune checkpoint signaling pathways re-activates tumor-reactive T cells, leading to anti-tumor immune responses in the tumor microenvironment and thereby inhibiting tumor growth [2, 3].
Oral squamous cell carcinoma (OSCC) is an aggressive malignant neoplasm, with an overall survival rate of 50% that has remained unchanged for decades [4]. It has been previously shown that blockade of immune checkpoint receptors in mouse OSCC models is an effective cancer prevention strategy that reverses the carcinogenesis process prior to malignant transformation by blockade of immune checkpoint receptors in mouse model [5, 6].
However, primary resistance to anti-PD-1 therapy is not only observed in human tumors, resulting in tumor relapse and poor prognosis [7, 8, 9, 10], but also in precancerous lesions during progression to OSCC in mouse models [11, 12]. For example, the non-response rate was reported to be 30.43% in a 4NQO-induced mouse oral squamous carcinoma model after administration of anti-PD-1 antibodies, and this was associated with increased levels of regulatory T cells (Treg), which promoted resistance of the anti-PD-1 therapy.
Monotherapies are unlikely to overcome the major mechanisms that impede antitumor immunity in patients because the induction, potency, and persistence of host immune responses reflect the complex interplay between the different immune-cell populations and the progressing tumor [8]. Indeed, the generation of durable and clinically effective antitumor immune responses is probably dependent on the successful reprogramming of several immune processes. Herein, we employed the 4-nitroquinoline-1-oxide (4NQO)-induced carcinogenesis mouse model to unravel the changing immune microenvironment in different pathological stages of oral cancer, as well as to identify key signaling pathways as therapeutic targets to reverse immunosuppression. We achieved this by performing morphological, biological, and bioinformatics analyses in our in vivo mouse model. Additionally, the therapeutic potential of targeted inhibition of identified therapeutic targets in combination with anti-PD-1 blockade was investigated in our mouse tongue cancer model. We hypothesized that the identification of key players of resistance to immune checkpoint blockade would bring us one step forward to overcoming resistance to single-agent PD-1 therapy, expanding the application scope of immune checkpoint blockade inhibitors (ICIs), and ultimately achieving improved response rates and prolonged survival in patients with oral cancer.
Materials and methods
Mouse specimens
