Abstract
BACKGROUND:
This study aimed to evaluate the relationship between survivin expression and melanoma after 5-aminolevulinic acid (5-ALA)-mediated sonodynamic therapy.
METHODS:
Immunohistochemistry was used to detect survivin protein expression in human melanoma clinical samples. Subsequently, the effects of 5-ALA-mediated sonodynamic therapy were determined by measuring the volume of melanoma xenografts and the bodyweights of melanoma-bearing nude mice. The MTT assay was used to detect the viability of melanoma B16-F10 cells under the action of 5-ALA-mediated sonodynamic therapy, and Western blotting and PCR were used to detect survivin expression in melanoma cells and in the melanoma-xenograft model.
RESULTS:
Survivin expression was significantly upregulated in human melanoma tissues compared with that of non-melanoma tissues. In the in vivo case, 5-ALA-mediated sonodynamic therapy significantly delayed tumor growth, prolonged the survival of mice, and inhibited the expression of survivin. In the in vitro case, 5-ALA-mediated sonodynamic therapy inhibited B16-F10 cell proliferation and decreased survivin expression at both protein and mRNA levels.
CONCLUSION:
Our results suggest that 5-ALA-mediated sonodynamic therapy inhibited B16-F10 cell proliferation and melanoma-xenograft growth and prolonged survival of melanoma-bearing nude mice, which might be through downregulation of survivin expression.
Introduction
Melanoma is a tumor of melanocytes and is the most aggressive and fatal cancer that is found primarily in the skin [1]. According to epidemiologic studies, the incidence of malignant melanoma has grown rapidly over the past few decades [2]. Although governments, industries, and academia have made extraordinary efforts in melanoma treatment, the low overall survival and poor prognosis of melanoma patients remain a challenge in clinical practice. In addition, researchers have achieved reductions in melanoma-cell death via the combined activation of anti-apoptotic factors, inactivation of pro-apoptotic effectors, and enhancement of the survival signals during melanoma progression; this has confirmed that melanoma is highly resistant to chemotherapeutic drugs [3, 4]. Thus, another common feature of melanoma is its tolerance to chemotherapy and/or radiotherapy. Overcoming tumor resistance against one or more of the above factors may help to improve the clinical outcomes of patients with malignant melanoma.
Survivin is an evolutionarily conserved eukaryotic protein that plays an important role in cell division. Survivin is the smallest and most powerful inhibitor of apoptosis protein (IAP) discovered thus far. Survivin plays important roles in tumorigenesis and tumor progression by inhibiting apoptosis and promoting cell-cycle progression, tumor metastasis and angiogenesis [5, 6]; it also plays an important role in autoimmune diseases [7]. Habibie et al. showed that survivin is essential for the survival of melanoma cells in vitro and in vivo, and the overexpression of survivin protects melanoma cells from apoptosis [8]. Reduced survivin expression inhibits melanoma growth and lung metastasis [9]. Another study reported that ribozyme-mediated downregulation of survivin enhanced the sensitivity of melanoma cells to chemotherapy [10]. Fenstermaker et al. demonstrated that the application of survivin antibodies provides specific targets for antibody-mediated tumor immunotherapy [11]. Therefore, survivin has become a new target for the treatment of malignant tumors. Other, targeting survivin may be an effective strategy for overcoming the drug resistance of melanoma, which may better control and treat melanoma.
Sonodynamic therapy (SDT) is a method used to sensitize cells by ultrasonic irradiation [12, 13]. The SDT process involves injecting an ultrasound sensitizer to selectively accumulate in the lesion and locally irradiate the tumor with low-intensity ultrasound. This process activates an ultrasound-sensitizing drug to induce apoptosis and necrosis through direct mechanical stress and indirect chemical reactions. SDT has achieved significant therapeutic effects in improving tumor-cell proliferation [14, 15], angiogenesis, and immunity [16]. Our previous study showed that ALA-mediated SDT utilizing reactive oxygen species (ROS) on the p53-miR-34a-Sirt1 axis inhibits the proliferation of melanoma cells [21]. Inhibition of miR-34a expression downregulates the expression of specific downstream targets, survivin, Ki67, and B-cell lymphoma 2 (Bcl-2), and arrests the cells in the G2/M phases of the cell cycle [17, 18]. However, relationships between SDT and survivin—and between 5-ALA-mediated SDT, surviving, and melanoma—have rarely been reported.
