Abstract
BACKGROUND:
Matrix metalloproteinase-9 (MMP-9) is an important mediator of invasion and metastasis in neoplasia. In thyroid cancer expression levels correlate with aggressiveness but data on peripheral MMP-9 levels are less definitive.
OBJECTIVE:
Prospective study evaluating serum MMP-9 in the diagnosis and prognosis of papillary thyroid cancer.
METHODS:
Serum samples of MMP-9 were drawn before surgery in 185 consecutively enrolled patients with nodular thyroid disease, stratified on pathology as benign disease (
RESULTS:
MMP-9 levels were not different between benign vs malignant pathology (
CONCLUSION:
Serum MMP-9 is not useful in the diagnosis of PTC, but preliminary data shows that high pre-surgical serum MMP-9 levels may identify patients at higher risk of persistent disease who require intensive treatment. Large volume prospective studies are required to confirm this observation.
Introduction
Thyroid cancer is the most frequent endocrine neoplasia and it’s incidence has risen dramatically in the last 40 years [1, 2]. In patients diagnosed with differentiated thyroid cancer (DTC), the prognosis is usually good, due mostly to the efficacy of radioiodine therapy. A subset of patients can have a more aggressive clinical course, become radioiodine resistant and head toward a fatal outcome. Early identification of this subset of patients would be useful to better guide management: the extent of surgery, the dose of radioiodine and the degree of TSH supression.
In this context, cancer research was focused on attempting to identify an easily measured serum molecule, to serve as a diagnostic and prognostic marker of neoplasia. One such marker, which has attracted a lot of interest in the past years, is Matrix metalloproteinase-9 (MMP-9) [3].
Matrix metalloproteinases (MMPs) are zinc- dependent endoproteinases whose enzymatic activity is primarily directed against components of the basement membrane and extracellular matrix (ECM). MMP-9 is physiologically expressed mainly in immune cells but also in fibroblasts and endothelium cells, and its expression can be induced or repressed by adhesion molecules, growth factors, cytokines and hormones that act through various signalling pathways to control transcription of the MMP-9 gene [3]; MMP-9 is overexpressed in almost all human cancers, and its expression is further augmented by the cross-talk signalling between the surrounding stroma and the inflamatory cells in the tumor microenvironment [3, 4]. It is believed that tumor cells use the MMP-9 in the tumor microenvironment for invasion and metastasis; MMP-9 further stimulates the release of VEGF (vascular endothelial growth factor) contributing to angiogenesis [3, 4].
In thyroid nodular disease, high MMP-9 expression was found to be able to distinguish malignant from benign tumors [5, 6]; in differentiated thyroid cancer, high expression of MMP-9 was associated with lymph node metastasis, invasion and generally higher tumor stages [5, 6, 7, 8]. Furthermore, mutational analysis suggested that MMP-9 might be one of the mediators of tumor invasiveness in BRAF
It is expected that changes in the tumor microenvironment will be reflected in the body fluids and, considering the relative ease of measurement in blood samples with immunometric assays, MMP-9 could be an ideal marker for helping with cancer diagnosis and prognosis. Indeed, studies have detected abnormal peripheral blood levels of MMP-9 in a variety of neoplasias, with high levels generally related to poor outcomes [4, 11, 12, 13, 14, 15, 16, 17]. There are few studies on MMP-9 levels in the blood of patients with thyroid cancer, and they are all limited by the small numbers of patients [18, 19, 20, 21].
In this context we aimed to assess serum levels of MMP-9 in benign and malignant thyroid disease of the follicular epithelium and i) evaluate its role as a diagnostic and prognostic marker in thyroid neoplasia and ii) evaluate its relationship with clinical and pathological features of aggressiveness of thyroid cancer associated with a worse prognosis.
Patients and methods
We assessed serum MMP-9 in 185 consecutively enrolled patients with nodular thyroid disease who underwent thyroid surgery between December 2012 and November 2016 at the “CI Parhon” National Institute of Endocrinology. Patients were informed about the study procedures and gave their written informed consent. The study was performed in strict agreement with the Helsinki declaration concerning ethical principles for medical research involving human subjects. The institutional ethics committee revised and agreed the study protocol.
Patients were divided into 2 groups, according to the pathology report: benign disease (BD) (
For patients with PTC, the pathology report included the histological subtype, the presence or absence of multifocality and invasion, size of primary nodule and pathology pT stage.
