Abstract
BACKGROUND:
Apoptosis inhibition is a major tumorigenic factor. Bcl-2 dysregulation and TP53 mutation status, which may correlate with autoantibody generation, contribute to impaired apoptosis.
OBJECTIVE:
This study aimed to investigate the prognostic value of circulating Bcl-2 and anti-p53 antibodies (p53Abs) in a 17.5-year follow-up of breast cancer patients. We also analyzed the correlations of Bcl-2 and p53Abs with various clinicopathological parameters in order to assess their impact on tumor aggressiveness.
METHODS:
Serum Bcl-2 and p53Abs levels were analyzed by the enzyme-linked immunosorbent assay (ELISA) in 82 patients with invasive breast cancer and twenty individuals without malignancy.
RESULTS:
Serum Bcl-2 and p53Abs levels in breast cancer patients were significantly higher than those in controls. Patients with high levels of Bcl-2 (cut-off 200 U/ml) had a poorer prognosis (17.5-year survival) than those with lower Bcl-2 values. In combined analysis the subgroup of patients with elevated p53Abs (cut-off 15 U/ml) and elevated Bcl-2 (cut-offs 124 U/ml and 200 U/ml) had the worse prognosis in 17.5-year survival. In correlation analysis p53Abs and Bcl-2 were associated with unfavorable clinicopathological parameters.
CONCLUSIONS:
Our results suggest that breast cancer patients with high serum levels of p53Abs and Bcl-2 present an especially unfavorable group in a long follow-up.
Introduction
Tumor growth is related to the imbalance between cell proliferation and cell death. Evading apoptosis is well recognized hallmark of cancer [1, 2]. Resistance to apoptosis appears to be critical factor for malignant potential. Cells that escape apoptosis accumulate regardless of their proliferative capacity.
Apoptosis or programed cell death may be mediated by membrane death receptors (the extrinsic pathway) or by changes in the mitochondrial permeabilization (intrinsic pathway) [3, 4]. The intrinsic pathway is regulated by B-cell lymphoma 2 (Bcl-2) family of proteins [4]. Balance between pro-apoptotic (e.g. Bax, Bak) and anti-apoptotic (e.g. Bcl-2, Puma, Noxa) factors plays a role in the cell destiny by affecting mitochondrial outer membrane permeabilization (MOMP). The Bcl-2 family has been named after its anti-apoptotic member Bcl-2, which exerts its effects by the binding and inhibition of pro-apoptotic proteins such as Bax and Bak and thus preventing MOMP [5]. The tumorigenic potential of Bcl-2 was first documented in hematologic malignancies [6]. Bcl-2 is also expressed in solid tumors, including breast cancer where it is associated with tumor growth [7, 8, 9]. In hematologic malignancies Bcl-2 was shown to have negative prognostic role [10, 11]. However, in most studies that analyzed Bcl-2 protein expression in breast cancer a seemingly contradictory effect was observed: a high Bcl-2 was associated with either favorable or poor outcomes [8, 12, 13, 14, 15]. Similarly, in a limited number of studies that aimed to evaluate the prognostic significance of serum Bcl-2 levels, variable results were obtained [16, 17, 18, 19].
p53 is a product of the tumor suppressor gene TP53. As a transcription factor it controls the cell cycle and the process of DNA repair [20]. The p53 response is in correlation with the intensity of cellular stress. In case of severe DNA damage p53 mediates apoptosis. The relationship between the mutation status of TP53 and the outcome of various cancers was analyzed in many studies and the association with poor survival was confirmed for breast cancer [21, 22]. Anti-p53 antibodies (p53Abs) are found in patients with various types of cancer, including breast cancer [21, 23]. Serum p53Abs were shown to be a negative prognostic factor in breast cancer [23, 24]. The most common type of TP53 mutation is missense mutation which leads to p53 stabilization and its accumulation in the cell. The accumulation of p53 is considered to be the most important factor in the generation of p53Abs [23]. One of the possible mechanisms of p53Abs generation is the escape of accumulated p53 from the nucleus to the cytoplasm and its presentation on the cell surface by MHC I molecules. The second mechanism is the necrosis of tumor cells with accumulated p53 and the release of p53 protein into the blood and lymphatics, inducing the immune response [24]. Autoantibodies are considered to be more sensitive biomarkers than the corresponding proteins because of their higher concentration and greater stability in the circulation [21]. p53 is implicated in the regulation of both the intrinsic and extrinsic apoptotic pathway [25]. p53 upregulates the transcription of pro-apoptotic genes such as Bax, Puma and Noxa and downregulates expression of anti-apoptotic genes such as Bcl-2 in some settings [25, 26]. Additionally, p53 may directly antagonize Bcl-2 function [26]. For this direct interaction, the DNA-binding domain of p53 is required [27].
Despite advances in diagnosis and treatment, breast cancer is still the second leading cause of cancer death among women [28].
Procedures less invasive than tumor biopsies, such as the identification of proteins and DNA released from the tumor in patient’s blood, have some advantages: they are easily available, feasible for follow-up and might overcome the potential problem of intratumor heterogeneity [29].
Providing that Bcl-2 and p53 represent the key factors regulating apoptosis, we designed this study to determine whether serum Bcl-2 and p53Abs correlate with the clinical outcome and long term survival of breast cancer patients (the follow-up period was 17.5 years). We also analyzed their relationship with various clinical, pathological and biochemical factors to assess a possible correlation with the tumor aggressiveness.
