Abstract
BACKGROUND:
The recognition of high-risk colon cancer patients prone to chemoresistant and recurrent disease is a challenge.
OBJECTIVES:
We aimed to assess the immunohistochemical expression of ERCC1, PARP-1, and AQP1 in 60 cases of stage II and III colon cancer who underwent curative resection and adjuvant chemotherapy. Their predictive role of tumor progression and disease-free survival (DFS) was analyzed.
METHODS:
The immunohistochemical expression of ERCC1, PARP-1, and AQP1 in 60 cases of stage II and III colon cancer who underwent curative resection and adjuvant chemotherapy was studied. The collected data on the overall survival (OS), disease-free survival (DFS), and the response to the chemotherapy were analyzed.
RESULTS:
Positive nuclear ERCC1 expression was identified in 58.3% of the patients, ERCC1 expression was significantly associated with left-sided tumors (
CONCLUSIONS:
ERCC1, PARP1, and AQP1 are adverse prognostic biomarkers in stage II–III colon cancer. Moreover, adjuvant chemotherapy may not be beneficial for patients with positive ERCC1, high PARP1, and AQP1-negative tumors. Therefore, we recommend that ERCC1, PARP-1, and AQP1 should be assessed during the selection of the treatment strategy for stage II–III colon cancer patients.
Introduction
Colon cancer (CC) is the third most common cancer worldwide, with a high mortality rate [1, 2]. In Egypt, it represents about 33.8% of whole GIT tumors and about 6.5% of all diagnosed cancer with a tendency toward younger ages [3]. Although surgical resection is effective in localized cases, nearly 20% of stage II and 40% of stage III progress within 5 years after surgery [4]. This gives the rationale for adjuvant chemotherapy after surgical resection. The 5-fluorouracil (5-FU)-based chemotherapy represents the standard adjuvant protocol for stage III patients [5]. A subset of stage II patients with high-risk of relapse may benefit from adjuvant therapy, however, its routine use in standard risk stage II is is not recommended. As well, there is a considerable heterogeneity amongst colon cancer patients leading to different prognosis and variable chemotherapeutic response. Therefore, it is essential to identify predictive biomarkers of recurrence and chemoresistance [6].
Oxaliplatin is a platinum-based chemotherapeutic agent that has efficacy against various tumor cell lines and became a part of the classical therapeutic protocol of CC [7]. Adding oxaliplatin to 5FU (FOLFOX regimen) improved the adjuvant treatment by reducing the risk of relapse and improving OS [8]. Oxaliplatin exerts its effect by forming DNA-platinum mono-adducts, inhibiting both DNA replication and transcription, and inducing apoptosis in actively dividing cells. The nucleotide excision repair (NER) pathway; a major DNA repair system in the mammalian cells; removes these bulky adducts produced by oxaliplatin. DNA repair efficiency may contribute to platinum-based cytotoxic drug resistance. The ERCC1 protein, which is a key component of NER pathway [9] repair the interstrand cross-links in the DNA beside the recognition and removal of cytotoxic agents as oxaliplatin [10]. The correlation of ERCC1 expression with the prognosis has been investigated in various tumors like gastric cancer [11], ovarian cancer [12], and lung cancer [13].
Poly (ADP-ribose) polymerase-1 (PARP-1) is a nuclear enzyme that is activated and engaged in the repair of DNA single-strand breaks through base excision repair pathway (BER), in addition to regulation of DNA transcription and cell cycle progression. PARP-1 inhibitors interrupt BER, rendering it inactive causing increased levels of persisting single-strand breaks, with DNA double-strand breaks (DSB) upon replication finally [14]. PARP-1 has a central role in CC carcinogenesis. Based on the preclinical studies which verified the improvement of radiosensitivity and chemotherapy response in experimental models of CC by PARP-1 inhibitors, a number of clinical trials have been initiated with the use of four PARP inhibitors in CC patients [15]. Thus, it is important to investigate the PARP-1 protein expression in clinical samples of CC. However, little is known about that [16].
Aquaporins (AQPs) are a group of basic membrane proteins that assist in water transport through the cell membrane. They are reported to play a major role in cellular proliferation, angiogenesis, apoptosis, cell migration, and mitochondrial metabolism. Although the role of AQPs in human pathology has been studied broadly, lately their roles in cancer have become an area of interest. Numerous studies have revealed that AQPs are overexpressed in different malignant tumors [17]. An in vitro research confirmed that AQP1-mediated plasma membrane water permeability is fundamental for colon cancer cell migration and may be associated with tumor invasion and metastasis [18]. Although the clinical and biological relevance of AQP1 has been investigated in numerous types of cancer, including CC, only a small number of studies have focused on its prognostic role, and these results were conflicting. Furthermore, it remains to be assessed whether the AQP1 expression is predictive of response to chemotherapy or not [19].
Several prognostic parameters have been analyzed in colon cancer. However, they are still inadequate to recognize the high-risk patients prone to chemoresistant and progressive tumor. Therefore, there is an actual need to identify new predictive biomarkers for relapse and chemotherapeutic resistance. These predictive markers can tailor the management of each individual patient, and consequently enhance the benefit from a specific therapeutic agent. Thus, the current study evaluated the potential clinical utility of ERCC1, PARP-1, and AQP1 as predictive and prognostic molecular markers in stage II and III CC patients receiving 5-FU-based adjuvant chemotherapy after curative surgery.
