Abstract
Ovarian cancer is the leading cause of death among gynecologic diseases in the USA and Europe. High-grade serous carcinoma (HGSC) of the ovary, the most aggressive type of ovarian cancer, is typically diagnosed at advanced stages when the 5-year survival is dismal. Since the cure rate for stage I HGSC is high, early detection of localized initial disease may improve patient outcomes. Serous tubal intraepithelial carcinoma (STIC) is considered to be a precursor lesion of HGSC. Discovery of biomarkers associated with STIC could aid in the development of an HGSC screening algorithm. Using immunohistochemical staining, we have demonstrated overexpression of UCHL1, ADAMTS13, and GAPDH in patients’ STIC lesions, but not in cancer-free fallopian tubes. We additionally demonstrated a marked increase of T cells in perineoplastic stroma surrounding STIC lesions (largely CD4
Keywords
Introduction
Ovarian cancer (OC) is the second most frequently diagnosed gynecological malignancy in the USA and Europe with the overall mortality higher than any other gynecological cancer [1]. In 2019, there will be more than 22,000 new cases of ovarian cancer diagnosed and more than 14,000 ovarian cancer deaths in the US (
There are two main types of OC: Type I carcinomas are generally slow-growing indolent neoplasms and include low-grade serous, low-grade endometrioid, mucinous, and clear cell carcinomas [2]. They are thought to develop in a stepwise progression, similar to the adenoma carcinoma sequence in colorectal cancer: from benign lesions via atypia and precursor lesions to noninvasive borderline tumors, and invasive low-grade carcinoma [3]. Typical mutations include KRAS, BRAF, ARID1A, PTEN, PIK3CA, CTNNB1, and PPP2R1A. Type II carcinomas represented by high-grade serous carcinoma (HGSC) that accounts for more than 60% of epithelial ovarian cancers and over 70% of all OC deaths [2, 4, 5, 6]. Other Type II tumors comprise endometrioid carcinomas, malignant mixed mesodermal tumors (carcinosarcomas), and undifferentiated carcinomas. Over 95% of HGSC harbor TP53 mutation [7, 8]. It is well accepted that OC, and particularly HGSC, needs to be diagnosed as early as possible, preferably in preinvasive stages of disease. However, currently, there are no reliable biomarkers for HGSC detection with a lead-time of more than 1 year, when curative intervention is still possible [9, 10, 11]. Identification of molecular alterations specific to HGSC or its precursor lesion could help identifying sensitive and specific biomarkers for detection of HGSC at its earliest stages.
The precursor lesions in the ovaries of Type II OC have been described [12] and are easily detected morphologically and histochemically. Originally epithelial OC have been thought to arise from ovarian surface epithelium. However, although numerous studies have explored the ovaries for possible precursor lesions of ovarian cancer, none have been found. Tumors arising from the fallopian tube (FT) had already been described in the 1950s [13], but it was not until the 2000s that the important role of fallopian tube epithelium in ovarian tumors carcinogenesis was further investigated. Recent evidence indicates that at least part of HGSC originates from the FT epithelium [14, 15, 16]. Potential precursor lesions of HGSC, including a TP53 mutant single-cell epithelial layer and serous tubal intraepithelial carcinoma (STIC) were identified in the fimbriae of the FT removed as part of prophylactic surgery and in patients with advanced stage sporadic HGSC of the ovary, FT and peritoneum [5, 16]. The morphological characteristics of STIC include disorganized, pleomorphic, hyperchromatic, and enlarged epithelial cells with highly atypical nuclei [4]. Regarding to terminology, others prefers the use of the term ‘high-grade tubal intraepithelial neoplasm’ (HGTIN) instead of STIC [17].
STIC lesions are associated with increased risk for HGSC [18, 5]. Identification of the biomarkers associated with STIC is therefore critical for identifying the mechanisms of early ovarian tumorigenesis and improving the early detection and prevention of OC. Until now, besides the established markers (TP53, p16, and Ki-67), several proteins were reported to be differentially expressed in STIC lesions including Stathmin 1 [19], signal transducer and activator of transcription 3 (phosphorylated STAT3 Tyr705) and suppression or loss of protein inhibitor of activated STAT3 (PIAS3) [20], Bcl-2 and
In this study, we have identified several candidate biomarkers that were differentially expressed in an in vitro model of STIC lesion and in pre-HGSC serum samples versus controls and validated overexpression of these candidate biomarkers in patients’ STIC lesions. We have additionally assessed immune cells in STIC lesions and correlated their level and location with biochemical markers.