Patients and methods
Patients and samples
We analyzed 82 patients with breast cancer operated at the University Hospital Center Zagreb, Croatia. The specimens were obtained through routine surgery between 1999 and 2000. None of the patients received any preoperative treatment. They received therapy in accordance with national guidelines based on St Gallen’s recommendations. The study was approved by the Ethics Committee of the University of Zagreb Medical School, and the Ethics Committee of the University Hospital Center Zagreb, Zagreb, Croatia. The follow-up period was 210 months (17.5 years). The following clinicopathological parameters of each patient were analyzed: age, tumor size, histological tumor grade of the tumors, axillary lymph node metastases, the concentrations of estrogen and progesterone receptors in the cytosol, serum concentration of HER-2/neu extracellular domain (ECD), and HER-2/neu receptor levels in tumor tissue. Tumor size, nodal status and histological grade (Scarff-Bloom-Richardson) were determined by standard procedure at the Department of Pathology.
Blood samples
Blood samples from 82 patients and 20 women without malignancy were taken and allowed to clot. The samples were retrieved before operation. After centrifugation (1250 g for 10 min), the serum was removed and stored at
Analysis of serum p53Abs, HER-2/neu ECD and Bcl-2
Serum p53Abs levels were assessed by enzyme-linked immunosorbent assay (ELISA, kitOncogene research product, Cambridge). HER-2/neu ECD concentration was analyzed by ELISA (Oncogene research product, Cambridge). Bcl-2 concentration was evaluated by ELISA (Endogen, Woburnm MA). The tests were performed according to the manufacturer’s instructions. The receiver operating curve (ROC) analysis was performed to determine the cut-off values. The cut-off value was defined as 15 U/ml for the p53Abs (sensitivity 95%; specificity: 72%; area under the curve (AUC) 0.858), 15 U/ml for HER-2/neu ECD (sensitivity 100%; specificity: 64%; area under the curve (AUC) 0.772) and 124 U/ml for Bcl-2 (sensitivity 100%; specificity: 78%; area under the curve (AUC) 0.943). The quantifications of p53Abs, HER-2/neu ECD and Bcl-2 in serum were achieved by the construction of the standard curve according to the manufacturer’s instructions.
Steroid receptors
Breast cancer tissue was stored in liquid nitrogen and then pulverized in the frozen stage. The tissue powder was suspended in the buffer. The suspension was centrifuged at 800 g for 15 minutes at 4
Statistical analysis
The results were analyzed using the MedCalc statistical software. The distribution of serum levels of Bcl-2 and p53Abs for the group of breast cancer patients and the control group was shown using the Kruskal-Wallis test. The association of Bcl-2 and p53Abs levels with clinicopathological parameters was studied using Spearman’s correlation coefficient. Univariate and multivariate analyses were performed by Cox proportional-hazards regression test. Overall survival (OS) curves were calculated by the Kaplan-Meier method and compared by long-rank tests. A
Results
The clinicopathological and biochemical characteristics of the patients and their tumors are shown in Table 1.
Distribution of clinical and pathological data in a group of 82 patients with breast carcinoma
Distribution of clinical and pathological data in a group of 82 patients with breast carcinoma
Luminal
Box plots showing the distribution of Bcl-2 (A) and p53Abs (B) in serum of breast cancer patients (p; N 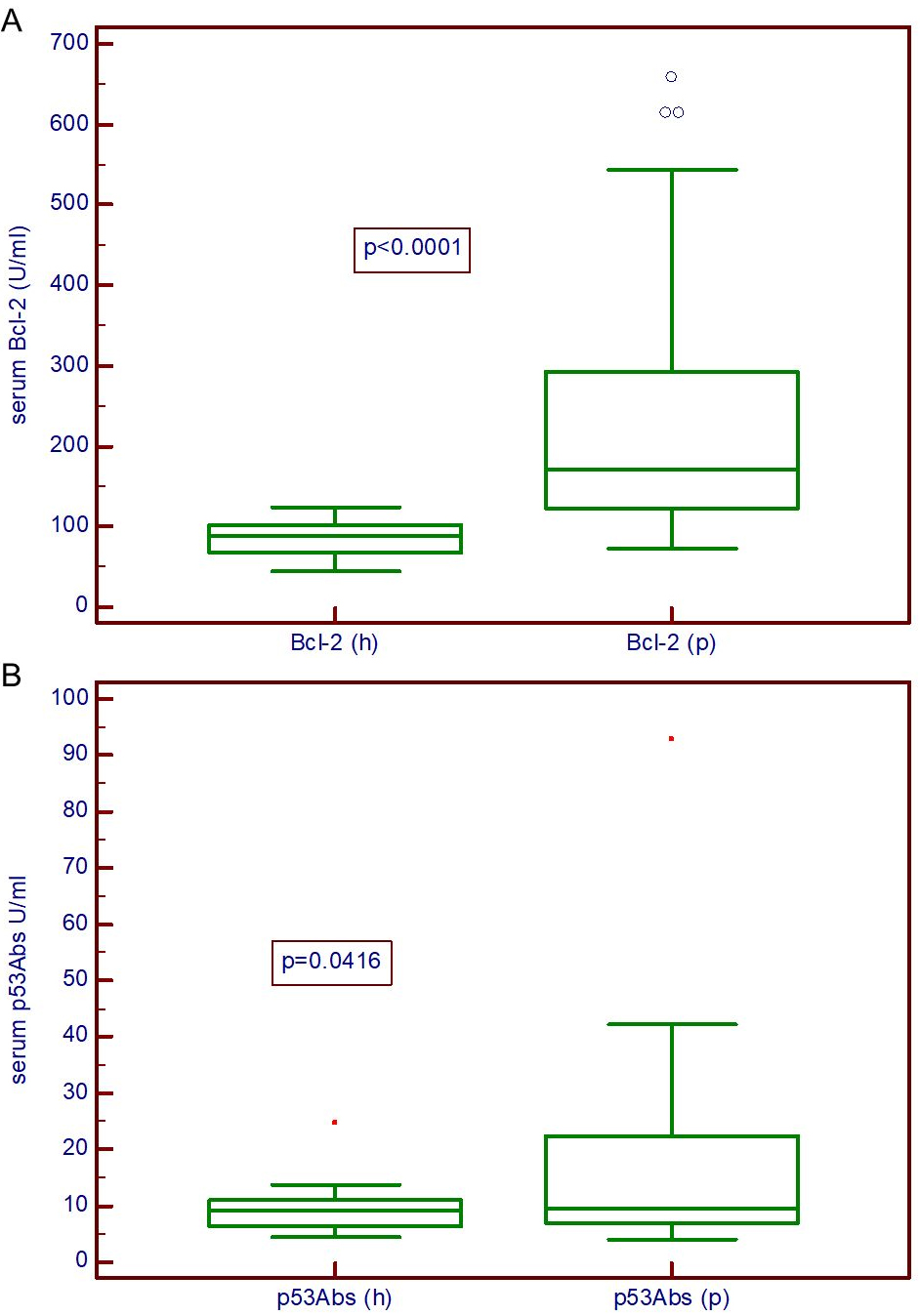
The serum concentrations of Bcl-2 (Fig. 1A) and p53Abs (Fig. 1B) of the patients with breast cancer were significantly higher than those in the healthy controls (
We found a statistically significant correlation between the serum concentrations of Bcl-2 and p53Abs in patients with breast cancer (Fig. 2).
