Abstract
BACKGROUND:
METCAM/MUC18 expression was increased with the malignant progression of prostate cancer and also a bona fide metastatic gene, capable of initiating and driving the metastasis of a non-metastatic human prostate cancer cell line to multiple organs.
OBJECTIVE:
We explored if METCAM/MUC18 was detectable in human serum and a novel biomarker to predict malignant propensity of prostate cancer.
MATERIALS AND METHODS:
Two antibodies were identified by Western blot analysis having the highest sensitivity and specificity to establish calibration curves from the recombinant METCAM/MUC18 proteins. They were used in ELISA and LFIA to determine the METCAM/MUC18 concentrations in serum samples from 8 normal individuals, 4 BPH patients, 1 with PIN, 6 with high-grade prostate cancer, and 2 treated cancer patients.
RESULTS:
Serum METCAM/MUC18 concentrations were statistically significantly higher in the patients with PIN and prostate cancer than those with BPH, the treated patients and normal individuals. The LFIA results were statistically better than ELISA and Western blot methods. Serum METCAM/MUC18 concentrations were in direct proportional to most of serum PSA concentrations.
Introduction
Prostate cancer accounts for the 1
We have previously shown that the expression of METCAM/MUC18 antigen is positively correlated with the malignant progression of clinical prostate cancer, suggesting that it may be a novel diagnostic biomarker for predicting the metastatic potential of human prostate cancer [13]. Furthermore, we showed that it is also a bona fide metastatic gene, capable of initiating and driving the metastasis of a non-metastatic human prostate cancer cell line to multiple organs [9, 10, 11]. As such, it very likely plays a role in converting indolent prostate cancer to an aggressive one [13]. The purpose of this research is to use three immunological methods, such as the Western blot (WB) analysis [14], an enzyme-linked immunosorbent assay (ELISA) [14, 15], and a lateral flow immunoassay (LFIA) method [16, 17, 18], to investigate the above possibility that METCAM/MUC18 may be used as a novel diagnostic biomarker for predicting the metastatic potential of human prostate cancer and for differentiating the indolent prostate cancer from the aggressive one. We found that the serum huMETCAM/MUC18 concentrations were statistically significantly higher in the patients with PIN and with prostate cancer at different pathological stages than those with BPH, the treated and normal individuals [15, 18]. We also found that the serum huMETCAM/MUC18 concentrations were also directly proportional to serum PSA concentrations when PSA was
Epitopes recognized by various antibodies
Epitopes recognized by various antibodies
Materials
Chicken anti-human METCAM/MUC18 antibody was home-made [20]. Rabbit anti-METCAM/MUC18 antibody (SC28667) and goat anti-METCAM/MUC18 antibody (SC18940) were from Santa Cruz Biotech. Rabbit anti-METCAM/MUC18 antibodies (MBS2756 88 and MBS2518792) were from MyBiosource (Table 1). Instant non-fat dry milk powder (NFM) (8 g of protein per 23 g of dry powder, Sunny Select) was purchased at the Lucky Super Market, Novato, CA, USA. PCR adhesive PCR sealing film (cat # BL-6174) was from Basic Life. For LFIA tests, nitrocellulose membrane (Cat # M10-00101), sample pad (Cat # M10-01401), conjugated pad (Cat # M10-00701), absorbent pad (Cat # M10-01701), and plastic backing pad (Cat # M10-00400) were from Rega Biotech. Colloidal gold (40 nm) (Cat # RE-30013001) was from Rega biotech.
Preparation of recombinant METCAM/MUC18 proteins
The recombinant METCA/MUC18 proteins were obtained as described [20, 21]. In brief, the intact human or mouse METCAM/MUC18 cDNA was PCR-amplified and cloned to pGEM-T or pGEM-T Easy, then to pcDNA3.1
Preparation of human serum samples
Human whole bloods were obtained from voluntary donors with a consensus at CYCU with an approved IRB (20130510-20) or from patients at Department of Urology, NTU Hospital with an approved IRB (201406098RIND). 15 or 30 ml of whole blood per person was withdrawn into one or two 16 ml conical centrifuge tubes without heparin and allowed to coagulate at room temperature for 30–60 min. Then serum was obtained by centrifugation at 1,000 rpm (180
Determination of METCAM/MUC18 concentrations in human serum samples by the Western blot method. Each human serum sample was analyzed by the Western blot (WB) method as described in “Materials and Methods.” The intensity of huMETCAM/MUC18 band in each serum sample was determined by Image J and compared to that of the SK-Mel-28 cell lysate. The METCAM/MUC18 band is indicated with an arrow. All the numbers on top of each lane were the PSA concentration and all the numbers under each lane were the % intensity compared with that of the SK-Mel-28 cell lysate (100%) in lane 1.