Flow cytometry analysis of FT cells. FT epithelial cells were isolated from healthy fallopian tube fimbriae as described in Methods. Newly isolated cells were allowed to reach confluence, were permeabilized and stained for pan-cytokeratin or PAX8 markers. A representative of three cultures is presented.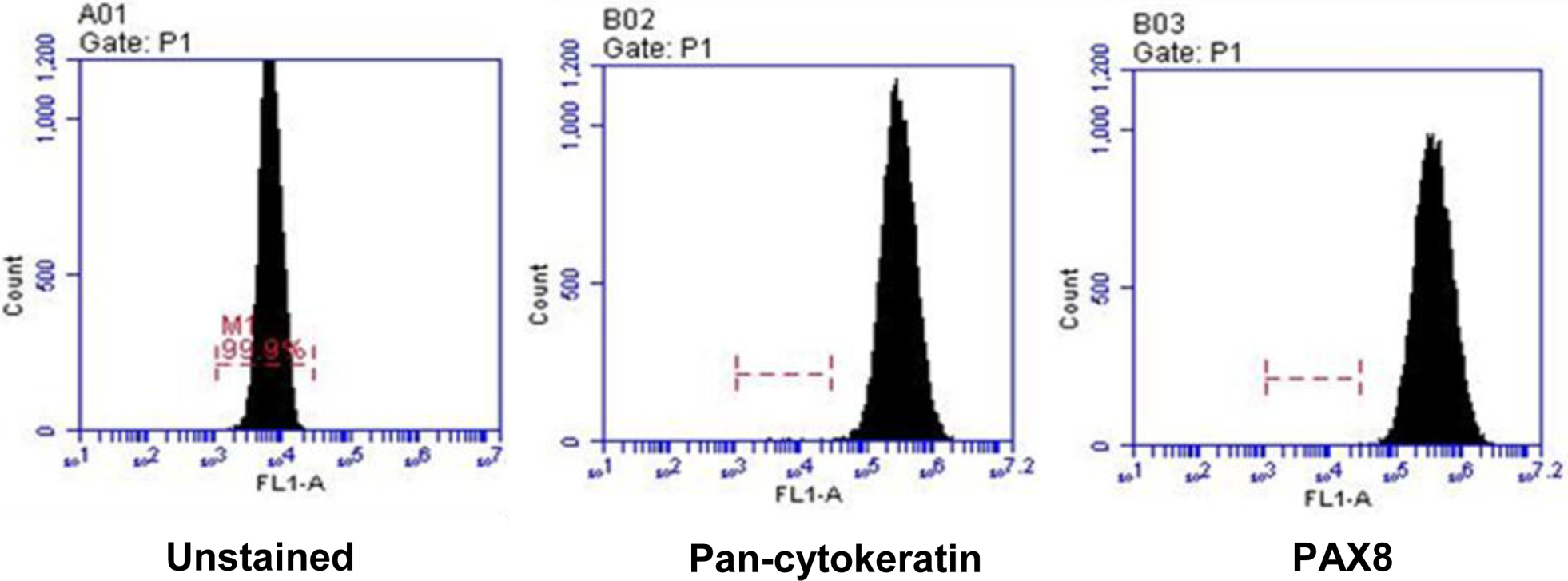
Patients and clinical data
Tissue samples were obtained from patients with the diagnosis of STIC in fallopian tubes, on either prophylactic salpingectomy, salpingo-oophorectomy or on debulking specimens performed for ovarian cancer at the UPMC Magee-Women’s Hospital between 2014 and 2016. Fallopian tube sections were submitted based recommended SEE-FIM protocol. Formalin-fixed paraffin-embedded tissue sections of fallopian tubes were stained with hematoxylin and eosin and reviewed by an independent pathologist to confirm the presence of invasive carcinoma, STIC or histologically normal tissue (Fig. 1). The presence of STIC was confirmed by positive p53 nuclear staining and by increase in proliferation index by ki67 immunohistochemical stains. A total of 10 patient were identified, however deeper sections for analysis did not contain sufficient tissue for evaluation, leaving a total of 6 final samples.
