Abstract
BACKGROUND:
Both mitochondria and the Nrf2/Keap1 pathway are targets of cancer therapy. Reactive oxygen species released from mitochondria can activate Nrf2, and the Nrf2/Keap1 pathway affects glycolysis, oxidative phosphorylation, mitochondrial biogenesis and mitophagy.
OBJECTIVE:
This study investigates the associations between the expressions of proteins in the Nrf2/Keap1 pathway and those related to mitochondrial function and glycolysis in colorectal cancer (CRC) with or without metastasis.
METHODS:
The protein levels of HO1, Nrf2, Keap1, Bach1, p21, p62, NRF1, LC3, ATP5B, HSP60 and GAPDH in the normal and tumor tissues of 60 CRC subjects were determined by Western blot.
RESULTS:
The Keap1 protein levels, the ATP5B/HSP60 ratio and the BEC index were higher in the tumor than in the normal tissues of CRC with or without metastasis. The following clusters were found in the dendrogram: Nrf2 and p21 with ATP5B and GADPH in all the tissues and with NRF1 in all except the tumor tissues with metastasis; Bach1 with ATP5B and GAPDH in the tumor tissues; Keap1 with p62 in all the tissues, with LC3 in the tumor tissues and with NRF1 and HO1 in the tumor tissues with metastasis.
CONCLUSIONS:
Nrf2, Keap1, Bach1 and p21 have the association with the proteins related to mitochondrial functions different among the tissues of CRC with or without metastasis.
Introduction
Mitochondria are bioenergetic, biosynthetic and signaling organelles, and they are important mediators of tumorigenesis due to their impacts on apoptosis, oxidative stress, mitochondrial biogenesis and others [1]. Mitochondrial DNA (mtDNA) mutations are frequently found in cancer and are consistent with reactive oxygen species (ROS)-related derivation [2, 3]. Subjects with D-loop mtDNA mutation, found in 38.3% of colorectal cancer (CRC), have a poorer 3-years survival than those without [4]. A low mtDNA copy number in CRC is associated with a short 3-years survival [5]. Many drugs targeting mitochondria are under development for treating cancer, including CRC [6].