Correlation between p53Abs and Bcl-2 concentrations in serum of breast cancer patients. The correlation between concentrations of Bcl-2 and of p53Abs is statistically significant. The association of Bcl-2 and p53 serum levels was studied using Spearman’s correlation coefficient.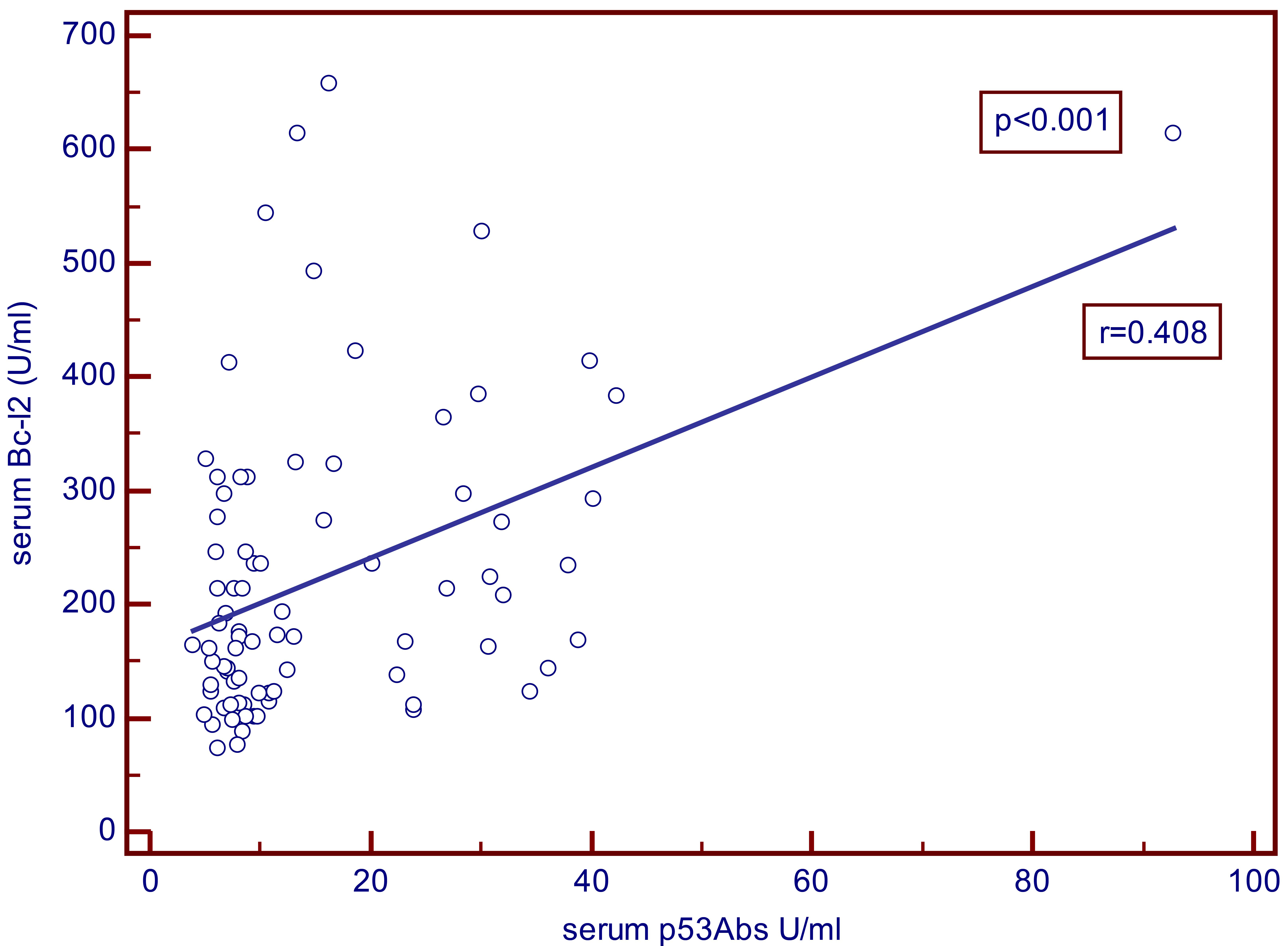
Spearman’s correlation among clinicopathological parameters
Statistically significant correlations are shown in bold (
As shown in Table 2, serum concentrations of p53Abs exhibited significant and positive correlation with tumor size (
According to univariate and multivariate Cox proportional-hazards regression analysis axillary lymph node metastases (
Prognostic significance of clinical and pathological parameters for overall survival determined by Cox proportional-hazards regression test
Significant correlations are shown in bold (
Prognostic significance of combination of different values of serum Bcl-2 and p53Abs for overall survival (17.5 years) determined by Cox proportional-hazards regression test
Combination of Bcl-2 (cut-off values 124 U/ml and 200 U/ml) with p53Abs (cut-off value 15 U/ml). Significant correlations are shown in bold (
The prognostic significance of the combination of different values of serum Bcl-2 and p53Abs for overall survival determined by the Cox proportional-hazards regression test are shown in Table 4. Only the combinations of higher values of Bcl-2 (cut-off values 124 U/ml and 200 U/ml) and p53 (cut-off value 15 U/ml) were significant in both: univariate and multivariate analysis.
