Abstract
BACKGROUND:
Runt-related transcription factor 2 (RUNX2) is an important gene that has been implicated in the progression of human cancer. Aberrant expression of RUNX2 predicts gastric cancer (GC) metastasis. However, the molecular mechanism of
OBJECTIVE:
We hypothesize that
METHODS:
The GEPIA database and immunohistochemical staining of 60 GC tissues were used to analyse the correlations between RUNX2 or COL1A1 expression and clinicopathological features, and the Kaplan-Meier method was used to evaluate survival. RT-PCR, western blotting and immunofluorescence were used to detect RUNX2 and COL1A1 expression in GC cells. Migration and invasion assays were performed to assess the influence of RUNX2 and COL1A1 on metastasis.
RESULTS:
RUNX2 and COL1A1 were highly expressed at both the gene and protein levels in GC, and patients who were positive for RUNX2 and COL1A1 had shorter survival. RUNX2 and COL1A1 expression linearly correlated with each other (
CONCLUSIONS:
RUNX2 may promote GC metastasis by regulating COL1A1. RUNX2/COL1A1 can be employed as a novel target for therapy in GC.
Introduction
Gastric cancer (GC) is one of the major causes of cancer-related death worldwide. Despite clinical progress in effective treatments for patients with GC, the 5-year survival rate remains poor. Many factors affect the prognosis of patients with GC, including the stage at diagnosis and lymphatic and distant metastasis [1]. Cellular stress results in changes in cell nutrition and development, leading to an atypical extracellular environment and changes in pH and oxygen levels [2]. Tumour relapse and metastasis after resection are great challenges in tumour therapy. The complicated mechanisms of GC invasion and metastasis should be examined to discover new biomarkers and therapeutic strategies.
The Runt-related transcription factor 2 (
Collagen type I alpha 1 chain (COL1A1), an extracellular matrix protein mapped to 17q21.33, is an important member of the collagen family. Oue et al. found that
This study provides ideas for the discovery of new targeting factors for GC metastasis and is of great significance for controlling the occurrence and development of this cancer.
Materials and methods
Clinical specimens
Tissue sections and the clinicopathological characteristics data were collected from patients undergoing surgical resection at the General Hospital of Tianjin Medical University between 2009 and 2014. The patients included 35 men and 25 women; the median age of the patients was 49.8 years (range, 35 years to 70 years). A total of 31 patients were considered lymph node metastasis-positive, and 29 were considered lymph node metastasis-negative. The median follow-up time was 97.1 months (range, 5 months to 150 months). Overall survival was calculated from the time of surgery to the time of death or date of last follow-up. Patients who were alive at the last follow-up were censored. Kaplan-Meier survival analysis was used for survival curves. The diagnosis of gastric adenocarcinoma was confirmed by two pathologists. This study was approved by the Ethics Committee of Tianjin Medical University. All patients agreed to the research use on their tissues and signed an informed consent agreement.
Immunohistochemistry and scoring system
Formalin-fixed GC tissues embedded in paraffin were dewaxed, rehydrated, and analysed by immunohistochemistry (IHC). Primary antibodies against RUNX2 (ab76956, Abcam, 1:200) and COL1A1 (sc-293182, Santa Cruz Biotechnology, 1:100) were used in accordance with manufacturer’s instructions. Brown positive signals of RUNX2 were found in the nuclei or cytoplasm, and positive COL1A1 staining was detected in the cytoplasm. Three separate high-power fields were chosen, and 100 adjacent cells were counted in the area with the highest density of positive cells for each slide. Immunostaining intensity was evaluated on a three-point scale (from 1 to 3). Positive cell numbers were scored as follows: 0, (
Cells
The GC cell lines MKN28, MGC-803, MKN74, and MKN45 were purchased from the Shanghai Cell Bank of the Chinese Academy of Sciences. Foetal bovine serum (FBS) was purchased from Biological Industries (Israel), and penicillin-streptomycin (PS) was purchased from Invitrogen (USA). RPMI-1640 medium was obtained from Gibco (USA). All cells were cultured in RPMI-1640 with 10% FBS and 1% PS at 37
Plasmids and transfection
Overexpression, siRNA, and negative control plasmids for RUNX2 and COL1A1 were constructed by GeneCopoeia (Guangzhou, China) and used to transfect MKN-28 and MGC-803 cells followed Lipofectamine 2000 (Invitrogen, USA). Puromycin (Sigma, USA) was used to screen stably transfected cells.