Materials and methods
Patients’ selection
The current prospective study was carried out at Pathology, Clinical Oncology, Medical Oncology, and General Surgery Departments, Faculty of Medicine, Zagazig University hospitals after written informed consent was taken from our participants. A total of 60 consecutive patients of stage II or III primary CC that underwent curative resection between July 2015 and December 2017 were enrolled in the study. All the hematoxylin and eosin-stained slides were revised to confirm the diagnosis, histological grade, and tumor stage. The tumor stage was pathologically confirmed according to TNM staging system (American Joint Committee on Cancer (AJCC), the 8th edition). Patients with distant metastases at the time of operation, preoperative chemotherapy and/or radiotherapy, or patients with rectal cancer were excluded from the study. Patients’ clinical data, including survival and follow-up information, were retrieved from the hospital registry, and the clinical patients’ records. Approval of the local research ethics committee in Zagazig University hospitals was obtained.
Treatment schedule and follow-up
All the patients were admitted to the General Surgery department and diagnosed by the clinical symptoms (e.g., bleeding per-rectum, obstructive manifestations), abdominal ultrasonography, computed tomography with oral contrast, and colonoscopy biopsies. For the twenty patients of right-sided colon cancer, right hemicolectomy and ileotransverse anastomosis was done. For the forty patients of left-sided colon cancer, left hemicolectomy with primary anastomosis was done after preoperative colonic preparation. All the patients were followed-up until stitches removal and then completed their treatment in the Oncology department.