Fallopian tube epithelial cells isolation and culture
Fresh, healthy fallopian tube fimbriae were obtained from 3 donors at UPMC Magee-Women’s Hospital. Specimens were collected in a 50 ml of DMEM F12 Ham medium (Sigma-Aldrich) supplemented with 10% FBS (Gibco) and 1% Penn/Strep (Sigma- Aldrich). Upon receipt, FT tissues were processed under sterile tissue culture conditions as described earlier [30, 31] and cultured in Prigrow cell culture medium (Applied Biological Materials Inc., catalog #TM004) supplemented with 2% Ultroser G serum substitute (Pall France). To immortalize FT epithelial (FTE) cells, we used lentiviral transduction with SV40 large plus small T antigen (FTE-Tag) that suppresses TP53 signal transduction pathway thus mimicking the genetic makeup of STIC lesion [31]. Unmodified lentivirus was used in control cells. Recombinant lentiviral expression vector (Catalogue # CILV01) and an empty lentiviral expression vector (Catalogue # LV000) were commercially purchased from ALSTEM and used to transduce primary FTE cells in the presence of transduction reagent TransPlus following the manufacturer’s protocol [32, 33, 34].
Flow cytometry
Isolated FT epithelial cells were cultured until they reached confluence. Cells were trypsinized, washed, permeabilized with cytofix/cytoperm buffer (Becton Dickinson), and resuspended with 0.3% saponin (Sigma) in FACS-buffer. Cells were incubated anti-human pan-cytokeratin AE1/AE3 antibody conjugated with Alexa Fluor
Mass spectrometry
Serum-free culture medium conditioned for 48 hours by SV40 transformed and primary FTE was analyzed using LC-MS/MS on an Orbitrap Fusion system using an Acclaim PepMap 100, 75
Study populations for serum biomarkers analysis
We have utilized serum samples collected in the course of a prospective Prostate, Lung, Colorectal, Ovarian (PLCO) clinical trial conducted by the NIH [35, 36, 37, 38, 39]. Serum samples were collected from 29 HGSC cases 6–18 months prior to diagnosis and from 203 healthy controls matched by age, race and time of sample draw [35, 40]. For all studies, written, informed consents were obtained from each participating subject or from patient’s guardian. All subjects were accrued under protocols approved by each site’s Institutional Review Board. Specimens were transferred in accordance with material transfer agreements between the shipping and receiving institutions regarding transfer of specimens without personal identifiers.
Luminex analysis
All proteins were analyzed using multiplex bead-based immunoassay using Luminex platform (Luminex, Inc.) as previously described [41]. Kits were purchased from EMD/Millipore.
Immunohistochemistry
Immunohistochemical staining (IHC) was per- formed on Benchmark Ventana system (Ventana Medical Systems). Staining for ADAMST13 was performed using polyclonal antibody (Pierce ThermoScientific), for AHSG by using polyclonal antibody (Pierce ThermoScientific), for UCHL1 (PGP9.5) by using polyclonal antibody (Cell Marque), and for GAPDH by using polyclonal Ab (Santa Cruz Biotechnology). Immunohistochemical staining recognizing different populations of T cells (CD3, CD4, CD8) was performed using CONFIRM anti-CD3, anti-CD4, and anti-CD8 rabbit monoclonal antibody (Roche Diagnostic) on Benchmark Ventana. The results were evaluated independently by two pathologists. Studies were interpreted in conjunction with appropriate negative (incubation without primary antibodies) controls. Additionally, staining in non-neoplastic epithelium was used an internal negative control.
Statistical analysis
Descriptive statistics for serum concentrations of each of the tested biomarkers were calculated for each subject group using GraphPad Prism version 6 (GraphPad Software, Inc.). Comparisons of average biomarker concentrations between OC cases and control groups were performed using the Mann-Whitney non-parametric U test. P values less than 0.05 were considered statistically significant.