Western blot was carried out [14] with slight modifications [7, 8, 9, 11, 12, 20, 21]. One volume of serum was mixed with 1/3 volume of 4X WB buffer and kept frozen till use. 10
ELISA
Standard direct ELISA [14] was used with slight modifications [15]. One volume of serum was mixed with an equal volume of 2X CB (1/2X serum) or with an 1/3 volume of 4X CB (3/4X serum). 1X CB was composed of 0.1 M carbonate buffer, pH 9.6, 2X CB of 0.2 M carbonate buffer, pH 9.6, and 4X CB of 0.4 M carbonate buffer, pH 9.6. Most ELISA was carried out by the direct method and some by an indirect method [14, 15]. In brief, 0.4–6.3 ng of M protein, 0.42–6.77 ng of NM-GST fusion protein, or 0.23–3.6 ng of C-terminal-GST fusion protein in 0.05 ml of 1X CB buffer, or a triplicate of 0.05 ml of 3/4X human serum in 1X CB was pipetted to each of the 96-well, covered with an adhesive PCR seal- ing film, fixed for overnight at 4
LFIA
LFIA is useful for immunoassay on a nitrocellulose strip/membrane [16, 17] and was carried out with slight modifications [18]. The test line contained about 26 ng (0.53
Statistical analysis
Standard deviation of each set of data and R2 of two different sets of data were analyzed by the build-in programs in the Excel. The one-way ANOVA method in the SSPS software was used for statistical analysis among several sets of data. The difference among different sets of data was considered statistically significant if
HuMETCAM/MUC18 concentrations in various human serum samples determined by the Western blot method and its relation with the status of patients. The average METCAM/MUC18 concentrations with standard deviations in the serum samples from the treated prostate cancer patients and patients with prostate premalignant PIN, or BPH, were compared to those from normal individuals. The number on top of each bar was the 
Relation of PSA to the huMETCAM/MUC18 concentrations determined by the WB method by using our home-made chicken antibody. The serum METCAM/MUC18 concentrations were compared to PSA. (A) shows 7 serum METCAM/MUC18 concentrations versus PSA (left panel) and PSA versus serum METCAM/MUC18 (right panel) when PSA was from 0.36 to less than 7 ng/ml, (B) shows 11 serum METCAM/MUC18 concentrations versus PSA (left panel) and PSA versus serum METCAM/MUC18 (right panel) when PSA was from 0.36 to less than 7 ng/ml, (C) shows 13 serum of that when PSA was from 0.36 to less than 25 ng/ml, and (D) shows 15 serum of that when PSA was from 25 to 1219 ng/ml.