Curative resection was done for all patients and followed by the chemotherapy regimen in 50 cases only (high risk stage II and stage III). The fifty cases were treated with the standard mFOLOFOX6 consisting of oxaliplatin (85 mg/m
The follow-up to detect early recurrence was completed according to a standard protocol that included colonoscopy, abdominal ultrasound, and chest radiography every 6 months/2 years then every year later on. Assessment of CEA level was done every 3 months/ 2 years, then every 6 months later on. The follow-up aimed to detect early treatment failure (tumor recurrence).
Immunohistochemistry (IHC)
Formalin-fixed, paraffin-embedded tissue (FFPE) blocks were serially sectioned into 3–5
Immunohistochemistry assessment
ERCC1 expression
ERCC1 assessment based on the percentage of positive tumor cells/10 HPF. Score 0: complete negative staining, score 1:
PARP-1 expression
Both intensity (0–3) and pattern scores (1–6) of nuclear PARP-1 expression were evaluated. Each intensity score was multiplied by its parallel pattern score (1
AQP1 expression
AQP1 membranous or cytoplasmic staining within the tumor cells was semi-quantitatively scored as 0 (0–5%), 1 (5–20%), 2 (20–50%) or 3 (
Clinicopathological features, immunohistochemical markers and outcome of 60 patients with colorectal carcinoma
Clinicopathological features, immunohistochemical markers and outcome of 60 patients with colorectal carcinoma
Categorical variables were expressed as number (percentage). Continuous variables were expressed as mean
Continuous variables were expressed as the mean
Results
Clinicopathological characteristics of the patients