The histology subtypes of PTC were grouped into: classic PTC (CLS), follicular variant (FV), oxyphil variant of PTC (OXY) and other aggressive forms (AGR), including poorly differentiated, tall cell, insular, solid and sclerosing subtypes. The presence of capsular invasion and gross extra-thyroidal invasion was evaluated based on pathology and imaging data, and patients were grouped into “No invasion”, “Microscopic invasion” and “Macroscopic invasion” groups. Lymphovascular invasion, thought to be a risk factor for adverse prognosis of PTC, was also evaluated and used as a criterion of recurrence risk assessment.
In terms of pTNM staging, according to the AJCC 7th Edition/TNM Classification System for Differentiated Thyroid Carcinoma [22], patients were classified as pT1, pT2, pT3 and pT4 stage. Cross-sectional imaging data and radioiodine scans were used to assess the presence of metastasis and establish the TNM stage.
Imaging evaluation performed before surgery (when indicated), pathological features, as well as information collected after surgery: ultrasound, stimulated thyroglobulin, radioiodine scans, were used to classify patients according to the 2015 American Thyroid Association risk stratification, into Low risk, Intermediate risk and High risk of reccurence [23].
Sera was collected in all patients 1–2 days before surgery. As inflammation after biopsy may influence serum MMP-9 levels, in those patients in which FNAB was performed serum samples were collected after more than one month after FNAB.
Blood samples were collected in serum tubes with clot activator (Kabevette
MMP-9 was measured by a specific immunometric assay supplied by R&D Systems (Quantikine human MMP-9 (total)). The assay uses the quantitative sandwich enzyme immunoassay technique and recognizes pro-MMPs, active MMPs and to different extents MMP/TIMP complexes [24].
After surgery thyroid cancer patients were prospectively followed-up in the Institute and radioiodine therapy was performed when indicated after risk assessment stratification. Clinical, biochemical and imaging data were collected after each visit and at the end of follow-up in order to evaluate the response to treatment, according to the recommended dynamic risk stratification system [23]. Complete follow-up data was available in 78 patients who were included in the analysis. At the end of follow-up, patients were classified as having no evidence of disease (NED) if they had a stimulated thyroglobulin of
Statistical analysis
Statistical analysis was performed with Medcalc software version 14.8.1. Normal distribution of all groups was evaluated with the Kolmogorov-Smirnov test; independent
Results
The demographical data of all patients and a summary of MMP-9 levels in patients with papillary thyroid cancer and benign nodular disease are presented in Table 1. Both cancer patients and patients with benign goiters were predominantly female (77.3% vs 82.9%), with no significant difference in gender balance between groups (
Demographical data of patients, average MMP-9 levels
Demographical data of patients, average MMP-9 levels
BD – Benign disease; PTC – Papillary thyroid carcinoma.
A. Box and whisker plot of MMP-9 values for benign vs malignant disease (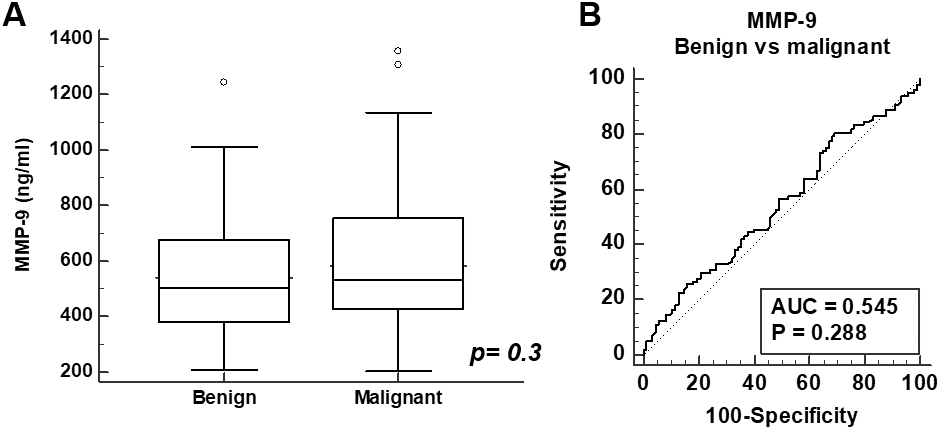
There was no difference in MMP-9 levels between patients with benign vs malignant pathology when evaluated as a group (541.6
MMP-9 levels tended to be lower in older patients when all samples were considered (Pearson
The clinical and pathological characteristics of patients with PTC are presented in Table 2.