The animal protocol was approved by the Animal Care and Use Committee of the First Affiliated Hospital Harbin Medical University. BALB/c mice were used for morphological analysis, ELISA, and survival analysis. For bioinformatic analysis BALB/c mice were used in DATA1 and C57BL/6 mice were used in DATA2. 4NQO (Sigma-Aldrich) was administered to the mice in drinking water at the dose of 50 mg/L for 16 weeks, as described elsewhere [13]. The mice were randomly allocated into control IgG2a group, PBS group, anti-PD-1-treated group, anti-IL-17
Tissue dissection and sectioning for macroscopic and microcosmic analysis
To harvest oral tissue from above four groups, the mice were sacrificed. The tongues were excised and macroscopic lesions were counted after death. Quantified macroscopic lesions comprised those lesions that were visible, with a diameter of
Histological staining
Sections of tongues from 4NQO-treated mice were subjected to H&E to identify histologic changes associated with oral tumorigenesis, including normal mucosa in untreated mice tongue, and hyperplasia, dysplasia, and tumor. Formalin-fixed, paraffin-embedded tongue tissues were sectioned (4
Computational Immunophenotyping
The total immune infiltrates in tongue samples from 4NQO-treated mice were analyzed. The deconvolution was achieved using CIBERSORT Jar version 1.05 (
Single-sample gene set enrichment analysis (ssGSEA)
Single-sample GSEA, a modified analysis of standard GSEA, was performed on RNA measurements for each sample using the GSVA package in R version 3.6.1. In order to identify significantly up and downregulated KEGG pathways, the
ELISA
IL-17
Statistical analysis
All comparisons of the differences between two groups were performed using Student’s
Dynamic immune infiltration status during oral tumorigenesis in the 4NQO mouse model (A) Histologic changes during oral carcinogenesis in the mouse model a. Normal mucosa; b. Hyperplasia; c. Mild dysplasia; d. Moderate dysplasia; e. Severe dysplasia (carcinoma in situ); f. Invasive carcinoma. (B–C) Fractions of immune cell subsets in tongue tissue of BALB/c and C57BL/6 mice treated with 4NQO inferred from gene-expression data using CIBERSORT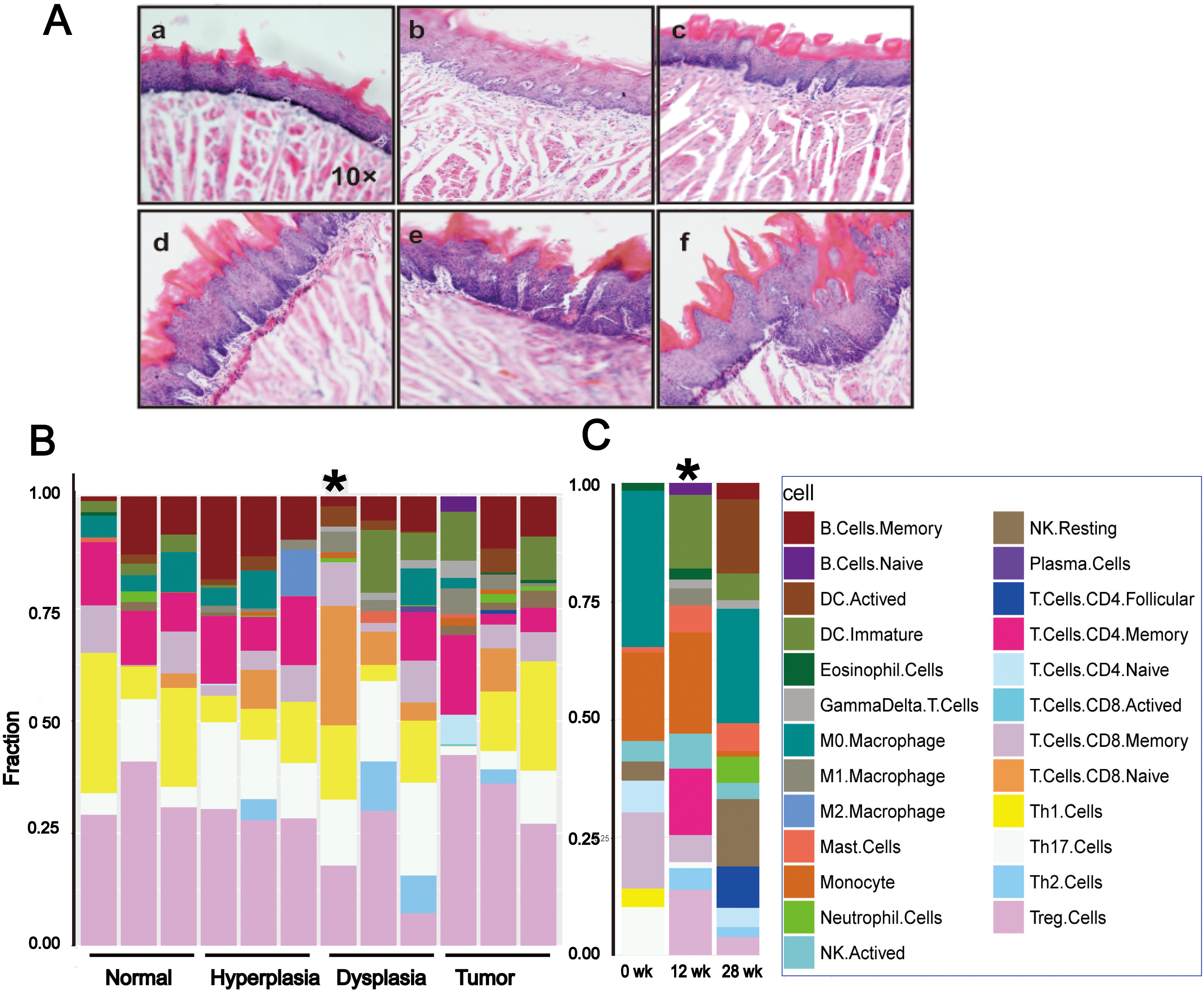
General information
We performed data mining of two publicly available RNA sequencing datasets acquired from Gene Expression Omnibus, GSE75421 and GSE101469, which for simplicity reasons will henceforth be referred to as Data 1 and Data 2, respectively, in this study. In Data 1, nine BALB/c mice were treated with 4NQO, which was added in drinking water at the dose of 100
At the same time we established a 4NQO-induced carcinogenesis model in immunocompetent BALB/c mice to assess the therapeutic effect of targeted agents combined with immune checkpoint inhibitor anti-PD-1 Histologic changes during oral carcinogenesis in our BALB/c mouse model are shown in Fig. 1A.