Kaplan-Meier curves for overall survival for 17.5 years of breast cancer patients according to serum concentrations of Bcl-2 (cut-off 200 U/ml). The Kaplan-Meier analysis showed significantly shorter 17.5-year survival in patients with Bcl-2 values 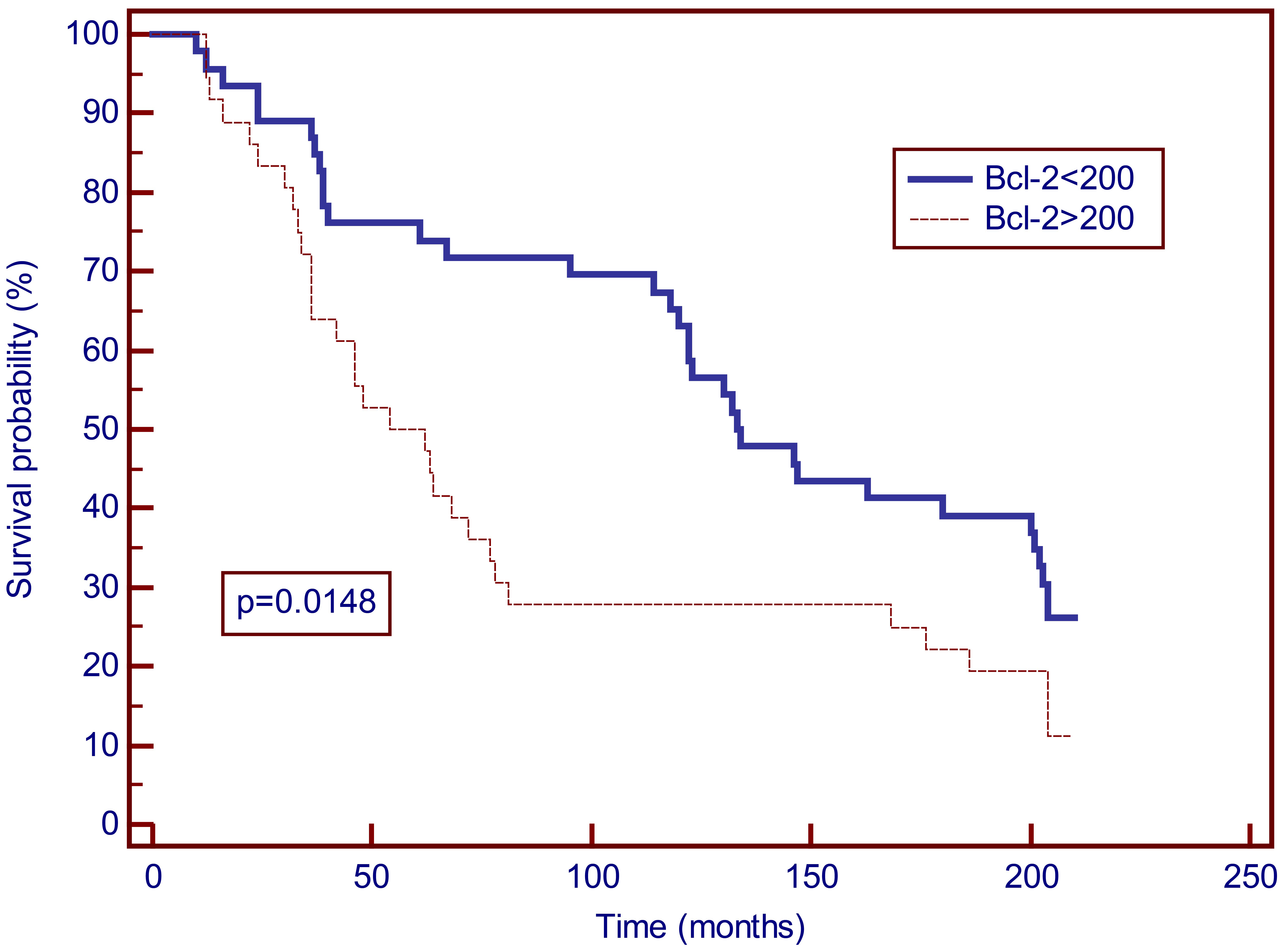
Kaplan-Meier curves for overall survival for 17.5 years of breast cancer patients according to serum concentrations of p53Abs (cut-off 15 U/ml). According to the Kaplan-Meier analysis the difference between the group of patients with p53Abs 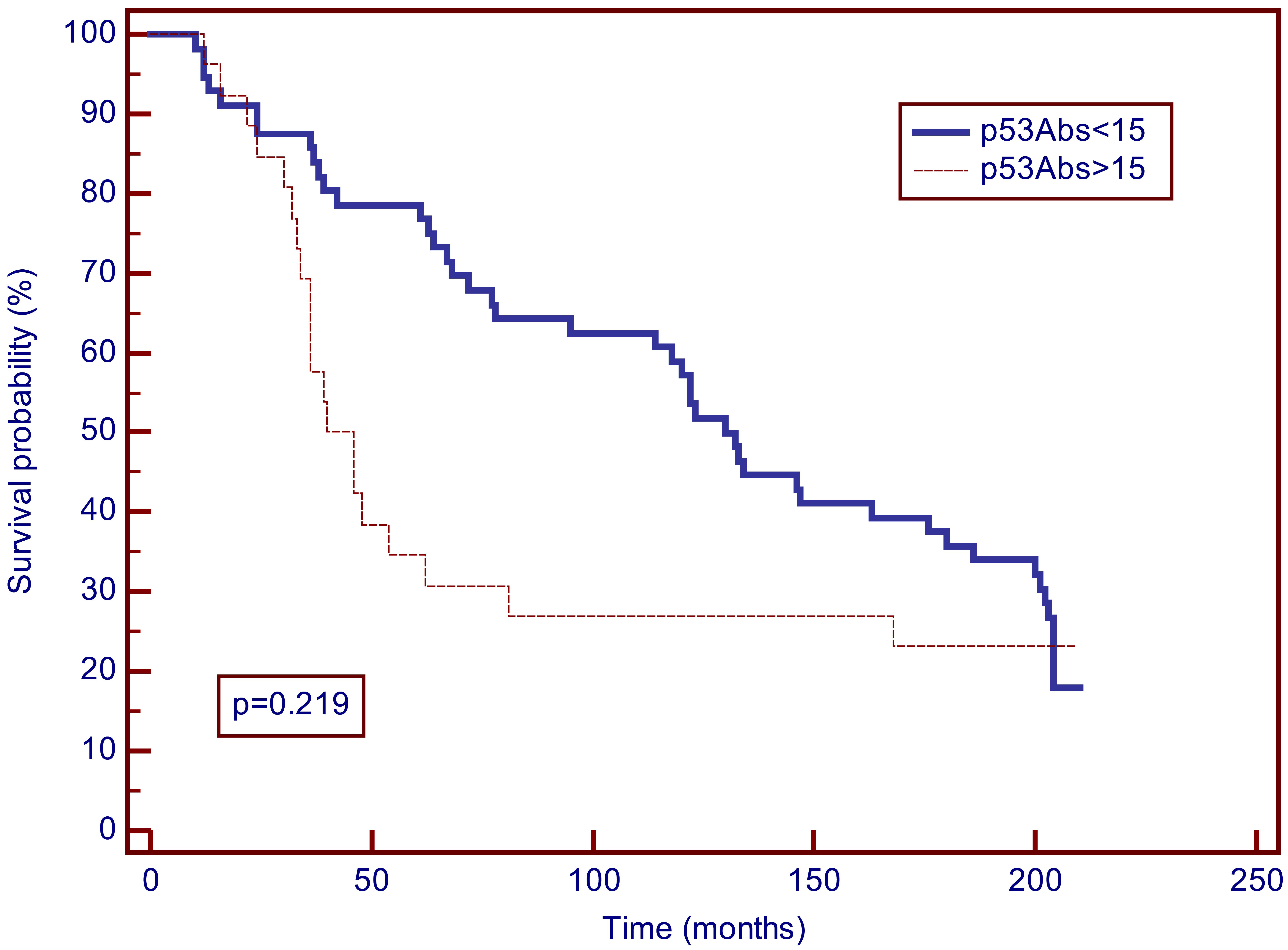
Kaplan-Meier curves for overall survival for 17.5 years of breast cancer patients according to serum concentrations of Bcl-2 (cut-off 124 U/ml) and p53Abs (cut-off 15 U/ml). According to the Kaplan-Meier analysis the group of breast cancer patients with Bcl-2 values 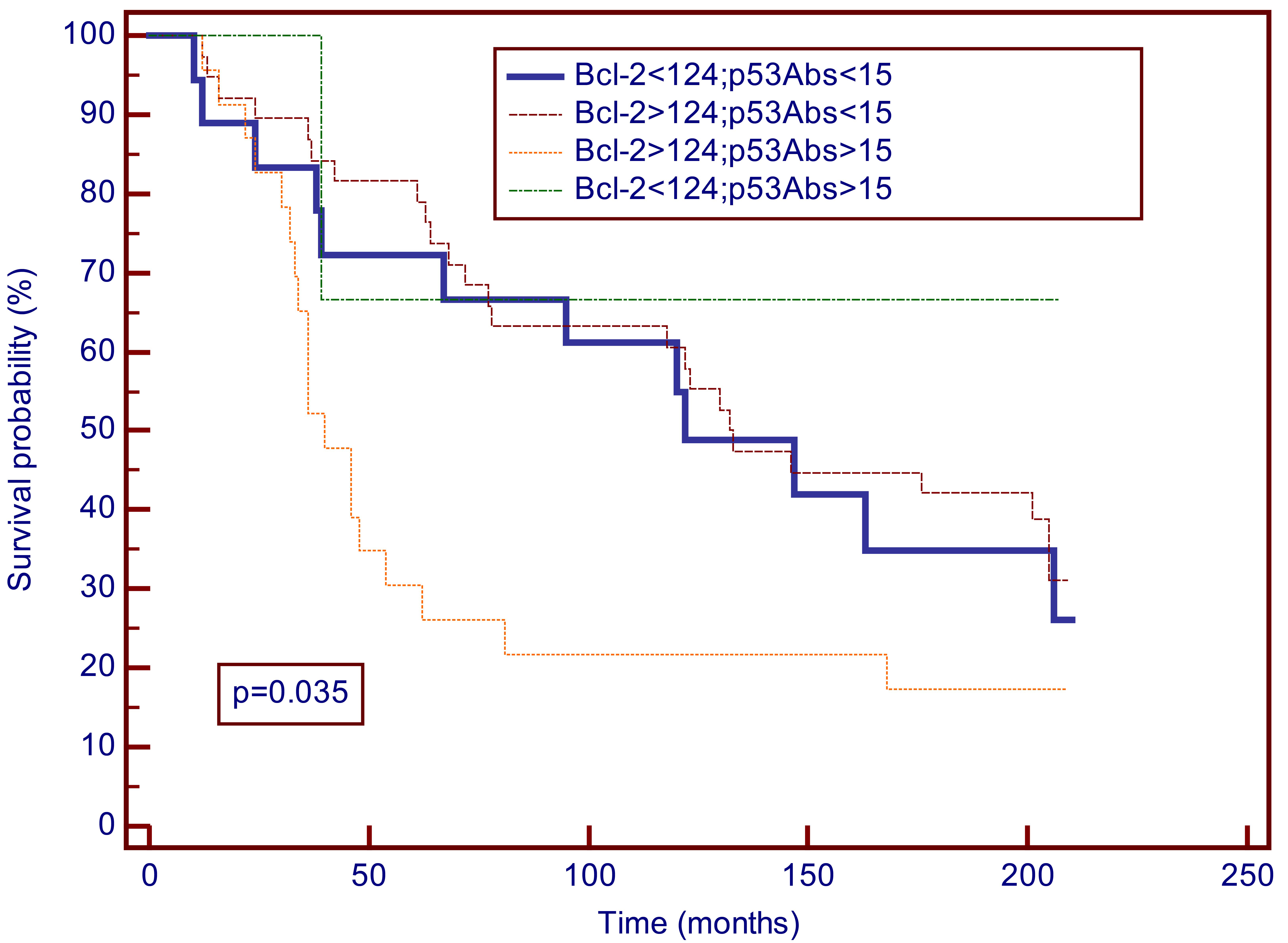
