Abstract
OBJECTIVES:
Thus far, biological roles of plasminogen activator inhibitor 1 (PAI1) in hepatocellular carcinoma (HCC) remain controversial. Moreover, its expression, clinicopathologic and prognostic significance in HCC have not been comprehensively investigated, therefore needing further evidence.
METHODS:
PAI1 expression was measured, using tissue microarray-based immunohistochemical staining, in matched HCC and adjacent liver samples from 178 patients with HCC after curative resection. The correlations of PAI1 H-scores with clinicopathologic variables and survival were further evaluated. Its prognostic value was finally confirmed in some public databases.
RESULTS:
It was found that PAI1 expression was significantly higher in HCC than in adjacent liver tissues. Moreover, high PAI1 expression was more frequent in those with multiple lesions. Univariate analyses showed that PAI1 expression was negatively associated with both overall and relapse-free survival. Although PAI1 expression was not statistically significant in multivariate Cox regression test, combination of it with TNM stage effectively distinguished survival and relapse, and served as an independent prognostic factor. In the online available datasets of HCC and liver cancer used, SERPINE1, the gene encoding PAI1, was also revealed to be prognostic.
CONCLUSIONS:
Our data suggested that high PAI1 expression was predictive for unfavorable biological behavior and long-term prognosis in HCC.
Keywords
Introduction
It has been well known that hepatocellular carcinoma (HCC) is a highly prevalent and poorly prognostic neoplasm [1]. Thus far, post-surgical recurrence of HCC remains to occur frequently [2], although favorable long-term prognosis after radical hepatectomy has been obtained in strictly selected patients [2, 3]. This has become one of main barriers of long-term survival of HCC [4]. Therefore, the underlying molecular mechanisms of HCC recurrence are of great clinical importance. Up to now, many invasion-related factors, such as tumor microenvironment, cancer stem cells and epithelial to mesenchymal transition (EMT) [5, 6, 7], and genes involved in them [8, 9], have been identified in HCC. Of course, novel findings are still needed.
In the proteolytic degradation of extracellular matrix (ECM), one of key processes in cancer spread [10], urokinase-type plasminogen activator (uPA) system was found, by many researchers, to function as an important player [11, 12]. As one of two serine protease inhibitors within the system, plasminogen activator inhibitor 1 (PAI1) was shown to play biological roles not only in benign cell disorders, such as endothelial cell apoptosis and skin fibrosis [13, 14] but also in many cancers [15, 16, 17, 18, 19, 20, 21]. Surprisingly, most papers showed that PAI1 contributed to malignant behaviors, such as cell cycle progression, anti-apoptosis, chemo/radio-resistance, cell proliferation and migration as well as recruitment and polarization of macrophages, through some uPA-independent mechanisms [16, 17, 18, 19, 20, 21]. These, different with its role as an inhibitor of uPA, suggested its pro-oncogenic effects, although opposite report was also shown [15]. In addition, it was revealed to be associated unfavorable prognosis in some malignancies [22, 23, 24]. In HCC, clues from in vitro experiments were also contradictory [25, 26]. We previously found that PAI1 expression was high in HCC tissues [27]. Moreover, it was also related to vascular involvement, poor differentiation and a tendency to dismal prognosis in HCC [28, 29]. However, these works were largely limited by smaller sample sizes and incomprehensive evaluation criteria. Thus, further evidence is needed.
In the present study, we aimed to explore clinicopathologic and prognostic significance in HCC, based on a Chinese cohort and the online publically available datasets, including the The Cancer Genome Atlas (TCGA) database.
Materials and methods
Patients
A total of 178 patients with HCC after curative resection (142 males and 36 females) were included in this study. Their general, etiologic and clinicopathologic parameters are shown in Table 1. This work was approved by the Institutional Ethics Committee and the informed consents were acquired from all patients.