Representative images of immunofluorescence staining of dysplasia tissues obtained from mice treated with 4NQO for 12 weeks. (A) Tregs (CD4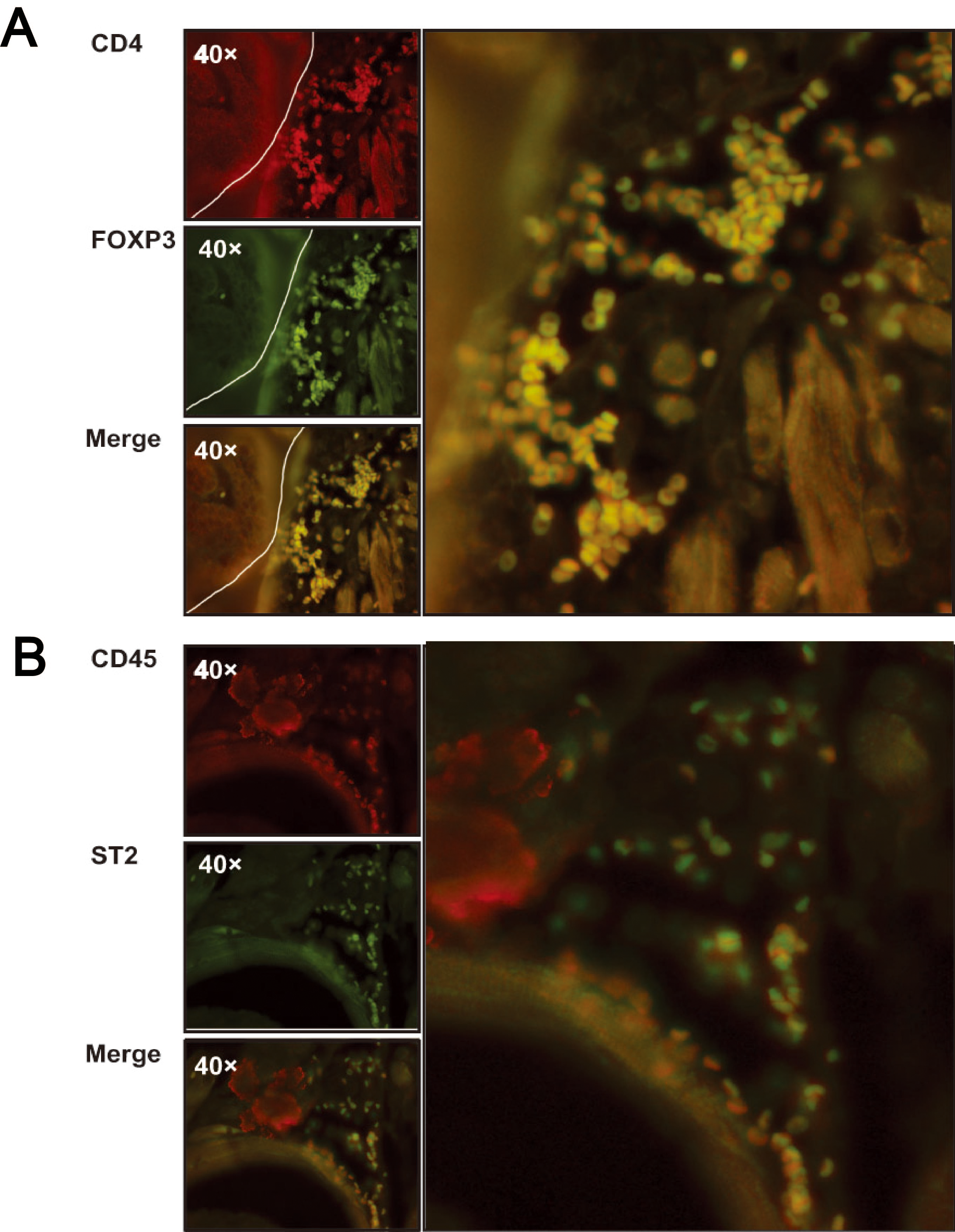
To get more insight into the changes in immune infiltration levels by different immune cell subsets during oral carcinogenesis progression from normal tongue tissue to hyperplasia, dysplasia, and tumor, we performed computational analysis of the immune phenotype in gene expression data using CIBERSORT [13]. This analysis suggested higher levels of infiltration by Th2 cells in dysplastic samples compared to control and tumor samples and that dysplastic lesions were significantly more infiltrated by CD8+ T cells (Fig. 1B). The larger immune cell components corresponded to memory CD8+ T cells, Th17 cells, Tregs, and Th2 cells at 12 weeks than at 28 weeks and 0 weeks, although this reached statistical significance only for dysplastic lesions in BALB/c mice and at 12 weeks in C57BL/6 mice treated with 4NQO (Fig. 1C, Tables S1 and S2).
Following this analysis, samples from our BALB/c mouse model were subjected to immunofluorescence staining for T cell markers, including CD4, FOXP3, CD45, and ST2. Consistent with the virtual immunophenotyping analysis based on transcriptomics data, three BALB/C mice treated with 4NQO for 12 weeks were found to show high infiltration of Th2 cell and Treg cells in dysplastic and hyperplastic lesions in tongue mucosa (Fig. 2A and 2B) and normal mice were not sacrificed as control samples due to little inflammatory reaction in normal mucosa.
