Abstract
BACKGROUND:
To investigate the feasibility of cerebrospinal fluid (CSF) CYFRA 21-1 levels as a therapeutic monitoring biomarker in leptomeningeal carcinomatosis (LMC) patients undergoing ventriculo-lumbar perfusion (VLP) chemotherapy.
METHODS:
The levels of CYFRA 21-1 in 42 CSF samples from 15 LMC patients were analyzed using an electrochemiluminescence immunoassay. Samples were collected at individual time points during VLP chemotherapy. Therapeutic outcomes were measured as improvements in the Karnofsky Performance Status (KPS) score and decreasing intracranial pressure (ICP) as the main endpoint of VLP chemotherapy. Changes in CSF CYFRA 21-1 levels, protein levels, and cytology results were also investigated. We subsequently evaluated whether these changes were correlated with KPS score and ICP.
RESULTS:
The CSF CYFRA 21-1 levels at individual time points were associated with KPS score and ICP. The KPS scores (
CONCLUSIONS:
CSF CYFRA 21-1 may have utility as a therapeutic monitoring biomarker to design personalized therapeutic strategies in LMC patients undergoing VLP chemotherapy.
Background
CYFRA 21-1 is a fragment of cytokeratin 19, a component of the structural proteins that maintain the stability of epithelial cells, and has been investigated as a serum biomarker in several cancers [1, 2, 3, 4]. Recent studies have reported evidence supporting CYFRA 21-1 in cerebrospinal fluid (CSF) as a diagnostic and prognostic indicator for leptomeningeal carcinomatosis (LMC), a multifocal involvement of malignant cells in the leptomeninges and/or CSF in metastatic solid tumors [5, 6, 7, 8, 9, 10, 11].
With current advances in understanding, and improvement in diagnostic tools and systemic cancer therapies, the survival of cancer patients has been prolonged and the incidence of LMC has also increased [11, 12, 13]. LMC was generally regarded to be a debilitating condition, leading to end-of-life care without an established optimal treatment. However, the development of new therapeutic methodologies, such as ventriculo-lumbar perfusion (VLP) chemotherapy, which decrease the disruption of CSF flow, have enabled us to expect longer survival of patients with LMC [14, 15, 16, 17]. For these reasons, evaluating treatment response in patients with LMC has become more important in decision making and designing personalized therapeutic strategies in clinical practice. Nevertheless, established biomarkers that can monitor the benefits of specific regimens in LMC patients, particularly in those involving new therapeutic options such as VLP chemotherapy are still unavailable.
In a previous study, we preliminarily reported the potential of CSF CYFRA 21-1 as a therapeutic monitoring tool for LMC patients undergoing VLP chemotherapy [9]. In the present study, we further evaluated the feasibility of CSF CYFRA 21-1 as a therapeutic monitoring biomarker through assessment at individual time points in additional LMC patients undergoing VLP chemotherapy.