Correlations between PAI1 expression and clinicopathologic features of HCC
Correlations between PAI1 expression and clinicopathologic features of HCC
Abbreviations: PAI1, plasminogen activator inhibitor 1; HCC, hepatocellular carcinona; HBV, b-type hepatitis virus; HCV, c-type hepatitis virus; TNM, tumor-node-metastasis; AFP,
Tissue microarray construction was performed using formalin-fixed paraffin-embedded (FFPE) blocks. After careful review and selection of representative regions, two cores (1.5 mm) of HCC and corresponding adjacent liver (AL) tissues of each patient were punched, by the manual tissue arrayer (Beecher Instruments, 686 Progress Way, Sun Prairie, WI, USA), to construct TMA.
PAI1 expression in resected hepatocellular carcinoma. (A) High expression in HCC tissue (original magnification 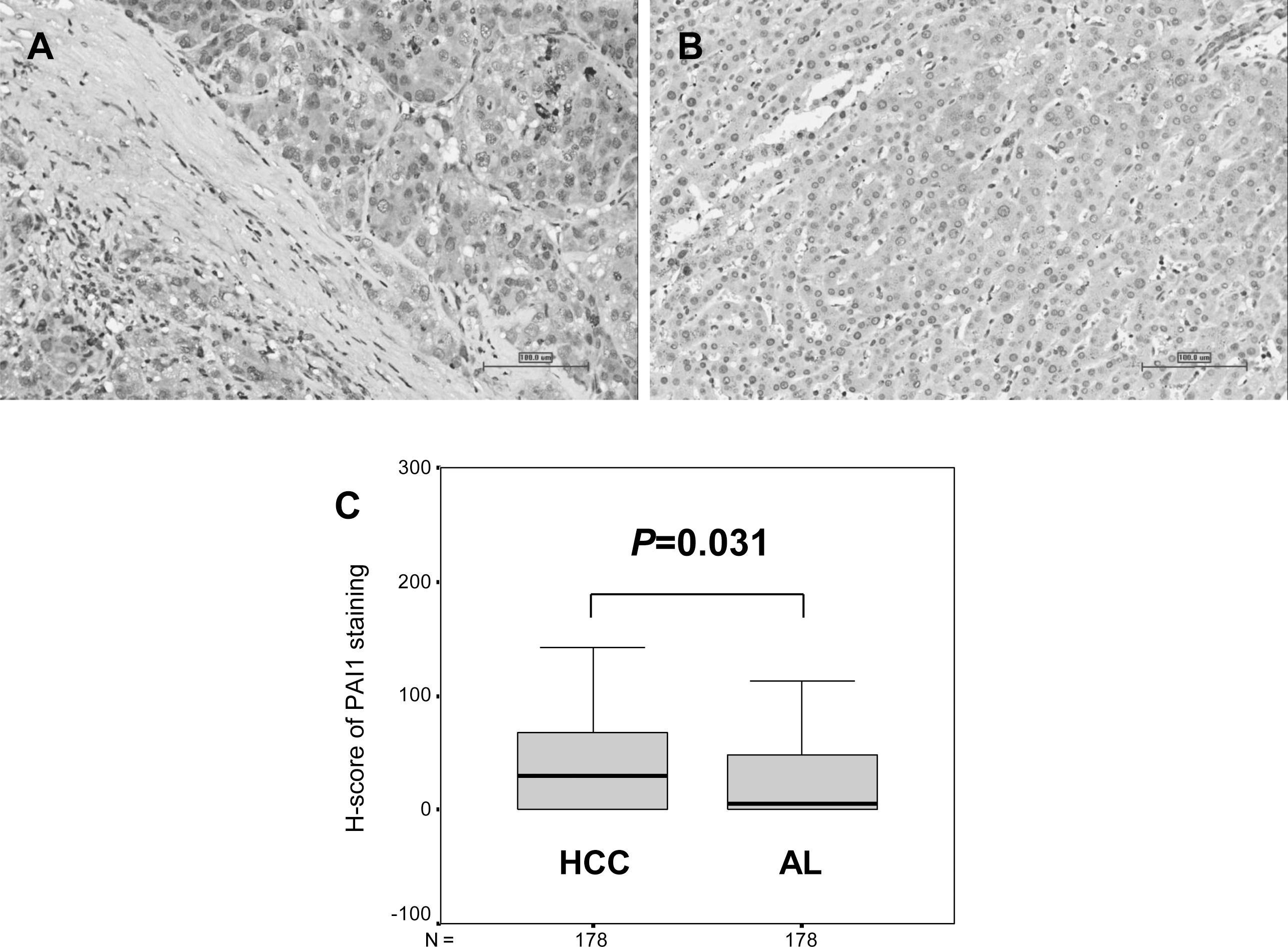
Univariate identification of prognostic factors in HCC