Changes in cytokine expression in the tumor microenvironment during 4NQO-induced oral cancer initiation and progression
Considering the relevance of tumor-associated inflammatory response in mice treated with 4NQO for 12 and 28 weeks, we also assessed for expression of interleukins, CC chemokines and their receptors, and inflammatory markers. Interestingly, we found an increase in IL-17
Expression of interleukins, CC chemokines, and their receptors after 4NQO treatment for 0, 12, and 28 weeks. The expression was compared between 0 and 12 weeks (in pink), 12 and 28 weeks (in green), and 0 and 28 weeks (in blue). Results were expressed as fold change relative to the control group. (A) Expression of interleukins. (B–C) Expression of CC chemokines and their receptors. 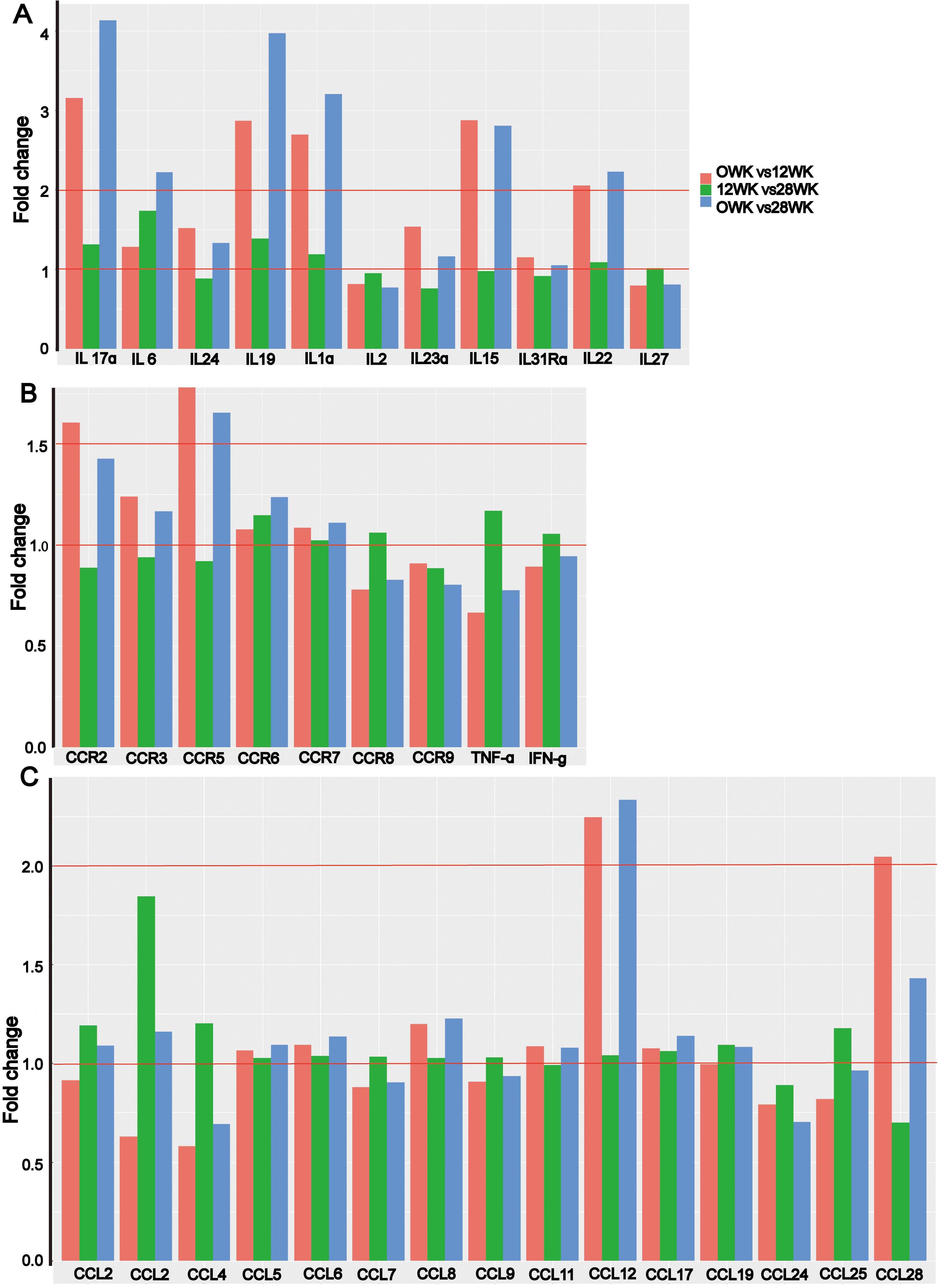
In order to assess the dynamics of immunerelated pathway expression in different tongue lesions and different durations of 4NQO treatment, we performed gene set enrichment analysis in Data 1 and Data 2 using single sample GSEA (ssGSEA). In Data 1, we identified complement and coagulation, as well as IL-17 signaling pathways to be highly associated with the immune state and were strengthened upregulated during the progression from normal mucosa to hyperplasia (Fig. 4A, Table S3 and Fig. S1). However, the platelet activation pathway was downregulated in tumors compared to dysplastic lesions (Fig. 4B, Table S3 and Fig. S2A). We also identified five pathways that were significantly differentially expressed between normal and tumor tissues: leukocyte transendothelial migration and platelet activation pathways were downregulated (