Wound-healing assay
Cells (MKN28, MKN28-RUNX2, MKN28-RUNX2-shCOL1A1, MGC803, and MGC803-shRUNX2, MGC803-shRUNX2-COL1A1) were plated in 12-well plates (3
Invasion assay and migration assays
Cell invasion and migration abilities were detected using a transwell assay (Corning, NY, USA). Cells were washed twice and resuspended to a density of 3
Immunofluorescence (IF) staining
Approximately 10
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from GC cells using TRIzol reagent (Tiangen Biotech, Beijing, China) and used for reverse synthesis of cDNA with a reverse transcription kit (TaKaRa, Shiga, Japan). GAPDH was used as an internal control. qRT-PCR was performed using SYBR
Western blot analysis
Proteins were separated by electrophoresis on an SDS gel for 90 min and then transferred to PVDF membranes (Millipore, Bedford, MA, USA). Non-specific binding was blocked with 5% bovine serum albumin (BSA), followed by incubation of the membrane with the primary antibody on a rotating shaker overnight at 4
Correlations of RUNX2 expression and clinicopathological features in gastric cancer
Correlations of RUNX2 expression and clinicopathological features in gastric cancer
Correlations of COL1A1 expression and clinicopathological features in gastric cancer
Immunohistochemical staining for the expression of RUNX2 and COL1A1 in patients with gastric cancer (GC). (A) RUNX2 expression in GC (magnification, 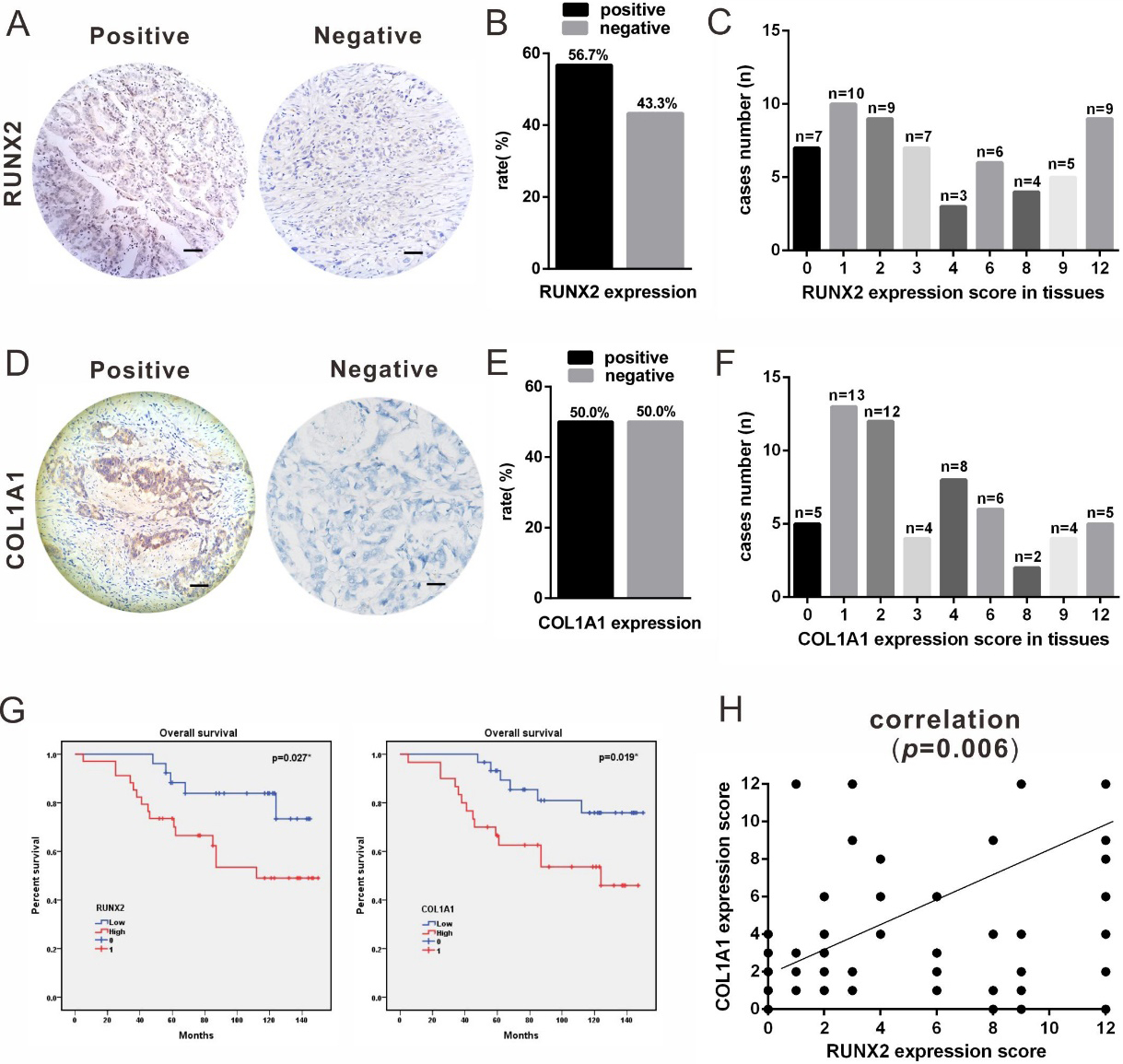
RUNX2 regulates COL1A1 expression in GC cells. (A) RUNX2 and COL1A1 expression in four GC cell lines. (B) RUNX2 mRNA expression in MKN28, MKN28-RUNX2, MGC803, and MGC803-shRUNX2 cells. (C) COL1A1 mRNA expression in MKN28, MKN28-RUNX2, MGC803, and MGC803-shRUNX2. (D and E) Effect of overexpression or downregulation of RUNX2 on COL1A1 as analysed by western blot. The results are from three repeated experiments. 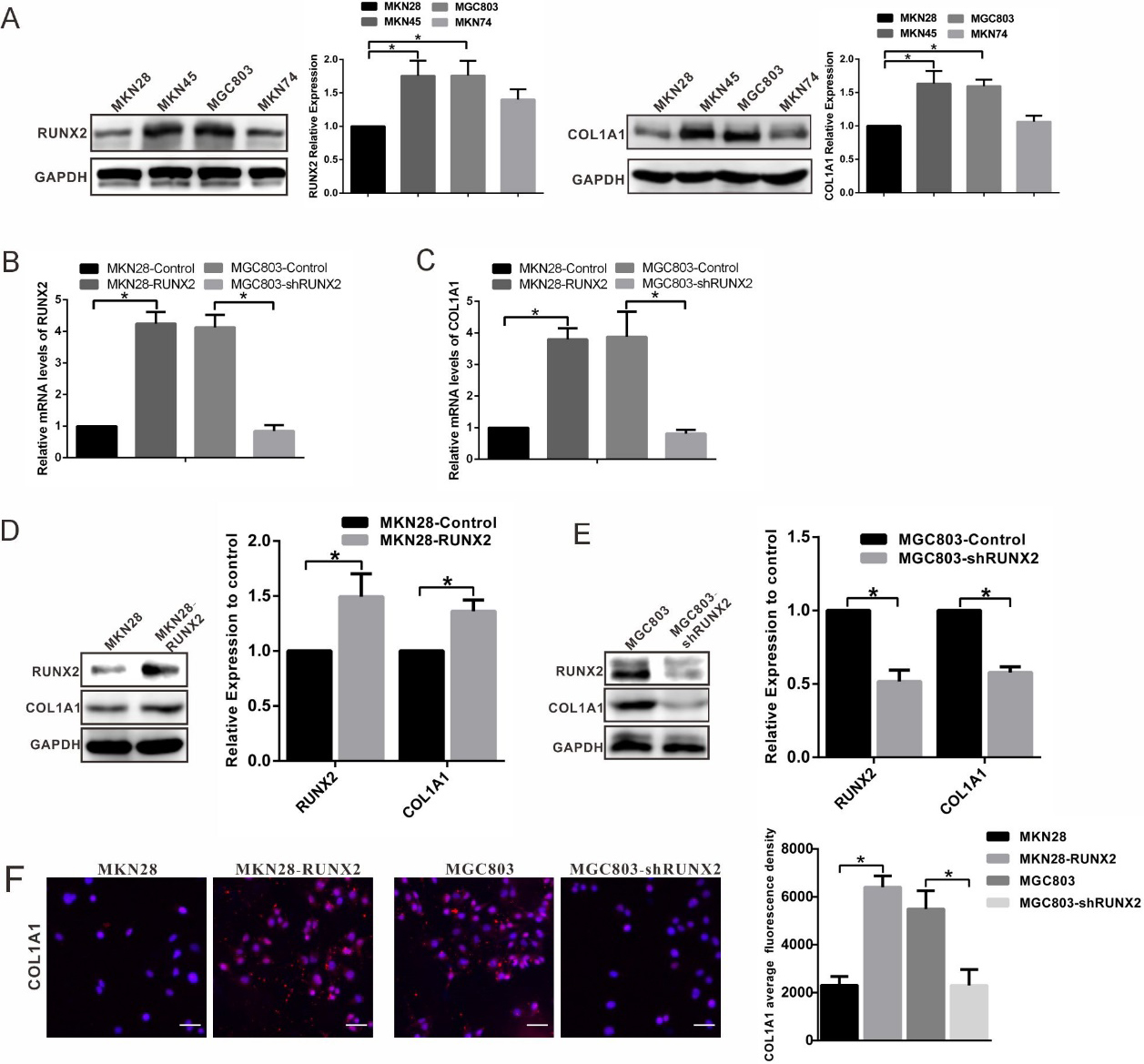