Established Standard curve of recombinant METCAM/MUC18 proteins determined by an ELISA by using our home-made chicken antibody (A) and the commercial rabbit antibody (MBS275688) (B). (A) shows the intensities of various recombinant METCAM/MUC18 proteins, versus their concentrations by using chicken antibody for ELISA. (B) shows those by using the rabbit antibody MBS275688 for ELISA.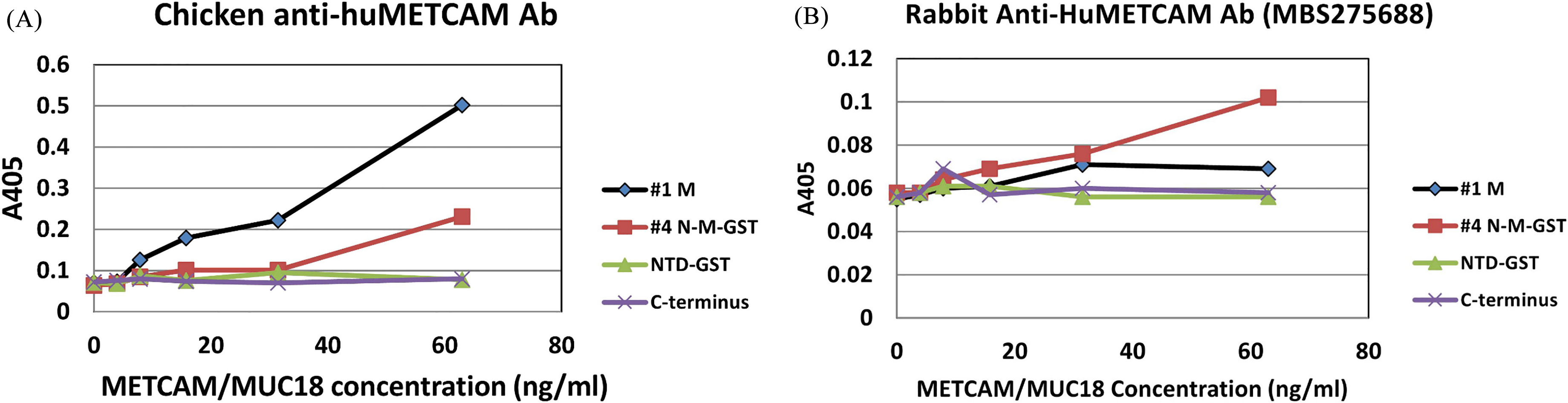
Determination of METCAM/MUC18 concentrations in human serum samples by using quantitative WB method
Figure 1 shows that human METCAM/MUC18 with a molecular weight of about 130 KDa was detectable in various human serum samples by WB analysis. Figure 2 shows the quantitative results that the METCAM/MUC18 protein concentrations in malignant prostate cancer patients were significantly higher than those in normal serums and BPH (
Patient (male) characteristics (
19)
Patient (male) characteristics (
HuMETCAM/MUC18 concentrations in various human serum samples determined by using chicken antibody in the ELISA and its relation with the status of patients. The average METCAM/MUC18 concentrations with standard deviations in the serum samples from the treated prostate cancer patients and patients with prostate cancer, or BPH, were compared to those from normal individuals. The number on top of each bar was the 
Relation of PSA to the huMETCAM/MUC18 concentrations determined by ELISA by using our home-made chicken antibody (A) and the commercial rabbit antibody MBS275688 (B and C). The serum METCAM/MUC18 concentrations were compared to PSA. (A) shows 16 serum METCAM/MUC18 concentrations versus PSA by using our home-made chicken antibody, when PSA was from 0.27 to less than 25 ng/ml. (B) shows 15 serum METCAM/MUC18 concentrations by using the rabbit antibody for ELISA versus PSA, when PSA was from 0.27 to less than 25 ng/ml. (C) shows 17 serum METCAM/MUC18 concentrations by using the rabbit antibody for ELISA versus PSA, when PSA was from 0.27 to 1219 ng/ml.
Established Standard curve of recombinant METCAM/MUC18 proteins determined by LFIA by using our chicken antibody. (A) shows the signals of NM-GST (the positive control protein) and C-terminus-GST (the negative control protein) on the test line of the membrane. The concentrations (ng/ml) of the recombinant proteins are indicated on the top of each lane. (B): The left panel shows the intensities of the two recombinant METCAM/MUC18 proteins (NM-GST and C-terminus GST) versus their concentrations by using chicken antibody for LFIA. The right panel shows the standard curve derived from the data in the left panel after subtraction of the intensity of the C-terminus GST from that of NM-GST.