As presented in Table 1, the mean age of the studied patients at the initial diagnosis was 46.17
A. Represenative case of colon adenocarcinoma (H&E, 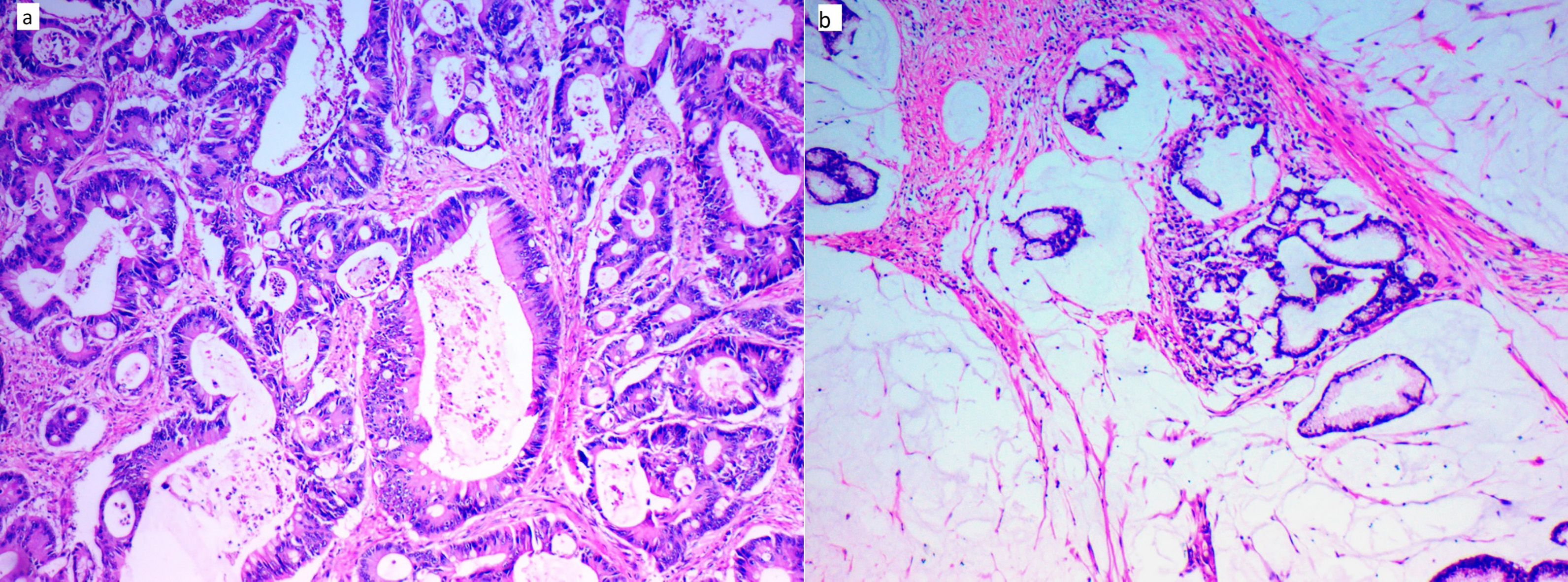
Representative images of ERCC1 immunohistochemical staining A. Nuclear ERCC1 expression in grade II colon adenocarcinoma (IHC, 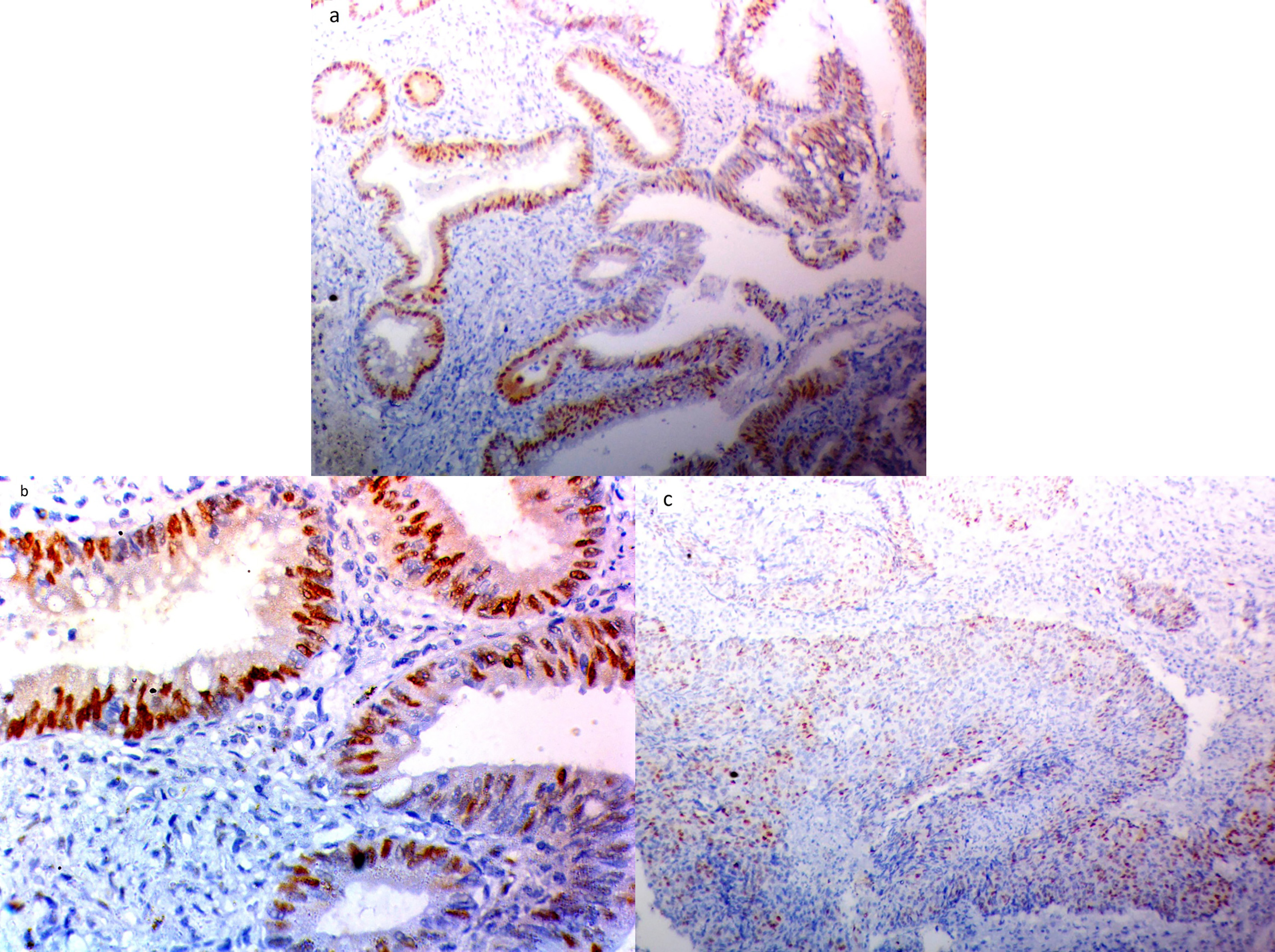
Representative images of PARP1 immunohistochemical staining A. Nuclear PARP1 expression in grade II colon adenocarcinoma (IHC, 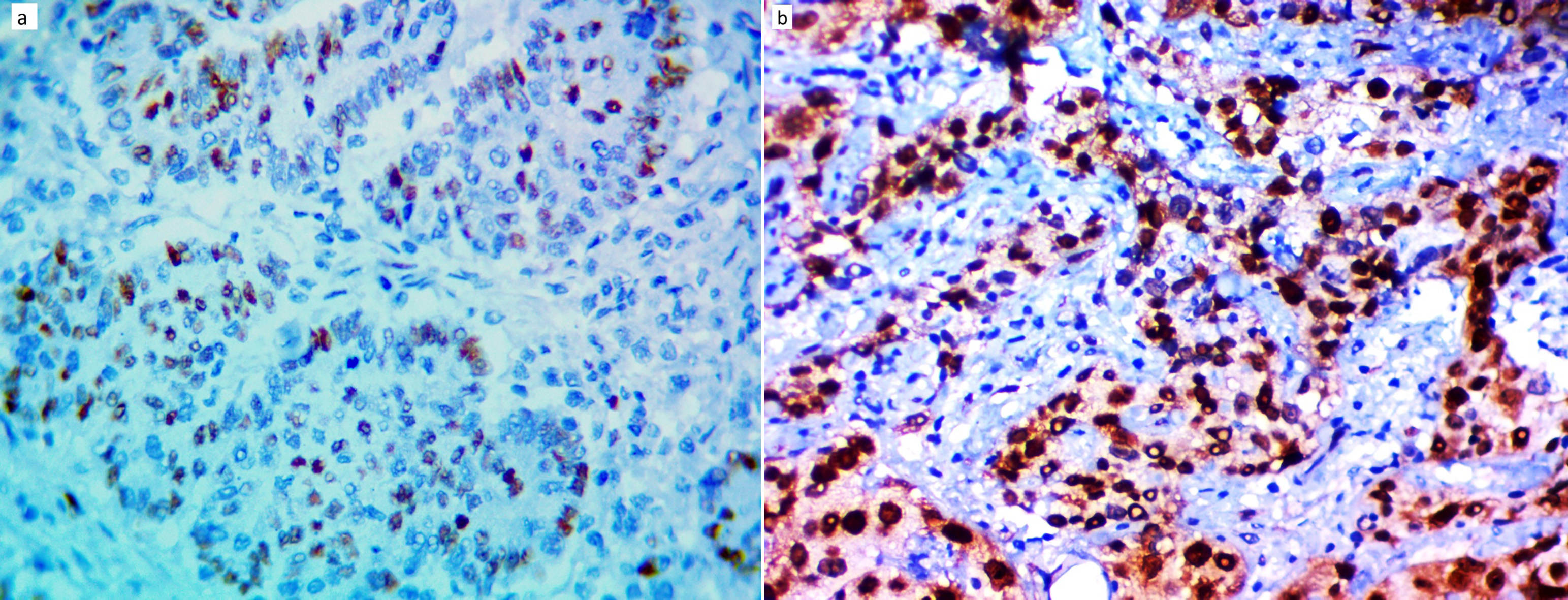
Representative images of AQP1 immunohistochemical staining A. Negative expression of AQP in normal mucosa (IHC, 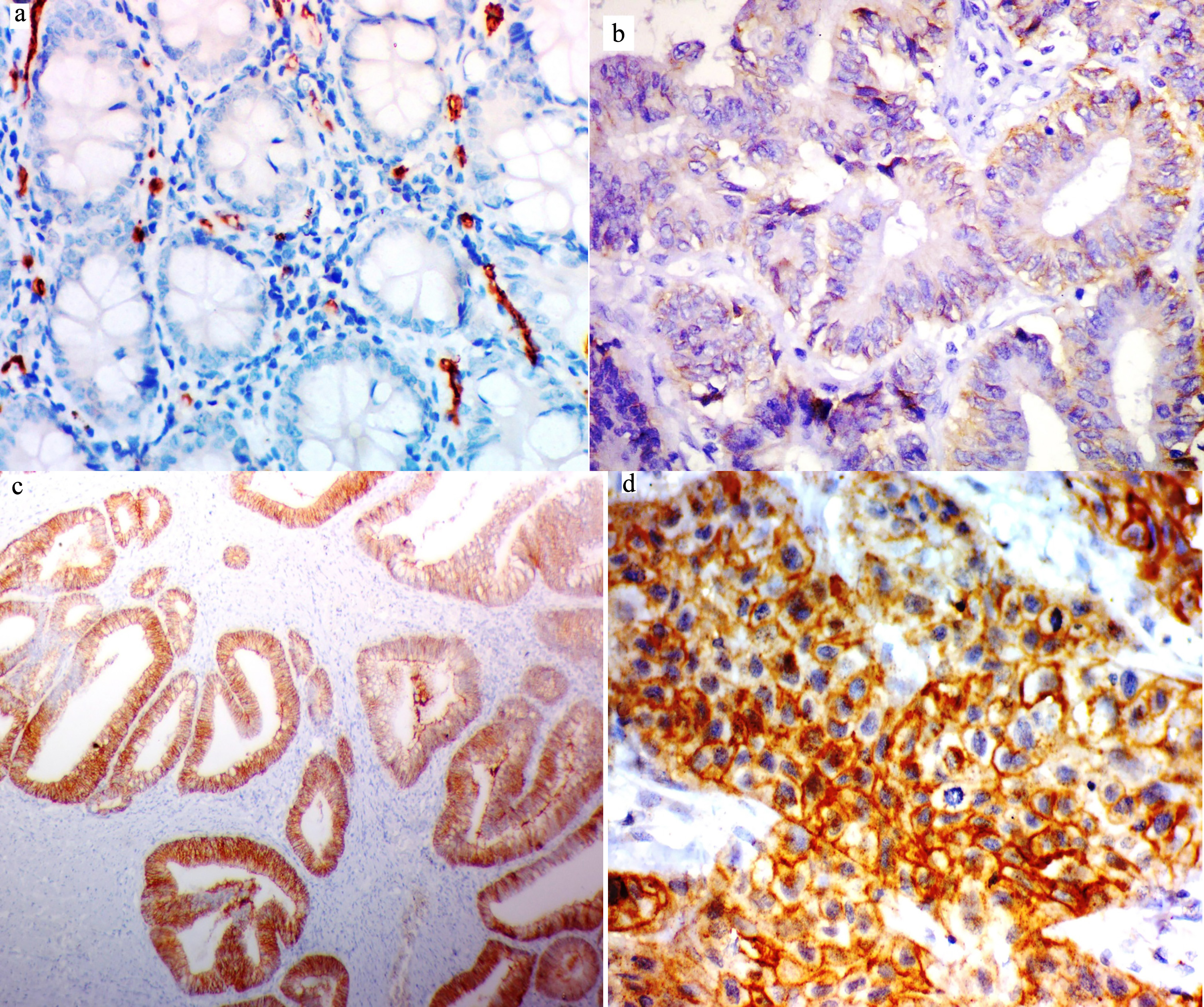
Positive nuclear ERCC1 expression was identified in 58.3% of the patients (Fig. 2). Positive ERCC1 expres-
Relation between clinicopathological features and immunohistochemical staining ERCC1, PAPR1 and AQP1 in colorectal carcinoma patients (
60)
Relation between clinicopathological features and immunohistochemical staining ERCC1, PAPR1 and AQP1 in colorectal carcinoma patients (
Categorical variables were expressed as number (percentage);
Relation between immunohistochemical staining ERCC1, PAPR1 and AQP1 and outcome in colorectal carcinoma patients received adjuvant chemotherapy (
Continuous variables were expressed as mean (95% CI); categorical variables were expressed as number (percentage);
sion was significantly associated with left-sided tumors (
High nuclear PARP-1 expression was observed in 63.3% of the patients (Fig. 3). High PARP-1 expression was significantly associated with the tumor grade and lymph node metastasis (
Positive membranous AQP1 expression was identified in 41.7% of the patients (Fig. 4). Negative AQP1 expression was significantly associated with the right-sided tumors vs. left-sided tumors (