Clinical and pathological characteristics of PTC patients
CLS – classic PTC; FV – follicular variant PTC; AGR – aggressive histology; MFoc – multifocal; UFoc – unifocal; ETE – extrathyroidal extension; No – no invasion; Micro – microscopic invasion; Macro – macroscopic invasion; LR – low risk; IR – intermediate risk; HR – high risk.
A. Box and whisker plot of MMP-9 values showing that patients with High-risk (HR) PTC had significantly higher pre-surgical MMP-9 levels compared to Low-risk – Intermediate-risk (L-IR) patients (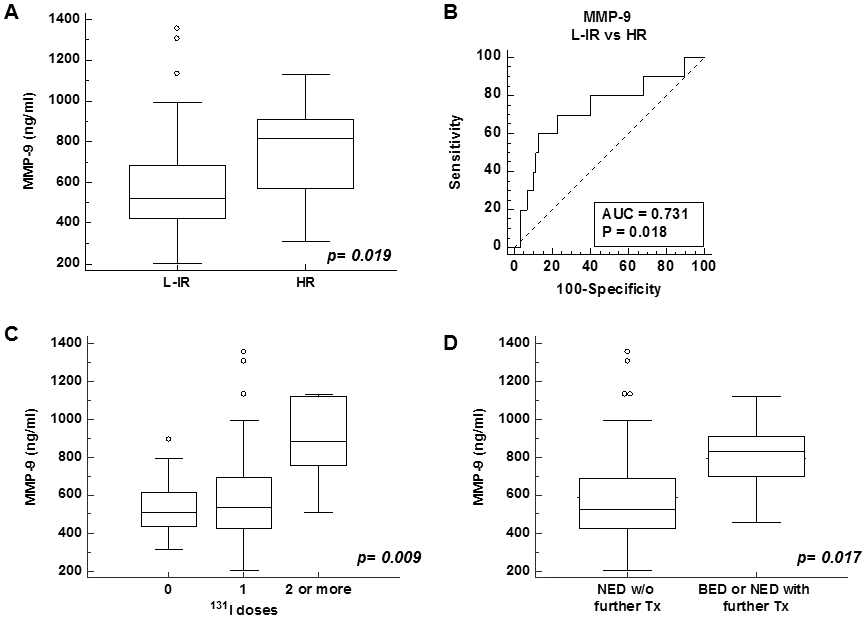
MMP-9 levels did not differ significantly between patients younger or older than 45 years old (
Complete data concerning response to treatment and outcomes was available in 78 cancer patients who were followed-up for a median of 58.5 months (range 3–82 months) – Table 3. Of the 78 patients, 53 were low-risk according to the ATA criteria, 20 were intermediate risk and 5 were high-risk papillary thryoid cancer.
Response to treatment in PTC patients
I
In 22 low-risk patients radioiodine ablation was not indicated. At the end of follow-up, of these 22 patients, 18 presented no evidence of disease (NED), while 4 presented with minimally elevated, stable stimulated thyroglobulin levels, median 4.32 ng/ml (range 4.11–5.96 ng/ml) most likely reflecting small thyroid remnants (classified for the purpose of this study as minimal biochemical evidence of disease).
The remaining 56 patients underwent radioiodine therapy, receiving a total median
Of the 56 patients who underwent initial radioiodine ablation, 47 patients (83.9%) showed no evidence of disease (NED) at the end of follow-up without further treatment. 3/56 patients (5.4%) achieved NED status after further
No patients were identified to have structural/ functional evidence of disease at the end of follow-up. No recurrences were identified (i.e. new biochemical, structural or functional evidence of disease after a period of NED) so the association between MMP-9 levels and the risk of recurrence could not be investigated.