The present study was designed to evaluate the antitumor effect of 5-ALA-mediated SDT on malignant melanoma in vitro and in vivo. Our experiments confirmed that SDT inhibited melanoma growth in mice by inhibiting survivin expression to better prolong the survival of melanoma-bearing nude mice compared to that of mice receiving monotherapy or without ultrasound therapy.
Materials and methods
Cell lines and reagents
The murine-melanoma B16-F10 cell line was obtained from the Shanghai Experimental Center at the Chinese Academy of Sciences. The cells were cultured in 10% fetal bovine serum (FBS)-supplemented Dulbecco’s modified Eagle’s medium (DMEM) in a humidified incubator at 37
Clinical samples
Archived paraffin-embedded human melanoma tissues and matched adjacent normal tissues were obtained from 24 patients who underwent operation in the affiliated hospital of Jining Medical University from 2014 to 2016, and melanoma was diagnosed pathologically. All specimens were anonymously handled according to the ethical and legal standards. This study was approved by the Ethics Committee of Jining Medical University.
Model of tumor therapy
Approximately 6–8 week-old male BALB/C athymic nude mice were obtained from the Slack’s Company of Shanghai (Shanghai, China). B16-F10 cell suspensions (1
Treatment protocols
Tumor-therapy experiments were performed as described previously [19], with minor modifications. B16-F10 cells were divided into four groups: control (Control), Ala alone (ALA), ultrasound alone (US), and ultrasound plus ALA (SDT). For the ALA and SDT groups, the cells were incubated with 1 mM ALA in the dark. Instead of ALA, an equivalent quantity of medium was used for the Control and US groups. After 4 h of incubation, the cells in the US and SDT groups were exposed to ultrasound (1.1 MHz, 1 W/cm
For the in vivo experiments, tumor-bearing mice were randomly segregated into four groups, with 24 mice in each group. ALA and SDT groups received 200 mg/kg ALA by intraperitoneal injection. After 4 h of administration of ALA, the mice in the US and SDT groups were irradiated by ultrasound (1.0 MHz, 1 W/cm