ROS generated by OXPHOS can free Nrf2 from the binding of Keap1 and activate p21 through p52. The nuclear translocated Nrf2 can increase the transcription of many genes (zigzag arrows), and its function on gene promoters can be regulated negatively by Bach1 but positively by p21. Nrf2 can regulate the expression of ME1, IDH1, G6PD and PGD, which affect the balance of glycolysis and OXPHOS that help to maintain 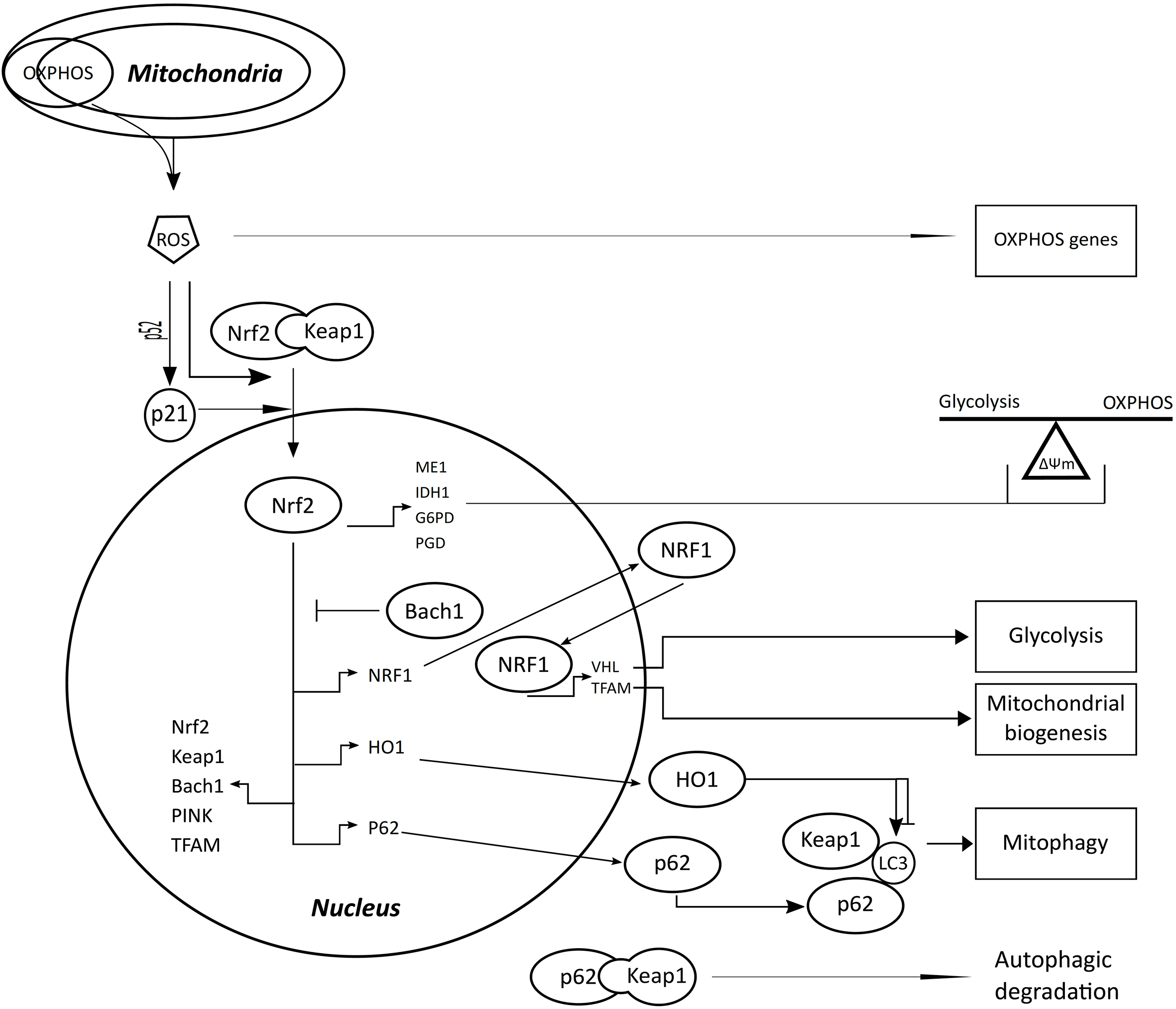
ROS has intimate relationships with cancer [7] and mitochondria [8]. Nuclear factor erythroid 2-like 2 (Nrf2, NFE2L2) is a key regulator of cellular antioxidant response [9] and is activated by oxidative stress [10]. The activation of Nrf2 leads to the transcription of Nrf2 itself, kelch like ECH associated protein 1 (Keap1), BTB domain and CNC homolog 1 (Bach1), p62 and many phase II detoxifying enzymes like heme oxygenase 1 (HO1) [11]. Nrf2 activity is negatively regulated by Keap1 in the cytoplasm [10] and by Bach1 in the nucleus [11]. Keap1 can sequester Nrf2 in the cytoplasm and targeting it for polyubiquitination by a Cullin3-based E3 ligase complex with subsequent proteasomal degradation [11]. The binding of p62 to Keap1 [12] can release Nrf2 and facilitate its nuclear translocation. Nrf2 binding to Keap1 is competed by p21, which can directly interact with DLG and ETGE motifs of Nrf2 [13]. Both p21 and p62 can interfere with the interaction between Keap1 and Nrf2, and promote the transcription of genes regulated by Nrf2 (Fig. 1). The Nrf2/Keap1 pathway molecules are correlated with tumor progression and become therapeutic targets in different types of cancer, including CRC [9, 14, 15, 16, 17, 18].