Screening for deregulated immune-related pathways depending on histological variation by single-sample GSEA (ssGSEA). Y axis represents the of ssGSEA score of various pathologic stages. (A) IL-17 signaling pathway, as well as complement and coagulation cascades were upregulated in hyperplasia compared to normal tissue (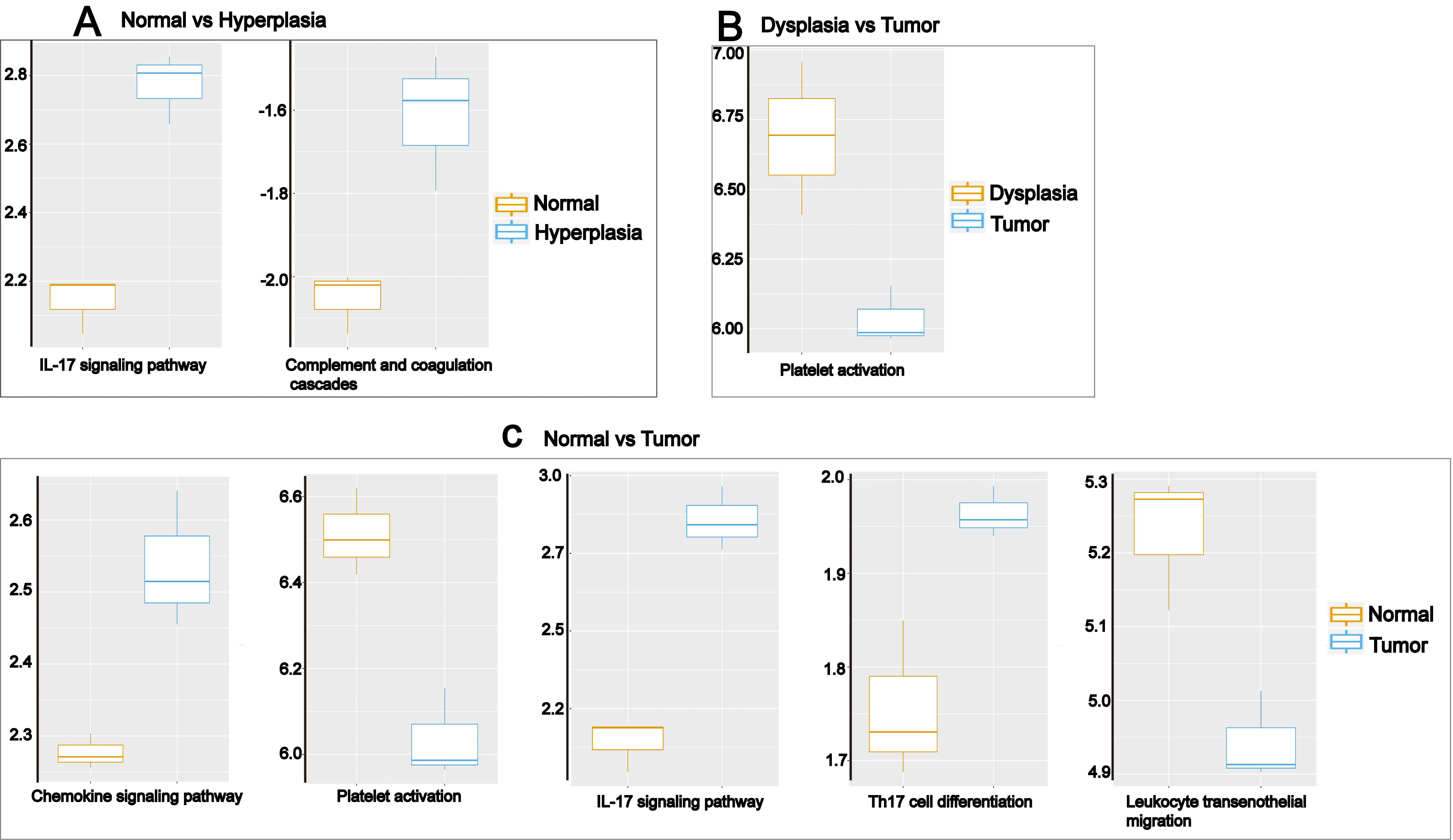
IL-1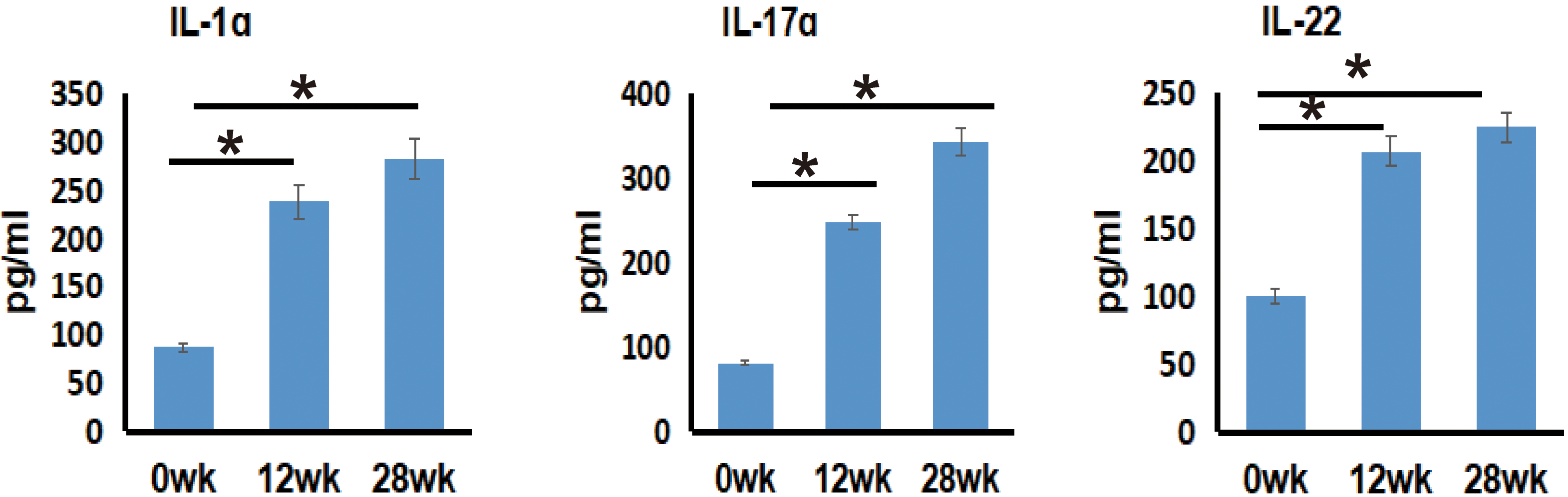
In Data 2, we found natural killer-mediated cytotoxicity, Fc epsilon RI, Toll-like receptor, and Antigen processing and presentation signaling pathways to be overexpressed (fold change
Considering that the IL-17 signaling pathway was significantly upregulated during progression from normal mucosa to hyperplasia and tumor in BALB/c mice and that it was bioinformatically identified to be expressed at higher levels in mice treated with 4NQO for 28 weeks compared to 12 or 0 weeks, we reasoned to evaluate the expression of IL-17
To morphologically assess the therapeutic potential of PD-1 immune checkpoint blockade combined with anti-IL-17
The mice were randomly allocated into treatment and control groups (11 mice per group). Mice were injected with anti-PD-1 antibody (200
PD-1, combined with IL-17 blockade prevents oral cancer development. (A) Quantification of the macroscopic lesions observed in control IgG2a, PBS, anti-PD-1, anti-IL-17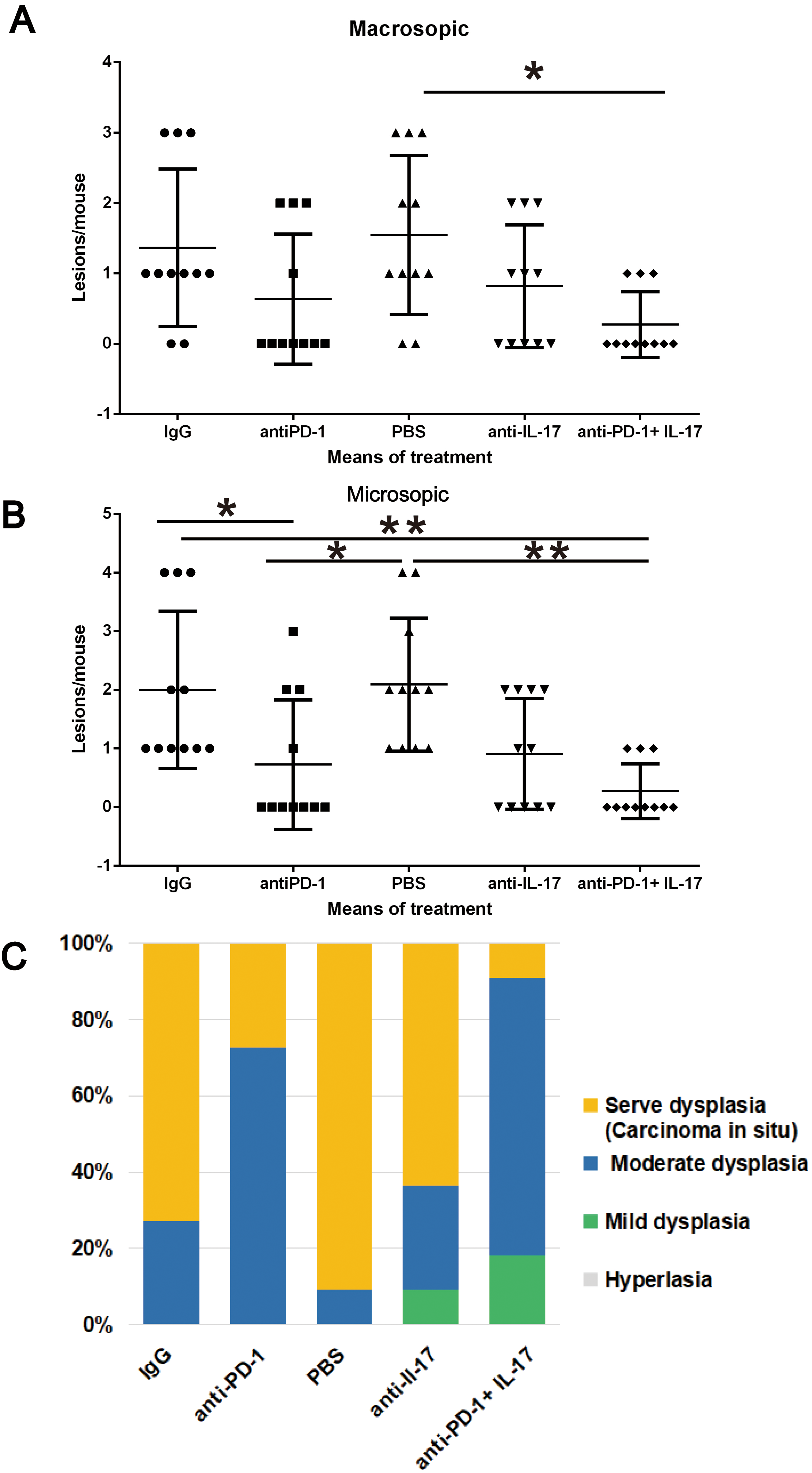
We found that only when anti-PD-1 was combined with anti-IL-17