Kaplan-Meier curves for overall survival for 17.5 years of breast cancer patients according to serum concentrations of Bcl-2 (cut-off 200 U/ml) and p53Abs (cut-off 15 U/ml). According to the Kaplan-Meier analysis the group of breast cancer patients with Bcl-2 values 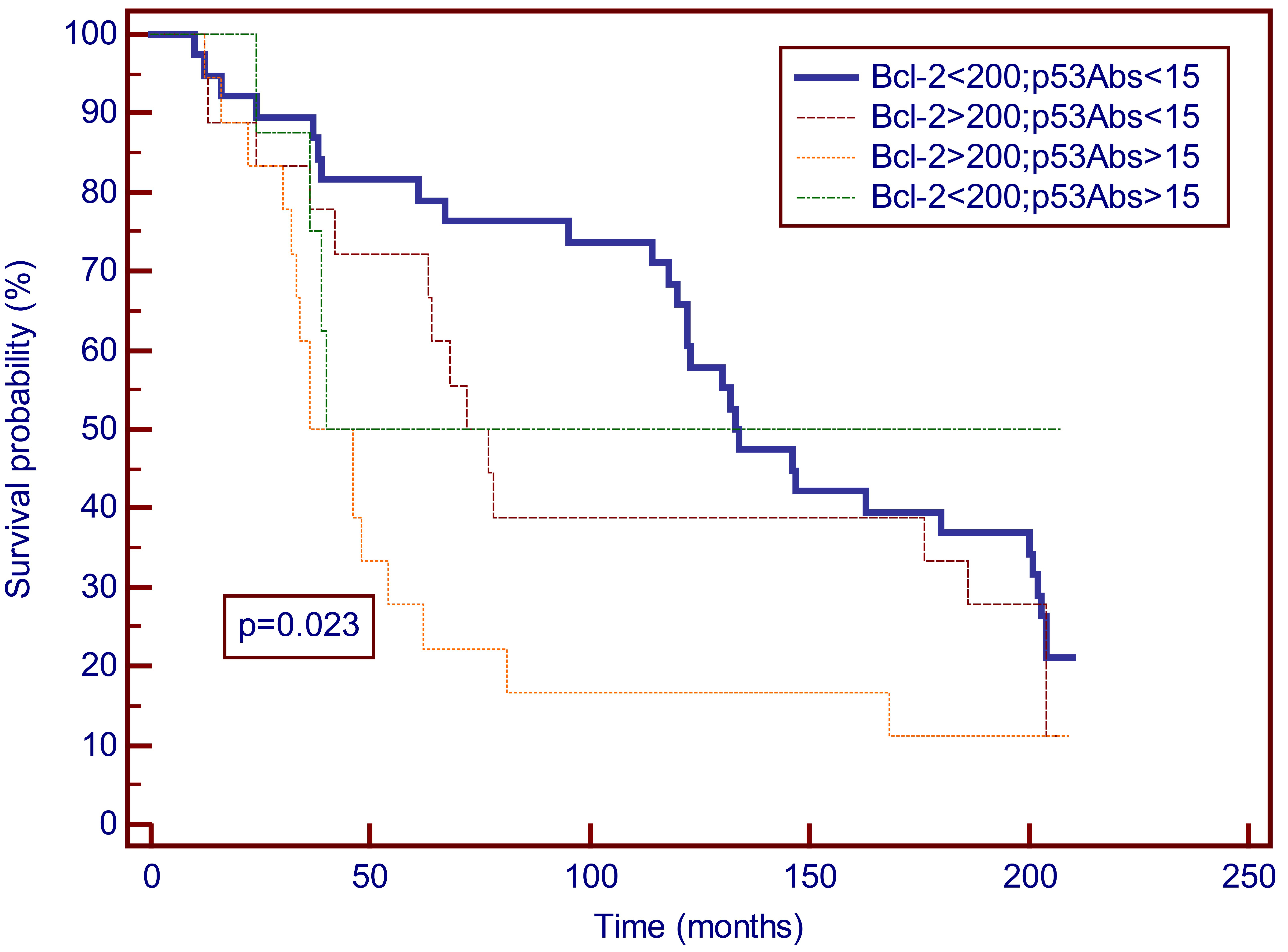
Kaplan-Meier curves for overall survival of breast cancer patients with negative estrogen (ER), progesterone (PR) and HER-2/neu receptors according to serum concentrations of Bcl-2 (cut-off 200 U/ml) and p53Abs (cut-off 15 U/ml). According to the Kaplan-Meier analysis the group of triple-negative breast cancer patients with Bcl-2 values 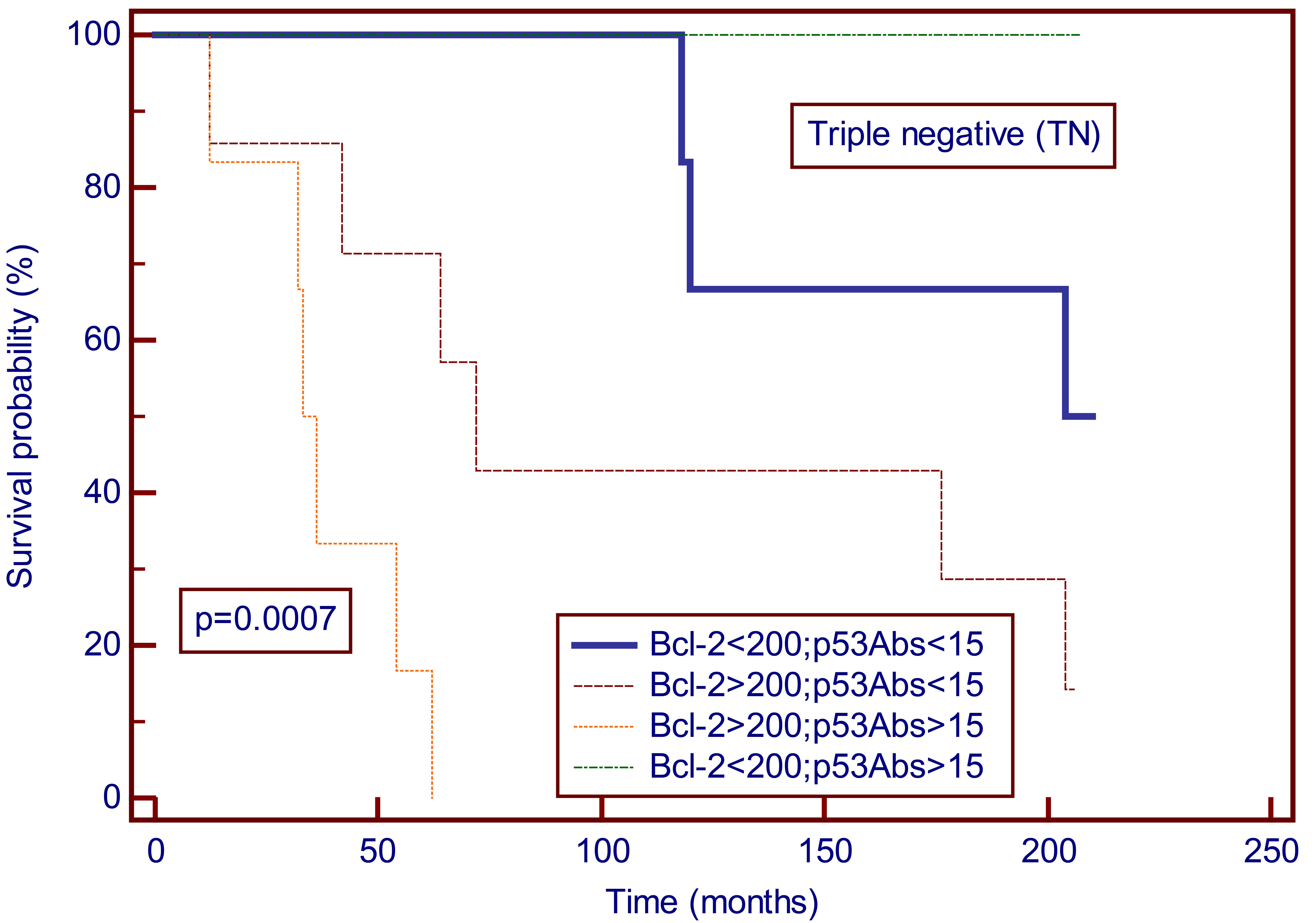