Immunohistochemistry
Analysis and evaluation of immunohistochemical results were performed as previously described [20]. The concentration of the primary antibody was 1:100 for survivin.
Cell-proliferation assay
B16-F10 cells were seeded into 96-well plates (2
Western-blot analysis
Cells and tissue were lysed in RIPA buffer (Beyotime Biotechnology, China). Protein concentrations were determined using a BCA Protein Assay Kit (Beyotime Biotechnology, China). For immunoblotting analysis, 50
Total RNA extraction and real-time PCR
Total RNA was extracted from the cells and tissues using Trizol reagent (Invitrogen) and cDNA was synthesized using the High Capacity cDNA reverse Transcription Kit (Applied Biosysterms). Real-time RT-PCR was performed using the Power SYBR Green PCR master mix (Applied Biosystems) on an ABI 7900HT System. GAPDH was used as an endogenous control.
All primers were obtained from Invitrogen (Shanghai, China). Sequences of primers were listed in Table 1.
Primers used for real-time RT-PCR
Primers used for real-time RT-PCR
Data are presented as the mean
Results
The expression of survivin in human melanoma tissues
To investigate the role of survivin in melanoma progression, we first conducted immunohistochemistry for the survivin in a panel of human melanoma tissues. Survivin expression was observed mainly in the cytoplasm or nucleus. As expected, the expression of survivin was significantly up-regulated in human melanoma tissues compared to that of no-melanoma tissues (survivin IOD: 13.23
Representative pictures of immunohistochemical staining of survivin in human non-melanoma and melanoma tissues (magnification: 200X). (A) The expression of survivin in non-melanoma, (B) The expression of survivin in melanoma, The black arrows show areas of positive expression. (C) Survivin is less expressed in the human non-melanoma group and more expressed in the melanoma group. Data are presented as the mean + SD (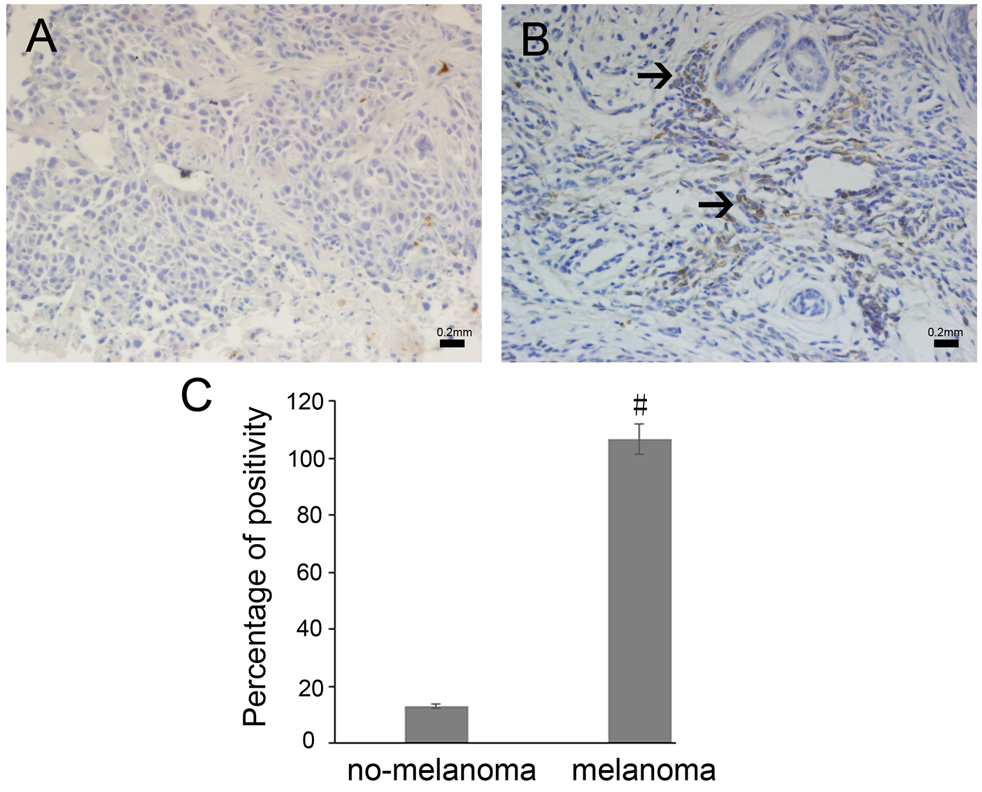