More specifically, 72.72% (8/11) of the mice treated with IgG2 antibody and 90.09% (10/11) of those injected with PBS were found to have high-grade lesions (severe dysplasia or carcinoma), whereas only 9.09% (1/11) of the mice that had received anti-PD-1 in combination with anti-IL-17
Combination of anti-PD-1 and anti-IL-17 resulted in prolonged survival in BALB/c mice treated with 4NQO. BALB/c mice were treated with 4NQO for 16 weeks. The mice were administrated a total of four doses, at one-week intervals with PBS, control IgG2a, anti-IL-17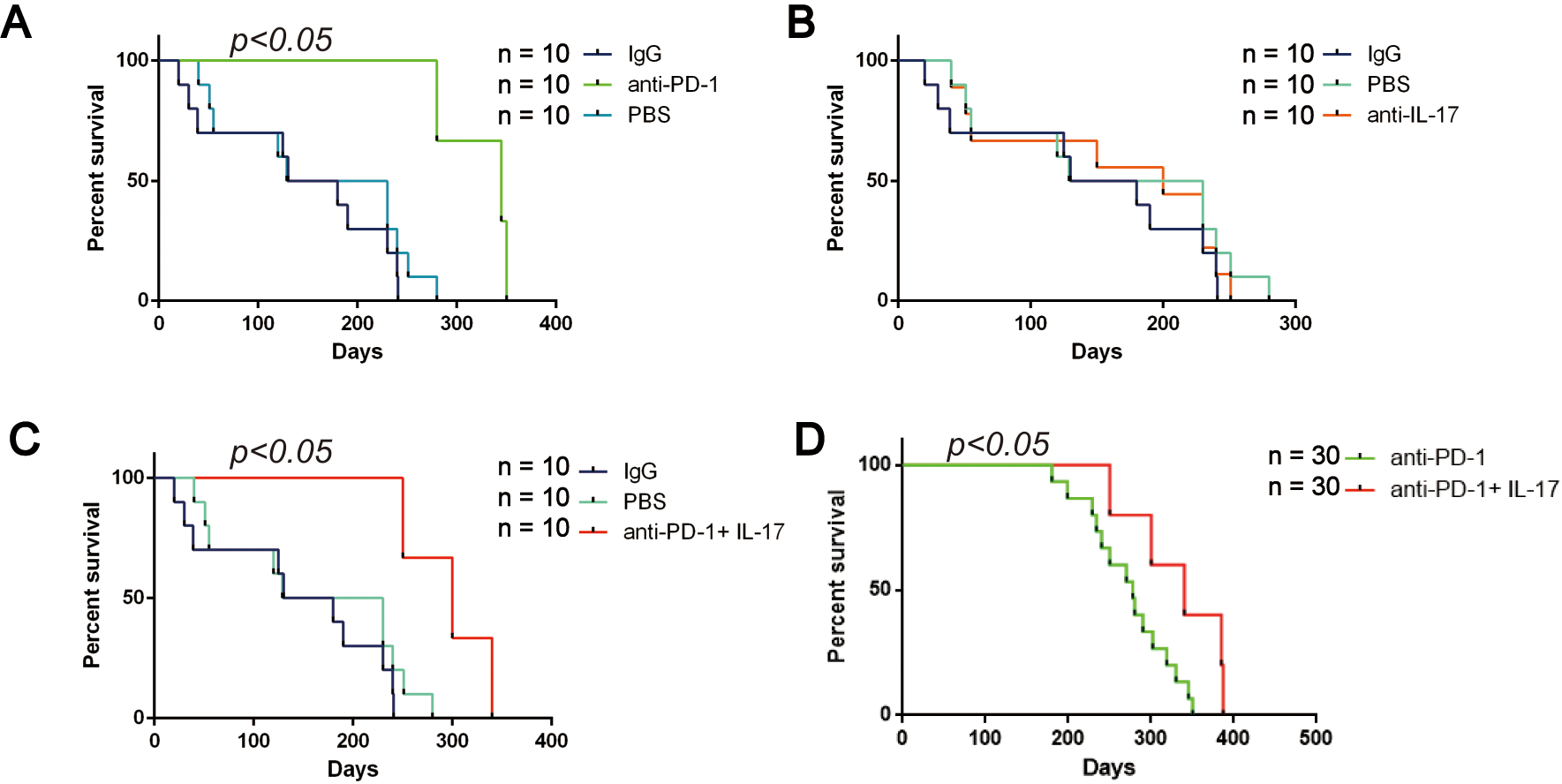
For survival analysis, the mice were randomly allocated into treatment and control groups (10 mice per group). Treatments were carried out as described earlier. As shown in Fig. 7A, mice treated with anti-PD-1 had a favorable survival compared to mice treated with PBS or IgG2a control (
Discussion
In this study, we dissected the dynamic immune microenvironment in the 4NQO-induced mouse model of oral tumorigenesis using BALB/c and C57BL/6 mice. We found evidence of immune surveillance in the precancerous lesions (mild dysplasia and hyperplasia) of C57BL/6 mice treated with 4NQO for 12 weeks. Therefore, we treated mice having precancerous oral lesions and early-stage oral cancer with immune checkpoint inhibitors (ICIs). However, previous reports demonstrated a 33% non-response rate in mice bearing 4NQO-induced oral squamous carcinomas treated with anti-PD-1 therapy [12]. In addition, Levingston and his colleagues found an initial response to PD-1 treatment at early stages; however, lesions continued to progress when anti-PD-1 was administered at later stages [18].