RUNX2 promotes gastric cancer cell migration and invasion by regulating COL1A1 expression. (A) The migration and invasion capacity of MKN28, MKN28-RUNX2, MKN28-RUNX2-shCOL1A1, MGC803, MGC803-shRUNX2 and MGC803-shRUNX2-COL1A1 cells was assessed using transwell assays. 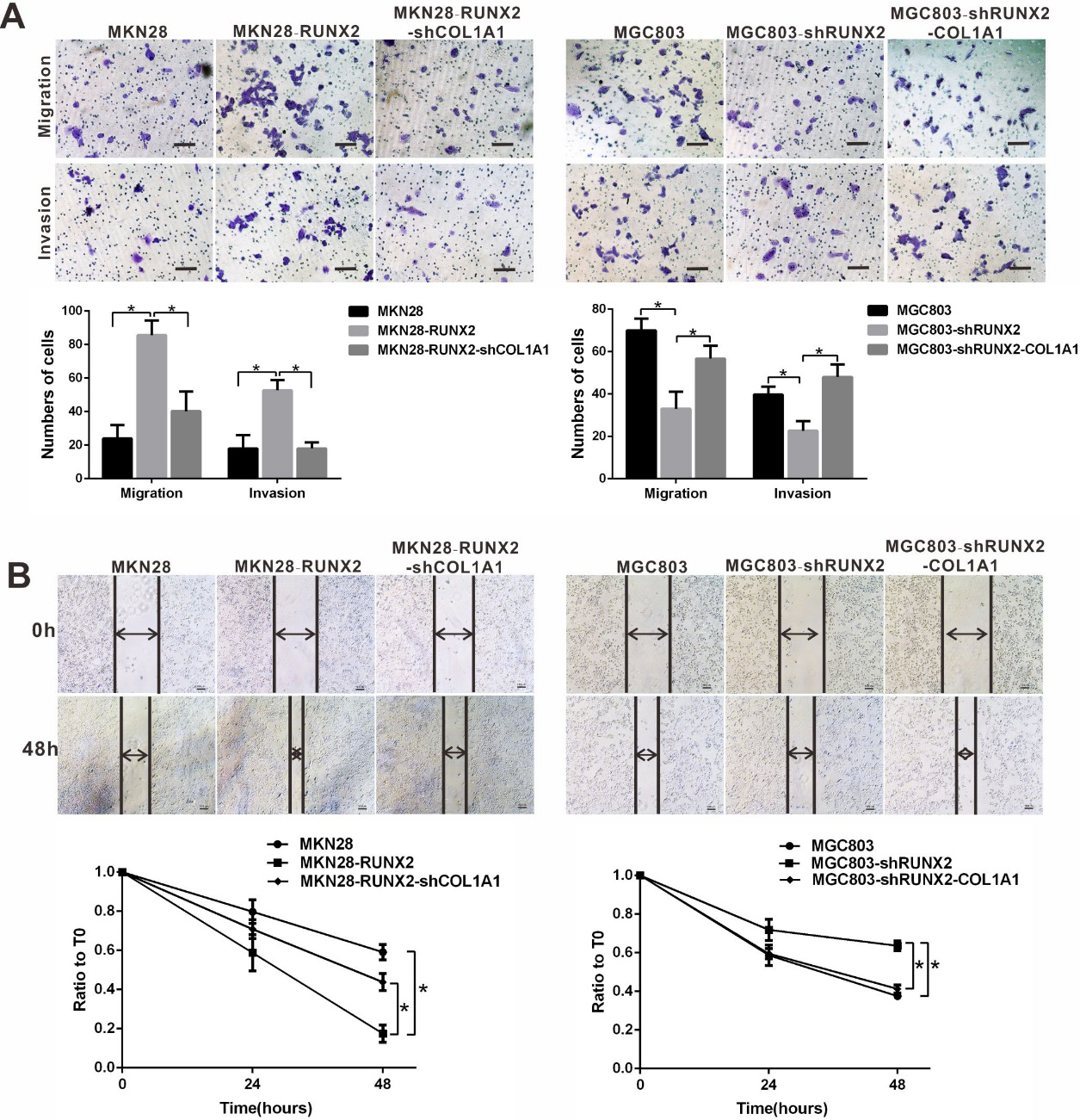
RUNX2 promotes cell metastasis through COL1A1 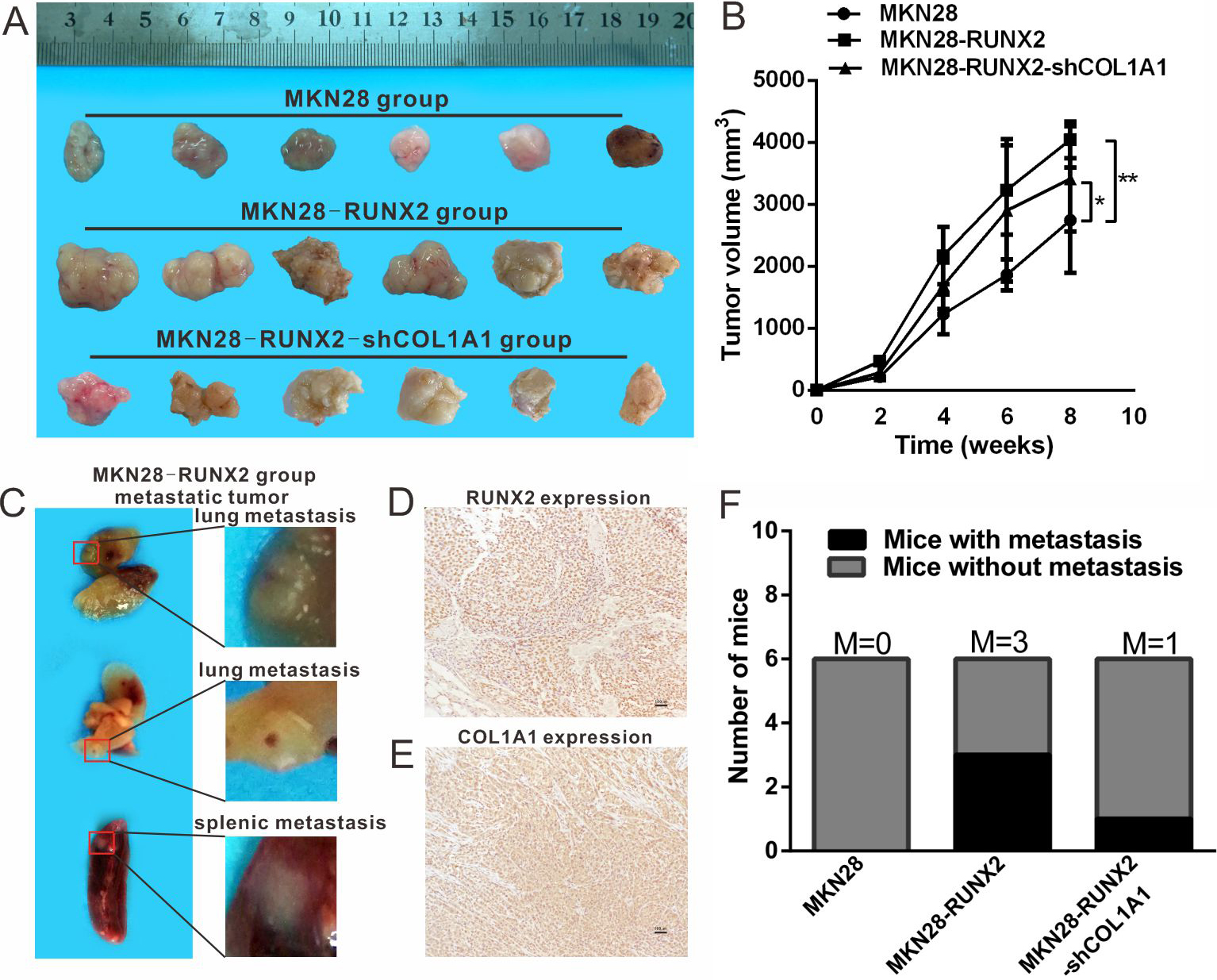
GEPIA (
Animal experiments
A total of 5
Statistical analysis
All statistical analyses were performed using SPSS 19.0 (Chicago, IL, USA) software. Analyses were repeated at least three times, and the results are reported as the mean
Results
RUNX2 and COL1A1 are upregulated in GC and are associated with poor prognosis
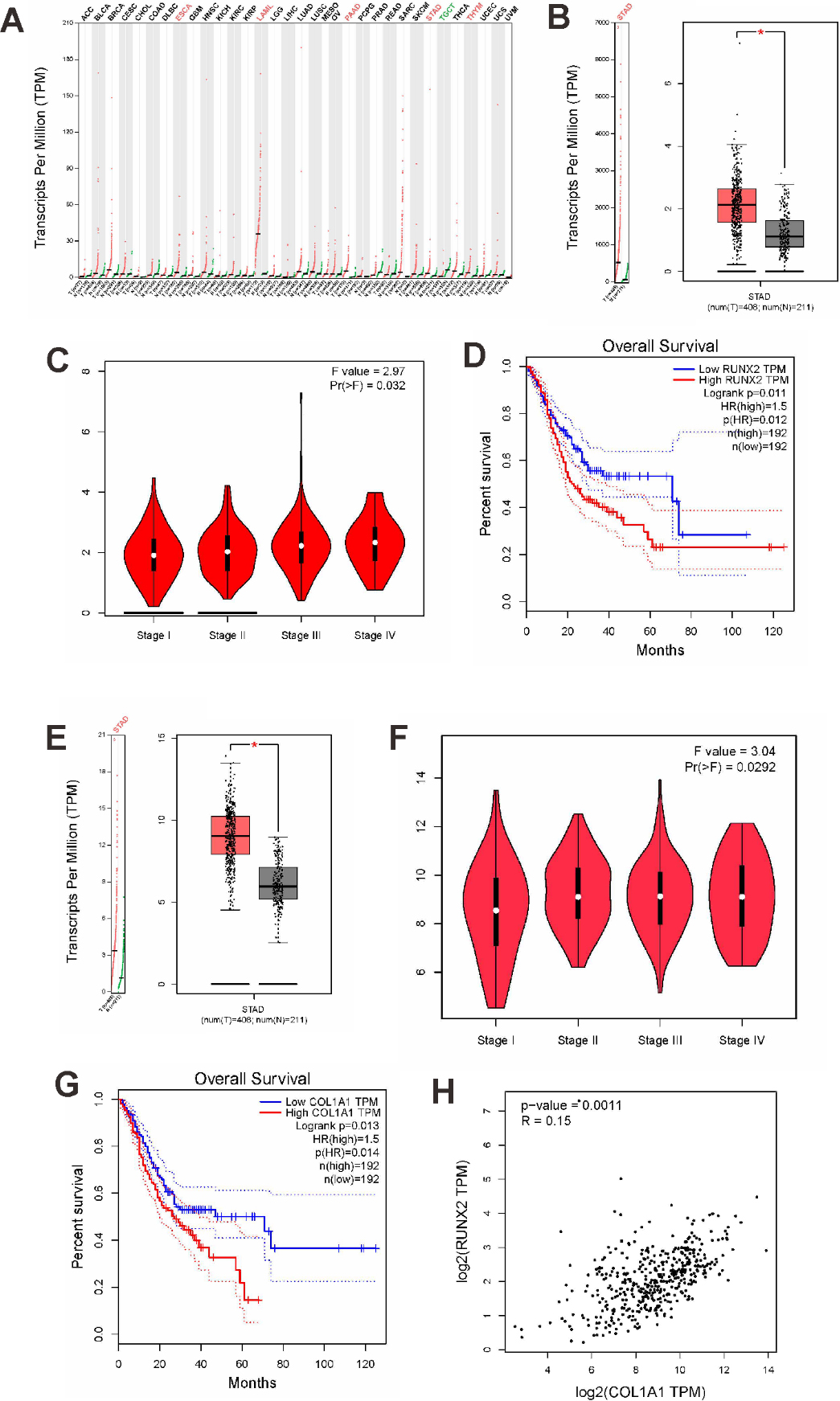
RUNX2 was found to be abnormally expressed in various tumours, including stomach cancer, in the GEPIA database (Supplementary Fig. 1A). A comparison of 408 GC tissues with 211 normal gastric tissues revealed that
Immunohistochemical staining of RUNX2 and COL1A1 in GC