Nrf2 contributes broadly to the regulation of mitochondria including biogenesis, respiration/ATP production and mitophagy [12]. Nuclear respiratory factor 1 (NRF1) is a transcription factor of many genes involving in mitochondrial biogenesis [19], which is induced by nutrient deprivation and oxidative stress [20]. Nrf2 promotes the transcription of NRF1 and mitochondrial transcription factor A (TFAM), and NRF1 also increase the expression of TFAM with the participation of peroxisome proliferator-activated receptor
ROS, primarily generated in mitochondria, plays a critical role in tumorigenesis and interacts with many oncogenic pathways [26]. In CRC, ROS contributes to the genesis and progression of cancer, and Nrf2 plays a dual role in the tumorigenesis [27]. This study uses Western blot to investigate the correlations among the protein levels of molecules in the Nrf2/Keap1 pathway and those related to biogenesis (NRF1), OXPHOS (ATP5B) and mitophagy (LC3) of mitochondria and glycolysis (GAPDH) in both the tumor and normal tissues of CRC with or without lymph node or distant metastasis.
The biobank of Chang Gung Memorial Hospital, Keelung routinely collects the resected specimens of subjects with a pre-operative diagnosis of CRC. This study selected CRC subjects after surgery from the biobank database randomly, retrospectively and blind to the applicant. The frozen tumor and normal colon tissues of 60 CRC subjects with their de-identified clinicopathologic features were obtained from the biobank. Histopathologic evaluation was performed based on the diagnostic criteria of the World Health Organization [28], and all of the tumors were diagnosed as adenocarcinoma. The staging assessment was carried out according to the American Joint Committee on Cancer TNM-classification (8th edition) [29]. A metastatic status denoted metastasis to lymph node or distant sites. The work was approved by the Institutional Review Board of Chang Gung Memorial Hospital (no. 201601829B0C601).