There is increasing evidence supporting the involvement of MMP-9 in the development and progression of neoplasia, including thyroid cancer. MMP-9 takes part in several dysregulated signalling pathways that lead to tumor cell proliferation and invasion, metastasis, inhibiting apoptosis, contributing to angiogenesis and escaping the immune surveillance [3, 4, 25]. High peripheral MMP-9 levels have been identified in various forms of neoplasia [4, 11, 12, 13, 14, 15, 16, 17]; there are, however, only a few studies concerning thyroid cancer [18, 19, 20, 21], usually with small numbers of patients included.
In our study the original assumption was that patients with thyroid cancer: i) would have higher levels of serum MMP-9 than those with benign nodular goiters; ii) patients with aggressive thyroid cancer would have higher MMP-9 levels compared to milder forms. The published literature on tissue expression of MMP-9 in thyroid cancer seemed to suggest that. High MMP-9 expression was noted in thyroid cancer tissue vs normal thyroid cells and vs benign nodular disease [5, 7, 26, 27, 28]. In PTC higher MMP-9 expression was noted in older PTC patients (age
Contrary to our expectations, we found no difference in MMP-9 levels between patients with cancer and benign nodules, suggesting that, as opposed to tissue MMP-9 expression, serum MMP-9 would not be useful in the diagnosis of PTC before surgery. Similarly, an early Japanese study by Maruyama et al. identified no difference in serum MMP-9 levels in PTC patients vs control, although tissue levels were significantly higher in PTC compared to benign thyroid disease or controls [21]. In contrast, Zhou et al. identified significantly higher serum levels of MMP-9 in PTC patients vs patients with bening thyroid disease and vs normal controls, as measured by both ELISA and RT-PCR [19]. Lin et al. identified significantly higher plasma VEGF and MMP-9 levels in patients with PTC vs bening thyroid disease and controls, but their levels in early stages of PTC (stage I and II) did not differ from the control or the benign group, suggesting their limited usefulness as diagnostic markers in early PTC [20]. However, in terms of prognosis, VEGF and MMP-9 levels were higher in PTC patients with larger tumors, local invasion, lymph node or distant metastasis [20].
In contrast, in our PTC patients we found no association between the MMP-9 level and tumor size, pT stage, TNM stage, presence or absence of multifocality. There was also no difference in MMP-9 levels between patients with classic PTC, follicular variant, oxyphil variant and other aggressive histologies, although it is worth noting that the “aggressive” group was heterogeneous, including patients with poorly differentiated, solid, sclerosing and oxyphil variants, too few to be evaluated statistically individually.
It is of note that most studies of differentiated thyroid cancer performed so far, whether concerned with tissue expression of MMP-9 [5, 6, 8, 27, 28] or peripheral blood levels [20], associate high MMP-9 levels with lymph node metastases. This is important, since PTC spreads mostly through lymph vessels and recurrences generally occur in the lymph nodes of the neck. In some of these studies, central and/or lateral neck dissection was performed routinely [6, 8]; however, there is evidence that nodal micro-metastases can be present in up to 80% of patients with PTC but adverse prognosis is limited to those patients with macroscopic, clinically evident node involvement [29]. So, although MMP-9 apparently correlates with lymph node metastasis, it is debatable whether this association is indeed relevant to prognosis. In our PTC patients we found no association between MMP-9 level and lymph node metastasis, but lymph nodes were evaluated at pathology in a minority of patients, as current guidelines recommend node dissection as part of PTC surgery only in cases with clinical node involvement [23].
In terms of invasion, it is strongly suggested that it is gross extra-thyroidal extension (based on radiological and clinical data showing a tumor extending beyond the thyroid capsule) that represents a negative prognostic factor, as opposed to microscopic extension, proven at pathology, which has minor significance [30]. Because of this, the 2018 update of the TNM staging system removed the presence of microscopic extrathyroid extension from the criteria for pT3 stage [31]. Our patients with macroscopical extra-thyroidal extension tended to have slightly higher MMP-9 levels, however the difference was not statistically significant.
According to the ATA risk stratification system, patients with macroscopical tumor invasion and incomplete resection are considered at high risk of recurrence. Other high-risk features are the presence of metastatic lymph nodes
In fact, looking at the response to treatment and final outcomes of our patients, we found that MMP-9 levels were significantly higher in patients who required repeated doses of
In interpreting our results several observations should be made. We expected to find higher serum MMP-9 levels in cancer patients vs benign disease and we also expected more clear correlations between MMP-9 levels and pathological features of aggressiveness in PTC patients. It is worth noting, however, that the strongest evidence of the connection between MMP-9 and PTC aggressiveness comes from histological studies of MMP-9 expression; serum or plasma measurements of MMP-9 seem to provide less clear, sometimes contradictory results. It is quite likely that blood levels of MMP-9 (and probably MMPs in general) only partially reflect the events occuring in the tumor microenvironment. Some of the causes relate to biological factors, such as secretion from the tumor into the bloodstream and MMP-9 leakage from blood cells, and some are related to artefacts from the measurement assay.