Proteins differentially present in the conditioned media of SV40 transformed vs. control FTE cells
Proteins differentially present in the conditioned media of SV40 transformed vs. control FTE cells
Biomarker discovery in an in vitro model
Secretory epithelial cells were isolated from healthy control FT obtained from three cancer-free patients at Magee-Women’s Hospital. The FTE nature of FT cells was confirmed by flow cytometry after staining for pan-cytokeratin and PAX8 markers (Fig. 1). From each primary culture we generated isogenic pairs of cells, one transfected with unmodified lentivirus and another- immortalized with SV40 large plus small T antigen (FTE-Tag) that suppresses TP53 signal transduction pathway thus mimicking the genetic makeup of STIC lesion [31]. Transfection with either unmodified or recombinant lentivirus did not change the pan-cytokeratin or PAX8 expression as measured by flow-cytometry (data not shown). We hypothesized that proteins secreted by cells with inactivated TP53 signaling correspond to those secreted by STIC. To test this hypothesis, and to establish a proof-of-principle of pre-neoplastic HGSC biomarker discovery, we analyzed serum-free culture media from 3 isogenic pairs in duplicates by mass spectrometry to identify several proteins whose levels were at least 2-fold higher in immortalized versus primary cells with levels of significance at
H&E staining of STIC lesion with adjacent superficially invasive tumor (A – STIC, B – invasive carcinoma); 200x magnification, A representative of 36 sections from 6 patients is presented.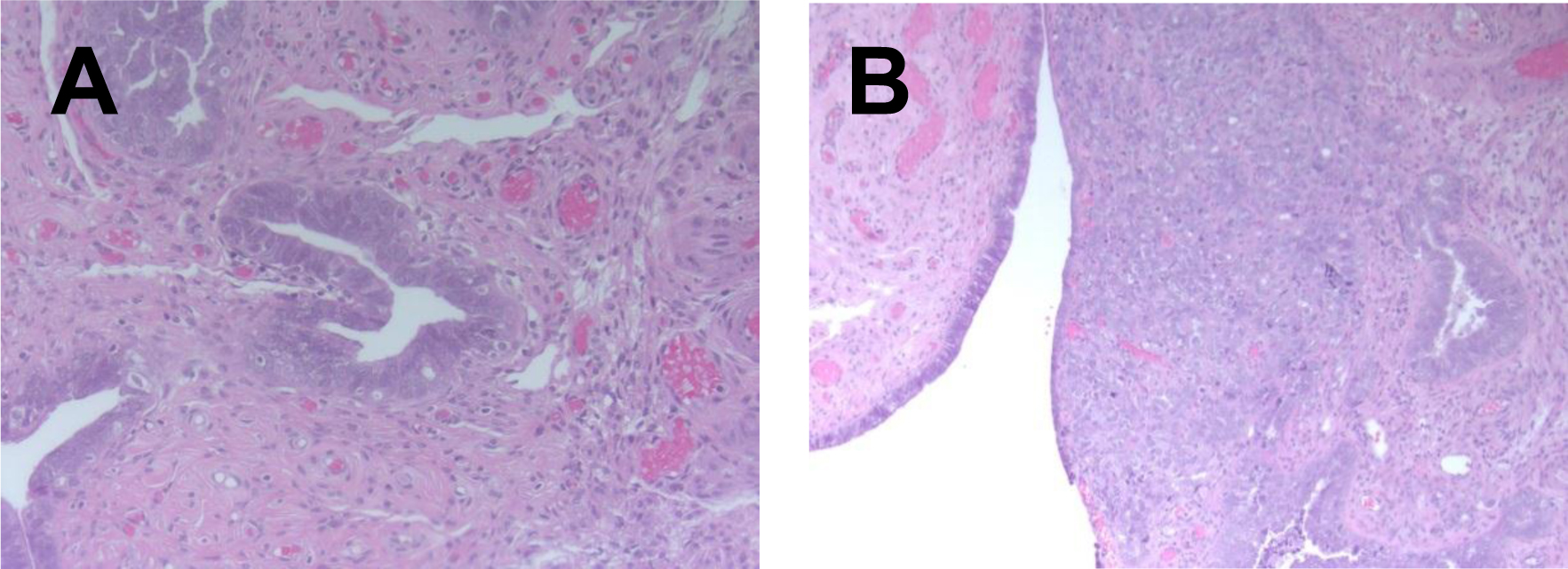
Elevated levels of ADAMTS13 in sera of HGSC cases collected 18–84 months before clinical diagnosis in PLCO prospective study. ADAMTS13 levels were measured using bead-based immunoassay.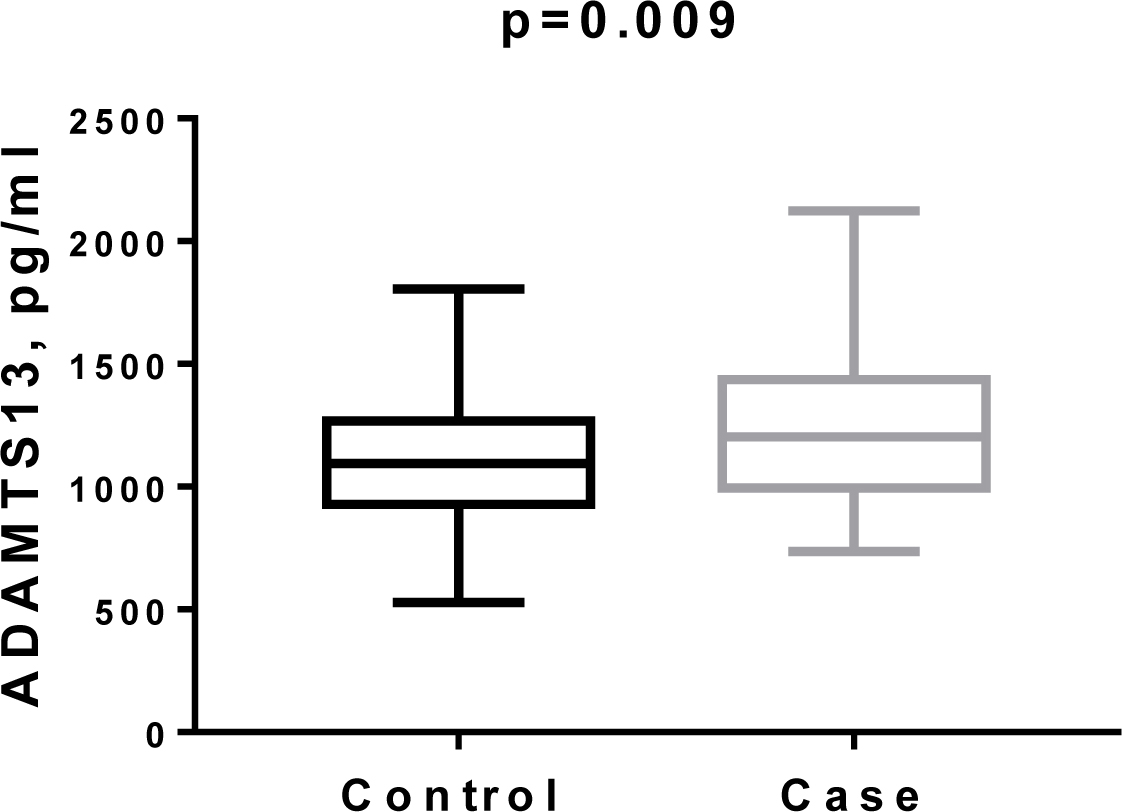
Immunohistochemical staining for ADAMS13, UCHL1, and GAPDH in normal fallopian epithelium (A) and in STIC (B), 200x magnification, A representative of 36 sections from 6 patients is presented.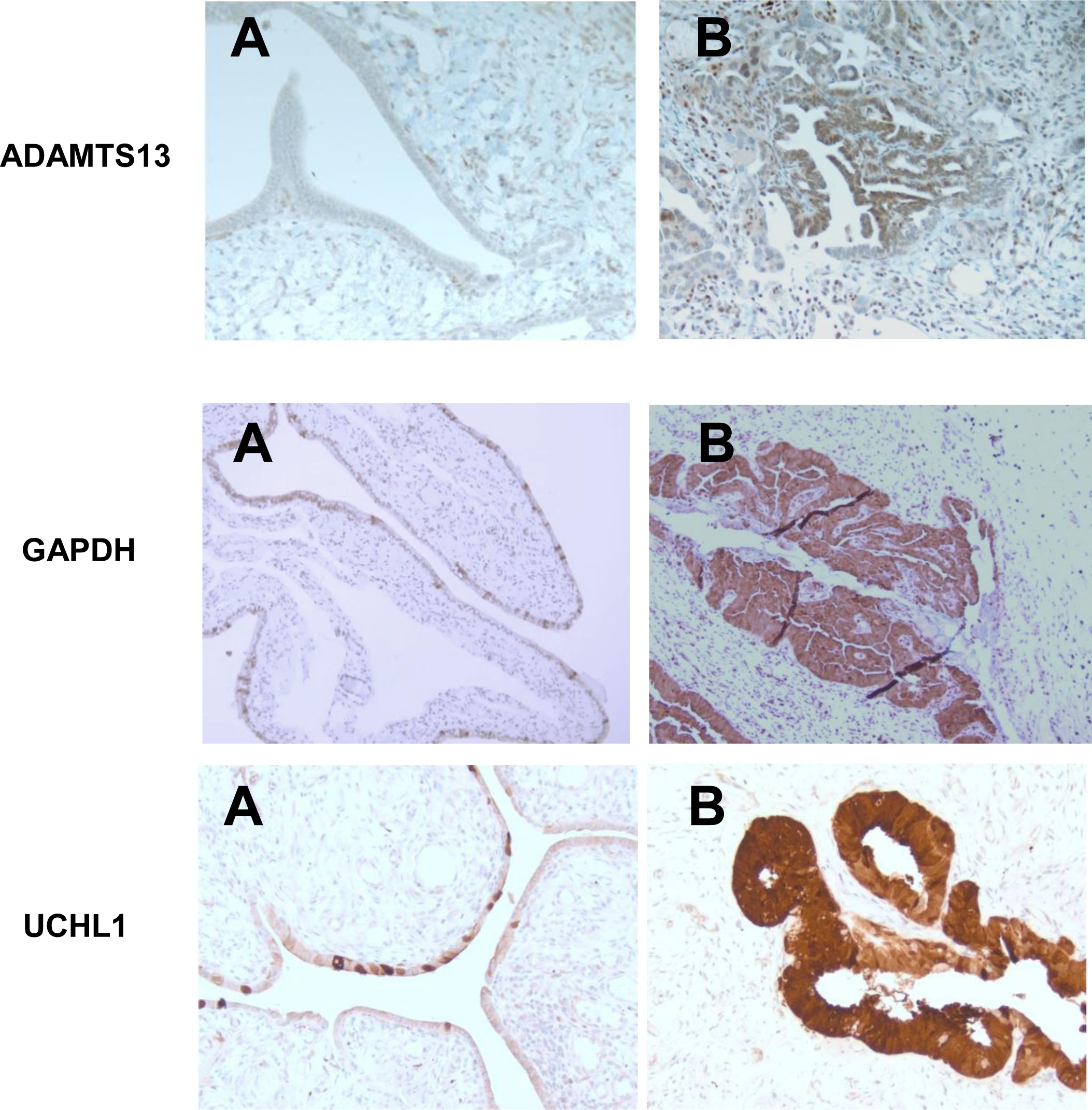
Immunohistochemical staining for CD3, CD4, CD8, and FOXP3 in normal FT, STIC, and HGSC. 200x magnification, A representative of 36 sections from 6 patients is presented.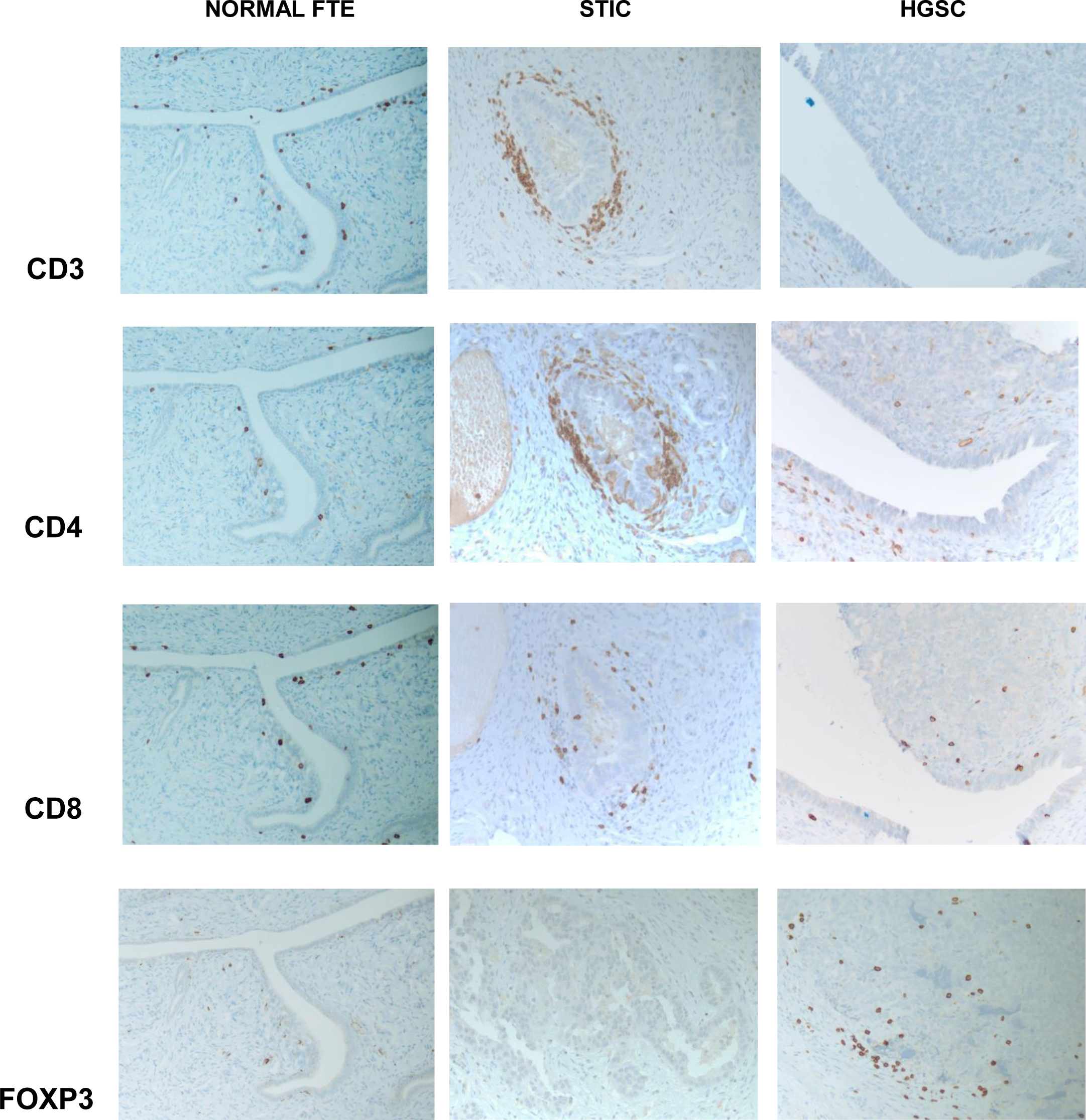
We performed evaluation of 58 serum markers in serum samples from PLCO participants collected 18–84 months before diagnosis with ovarian cancer [35, 40] and found that, along with CA125 and HE4, ADAMTS13 and fibrinogen (FBG) were significantly elevated (Fig. 3 for ADAMTS13).