HuMETCAM/MUC18 concentrations determined by using chicken antibody for the LFIA in various human serum samples and its relation with the status of patients. (A) shows the signals of each serum sample on the test line of the membrane. The numbers on the top of each lane were the PSA concentrations (ng/ml). (B) shows the average METCAM/MUC18 concentrations with standard deviations in the serum samples from the treated prostate cancer patients, patients with prostate cancer, or BPH, were compared to those from normal individual. The number on top of each bar was the 
Relation of PSA to the huMETCAM/MUC18 concentrations determined by LFIA by using our home-made chicken antibody. (A) shows 6 serum METCAM/MUC18 concentrations versus PSA, when PSA was from 0.02 to 0.85 ng/ml. (B) shows 10 serum METCAM/MUC18 concentrations versus PSA, when PSA was from 0.02 to 1219 ng/ml.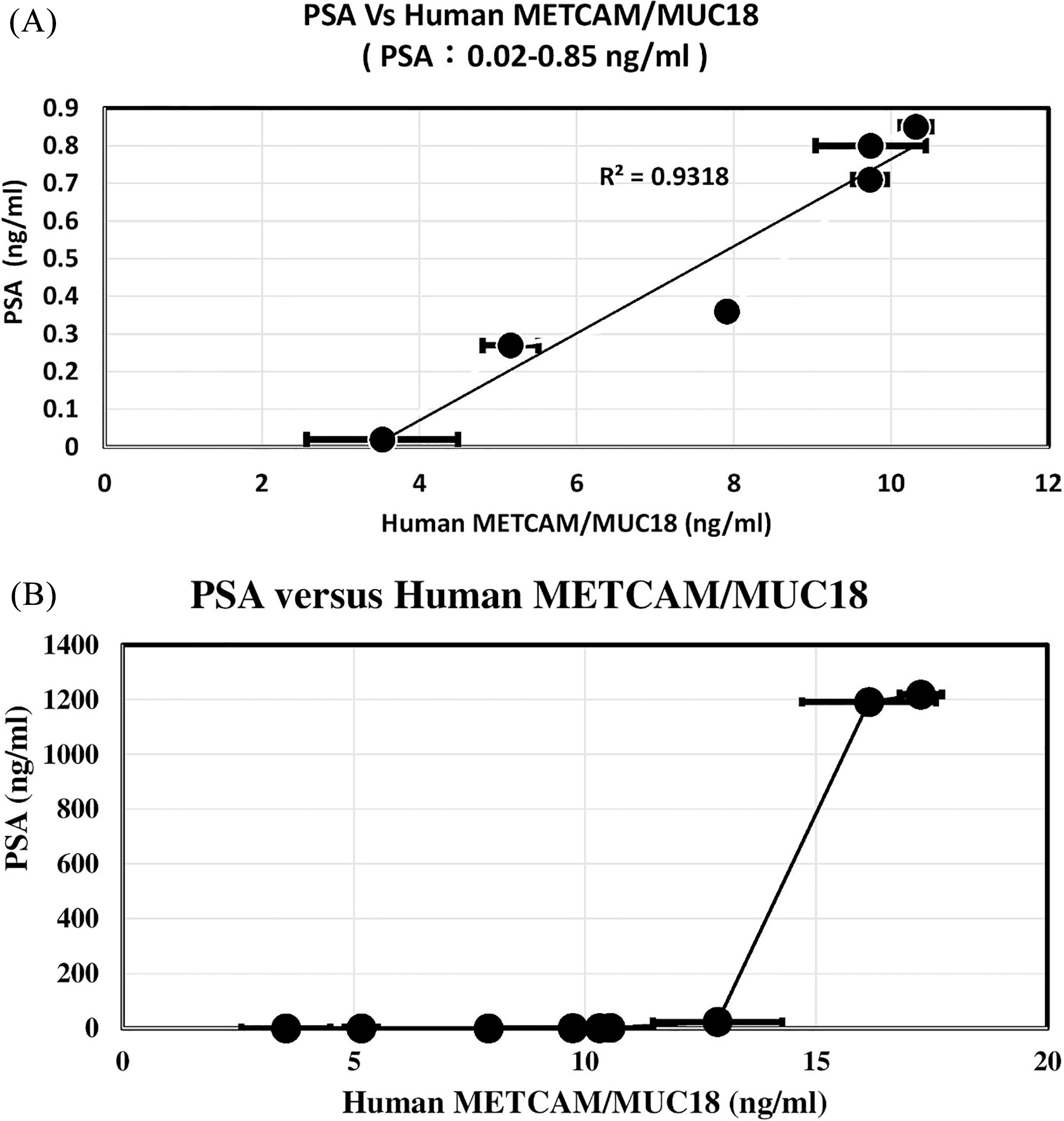
PSA concentrations in various patients. The figure shows the average PSA concentrations with standard deviations in the serum samples from various persons. The number on top of each bar was the 
A good antibody should recognize the full size huMETCAM/MUC18 in the lysates made from tumor cell lines of SK-Mel-28, DU145 and PC-3, and the proper recombinant proteins in the WB analysis. Table 1 shows the summary of the epitopes recognized by our home-made chicken antibody and four commercial antibodies used for the test. Our home-made chicken antibody and the rabbit antibody MBS275688 met our criteria and were further used for the following ELISA and LFIA. However, the other two commercial antibodies did not meet our criteria and were not used.