Abbreviations: HCC, hepatocellular carcinoma; HR, hazard ratio; CI, confidence interval; HBV, b-type hepatitis virus; HCV, c-type hepatitis virus; TNM, tumor-node-metastasis; AFP,
Prognostic value of PAI1 expression in resected hepatocellular carcinoma. (A) Overall survival of patients according to PAI1 expression (Log-rank test; 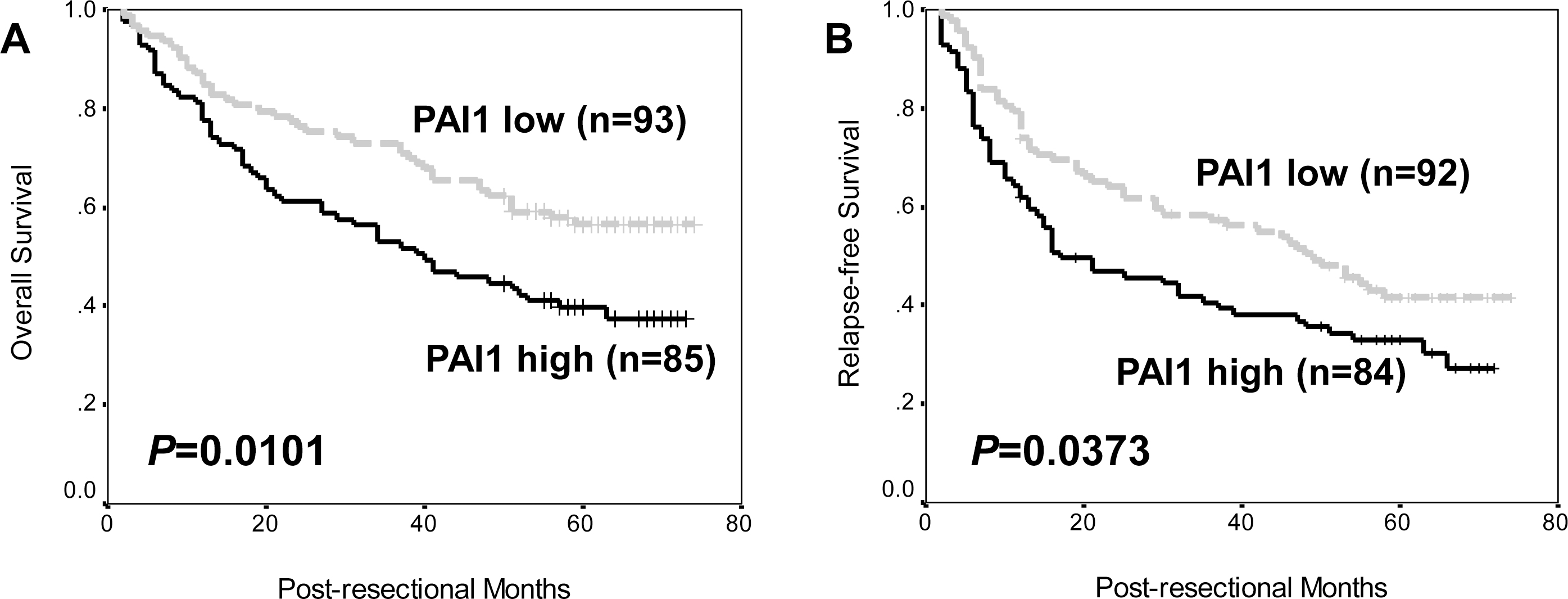
Immunohistochemistry was carried out, using the mouse monoclonal antibody for human PAI1 (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) and a two-step staining kit (EnVision
Staining evaluation
Two senior pathologists who were blinded to clinicopathologic and follow-up information (Z. Y. L. and W. X. Z.) evaluated the slides, applying the H-score [30], a comprehensive staining evaluation method that considered both positive cell ratio and staining intensity (formula: positive cell ratio
Follow-up
Our follow-up, ranging from 2 to 74 months (median: 51 months), was carried out for all the patients. At the last follow-up, 86 patients (48.3%) were still alive, while 92 (51.7%) have died.