In order to test the in vivo efficacy of SDT, B16-F10 cells were injected subcutancously into the right flanks of BALB/C athymic nude mice. The treatment was initiated at seven days after melanoma-cell inoculation. At the end of the two-week treatment, the tumor volume of the control group was 767.99
Inhibitory effects SDT treatment in vivo. (A) SDT suppresses mouse melanoma B16-F10 tumor-xenograft growth in BALB/c athymic nude mice. (B) The bodyweights of mice were recorded and plotted each day. (C) Overall survival-analysis results by the Kaplan-Meier survival curves (long-rank test, 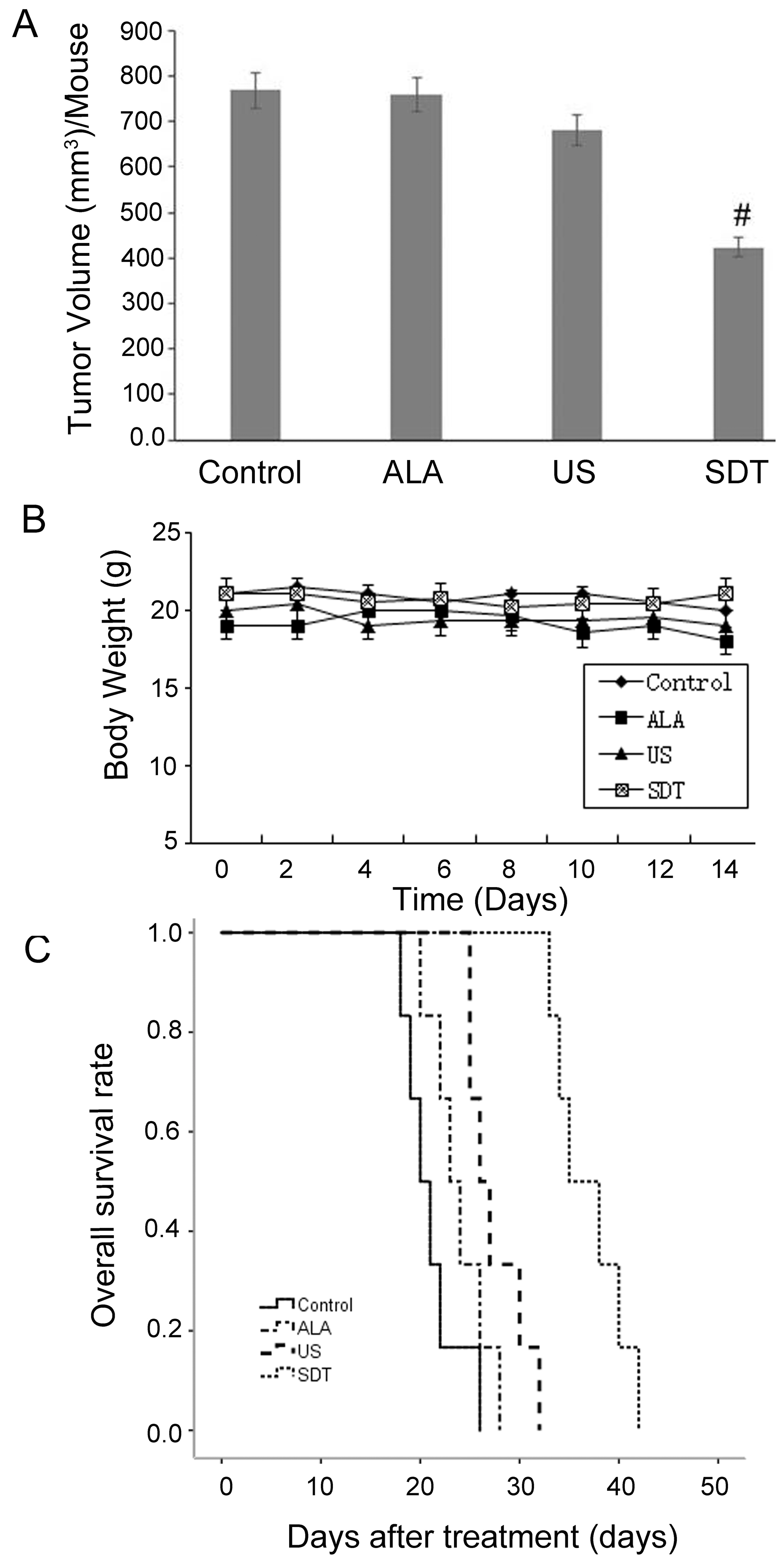
Based on our previous study, SDT can suppress tumor growth through inhibiting the expression of cyclin D1 (CCND1), cyclin-dependent kinase 6 (CDK6), and silent information regulator 1 (SIRT) [16]. We hypothesized that melanoma tumor-growth suppression by SDT treatment is associated with the inhibition of survivin, as this protein is known to be aberrantly activated in melanoma. In order to test our hypothesis in vivo, tissue from each experimental group was lysed and levels of survivin were examined by Western blotting. Our results showed that SDT treatment significantly reduced the protein levels of survivin, as compared with that of untreated control tumors. There were no significant differences among the levels of survivin in the Control, ALA, and US-alone groups (Fig. 3A).
Expression levels of survivin protein and mRNA in vivo. (A) Survivin protein expression was inhibited after SDT treatment. 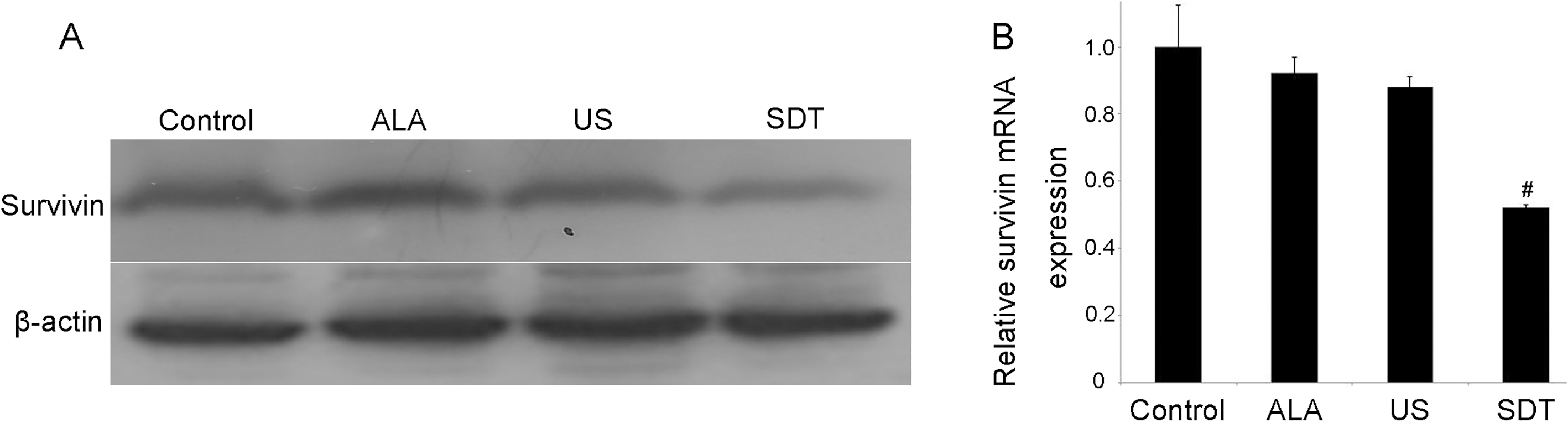
To confirm our Western-blotting results, we analyzed the mRNA levels of survivin in four groups. Our results showed that the mRNA levels of survivin were significantly lower in the SDT group than in the other three groups (
Using microarray analysis, we found that the expression of survivin was decreased significantly after treatment with SDT compared with that of the Control, ALA, or US groups (data not shown).