In Kaplan-Meier analysis for 17.5 years there was a significant difference in survival between breast cancer patients with Bcl-2 values above 200 U/ml and those with lower values (
In our study we found the diagnostic and prognostic values of circulating Bcl-2 and p53Abs. Circulating levels of p53Abs and Bcl-2 were significantly higher in the patient group compared with the control group. According to the Kaplan-Meier analysis high levels of Bcl-2 (higher than 200 U/ml) were associated with adverse outcomes (worse OS) in the 17.5-year follow-up.
When we analyzed combinations of p53Abs and Bcl-2, the subgroup of patients with high p53 and serum Bcl-2 values higher than 124 u/ml (estimated by ROC analysis) had a worse prognosis in the follow-up period. Furthermore, there was a statistically significant correlation between p53Abs and serum Bcl-2 suggesting a possible association and cumulative effect of these two parameters: both Bcl-2 and dysfunctional p53 (TP53 mutation is related to autoantibody generation) may have anti-apoptotic, pro-tumorigenic effects. In multivariate analysis p53Abs along with tumor size and serum HER-2/neu ECD were shown to be independent prognostic factors for 17.5-year survival.
The relation between p53Abs and a worse prognosis was found in many studies including our previous report [31, 32]. Mutated TP53 leads to its loss of function as a guardian of the genome and contributes to malignant phenotype [33, 34]. p53Abs reflect the status of mutant TP53 in the tumor and the negative prognostic impact is in accordance with the loss of function of p53 [21].