Relation between immunohistochemical staining ERCC1/PAPR1/ AQP1 and outcome in colorectal carcinoma patients (
Continuous variables were expressed as mean (95% CI); categorical variables were expressed as number (percentage);
During the follow-up period, 23 patients (38.3%) suffered progressive disease after curative surgery (failure of therapy) and 11 patients (18.3%) died. Out of the 60 patients, 50 patients received adjuvant chemotherapy following curative surgery, while 10 patients were treated with surgery alone. Among the patients that have received adjuvant chemotherapy, relapse was detected in 20 cases (40%). Relapse had a positive association with the ERCC1 expression (
Statistical analysis of the survival data revealed that shorter DFS was significantly associated with positive ERCC1, high PARP-1, and positive AQP1 expression (
Kaplan Meier plot for overall survival (OS): A. Stratified by ERCC1 IHC staining (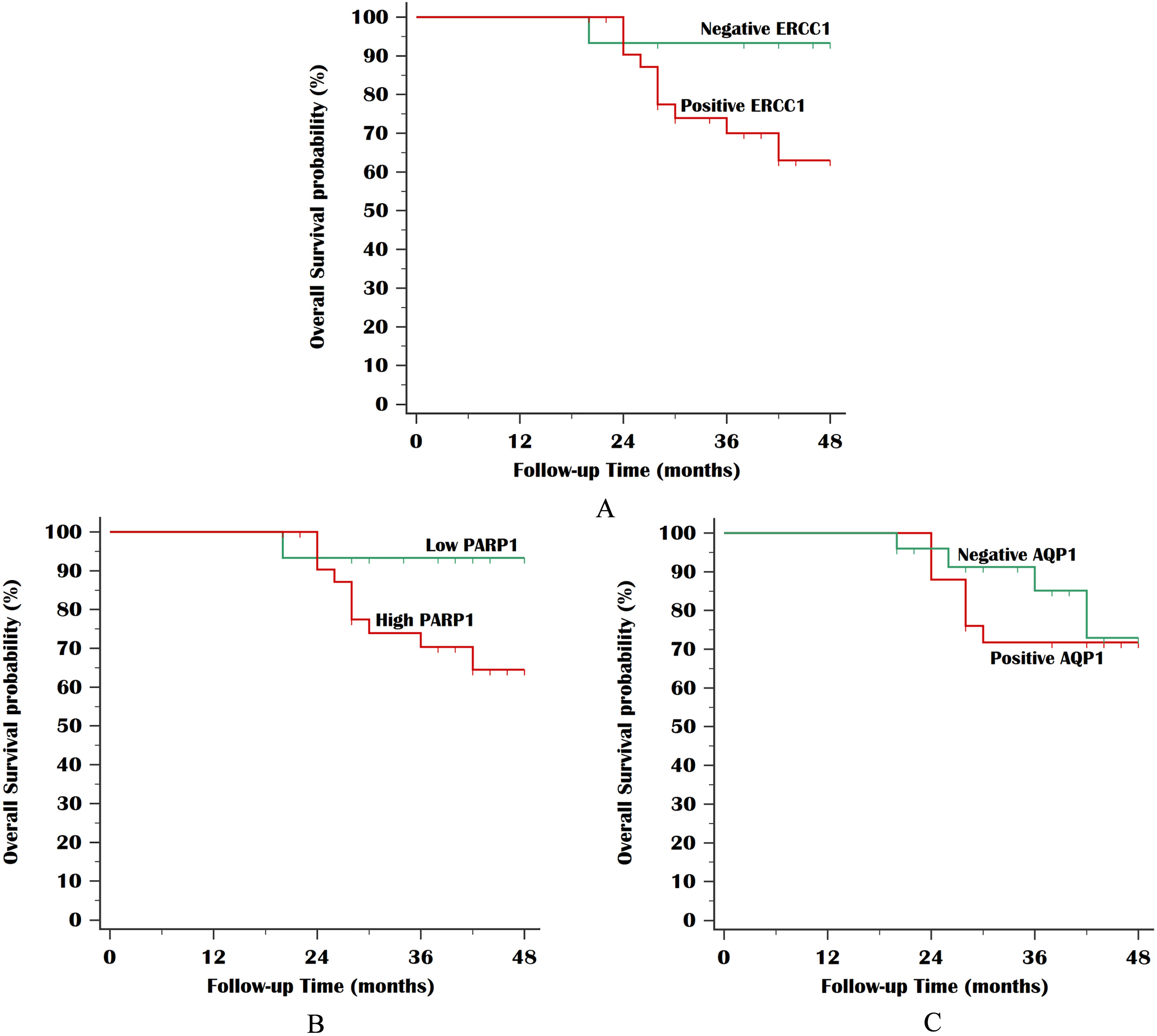
Kaplan Meier plot for disease-free survival (DFS): A. Stratified by ERCC1 IHC staining (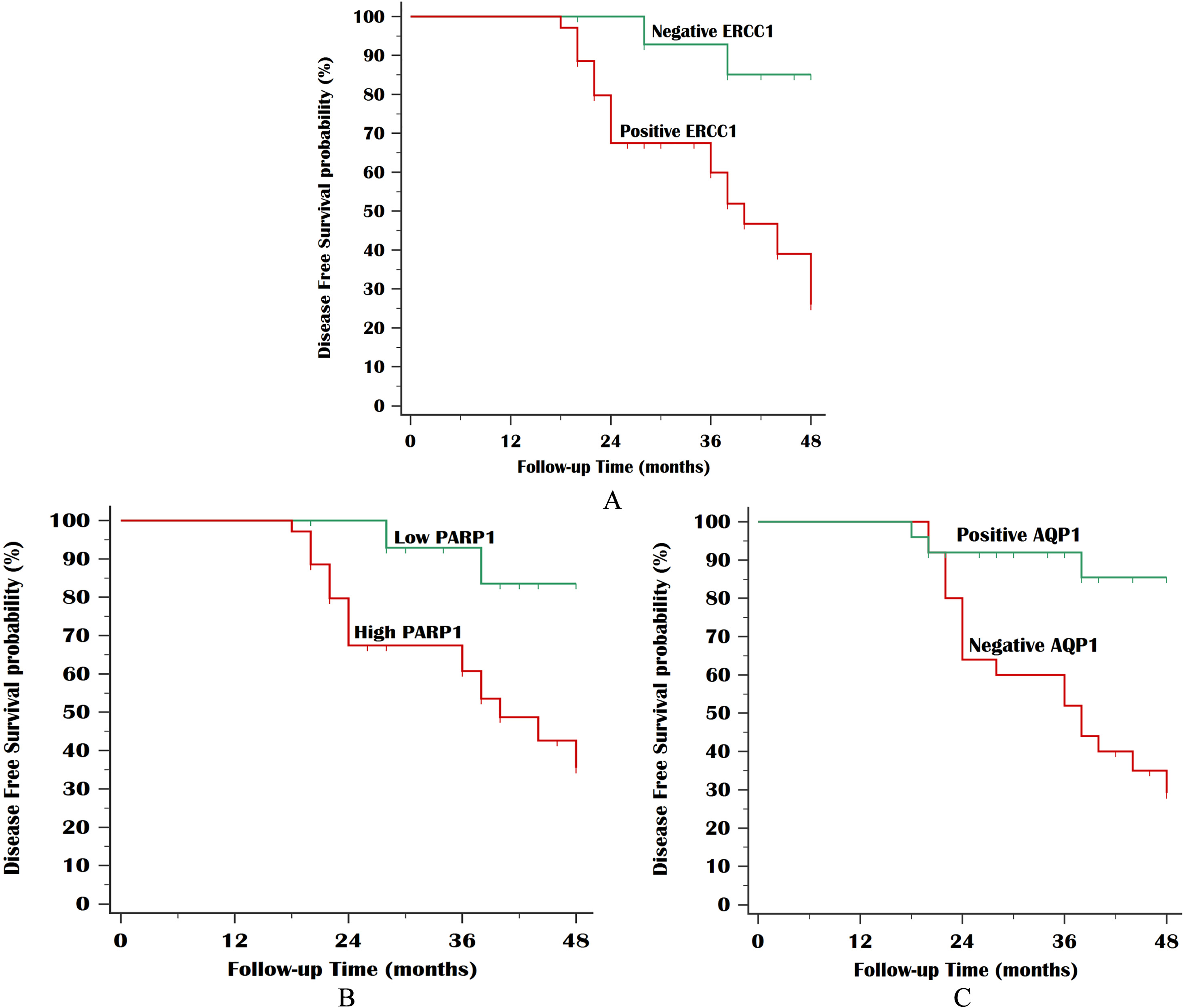
Colon cancer prognosis and therapy choice basically depend on pathology-related staging that infrequently predicts the CC clinical outcome perfectly. About 20–40% of patients with relatively low-grade, stage II rapidly progress and die [23].
Colon cancer therapy includes surgery, with or without adjuvant chemotherapy that is mainly based on 5-fluorouracil (5-FU). 5-FU results in thymidylate synthase inhibition, reduction of thymidine triphosphates available for DNA synthesis, and misincorporation of FdUTP (5-FdUrd triphosphate) into DNA [24]. Even with therapy, tumor progression is common within a few years, indicating drug resistance [25].