The human METCAM/MUC18 proteins in serum samples of various patients determined by an ELISA
We then determined the best concentrations of the two primary antibodies and secondary antibodies for obtaining optimal ELISA signals. We found that 25
The human METCAM/MUC18 proteins in serum samples of various patients determined by an LFIA [18]
Figure 7A shows that the intensity of the images in the test line of LFIA for the positive control recombinant protein was visually significantly stronger than that of the negative control protein. Figure 7B shows the quantitative results that our home-made chicken antibody could differentiate the positive control protein (NM-GST) from the negative control protein for a five-fold difference in intensity (
Discussion
In this report, we presented evidence to show that it was possible to use three immunological methods to determine the serum METCAM/MUC18 concentrations and to differentiate those in the prostate cancer from normal individuals and patients with BPH. Furthermore, there was a positive correlation of METCAM/MUC18 with most of PSA in human serum. Our home-made chicken antibody was better than the commercial rabbit antibody MBS275688. The reason for our success was due to using chicken antibody for the tests instead of using antibodies made in mouse or rabbit, which had also been observed by one group [22]. In the near future we should expand our survey cohort in Taiwanese males and to increase sensitivity of our tests by using biotinylated antibodies [23, 24] to further improving the LFIA and by exploring the use of streptavidin-coated magnetic beads to enrich the antigen and removing the interference materials in the serum sample [24, 25]. Eventually, we may be possible to use the further improved LFIA to detect the presence of METCAM/MUC18 antigen in urine samples [26].
Each of the three methods has advantages and disadvantages. The major advantage of using the WB analysis was that the correct size of METCAM/MUC18 could be observed and quantified; however, the major disadvantages were that the quantitation was limited by the resolution of the Image J software, several costly electrophoretic equipment were required, and the procedures were tedious, time-consuming and not easily reproducible even with the trained personnel. The major advantages of using the ELISA method were that a large number of samples could be simultaneously processed on the 96-well plates and the quantitation sensitivity was excellent and highly reproducible, which was the gold standard for most diagnostic methods; however, the major disadvantages were that several costly equipment were also required and the procedures were time-consuming, tedious, and not easily reproducible with the train personnel. The major advantages of using LFIA were that the procedures were relatively simple and easy operation, user friendly and easily trained, rapid visual results with naked eyes, high reproducibility, and very cost-effective in comparison to the other two methods since no expensive equipment required; however, the major disadvantages were that the results were variable among materials from batch to batch, qualitative results were obtained rather than quantitative results (semi-quantitative), and resolution may be limited by different antibodies used.
In our hands, it was possible to use the LFIA for quantitative analysis, since among the three methods, the LFIA method was better than ELISA, which in turn was better than WB analysis, in that the standard deviations of the data in LFIA were much smaller and data were statistically better than the two other methods and the PSA test (as shown in Fig. 10). Furthermore, the linear proportionality between the METCAM/MUC18 and PSA concentrations in LFIA was better than the two other methods. Thus, LFIA has the high potential to be further improved to become a reproducible, simple, cost-effective diagnostic test for clinical purpose. At this time, we still could not differentiate malignant prostate cancers from indolent ones and also to monitor treatment outcome of the patients, which should await many years of follow-up checking our patients in the future.
Taken together, it is feasible and promising to use the METCAM/MUC18 as a diagnostic biomarker for developing a reliable, cost-effective, and rapid test for predicting the malignant progression of prostate cancer. The potential limitation for using the METCAM/ MUC18 as a diagnostic biomarker for predicting malignant potential of prostate cancer is that it is not prostate organ-specific, since it is also overly expressed in other cancers, such as angiosarcoma, breast cancer, gastric cancer, hepatocellular carcinoma, lung cancers, most melanoma, nasopharyngeal carcinoma type III, osteosarcomas, and pancreatic cancer; however, it is under expressed in some cancers, such as colorectal cancer, nasopharyngeal carcinoma type I, and ovarian cancer [27]. Nevertheless, it may be feasible to use the METCAM/MUC18 to at least complement the current biomarker PSA and to combine it with other prostate cancer biomarkers for predicting the malignant potential of prostate cancer [4].
Footnotes
Acknowledgments
We thank the supports from NSC (NSC-101-2320-B-033-003), Center for Biomedical Technology and Research Center for Circular Economy at CYCU, and the joined project of CYCU and Ten Chan General Hospital, Taiwan.
Conflict of interest
All the authors declare no conflict of interest.