Multivariate analyses for overall and relapse-free survival of HCC
Multivariate analyses for overall and relapse-free survival of HCC
Abbreviations: HCC, hepatocellular carcinoma; HR, hazard ratio; CI, confidence interval; TNM, Tumor-Node-Metastasis; HCV, c-type hepatitis virus; AFP,
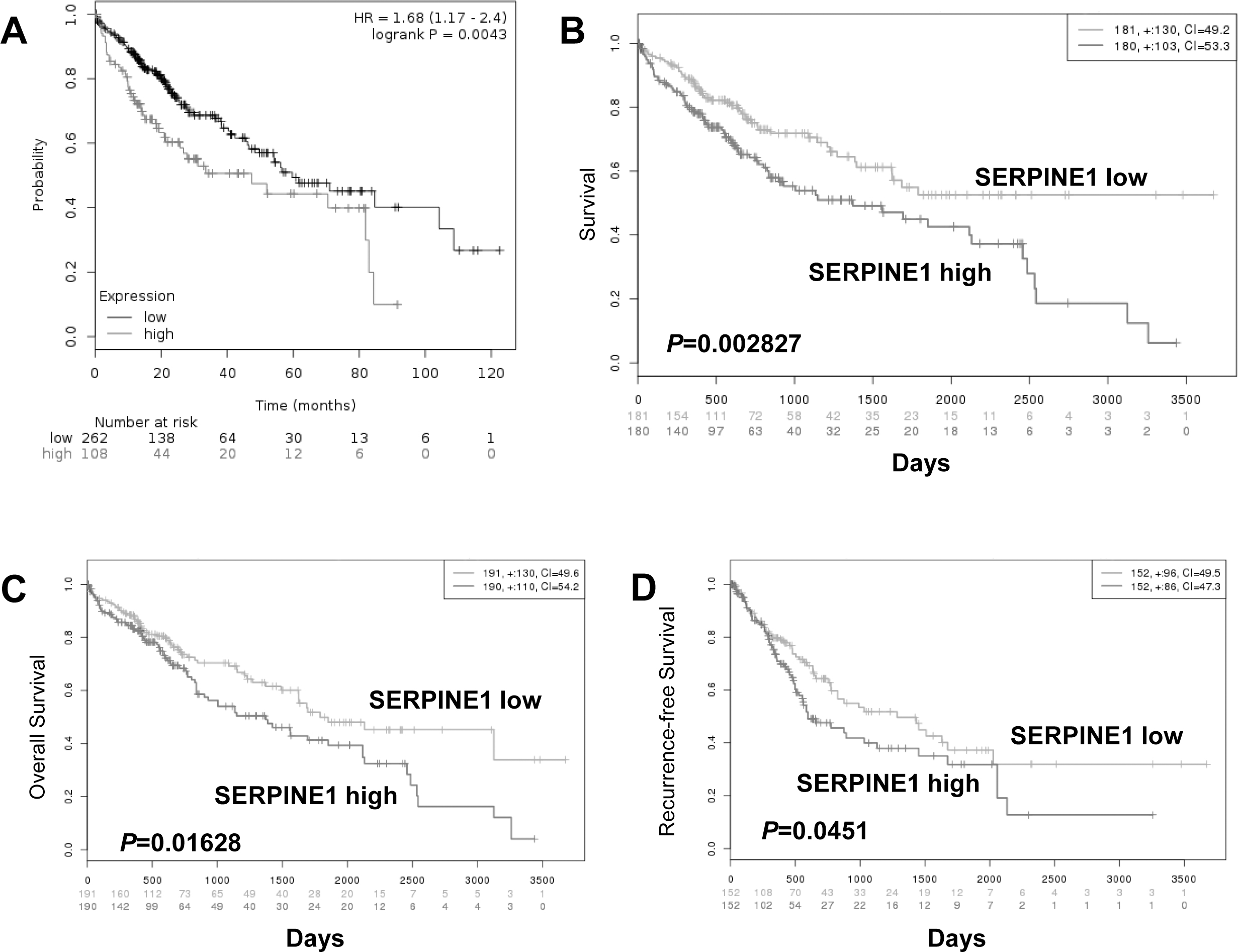
The prognostic value of SERPINE1, the gene encoding PAI1, was analyzed in the publically available Kaplan-Meier plotter (
Statistical analyses
The PAI1 H-scores in HCC and AL tissues were compared using Mann-Whitney
Results
PAI1 expression in HCC tissues
It was shown in Fig. 1A and B that the positive PAI1 staining signal was mainly observed in the cytoplasm of both HCC and adjacent liver (AL) cells, especially in the invasive fronts of tumor nodules to the parenchyma (Fig. 1A). The H-score in HCC tissues was statistically higher than that in AL ones (Fig. 1C,
Correlations of PAI1 expression with clinicopathologic parameters in HCC
According to the ROC curve (Fig. S1), the H-score of PAI1 in HCC tissues at the largest YI for survival status (38.75) was identified as the cut-off point. Using Chi-square test, high PAI1 expression was significantly more frequent in multiple HCC lesions than that in single ones (Table 1,
Prognostic significance of PAI1 in HCC