To further investigate the role of RUNX2 and COL1A1 in GC, we detected the protein expression of RUNX2 and COL1A1 by IHC. RUNX2 was mainly expressed in the nucleus (Fig. 1A), and positive expression was observed in 34 of the 60 cases (Fig. 1B and C). COL1A1 was mainly expressed in the cytoplasm (Fig. 1D), and positive expression was observed in 30 of the 60 cases (Fig. 1E and F). Analysis of the medical records of the GC patients revealed a correlation between RUNX2 expression and TNM stage (Table 1). Patients with high RUNX2 expression had a tendency towards a poor prognosis (Fig. 1G). High COL1A1 expression was related to TNM stage and lymph node metastasis (Table 2) as well as poor survival prognosis (Fig. 1G). RUNX2 expression was positively related to that of COL1A1 in patients with GC (
RUNX2 regulates COL1A1 expression in GC cells
RUNX2 and COL1A1 expression levels were detected in different GC cell lines (Fig. 2A). MGC803 (high RUNX2 expression) and MKN28 (low RUNX2 expression) cells were selected from among several GC cell lines (MKN28, MKN74, MGC803, and MKN45) (Fig. 2A). Plasmid transfection was used to overexpress RUNX2 in MKN28 cells and knock down RUNX2 in MGC803 cells. Knockdown of RUNX2 by hairpin RNA in MGC803 cells significantly reduced RUNX2 mRNA levels (Fig. 2B). By contrast, the level of RUNX2 mRNA increased in MKN28-RUNX2 cells (Fig. 2B), as did the expression of COL1A1 mRNA (Fig. 2C). The expression of COL1A1 mRNA was reduced by the knockdown of RUNX2 in MGC803 cells (Fig. 2C). Western blot analysis revealed that RUNX2 had a significant promotion effect on COL1A1 protein expression. The upregulation of RUNX2 increased COL1A1 protein expression in MKN28 cells, whereas the loss of RUNX2 in MGC803 cells decreased COL1A1 protein expression (Fig. 2D and E).
Immunofluorescence analysis showed that RUNX2 overexpression promoted COL1A1 expression in the cytoplasm and nucleus, whereas RUNX2 knockdown reduced COL1A1 expression in the cytoplasm or nucleus (
RUNX2 regulates COL1A1 to promote GC invasion and migration
Transwell assays verified the effect of RUNX2 and COL1A1 on gastric tumour metastasis. As shown in Fig. 3A, overexpression of RUNX2 in MKN28 cells increased cell migration and invasion compared with the blank control group (
RUNX2 overexpression increases cancer cell metastasis through COL1A1 in vivo
To validate the essential functions of RUNX2 and COL1A1 in GC cell metastasis, xenograft mouse models were used. As shown in Fig. 4A, all three groups of cells (MKN28, MKN28-RUNX2 and MKN28-RUNX2-shCOL1A1) resulted in tumour formation when subcutaneously injected into BALB/c nude mice. The tumour volume was significantly larger in mice injected with MKN28-RUNX2 and MKN28-RUNX2-shCOL1A1 cells than in mice injected with MKN28 cells (Fig. 4B). Eight weeks later, the mice were acrificed, and lung or splenic metastatic tumours were found in three mice in the MKN28-RUNX2 group (Fig. 4C). The metastatic tumour tissue highly expressed RUNX2 and COL1A1 as shown in Fig. 4D and E. Lung metastasis was found in one mouse in the MKN28-RUNX2-shCOL1A1 group, and in the MKN28 group, no metastasis was found in any organ (Fig. 4F).