Protein extraction
Tissue protein was extracted as previously described [30]. In brief, the liquid nitrogen frozen tissues thawed at room temperature were washed and ground by a homogeniser. The ground tissues were treated with a phosphatase and protease inhibitor cocktail, vortexed and centrifuged. The supernatant was collected, quantitated and stored at
Western blot
The primary antibodies used in this study were anti-HO1 (ab-13243; Abcam, Cambridge, MA; 1: 600 dilution), anti-Nrf2 (sc-722; Santa Cruz Biotech, CA, USA; 1: 900), anti-Keap1 (sc-33569; Santa Cruz; 1: 600), anti-Bach1 (NBP1-88722; Novus Biologicals, Littleton, CO; 1: 250), anti-p21 (ab-109520; Abcam; 1: 1000), anti-p62 (ab-109012; Abcam; 1: 1000), anti-GAPDH (sc-47724; Santa Cruz; 1: 1000), anti-NRF1 (ARG58207; Arigo Biolaboratories, Hsinchu, Taiwan; 1: 1000), anti-LC3 (sc-271625; Santa Cruz; 1: 1000), anti-ATP5B (sc-55597; Santa Cruz; 1: 2000), anti-HSP60 (heat-shock protein 60) (sc-59567; Santa Cruz; 1: 1000) and anti-
Examples of protein electrophoresis. N: normal tissue, T: Tumor tissue.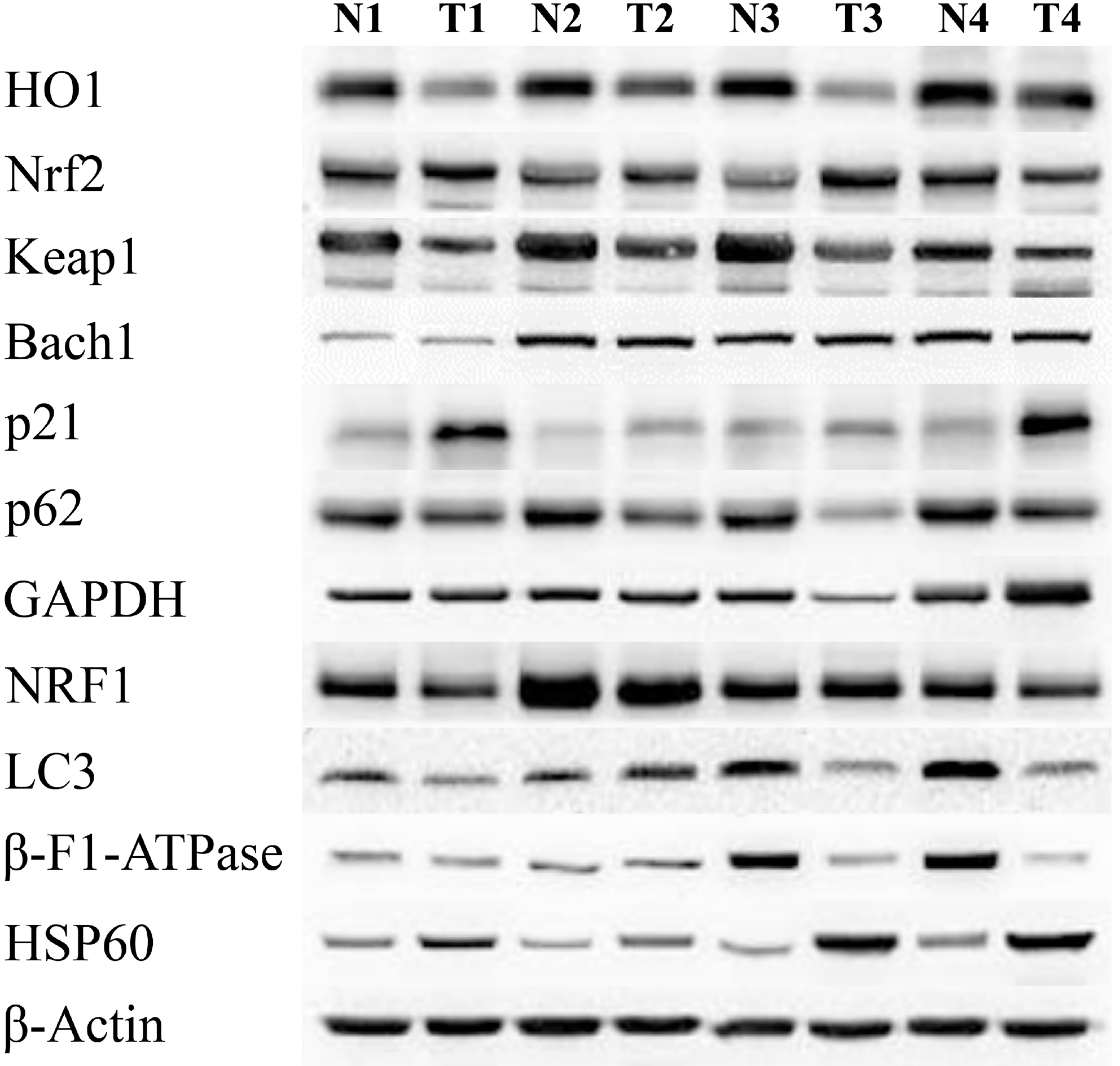
HSP60, which is mainly located in mitochondrial matrix, assists protein folding, unfolding and degradation in mitochondria [31]. The bioenergetic index of the cell (BEC) index was derived with ATP5B/HSP60/ GAPDH [22].
Statistical analysis
The paired or unpaired Wilcoxon signed-rank test was used to detect the differences of protein levels between tissues. The correlations between protein levels were assessed by Spearman’s rho test. Hierarchical clustering with nonparametric multiscale bootstrap resampling was performed using the R statistical package (
Results
Table 1 shows the characteristics of 60 subjects with CRC.
