Abstract
BACKGROUND:
Recent evidence support that netrin-1 involves in colorectal carcinogenesis.
OBJECTIVE:
This study was to evaluate the performance of serum netrin-1 for detection of colorectal cancer (CRC) in both clinical/screening sets.
METHODS:
A total of 115 consecutive patients with CRC and matched healthy controls were included in Clinical Set. Fifty subjects with CRC, 50 subjects with advanced adenoma (AA), and 150 matched control participants free of neoplasia were included in Screening Set.
RESULTS:
In Clinical set, subjects with CRC presented higher levels of serum netrin-1 (513.9
CONCLUSIONS:
It suggests netrin-1 as a potential biomarker for CRC detection.
Abbreviations
Flow chart of participants’ inclusion.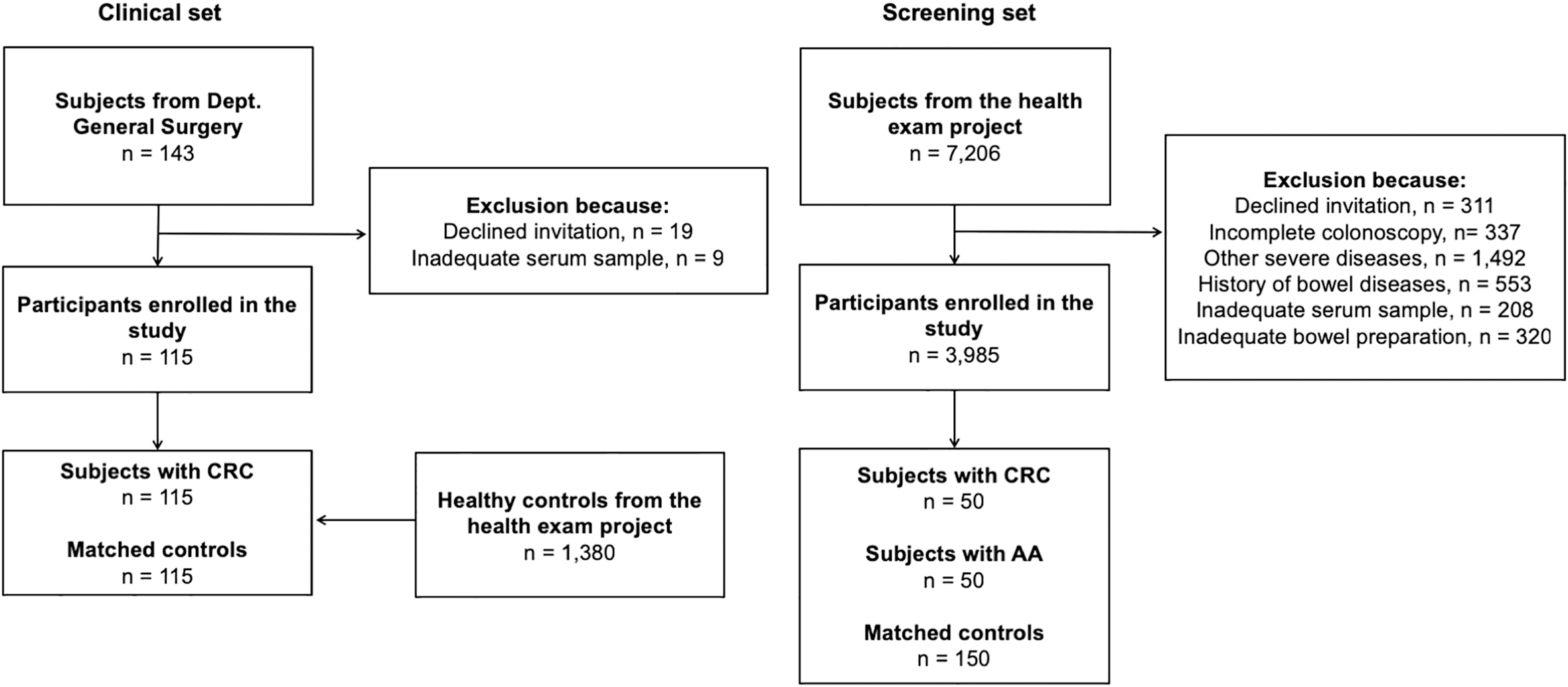
In western countries, colorectal cancer (CRC) is now the second most common cancer in men and the third in women [1]. In China, the incidence and mortality of CRC increase steadily during the past two decades, mostly because of the aging population and industrialized lifestyle [2].
As secreted and membrane-bound proteins, netrins play key roles in biological processes, e.g. neural cell migration, differentiation and apoptosis. Netrin-1 was found initially to involve in axonal guidance in enteric nervous system and central nervous system [3, 4]. Classical netrin-1 functions via two receptors: the deleted in colorectal cancer (DCC) family and the uncoordinated5 (UNC5) family [5, 6]. Furthermore, netrin-1 signaling has been presented to be upregulated in various cancer types. The evidence raises questions if netrin-1 involves in cell motility in cancer biology [7].