Using the Kaplan-Meier method and the log-rank test, high PAI1 expression in HCC tissues was significantly associated with poorer overall and relapse-free survival in our patient cohort of resectable HCC (Fig. 2,
Prognostic stratification by combination of PAI1 expression and TNM stage in resected hepatocellular carcinoma. (A) Overall survival; (B) Relapse-free survival.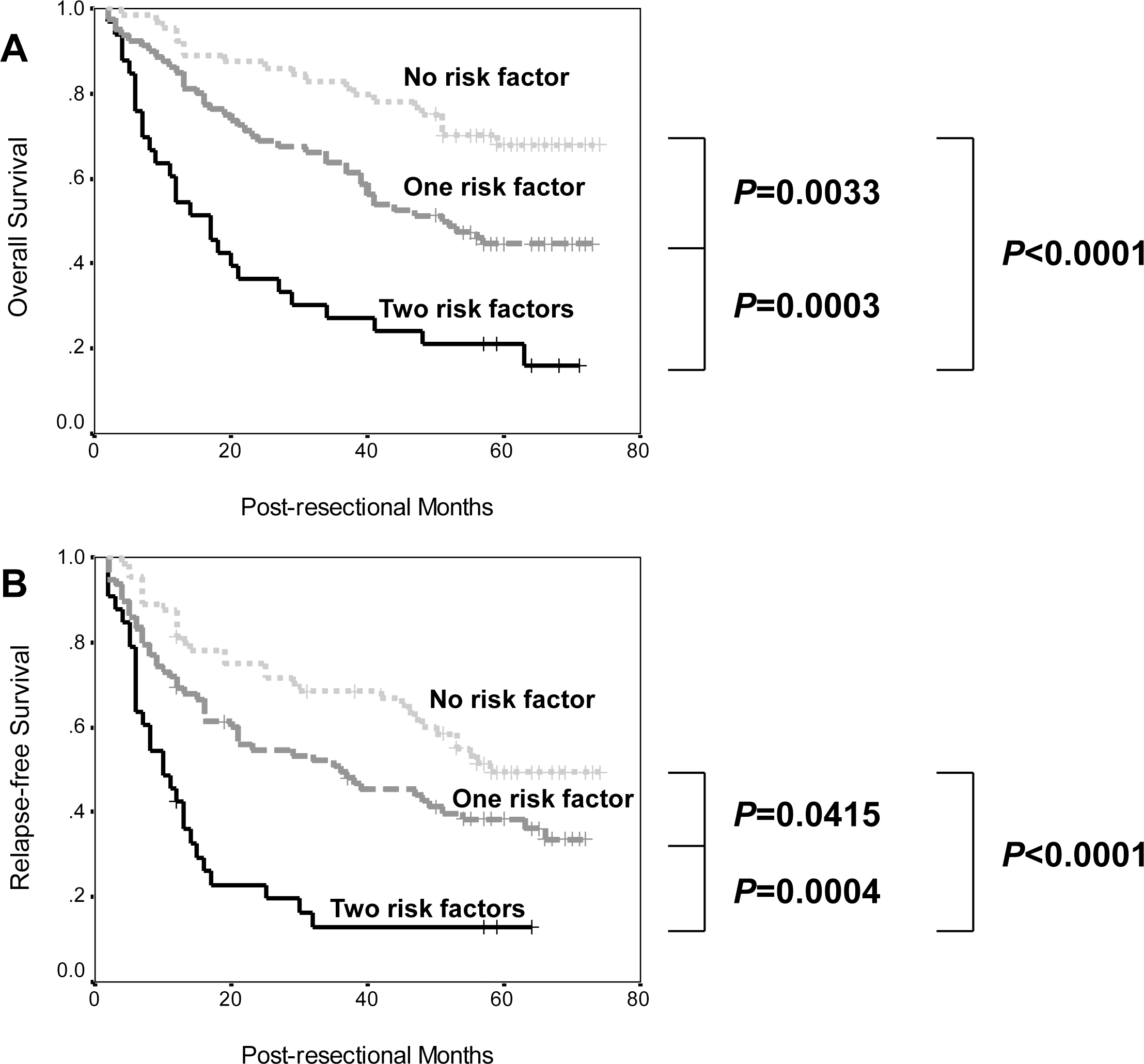
Univariate Cox regression analyses showed that HCV infection, tumor size, tumor number, TNM stage,
Combined evaluation of TNM stage and PAI1 expression in HCC
In order to get more accurate prediction of patient survival, TNM stage, the strongest prognostic indicator in the cohort, and PAI1 expression were combined. As shown in Fig. 3, the differences between no risk factor (stage I and PAI1 low), one risk factor (stage II-III or PAI1 high) and two risk factors (stage II-III and PAI1 high) were all univariately significant. In addition, this combination was also significant in multivariate analyses (Overall survival: HR
Prognostic value of SERPINE1 in public datasets for HCC and liver cancer
In the Kaplan-Meier plotter, SERPINE1, the PAI1-encoded gene, was found to be prognostic (Fig. S2A,
Discussion
As one of important hallmarks in cancer [34], invasion/metastasis is a complicated process, in that ECM degradation plays a key role [10], and depends on many active molecules, such as uPA that results in plasminogen activation, so as to accelerate the proteolysis [11, 12]. It has long been established that uPA has two specific inhibitors, PAI1 and PAI2. Among them, PAI1 was found by most investigations to regulate molecular events in benign conditions [13, 14] and to boost proliferation, angiogenesis, EMT and chemoresistance of cancer cells [16, 17, 18, 19, 20], even to modulate tumor microenviroment [21]. This indicated its proto-oncogenic nature, quite different its speculated effects. In the tissue level, PAI1 was also identified as a poor prognostic marker in some cancers, including clear cell renal cell carcinoma, differentiated thyroid carcinoma and pancreatic ductal adenocarcinoma [22, 23, 24]. In HCC, its biological roles remain contradictory. For example, PAI1 were shown to inhibit invasion and proliferation of HCC cells [25], but to promote angiogenesis [26], an important event in the development and pathogenesis of HCC [35]. The previous literatures for high expression of PAI1 and its associations with unfavorable behaviors and mortality supported its function as a tumor-promoting factor in HCC [27, 28, 29]. However, there were obvious limitations for the studies in sample size and evaluation criteria.