To confirm our results in vivo, the anti-cancer efficacies of ALA and US were investigated in the murine-melanoma B16-F10 cell line. An MTT assay showed that the cell-viability rate in the ALA group was 96.4%, and ALA alone showed no apparent cytotoxic effect on B16-F10 cells compared with that of the control. The cell-proliferation rate in the US group was 81.6%, which declined to 70% in the SDT group. This finding indicated that SDT significantly suppressed cell proliferation in the B16-F10 melanoma cell line (
Inhibitory effects of SDT treatment in vitro. Cells were subjected to the indicated treatments in the Materials and Methods. After 24 h, cell proliferation was assessed by the MTT assay. Data are presented as the mean 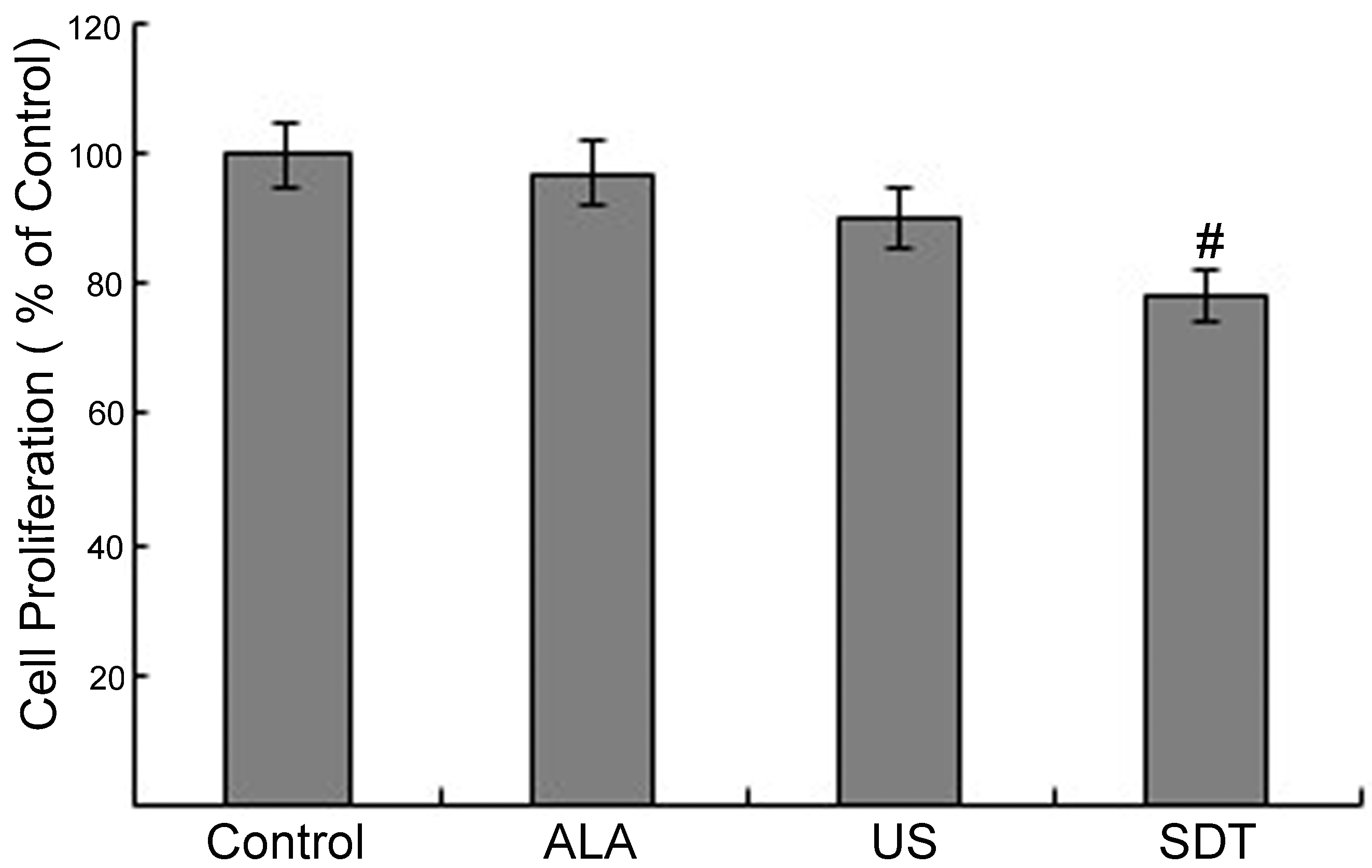
After confirming cell-growth inhibition by ALA and US treatments, we analyzed whether this inhibition was associated with the modulation of survivin. We evaluated the levels of survivin in B16-F10 melanoma cells by Western Blotting and RT-PCR. ALA treatment alone did not change the protein and mRNA levels of survivin as compared to those of the control group, whereas US-alone treatment slightly reduced the levels of survivin. Furthermore, the SDT treatment strongly suppressed the protein and mRNA levels of survivin in the B16-F10 melanoma cell line (Fig. 5). Collectively, these findings suggest that SDT may suppress tumor-cell proliferation through decreasing survivin in B16-F10 melanoma cells.
Expression levels of survivin protein and mRNA in vitro. (A) Survivin protein expression was inhibited after SDT treatment. 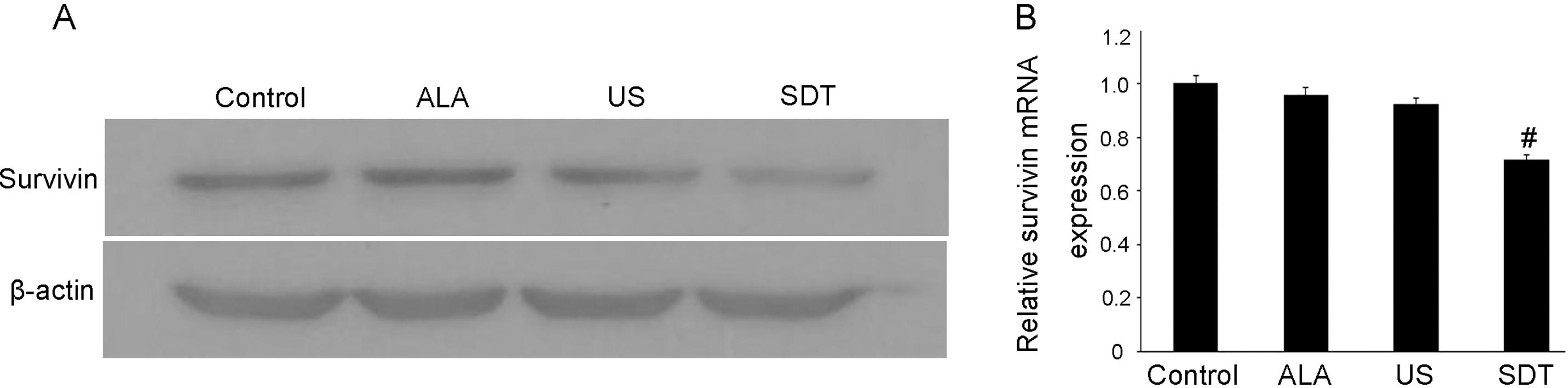