Characteristics of 60 subjects with colorectal adenocarcinoma
Characteristics of 60 subjects with colorectal adenocarcinoma
Differences of protein levels in the tissues of CRC with or without metastases
BEC: bioenergetic index of the cell
Spearman’s correlation coefficients of protein levels and BEC between molecules in the normal or tumor tissues of CRC subjects with or without metastasis. A bold italic number denotes a correlation with 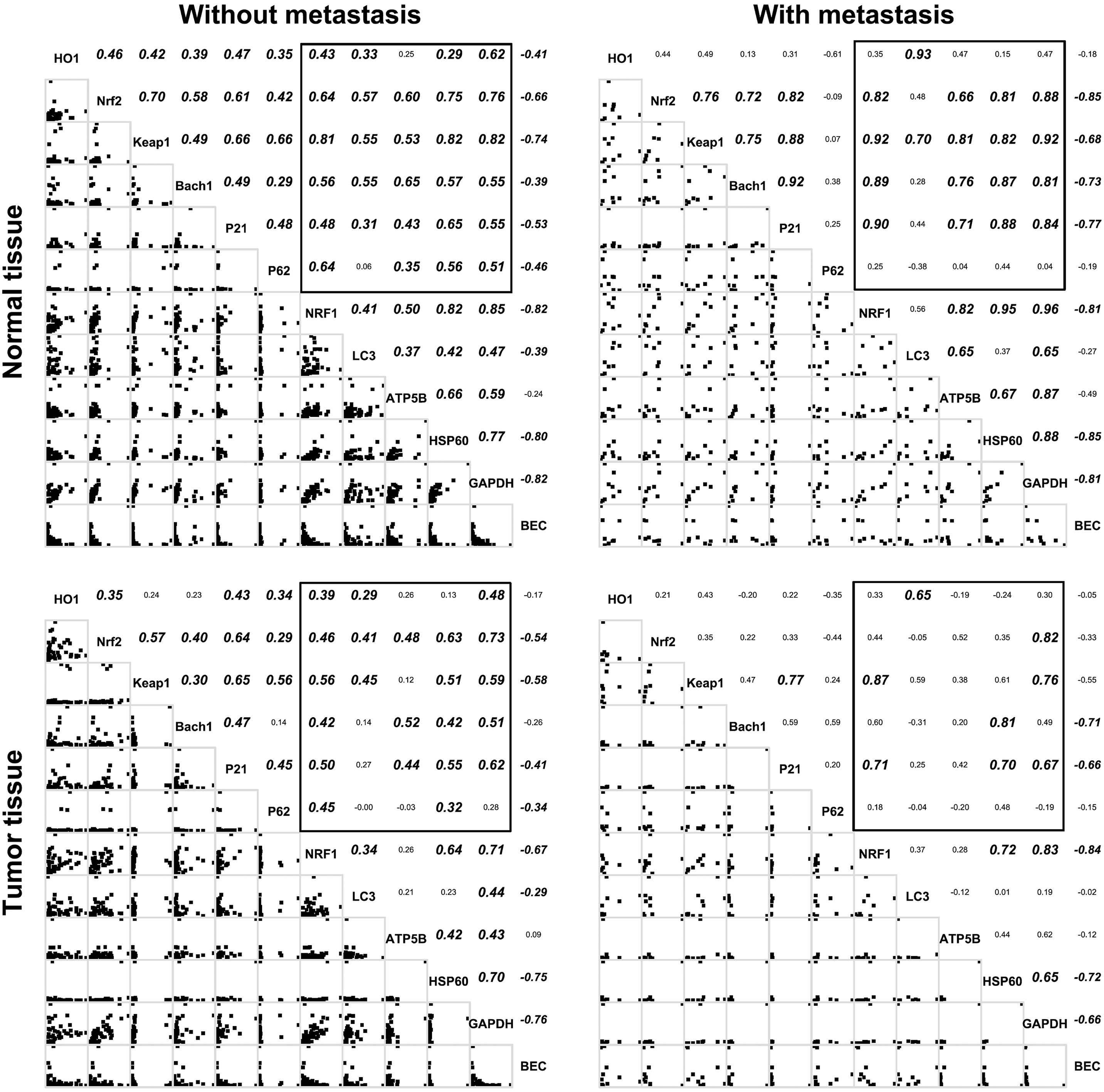
Cluster dendrograms showing the approximately unbiased probability (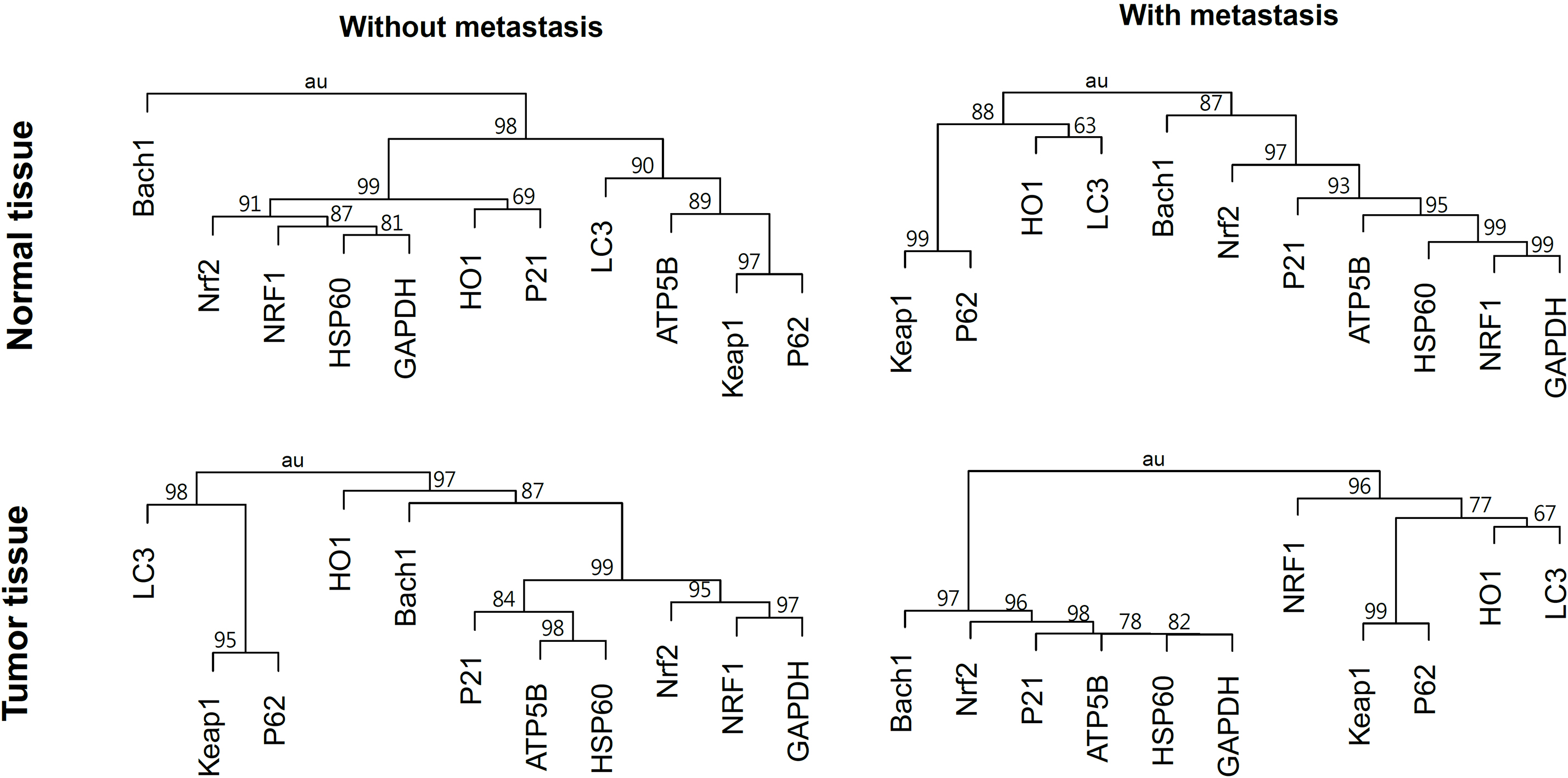
Table 2 lists the protein levels in the normal and tumor tissues of CRC subjects with or without metastasis. The tumor had higher HO1 and Keap1 protein levels, and lower p21 and HSP60 protein levels, than normal tissue in CRC with or without metastasis. The protein levels of p62 and ATP5B were higher in the tumor than in the normal tissues of CRC with and without metastasis respectively. The protein levels in the tissues of CRC subjects with or without an advanced stage (III or IV) had the same pattern of differences, and those with tumor located in colon or rectum had a similar trend (data not shown).