Oxaliplatin resistance is commonly seen in CC patients and so the recognition of sensitive predictive biomarkers is needed before the start of therapy to define patients who are prone to benefit from oxaliplatin. Understanding the mechanism of action of oxaliplatin-based chemotherapy on a molecular level combined with well-developed detection techniques of the genetic profile of the patients had led to great progress in individualizing treatment in advanced CC [7]. The genetic diversity between different CC cases may explain a superior response to one regimen than the others [26].
ERCC1 is a key enzyme in the NER pathway that is essential for repair of platinum-DNA adducts, and so associated with resistance to therapy containing platinum compounds. ERCC1 was suggested as a promising marker in the CC. ERCC1-overexpressing tumor cells are thought to be more resistant to platinum-based chemotherapy, suggesting that chemotherapy would be more effective in ERCC1-negative cancer [27].
In the present study, positive ERCC1 expression was noted in 58.3% of our cases. Its expression was significantly associated with the left-sided tumors and the aggressive tumor characteristics, including high grade, lymph node metastasis, and advanced tumor stage. Likewise in previous studies, ERCC1 positivity was observed in 45% and 55% of patients with colorectal cancer stage III, respectively [28, 21]. In a previous study, ERCC1 expression was detected in 72% of the studied cases and showed no significant association with the clinicopathological parameters apart from a tumor site [29]. On the other hand, Liang et al. reported that patients with nasopharyngeal carcinoma, the ERCC1 expression increased significantly with higher tumor stages and clinical stages (