In clinics, detection at early stages could significantly lower incidence and mortality of CRC [8]. Now, colonoscopy is the gold standard for CRC and its precursors, however its clinical practice as a screening test is limited by patients’ low adherence and high expense [9, 10]. Nevertheless the stool-based tests, e.g. fecal immunochemical tests, are regarded as alternatives, sample collection and storing still limit their application [11]. Therefore, blood-based markers might be favorable for CRC screening, because: 1) samples collected easily in health checkup; 2) minimal invasion; and 3) affordable costs.
Inspired by previous experimental findings [7], Ramesh et al. [12] evaluated the plasma levels of netrin-1 in patients with various cancers. It found that plasma netrin-1 concentrations increased in breast, renal, prostate and liver cancers, compared with controls. In the study, netrin-1 also presented higher levels in colon carcinoma (
Due to the low prevalence of CRC in screening populations, most studies were designed to evaluate novel biomarkers in clinical settings [15, 16]. In this study, we measured serum levels of netrin-1 in CRC patients recruited from the department of general surgery and healthy controls from the health-exam projects. Moreover, we also evaluated in a screening set, including subjects with CRC and advanced adenoma (AA) and matched controls.
Methods
Study design and population
The clinical set: a total of 143 consecutive patients with CRC were recruited from Department of Gastrointestinal Surgery, China-Japan Union Hospital of Jilin University (from February to November 2018). Meanwhile, we also recruited healthy controls (
Comparisons of serum netrin-1 (pg/ml, median [IQR]). A: Clinical Set; B: Screening Set; AA, advanced adenomas; CRC, colorectal cancer.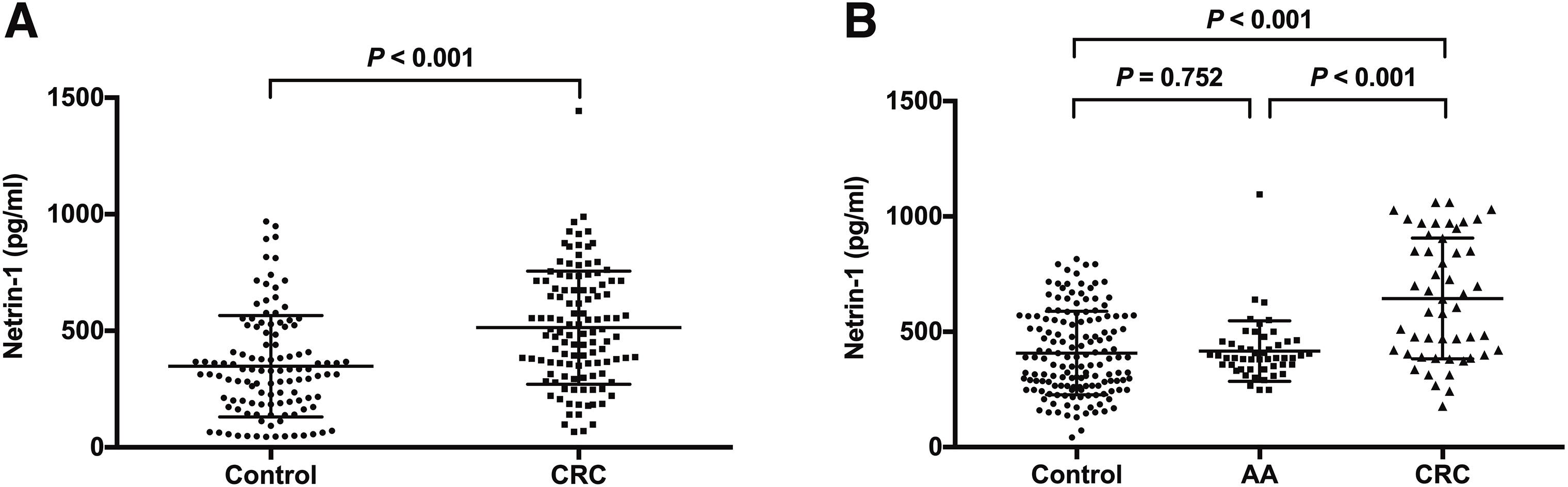
The screening set: both cases and control subjects were selected from The First Affiliated Hospital of Soochow University and Suqian Affiliated Hospital of Xuzhou Medical University. Of 7,206 eligible participants from the health exam project recruited between 2017 and 2018, 50 subjects with CRC were included in the study. One subject with AA and three control participants free of neoplasia were matched to each CRC case by age and sex.