IHC evaluation of GAPDH, ADAMTS13, and UCHL1 in STIC lesions and invasive HGSC
To confirm that STIC cells are the source of proteins identified in an in vitro model and in clinical specimens, we performed IHC in human samples of STIC, normal FT and invasive high-grade serous carcinoma. We found that GAPDH, ADAMTS13 and UCHL1 are significantly overexpressed in malignant cells (both STIC and invasive HGSC), while we did not observe the differential expression of ENO1 and FBG. For ADAMS13, tissue slides showed negative immunostaining in normal epithelium and stroma. In contrast, neoplastic epithelium in STIC and invasive carcinoma showed strong diffuse staining for ADAMTS13 with cytoplasmic and nuclear distribution (Fig. 4). For UCHL1, tissue slides showed weak patchy immunostaining in normal epithelium, while neoplastic epithelium in STIC and invasive carcinoma demonstrated strong diffuse staining for UCHL1 with predominantly cytoplasmic distribution (Fig. 4). The same pattern was observed for GAPDH: weak staining of occasional normal epithelial cells and diffuse strong staining in STIC and invasive carcinoma (Fig. 4).
Immune cells IHC
Since the aberrant protein expression may alter immune cells profiling and homing and because lymphocyte infiltrates have been observed in pre-neoplastic lesions [42, 43], we raised the question whether immune cells may recognize the emerging malignant cells of STIC lesion and infiltrate the early lesions. To that end we studied different subsets of T cells in normal FT, STIC, and invasive carcinoma by immunohistochemistry (Fig. 5). Tissue analysis revealed occasional CD3 positive T lymphocytes in benign tubular tissue, associated with epithelial layer and surrounding stroma. There is a mild predominance of CD8 positive subset of T cells. In STIC, there is a marked increase of CD3 positive T lymphocyte associated mainly with peri-neoplastic stroma. Both subtypes (CD8 and CD4 positive) of T cells were increased in STIC tissue. In contrast, the numbers of tumor-associated T lymphocytes were not increased in invasive carcinoma (Fig. 5). There were occasional CD3 positive T cells seen in the tumor mass, as well as in peri-tumoral stroma. It appears that there is a predominance of CD4 positive cells (Fig. 5). Immunostaining for FOXP3, T regulatory cell marker, reveals their increase in invasive carcinoma, compared to benign FT and STIC (Fig. 5).