To identify novel resistance mechanisms to anti-PD-1 at the early stages of tongue carcinogenesis, we analyzed changes in immune-related pathways during tongue carcinogenesis in BALB/c mice and and point-in-time of carcinogenesis in C57BL/6 respectively according to pathological classification and 4NQO exposure duration. We identified IL-17 signaling pathway to be a key pathway involved in the carcinogenesis, regardless of the stage in the pathologic process (tumor initiation, or progression from early stages to more advanced stages) or the period of exposure to the carcinogenic agent. In DATA 1, IL-17 signaling pathway was up-regulated during the transformation from normal tongue to hyperplasia and from normal tongue to tumor. Consistently, in DATA 2, the IL-17 signaling pathway was elevated in mice treated with 4NQO for varying periods. IL-17 is the founding member of a novel family of inflammatory cytokines. While the pro-inflammatory properties of IL-17 are key for its protective function, unrestrained IL-17 signaling is associated with immunopathology, autoimmune diseases, and cancer progression.
Interestingly, increased cytokine levels have been associated with resistance to targeted therapies [19, 20]. High IL-17 levels were associated with resistance to VEGF inhibitors [21]. Akbay et al. also suggested IL-17 to be a key molecule suppressing T cells and thereby promoting resistance to PD-1 immune checkpoint blockade [22]. Another research found that IL-17 could activate monocytes to express B7-H1 in a dose-dependent manner. These IL-17-exposed monocytes suppressed cytotoxic T cell immunity, which expressed high