Regarding the predictive value of ERCC1, some authors concluded that the high ERCC1 expression predicted bad response to chemotherapy and survival outcome [31], while other authors didn’t find any significant association between ERCC1 expression and clinical prognosis [32].
ERCC1 expression has been assessed by IHC in some previous clinical studies including patients who received oxaliplatin-based chemotherapy and showed a significantly longer DFS and OS in patients with low ERCC1 protein expression compared to those with high ERCC1 expression [21, 33, 34]. In the present study, we confirmed these findings where ERCC1 expression was related to tumor relapse (
In contrast to these results, some studies couldn’t reach the same conclusion, where Kim et al. found that ERCC1 expression was not significantly correlated with DFS in patients with stage II–III colon cancer treated with FOLFOX adjuvant chemotherapy [36]. Moreover, Lenz et al. didn’t find a significant predictive value of ERCC1 in cases received oxaliplatin-based regimens, but he remarked a suggestion that low ERCC1 expression was associated with longer PFS compared to those with high expression [37]. The contradictory results reported in these studies may be explained by differences in their design, heterogeneity in the target patients, the methods used to detect ERCC1 (IHC, RT-qPCR or SNP), different cutoff to stratify low vs. high besides differences in their endpoints. Furthermore, in some studies, due to the short follow-up period of 15 months, no correlation of DFS and OS with ERCC1 expression was observed [29].