The relation between Bcl-2 and prognosis in breast cancer patients is more complex. In contrast to hematological malignancies Bcl-2 expression has been related to favorable prognosis in many studies [8]. A meta-analysis that included 17 studies showed Bcl-2 to be positive prognostic factor in breast cancer independent of classical prognostic parameters such as lymph node status, tumor size and tumor grade [12]. This paradox has been explained by a functional association between estrogen and Bcl-2: transcription of Bcl-2 is regulated by estrogen [8, 35]. Additionally, non-apoptotic functions of Bcl-2 may play a role. These functions include the regulation of mitochondria, endoplasmic reticulum, cell cycle, DNA damage response, cell metabolism and autophagy [36]. Furthermore, the balance between Bcl-2 protein and other members of the Bcl-2 family may be important. “Mitochondrial priming” may contribute to the sensitivity to endocrine therapy in hormone dependent tumors [8]. In a large prospective study that included more than 11000 patients Bcl-2 was found to be a favorable prognostic factor in all molecular subtypes of breast cancer [13].
However, studies that focused on TNBC obtained variable results. In the study of Tawfik et al. Bcl-2 positivity was associated with poorer survival of TNBC patients [14]. These results differ from those reported by Kallel-Bayoudh [37]. The discrepancies between these two studies may be related to differences in the defining criteria for defining triple negative status (threshold for ER and PR positivity) and in the cut-off values for Bcl-2 positivity (1% versus 5% positive cells). More recent studies in ER, PR negative tumors also obtained opposite results. Abdel Fatah et al. found Bcl-2 to be a positive prognostic factor in TNBC [38], while Ruibal et al. analyzed “hormone independent” tumors (ER negative, PR negative) and found a correlation of Bcl-2 expression with a worse clinical outcome [15]. In the study of Ozretic et al. Bcl-2 was an independent prognostic marker for decreased OS in patients with TNBC [39]. Bcl-2 was also reported to have a predictive value: Bcl-2 expression in primary breast cancer was shown to be associated with a benefit from CMF chemotherapy in comparison with the anthracycline-based protocol [40]. Additionally, in a study of basal-like TNBC treated with anthracycline-based adjuvant therapy Bcl-2 expression was related to poor outcome [41].
During malignant transformation transcriptional control, including estrogen signaling, may be changed. Taking into account that ER negative tumors may have different transcriptional control of Bcl-2 our results may be related to the fact that the proportion of ER negative tumors in our sample was higher than the proportion of ER positive tumors. When we analyzed the OS in our subgroup of TNBC patients (N
The follow-up period may also play a role. In the study of Bozovic-Spasojevic et al. Bcl-2 and p53 protein expression were significantly related to OS only in the first 5 years of follow-up [40]. Time dependent effect was also reported in the study of Callagy et al. [42].
In the studies in which Bcl-2 expression was correlated with ER and PR expression, an association with the favorable phenotype was also found: a positive correlation with smaller tumor size, lower histological grade, lower number of positive lymph nodes and absence of HER-2 overexpression, as well as an inverse correlation with proliferative markers such as Ki-67 [40, 43, 44].
In our study there was no correlation between ER positivity and serum values of p53Abs and Bcl-2. Both analyzed markers were associated with the unfavorable phenotype: we found a positive correlation with tumor size, tumor grade, age and lymph node involvement. The relationship with ER seems to be important for the association of Bcl-2 with other positive prognostic factors in breast cancer.
The number of circulating Bcl-2 analyses in breast cancer is limited, as well as those that compared tissue and blood Bcl-2 levels. Alireza et al. found a correlation between Bcl-2 serum levels and Bcl-2 expression detected in breast tissue by IHC [19]. In the study of Samy et al. Bcl-2 serum levels were significantly higher in the patients compared with the values in the control group [16]. Bcl-2 in serum was among the parameters that were shown to be useful for predicting recurrence in a short follow-up period. Hamed et al. found Bcl-2 plasma levels to be significantly higher in benign breast lesions and breast cancer in comparison with controls, and in breast cancer compared with benign lesions [17]. In that study the plasma levels of Bcl-2 were significantly higher in stages III and IV compared with stages I and II. Gaballah et al. analyzed plasma levels of Bcl-2 in metastatic setting and found a difference between breast cancer patients and the control group [18].
However, the results regarding circulating levels of Bcl-2 are not uniform. Alireza et al. did not find difference in Bcl-2 serum levels between cancer patients and controls [19].
Further research including larger clinical cohorts is warranted in order to evaluate the significance of Bcl-2 circulating levels in breast cancer patients, especially in a long follow-up.
An important mechanism of action of many anticancer drugs is induction of apoptosis [33]. Therefore impaired apoptotic pathways may affect the therapeutic response [3]. Both increased expression of Bcl-2 and mutated TP53 may play a role in tumor chemo and radio-resistance [7, 45]. Anti-apoptotic members of the Bcl-2 family are implicated in resistance to anti-HER-2 and anti-ER therapies [7].
Various therapeutic approaches related to the induction of apoptosis have been studied, including the antisense RNA approach and small molecule inhibitors [8, 46, 47]. In hematologic malignancies BH3 mimetics have been approved and some clinical trials are studying this group of drugs in solid tumors [4, 47].
In conclusion our results suggest that patients with circulating p53Abs and high Bcl-2 present an unfavorable group of patients in a long follow up. Additionally, analysis of circulating Bcl-2 and p53Abs may be useful in defining the group of patients that may benefit of some therapeutic modalities.
Footnotes
Acknowledgments
The authors wish to thank Aleksandra Žmegač Horvat for the English language editing of the manuscript.
Conflict of interest
The authors declare no conflict of interest.