Methods
LMC patients (
Analysis of therapeutic responses during ventriculo-lumbar perfusion (VLP) chemotherapy in patients with leptomeningeal carcinomatosis. The black squares represent the serial levels of cerebrospinal fluid (CSF) CYFRA 21-1, the gray circles represent the serial levels of intracranial pressure, and the linear bars above the graph represent VLP chemotherapy. The Karnofsky performance status (KPS) score and CSF cytology results are presented below the graphs. (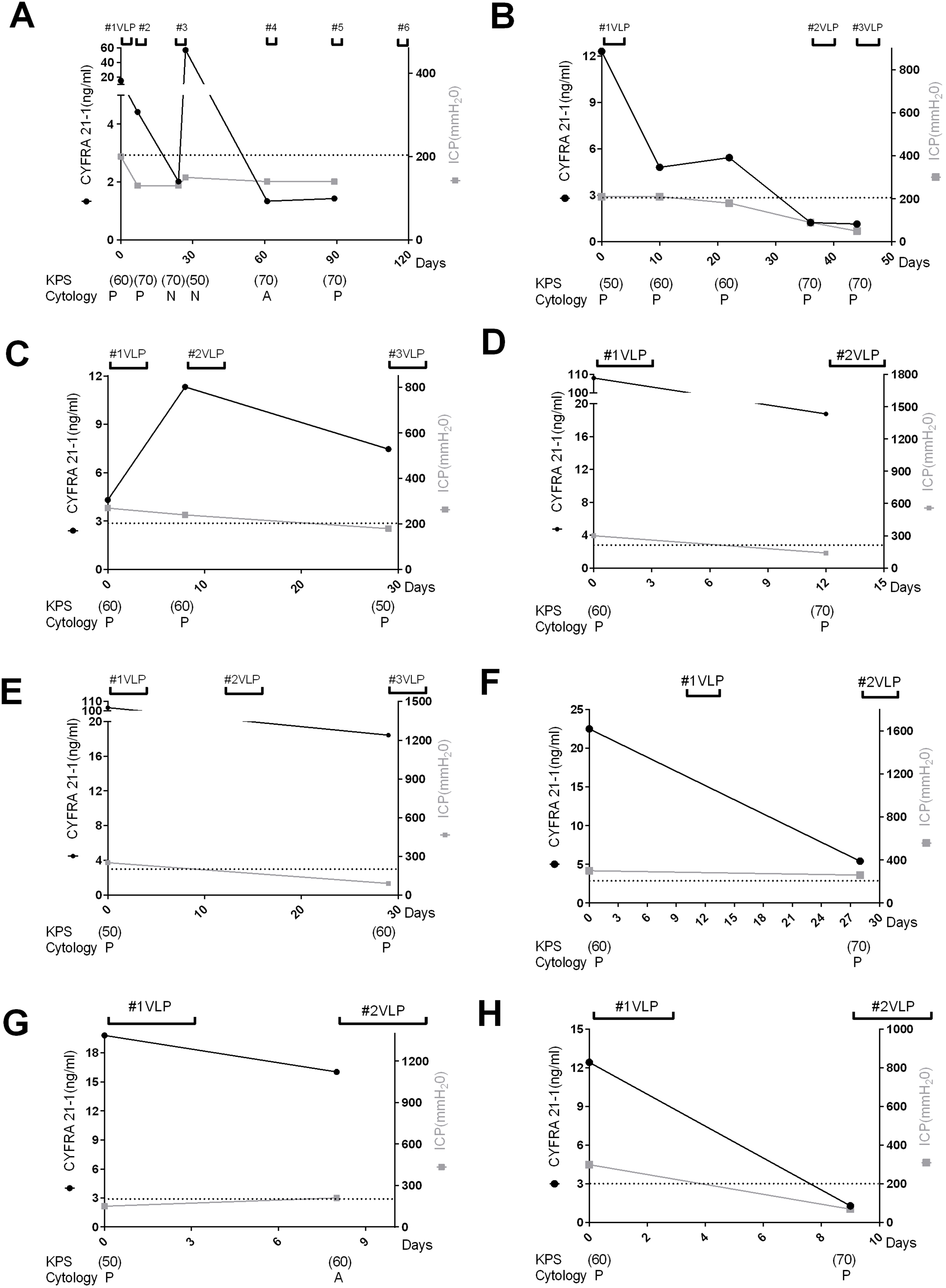
continued.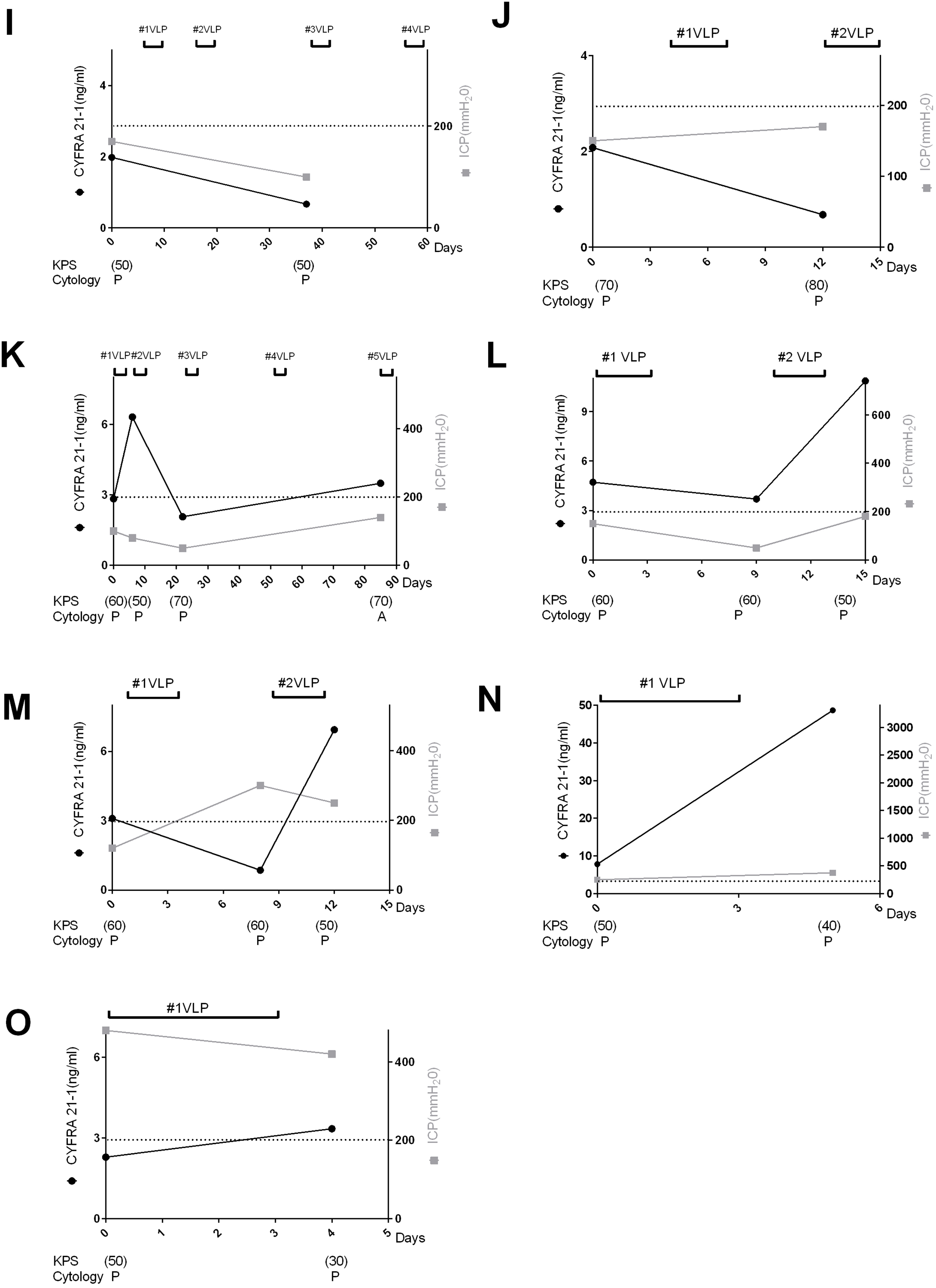
Analysis of therapeutic responses during ventriculo-lumbar perfusion (VLP) chemotherapy in patients with leptomeningeal carcinomatosis. The black