levels of the B7-H1 receptor programmed death 1 (PD-1) and exhibited an exhausted phenotype [23]. Therefore, IL-17-mediated immune tolerance should be considered as a therapeutic target for the design of effective immune-based anti-cancer therapies.
We demonstrated that targeting IL-17
Additionally, we believe that the ratio of Th17/Treg cells might be associated with the resistance anti-PD-1. It is certain that anti-IL-17
One major limitation of this study lies in that we only investigated the role of IL-17
Conclusions
Anti-PD-1 and PD-L1 are emerging as one of the routine methods of treating several cancers. Considering the prevalence of resistance to these therapies, research efforts aiming to identify combinational approaches are currently very active. In this study, we showed that IL-17
Author contributions
Conception: S-W were responsible for conceptualization
Interpretation or analysis of data: S-W, HX-F, XR-Y, F-L and Z-H collected, interpreted the animal data, were responsible for funding, and methodology of the manuscript. XR-Y analyzed bioinformation data
Preparation of the manuscript: S-W and HX-F wrote the manuscript
Revision for important intellectual content: S-W and E-Z were responsible for revision for important intellectual
Supervision: S-W were responsible for supervision
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-203092.
Footnotes
Acknowledgments
This work was supported by grants from Natural Science Foundation of Heilongjiang Province of China (No. YQ2019H020), and Foundation of First Affiliated Hospital of Harbin Medical University (No. 2019M20).