Figure 3 presents the correlations between protein levels and the BEC index in the tissues of CRC subjects with or without metastasis. The protein levels of NRF1 were correlated with those of Nrf2, Keap1 and Bach1 in all except the tumor tissues with metastasis, where they were not correlated with those of Nrf2 and Bach1. The protein levels of LC3 were correlated with those of Keap1 in all the tissues, and those of HO1 in the tissues with metastasis. The protein levels of ATP5B were correlated with those of Nrf2, Keap1, Bach1 and p21 in all except the tumor tissues, where they were not correlated with those of Keap1. The protein levels of HSP60 were correlated with those of Nrf2, Keap1, Bach1 and p21 in all except the tumor tissues without metastasis, where they were not correlated with those of Keap1. The BEC index was negatively correlated with the protein levels of Nrf2, Keap1 and p21 in all the tissues.
Figure 4 shows the hierarchical cluster dendrograms for the protein levels in the normal and tumor tissues of CRC subjects with or without metastasis. Nrf2, p21, ATP5B, HSP60 and GAPDH formed a cluster in all the tissues with Bach1 joined in the tumor tissues of CRC with or without metastasis. NRF1 joined the cluster formed by Nrf2, p21 and others in all except the tumor tissues of CRC with metastasis, where it had a close proximity to Keap1, p62, HO1 and LC3. Keap1 had a proximity close to p62 in all the tissues and to LC3 in all except the normal tissues of CRC with metastasis.
The tumor had the protein levels of HO1 and Keap1 higher and those of p21 lower than the normal tissues in CRC with or without metastasis. The ATP5B/HSP60 ratio and the BEC index were higher in the tumor than the normal tissues of CRC with or without metastasis. The hierarchical cluster dendrograms had shown the molecules of Nrf2/Keap1 pathway had different proximities to the proteins related to glycolysis/OXPHOS, mitochondrial biogenesis and mitophagy in different tissues with different metastasis status.
Microsatellite instability, a result of impaired DNA mismatch repair system (MMR), and K-Ras are frequently found in colorectal cancer [34]. Oxidative stress can inactivate MMR [35] and activate K-Ras [36] which can in turn upregulate the Nrf2/Keap1 pathway [37]. Cancer may accelerate metabolism, resulting in large amount of ROS, and develops mechanisms to resist excessive oxidative stress including, the guidance of glycolysis into the pentose phosphate pathway [7]. Mitochondria affect many aspects of cellular metabolism by producing precursors for macromolecules like lipids, proteins, DNA and RNA, generating metabolic by-products such as ROS and ammonia, and clearing or utilizing waste products [38]. The Nrf2/Keap1 pathway affects several functions of mitochondria, including biogenesis, bioenergetic and mitophagy [12, 21, 23]. The ROS produced by mitochondria can activate Nrf2 by freeing it from Keap1 binding [8, 9, 10, 27, 38]. Mitochondria and the Nrf2/Keap1 pathway contribute the tumorigenesis of CRC [5, 6, 14, 15, 16, 27].
ROS can promote proliferation, metastasis and angiogenesis in tumor, and activate Nrf2, which is the master regulator of the antioxidant response [7]. HO1 is anti-oxidative, and its overexpression in cancer, including CRC, is influenced by factors other than Nrf2, and promotes tumor proliferation and survival [39]. The protein levels of HO1 were higher in the tumor than the normal tissues in CRC with or without metastasis. Keap1 and p21 can down-regulate and up-regulate Nrf2 respectively [10, 13]. However, the tumor had higher Keap1 protein levels and lower p21 protein levels than the normal tissues, without significant difference in Nrf2 levels between the two tissues. Nrf2 is regulated by a complex network in which Keap1 and p21 exert their influences only at the post-transcriptional level [40]. The protein levels of p62 were significantly higher in the tumor than normal tissues in CRC with metastasis, and a similar trend was seen in those without metastasis. Keap1 can be degraded, as it is bound by p62 [41], possibly contributing to the similar protein levels of Nrf2 in the tumor and normal tissues of CRC with or without metastasis.