Discussion
Accumulating evidence has indicated the potential of RUNX2 to control the transcription of oncogenes and regulate various signalling pathways to promote tumour development. The extracellular matrix is a critical component of the tumour microenvironment and is involved in tumour formation and invasion [22]. RUNX2 can provoke extracellular matrix remodelling in tumours by regulating MMP13, MMP9, and SPP1. Through GEPIA database visualization analysis, we found that the expression of the extracellular matrix component COL1A1 was abnormally high in GC tissues and correlated with RUNX2 expression. These observations prompted us to hypothesize that RUNX2 promotes tumour metastasis by regulating COL1A1 in GC. To experimentally test this hypothesis, we analysed tissues from patients with GC by IHC. Our data demonstrated that RUNX2 and COL1A1 were highly expressed in GC and were associated with a poor prognosis. RUNX2 and COL1A1 expression levels were also significantly correlated. The critical involvement of RUNX2 in regulating COL1A1 expression in GC cells was further determined by a specific knockdown assay. Both the invasive and migratory capacities of GC cells were weakened after knockdown of the RUNX2 gene. Western blotting revealed that RUNX2 knockdown also decreased COL1A1 protein expression. RUNX2 is a powerful and complex member of the RUNX family. During osteoblast differentiation and development, RUNX2 molecules are considered a “platform molecule” or “master switch” that regulates a series of key molecules in differentiation and developmental processes [23, 24]. A RUNX2 mutation was first discovered in human clavicle dysplasia. Experiments have confirmed that RUNX2 is closely related to human osteoblast differentiation and chondrocyte maturation [25]. Emerging evidence has indicated that RUNX2 participates in tumour formation and invasion through functional modulation of extracellular matrix remodelling. Intriguingly, recent reports indicate that RUNX2 promotes the expression of bone sialoprotein (BSP) to mediate human breast cancer cell metastasis [26]. Bone sialoprotein is an extracellular matrix protein of mineralized tissue and plays a very important role in the early calcification of bone tissues [27]. Consistent with our experimental results, the transwell assay revealed that cells in which RUNX2 was knocked down were less invasive than control cells. These results are also consistent with the bioinformatics analysis. MMPs, a class of endopeptidases, are secreted as zymogens that are activated by cleavage of the amino-terminal propeptide. MMPs are involved in cancer cell growth, metastasis, and remodelling of extracellular matrices. Pratap et al. reported that RUNX2 expression in prostate cancer positively correlates with MMP9. RUNX2 activates the transcription of the MMP9 gene by directly binding to the proximal promoter of the MMP9 gene [28]. Galectin-3 is a member of the
Vascular smooth muscle cell-specific expression of RUNX2 in transgenic mice increases the expression of its target genes, namely, COL1A1 and COL1A2 [20]. RUNX2 and COL1A1 play important roles in atrophic nonunion pathogenesis [34]. According to our experiments, the downregulation of RUNX2 might affect COL1A1 expression. COL1A1 has been suggested to be a pro-metastatic extracellular matrix gene [35, 36]. Liu et al. confirmed that the ability of breast cancer cells to metastasize can be suppressed by the knockdown of COL1A1, suggesting that COL1A1 promotes breast cancer metastasis [37]. A previous study also suggested that COL1A1 contributes to tumour differentiation, proliferation, and invasion [38, 39].
Based on the results of bioinformatics analysis, RUNX2 and COL1A1 play a major role in GC metastasis. Further
Footnotes
Acknowledgments
This work was supported by Scientific Research Foundation of Tianjin Education Commission (No. 2017KJ225).
Author contributions
Conception: Yanlei Li
Interpretation or analysis of data: Ran Sun, Xiulan Zhao
Preparation of the manuscript: Yanlei Li
Revision for important intellectual content: Baocun Sun
Supervision: Baocun Sun
Supplementary data
Expression levels of