Furthermore, we evaluated PARP-1 expression in the studied cases to address detailed information and expand our shortened knowledge about its expression level in this prevalent cancer. Despite considerable progress in the estimation of the PARP-1 role in DNA repair and various biological processes, the level of PARP-1 expression in different tumors, including CC is largely unknown but intensive research is ongoing. This knowledge is actually crucial to properly evaluate the outcome of clinical trials concerning targeted treatment with the PARP-1 inhibitors [38].
PARP-1 has a fundamental task in the maintenance of genome stability and the chromatin structure. PARP-1 has a role in different DNA repair pathways, as base excision repair (BER) and DNA single-strand break (SSB) repair [39]. Therefore, PARP-1-deficient mice show hypersensitivity to DNA damaging agents, with an enhanced genomic instability [40].
In our study, high PARP-1 expression was found in 63.3% of the studied cases. However, in other studies the expression was 97.7% [41], and 82.1% [42]. These findings may have a clinical implication because the estimation of PARP-1 expression in the tumor may improve the selection of patients with colon cancer for PARP inhibitor therapy. In a previous study [22], it was noted the high PARP-1 expression in 68.2% of the studied CC cases and there was a significant association of PARP-1 expression with the site of CC (
PARP-1 inhibition emerged as a therapeutic target, and recent studies verified dramatic enhancement of chemotherapeutic agents in the presence of PARP inhibition [43]. New data indicate that besides the catalytic inhibition of PARP activity, PARP inhibitors (PARPi) provoke cytotoxic PARP-DNA complexes through PARP “trapping” that augment the cytotoxicity of alkylating agents. It is therefore of extreme significance to spot the molecular character that acts not only as biomarkers for the patient stratification, but also proposes insights into the mechanisms of chemotherapy resistance [44].
Although numerous studies have analyzed the prognostic significance of AQP1 in human cancer, only few clinical studies have assessed the association between AQP1 expression and the response to chemotherapy. Therefore, the present study focused on evaluating the potential contribution of AQP1 expression to chemotherapeutic outcomes in patients with stage II–III colon cancer who received 5-FU-based adjuvant chemotherapy. In the current study, we noted positive expression of AQP1 in 41.7% of the cases with a significant relation to aggressive tumor features including high grade, lymph node metastasis, and tumor stage. In accordance with this, previous studies [45, 46] have revealed AQP1 expression in 41.8% and 35.8% of patients, respectively. This was significantly associated with tumor grade, advanced tumor stage, and lymph node metastasis. These findings suggest that a tumor-promoting role of AQP1 that particularly affect lymph node metastasis and vascular invasion. In vitro studies have also reported that AQP1 increases the potential of invasion and migration in CC cell lines [47, 48]. This may suggest that AQP1 has the potential to contribute to tumor progression, particularly in left-sided colorectal tumors. However, Kang et al. reported that marked positive AQP1 expression was negatively associated with lymph node metastasis in stage I–III colon cancer. Therefore, further studies are required to specifically clarify the clinical and biological significance of AQP1 in colon cancer [49].
Regarding the prognostic significance of AQP1 expression, Yoshida et al. concluded that AQP1 is a poor prognostic factor for overall survival (OS) in patients with stage II–III CC using multivariate analysis [46], whereas Kang et al. identified that AQP1 expression had no significant impact on OS or DFS in patients with stage I–III CC [49]. In contrast to these findings, in the present study, we identified a significant survival impact of AQP1 expression on DFS. Furthermore, our results revealed that patients with AQP1 positive cancer had improved therapeutic outcomes. Similar to our findings, Imaizumi et al. reported that patients with AQP1-negative CC exhibited an inferior response to chemotherapy and may even be harmed by 5-FU-based treatment as demonstrated by the reduction in DFS in the AQP1-negative CC chemotherapy-treated group [45]. These findings suggest that AQP1-positive colon cancer have a superior sensitivity to 5-FU-based adjuvant chemotherapy compared with AQP1-negative cases. Furthermore, there is currently no sufficient explanation as to why AQP1-negative tumors have a poorer prognosis following treatment with adjuvant chemotherapy, suggesting the requirement for underlying mechanistic investigation in future studies [45].
On the other hand, in ovarian cancer cell lines, AQP1 expression was associated with sensitivity to cisplatin [50]. Liu and Zhu indicated that AQP1 expression was downregulated by combination therapy of celecoxib and afatinib in a lung cancer cell line. In view of these results, it is hypothesized that AQP1 may potentially modulate the sensitivity to anticancer drugs. A potential explanation for this discrepancy in the clinical impact of AQP1 between cancer types may be due to the multifaceted roles of AQP1 across various organs and tumor cells [51].
In conclusion, ERCC1, PARP1, and AQP1 are adverse prognostic biomarkers of aggressive colon cancer. Moreover, oxaliplatin usage in adjuvant chemo- therapy may not be beneficial for patients with positive ERCC1, high PARP1, and AQP1-negative tumors. Therefore, selecting alternative chemotherapeutic regimens or even adding targeted agents as PARP inhibitors may present a novel strategy. We hope that subsequent investigations may confirm our findings, paving the way to the implementation of these biomarkers in routine clinical practice.
Footnotes
Conflict of interest
The authors declare that they have no conflict of interest.