The protein levels of ATP5B, an essential component of OXPHOS [22], the ATP5B ratio and the BEC index were higher in the tumor than the normal tissues of CRC without metastasis and a similar trend was observed in those with metastasis. The ATP5B/HSP60 ratio and the BEC index are lower in the tumor than the normal tissues in a cohort of CRC without lymph node and distant metastasis [22], and the different results in this study may be due to the inclusion of subjects with advanced stages. The peri-tumor glycolytic stromal cells may produce mitochondrial fuels to drive OXPHOS and mitochondrial metabolism in the tumor cells, in the so-called “reverse Warburg effect” [42]. The tumor cells do not exhibit the Crabtree effect, and need high rates of both glycolysis and OXPHOS to meet the demand for anabolic processes [43]. Cancer cells undergo aerobic glycolysis with mitochondrial respiration preserved to a various extent in different tumors [1, 43]. Research has shown that the metastatic phenotype requires OXPHOS and increased mitochondrial metabolism [44]. Although glycolysis is preferred over OXPHOS in CRC [44], both may be needed for tumor growth and metastasis.
Nrf2 can upregulate the transcription of NRF1 by binding the antioxidant response element of its promoter [12]. The regulation of Nrf2 activation by Keap1 and Bach1 is modified by p21 and p62 [10, 11, 12, 13]. In the cluster dendrogram, NRF1 had proximity close to Keap1 in the tumor tissues with metastasis, but to Nrf2 and p21 in all the others. The autophagic adaptor protein p62 can bind Keap1 and activate mitophagy [12]. Keap1 and p62 were within the same cluster in all the tissues. LC3 may form a complex with Keap1 and p62 [24], which may be not present in the normal tissues. The control of LC3-mediated autophagy by HO1 and carbon monoxide is complex and varied in different cell types [25]. This study found a close proximity between HO1 and LC3 in the tumor tissues with metastasis. The intricate interplay between biogenesis and mitophagy is important for mitochondrial homeostasis, which is important for cellular adaption to stress [45]. The protein levels of Keap1, NRF1, LC3, p62 and HO1 in the tumor tissues with metastasis formed a cluster in the dendrogram. The experimental results imply that Keap1 may play an important role in balancing mitochondrial biogenesis and mitophagy in the tumor tissues of CRC with metastasis. Nrf2 can increase the transcription of HO1 [11], and HO1 can indirectly activate Nrf2 via mitochondrial H
Conclusions
Numerous agents targeting mitochondria and the Nrf2/Keap1 pathway in cancer are under development [6, 16, 18, 23, 43]. The Nrf2/Keap1 pathway has intensive interactions with mitochondria [12]. The close association of glycolysis/OXPHOS with Bach1, in addition to Nrf2 and p21, in the tumor tissues and that of Keap1 with mitochondrial biogenesis/mitophagy in the tumor of CRC with metastasis suggests the possibility of combining agents targeting mitochondria and the Nrf2/Keap1 pathway in CRC treatment. However, the strengths of this study are compromised due to the retrospective and correlational nature of this study with additional limitations of a small sample size and limited clinicopathological information. A large-scale prospective study with the exploration of molecular mechanisms is needed to verify the preliminary results found in this one.
Footnotes
Acknowledgments
We thank the Biobank of Chang Gung Memorial Hospital at Keelung for providing the tissues and de-identified clinicopathologic features of CRC subjects. This investigation was supported by grants from Chang Gung Memorial Hospital, Keelung (CMRPG2H0041).