The main differences between the two sets: 1) subjects from the clinical set were recruited and blood samples were collected after diagnosis, 2) in the screening set, cases were newly diagnosed by colonoscopy and blood draw was completed before colonoscopy. The selection process of participants was presented in Fig. 1.
Written informed consent was obtained from each participant. This study was approved by the Ethics Committee of each center, in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki), printed in the British Medical Journal (18 July 1964).
CRC is colonoscopy-detected and biopsy-confirmed. The diagnosis of AA contains: high-grade dysplasia, villous, or tubovillous architecture, or tubular adenoma with at least 1 cm. The inclusion criteria for healthy controls were gastrointestinal-healthy without other systematic diseases or malignant diseases.
Laboratory examination
The clinical set: blood samples were taken before any treatment for the CRC cases in hospitals. The screening set: the blood samples of all the participants were collected when they attended to the health-exam projects. All the blood samples were centrifuged and stored at
Statistical analysis
The Student’s
Results
The characteristics of all participants from Clinical set and Screening set were indicated in Table 1. The calculation of sample power (post hoc) was in Supplementary Fig. 3.
Study population characteristics
Study population characteristics
Data are mean
As shown in Fig. 2A (Clinical set), subjects with CRC presented higher levels of serum netrin-1 (513.9
Odds ratio of netrin-1 for CRC
In Table 2, the optimum cutoff value in Clinical set was 367.1 pg/mL, based on the Youden index. The participants were divided into two groups, according to the cutoff value. Binary logistic regression found that compared with the low netrin-1 group, the high group presented increased risk of CRC (OR
Odds ratio of serum netrin-1 for colorectal cancer, by sex, location and stage
Odds ratio of serum netrin-1 for colorectal cancer, by sex, location and stage
CI, confidence interval.
As indicated in Table 3, the AUC of netrin-1 in Clinical set was 0.703 [95% CI 0.636–0.770], sensitivity 33.9% at 90% specificity). To further validate the panel, the performance was evaluated in Screening set. The AUC was 0.759 [95% CI 0.680–0.837], and sensitivity at 90% specificity was 46.0%.
Performance of netrin-1 in the diagnosis of colorectal cancer, by sex, location and stage
Performance of netrin-1 in the diagnosis of colorectal cancer, by sex, location and stage
AUC, area under ROC curve; CI, confidence interval.
This study reveals that the serum levels of netrin-1 are higher in subjects with CRC than healthy controls or subjects with AA, both in a clinical set and a screening set. Furthermore, higher levels of netrin-1 are associated with increased risk of CRC. Lastly, circulating netrin-1 presents a fine performance for CRC detection in both two sets.
The enteric nervous system (ENS) is the intrinsic neural network of the gastrointestinal tract, which plays a fundamental role for gut functions and homeostasis [17]. The association between the ENS and CRC remains poorly understood, nevertheless perineural invasion and neoneurogenesis have been found to be involved in the development of CRC [18]. Recent evidence supports the netrin-1 signaling pathway links the ENS to colorectal carcinogenesis [3].
Mehlen et al. [19, 20] found the apoptotic functions of netrin-1 receptors could be aborted by binding with netrin-1. Mazelin et al. [21] presented the increased expression of netrin-1 could benefit survival of various cancers, including colorectal cancer. Furthermore, Paradisi and colleagues [22] discovered that netrin-1 involved in the motility of epithelial cancer. They found that NF
In consideration of the diversities between clinic-diagnosed patients and screening-detected patients, e.g. stage distribution or histological features, the performance of blood-/stool based markers in screening sets might differ it in clinical sets [26, 27]. Furthermore, we evaluated the serum levels of netrin-1 in both an independent clinical set and a true screening set, which offered the parallel comparison of diagnostic performance between two situations [28].
Our study has some limitations. To begin with, blood samples are collected at one time-point, which fails to assess the dynamic change of circulating netrin-1. Secondly, given the previous findings, the serum levels of netrin-1 in lung cancer, renal cancer, and gastric cancer should be evaluated in further studies to assess the specificity. Furthermore, this study is a retrospective design and thus large prospective studies to assess the role of netrin-1 in the development of CRC are needed. Lastly, comparisons with other markers for CRC, e.g. carcinoembryonic antigen, are needed.
Conclusions
Our study reports circulating netrin-1 levels in CRC in both a clinical set and a screening set. It suggests netrin-1 as a potential biomarker in the screening and detection of CRC.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-190340.
sj-docx-1-cbm-10.3233_CBM-190340.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-190340.docx
Footnotes
Acknowledgments
We thank the participants and relevant research staff for their contributions to the study. This study was funded by Medical and Health Project of Jilin Province (No. 20190304047YY) and Municipal Directive Technological Plan of Suqian (Z2018198).
Conflict of interest
The authors declare that they have no conflict of interest.