Discussion
We have used two different approaches, Luminex analysis of pre-diagnostic HGSC serum samples and MS analysis of proteins secreted by cultured STIC-like cells, to ascertain whether these proteins might originate from patients’ STIC lesions. In this study, we report five proteins elevated in either pre-diagnostic sera or conditioned medium, ADAMTS13, FBG, GAPDH, ENO1, UCHL1. Three of them – ADAMTS13, GAPDH, and UCHL1, were overexpressed by IHC in both STIC lesions and invasive carcinoma. At the same time, no differential expression of FBG and ENO1 was observed indicating that these two proteins could originated from sources other than ovarian malignant cells or associated stromal cells.
Aberrant protein expression in early ovarian tumorigenesis may induce immune response as the immune contexture plays a key role in cancer development, prognosis, and treatment [44]. Thus, we have assessed immune cells homing in STIC samples in comparison with normal FT and invasive HGSC. Our findings show a marked increase of T cells in peri-neoplastic stroma surrounding STIC lesions, which indicates the immunogenic properties of neoplastic cells, with both CD4 positive and CD8 positive cells being present. In contrast, T cells are sparse in invasive carcinoma, indicative of immunosuppressive microenvironment. In addition, immunostaining for FOXP3, T regulatory cell marker, reveals the absence of these cells in benign FT and, most importantly in STIC lesions, and their presence in invasive carcinoma, again confirming the immune promoting microenvironment of STIC and immune suppressive or tolerogenic microenvironment of invasive carcinoma. This is an important observation since it provides an opportunity to distinguish between immunological profile of premalignant and malignant lesions and establish a marker for prognostic and diagnostics predictions. Additional studies are needed to confirm the relationship between the immune cell subtypes associated with histologic progression/persistence/regression of STIC, including dendritic cells, macrophages, MDSC, B and NK cells.
Although this is an observational study and does not include mechanistic experiments, the overexpression of ADAMTS13, GAPDH, and UCHL1 in women’s STIC lesion has not been previously reported. In addition, these data demonstrate that (i) cancer-associated elevated blood levels of some proteins (in this case ADAMTS13) could results from the premalignant lesions, and that (ii) in vitro modelling of early stage of HGSC tumorigenesis by inactivating p53 in normal FTE could be a useful tool for discovery of mechanisms of early HGSC development and potentially identifying early biomarkers of HGSC.
Altered STIC expression of some proteins could be directly upregulated in response to TP53 knockdown. Tumor suppressor, TP53, that is mutated in
ADAMTS13 is mainly known for prevention of microvascular thrombus formation by decreasing thrombotic activity through proteolysis of its only known substrate, von Willebrand factor (VWF) [57, 58, 59]. Recent studies demonstrate that ADAMTS13 also plays a role in the down-regulation of inflammation, regulation angiogenesis, and degradation of extracellular matrix [60, 61, 62]. There is not enough experimental evidence to speculate on possible functional effects of ADAMTS13 overexpression in malignant ovarian cells.
Ubiquitin carboxyl terminal hydrolase 1 (UCHL1) has two opposing biologic functions, functioning as both a ubiquitin carboxyl-terminal hydrolase and ligase [63]. It has been reported as either an oncogene or a tumor suppressor [56, 64, 65, 66, 67, 68, 69, 70]. Knockdown of UCHL1 in OC cell lines promoted cell proliferation and reduced cell apoptosis [66] UCHL1 also promotes prostate cancer metastasis through EMT induction [71]. Conversely, in other publications, UCHL1 was described as a tumor suppressor and its overexpression was attributed to a response to tumor growth [56, 66, 68, 72]. In this role, UCHL1 forms a complex with p53/MDM2/ARF to promote p53 signaling and mediate apoptosis through stabilizing p53 [56, 73, 74]. Thus, an experimental strategy has to be devised to ascertain a role of UCHL1 expression in STIC lesions for HGSC progression.
GAPDH was initially identified as a glycolytic enzyme, but emerging evidence indicates that GAPDH is implicated in several other functions including regulation of apoptosis (reviewed in [75]) GAPDH is frequently upregulated in various cancers. TP53 not only modulates GAPDH gene expression but also affects its functions (reviewed in [75]). High GAPDH expression indicate early disease progression in advanced serous ovarian cancer [76, 77].
Currently, there is lack of efficient markers to identify pre-invasive lesions at highest risk of progression to invasive carcinoma. The development of validated tools to determine markers of disease progression will identify patients at high-risk, suggest novel OC chemoprevention and immunoprevention agents, and provide clinically relevant cellular and molecular biomarkers for monitoring outcome in OC prevention trials. Our results here offer novel potentially effective and useful markers associated with high-grade histology and malignant lesion progression.