To overcome these, we conducted on this work enrolling more patients and using H-score, a more comprehensive and widely used staining evaluation method [30, 31, 32]. The first finding was that PAI1 expression was significantly higher in HCC tissues than in AL ones, similar with previous ones [27, 29]. Also, a positive relationship between high PAI1 expression and multiple tumor nodules, a risk factor of poor survival and post-operative recurrence [36, 37, 38], was found. The results preliminarily support its oncogenic role in HCC.
Then, Kaplan-Meier method and log-rank test show-ed that high PAI1 expression in HCC tissues was predictive for poor overall and relapse-free survival. In univariate Cox regression analysis, PAI1 expression remained to be a predictor of poor prognosis, along with some clinicopathologic parameters. These data indicated the prognostic implication of PAI1 in HCC. Although being different with results by Itoh et al. [29], they were obtained based on a larger sample size and basically confirmed by the positive prognostic value of SERPINE1, the PAI1-encoded gene, for HCC and liver cancer in online available datasets. Therefore, our findings might be solid and easily understood.
However, it might be a bit pity that PAI1 expression was not significant in multivariate analyses. To enhance its prediction power, we attempted to combine it and TNM stage, the most strong prognostic marker in our cohort, that was also proven to be independently significant in other papers [39, 40], to form a risk score. The results revealed that this combination successfully stratified both overall and relapse-free survival. More importantly, it remained significant in the multivariate analyses, while other factors didn’t alter their prognostic value. This finding strengthened the potential of this combination as a promising biomarker for long-term prognosis of HCC. Of course, further validations are quite needed.
Taken together, our data suggested that high PAI1 expression was predictive for unfavorable biological behavior and long-term prognosis in HCC.
Footnotes
Acknowledgments
This study was supported by the National Natural Science Foundation, China (81301845).
Conflict of interest
The authors declare no conflict of interest.
Supplementary data
Cut-off point determination by the ROC curve of PAI1 staining H-scores for patient overall survival status in resected hepatocellular carcinoma.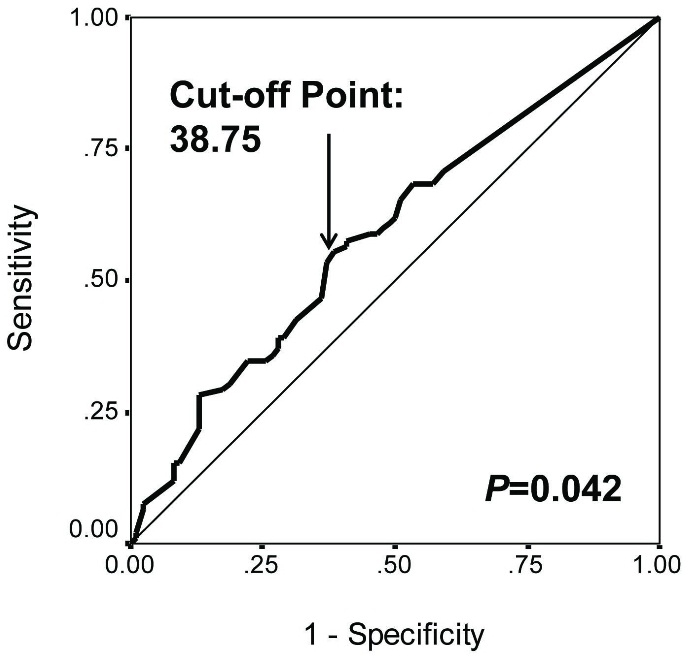
Prognostic value of SERPINE1 gene (PAI1-encoded gene) in hepatocellular carcinoma and liver cancer in the online datasets. (A) Patient survival of hepatocellular carcinoma in the Kaplan-Meier Plotter (Log-rank test;