The rationale for studying survivin as a prognostic marker for malignant tumors is based on its potential to inhibit apoptosis, promote cell proliferation, and promote angiogenesis. Therefore, survivin may be involved in tumor progression, and inhibition of high survivin expression would be expected to interfere with the progression of the invasive disease. Many studies have shown that high survivin expression is associated with more aggressive tumor behavior and poor survival in patients with various malignancies [5, 21]. In the study by Chen et al., survivin was shown to be highly expressed in liver cancer and was associated with histological grading and tumor size, as well as lymph node and metastasis (TMN) staging [22]. Similarly, Wei et al. confirmed that inhibition of survivin expression inhibits melanoma growth and lung metastasis [23]. In the present study, survivin expression in melanoma tissues was significantly higher than the adjacent non-melanoma tissues collected clinically, suggesting that inhibition of survivin expression in melanoma tissues may provide a basis for the clinical treatment of melanoma. The present study confirmed that the combined treatment with 5-ALA and SDT significantly inhibited the expression of survivin protein and mRNA in melanoma cells and the melanoma-xenograft model and prolonged the survival of melanoma-bearing nude mice. These results suggest that 5-ALA-mediated SDT may also inhibit survivin expression in clinical samples. However, the specific potential therapeutic effects and underlying mechanisms still require further experiments for their elucidation.