Moreover, when evaluating blood levels of MMPs, the existence of different sources of secretion must be taken into account. Increased levels of MMP-9 have been identified in a variety of disease processes, including vascular, inflammatory, degenerative and autoimmune disorders, apart from neoplasia, which lowers the MMP-9 specificity for cancer [3]. This could be particularly important in the evaluation of thyroid nodular disease, considering the high prevalence of autoimmune thyroiditis which is characterized by episodes of inflammation and tissue remodelling.
Another general limitation of studies on peripheral MMP-9 levels relates to the measurement assay. If measured by ELISA, MMP-9 levels reflect immunoreactivity in the biological sample being studied, which may include proMMP-9, activation forms (representing 1–2% of pro-MMPs), degradation products, monomers, oligomers, heterocomplexes and even non-covalent complexes with TIMP-1 [3, 24]. These different MMP-9 forms have distinct and overlapping biological activites in normal physiological conditions as well as cancer. This is an important aspect when comparing results from different studies, because different ELISA assays can detect different active forms/complexes of MMPs.
Finally, the method of collection and storage as part of the pre-analytical stage of MMP determination is essential [24]. There is ample evidence that the type of sample used for the determination, serum or plasma, can provide significantly different results, generally with higher MMP levels in serum vs plasma [32]. This is particularly true for MMP-9 and TIMP-1 due to variable release from platelets, as these contain high levels of MMP-9 and TIMP-1 [13, 32, 33]. The presence of coagulation accelerators in serum samples or anticoagulants in plasma samples further influence the results, mainly as the consequence of the MMP secretion/release in the coagulation/fibrinolisys pathways [13, 34]. Several authors noted that proMMP-9 and active MMP-9 levels in serum and plasma are only weakly correlated [33] or not correlated [35], which led them to call into question the validity of measuring MMP-9 in serum samples. On the other hand, it has been proved that serum and plasma total MMP-9 levels are significantly correlated in both healthy controls and diseases characterized by high MMP-9 levels, so MMP-9 can be regarded as a valid biomarker even when measured in serum samples [17, 35, 36]. In fact, there are several studies that have identified a positive diagnostic and prognostic role of high MMP-9 measured in the serum in different neoplasias. In colorectal cancer a significant association was found between high serum MMP-9 and neoplasia [15, 16], although a later larger scale study failed to confirm this association [37]. In patients with breast disease, higher serum MMP-9 levels could distinguish breast cancer from benign disease; MMP-9 was associated with more advanced disease, lymph node involvement and metastatic presentation [11, 14]. In contrast, in patients with gastric cancer, only plasma and not serum MMP-9, was able to distinguish cancer patients from controls and correlated significantly with lymph node metastasis, venous invasion and poor survival [17].
In the end, although its role in cancer development seems certain, whether it is in serum or plasma samples, and whether active MMP-9, proMMP-9 or total MMP-9 is better in terms of diagnostic and prognostic value is still to be determined.
Conclusion
There is a wealth of evidence suggesting that MMP-9 is an important mediator of cancer growth, invasion and metastasis. In thyroid cancer there are few studies of peripheral levels of MMP-9 and these are small and show conflicting results. Ours is the first large-scale study to investigate the role of MMP-9 as a circulating marker in differentiated thyroid cancer.
Our data suggest that serum MMP-9 is not a useful diagnostic marker for papillary thyroid carcinoma, as MMP-9 levels were not different in patients with benign nodular disease and papillary thyroid cancer. However, we found higher MMP-9 levels pre-operatively in patients with high risk papillary thyroid carcinoma, in those who required repeated doses of
Footnotes
Acknowledgments
This study was supported by UEFISCDI grant PN-II-PT-PCCA-2011-3.2 no.135/2012.
Conflict of interest
No competing financial interests exist.