Studies have shown that different ultrasound intensities combined with different sonosensitizers have different biological effects on specialized cells and tissues [24, 25]. In this study, we used 5-ALA to mediate low-intensity ultrasound-combination therapy to significantly inhibit survivin protein and mRNA expression in melanoma cells and a melanoma-xenograft model and to prolong the survival of melanoma-bearing nude mice. Our previous study showed that SDT induces mitochondria-dependent apoptosis by producing ROS, and that this apoptosis is related to the upregulation of p53 and the downregulation of Bcl-2, CCND1, silent information regulator 1 (SIRT1), and mRNA-34a [16]. Thus, we hypothesize that the downregulation of survivin expression is also involved in SDT-induced apoptosis.
Several studies have shown that survivin has a potential replacement effect in melanoma metastasis and angiogenesis, independent of apoptosis or mitosis regulation. McKenzie et al. suggested that survivin promotes melanoma metastasis by upregulating a-5 integrin [26]. In our previous study, we showed that SDT significantly inhibits endothelial cell proliferation, migration, invasion, and tube formation [19]. Fernandez et al. showed that survivin expression in B16-F10 melanoma cells enhances vascular endothelial growth factor (VEGF) release and promotes vascularization. In addition, knockdown of survivin expression in B16-F10 cells reduces the number of blood vessels and VEGF expression in the tumors of C57BL/6 mice [27]. A study by Blanc-Brude et al. suggests that survivin is a novel upstream regulator of mitochondrial-dependent apoptosis. The molecular targeting of this pathway has anti-cancer activity by inducing tumor-cell apoptosis and inhibiting angiogenesis [28]. Thus, we speculate that the inhibitory effect of SDT on melanoma may not only be related to the apoptotic function of survivin, but may also be closely related to the role of survivin in angiogenesis.
Although the present study did not detect the specific mechanism of 5-ALA-mediated SDT in the inhibition of survivin expression, previous studies have shown that miRNA-203, miRNA-34a, and miRNA-16 inhibit tumor-cell proliferation by targeting survivin [29, 30]. Combined with the results and microarray analysis of our previous study, miRNA-34a is synergistically upregulated under SDT stimulation to downregulate target genes involved in cell proliferation and apoptosis [16]. Therefore, survivin downregulation by SDT may be associated with abnormal miRNA-34a expression. However, we cannot identify how miRNA-34a is involved in the regulation of survivin expression; further studies are needed to confirm this potential interaction.
In conclusion, our clinical sample results showed that survivin expression was significantly increased in patients with malignant melanoma, suggesting that survivin may be involved in the poor prognosis of melanoma. In vitro and in vivo experiments confirmed that SDT effectively inhibited the proliferation of B16-F10 cells, which might be related to the inhibition of survivin. The above two conclusions suggest that SDT has potential for the clinical treatment of malignant melanoma and for addressing anti-melanoma drug resistance, as this mechanism may be closely related to the effective inhibition of survivin. However, the specific putative therapeutic mechanism still requires further experimental verification.
Footnotes
Acknowledgments
This work was supported by grants from the Lin He’s Academician Workstation of New Medicine and Clinical Translation in Jining Medical College (No. JYHL 2018MS19) and the Research Support Funds for Doctor of Jining Medical College.
Conflict of interest
The authors declare that they have no conflicts of interest.