squares represent the serial levels of cerebrospinal fluid (CSF) protein, the gray circles represent the serial levels of intracranial pressure, and the linear bars above the graph represent VLP chemotherapy. The Karnofsky performance status (KPS) score and CSF cytology results are presented below the graphs. (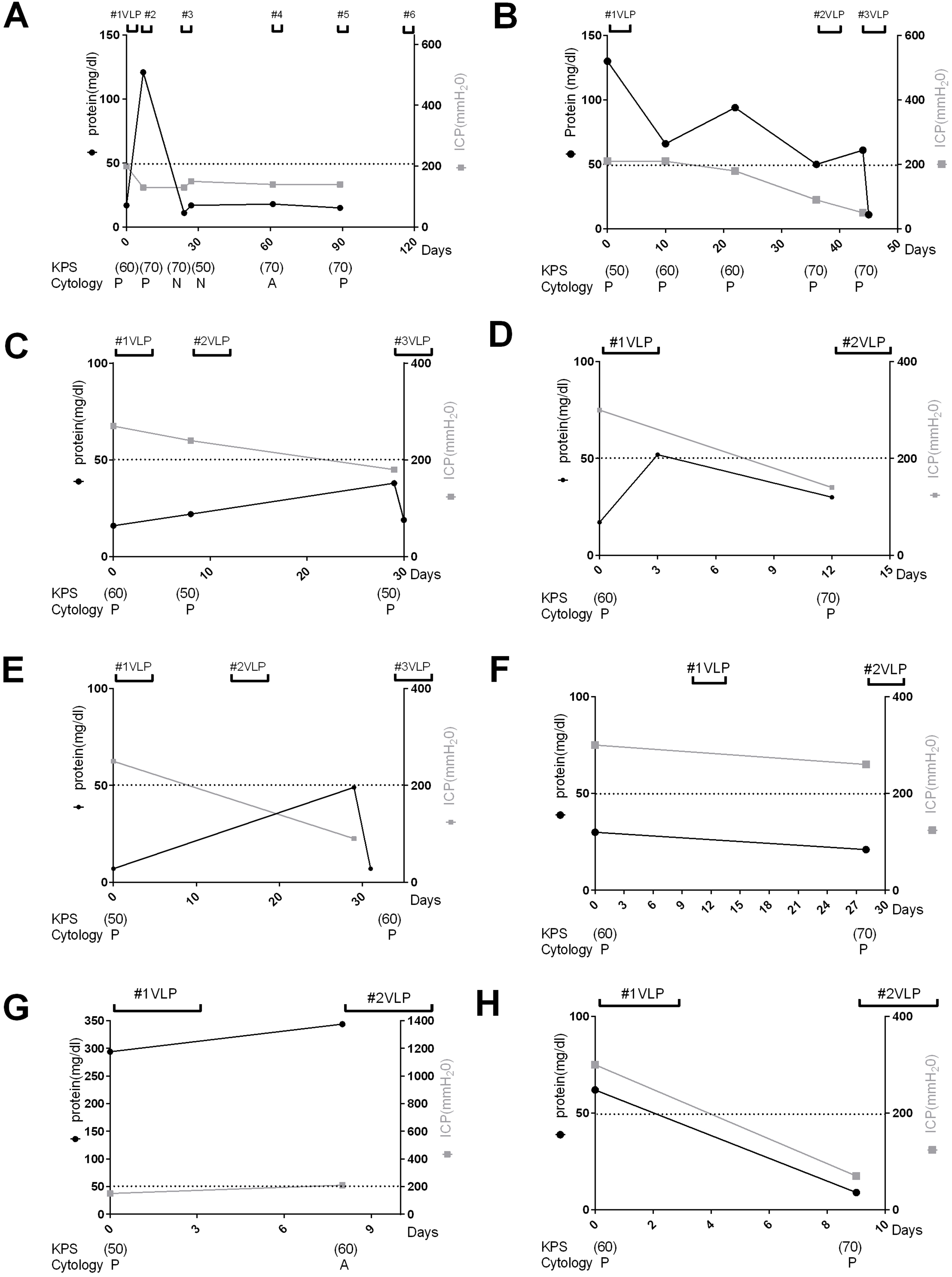
continued.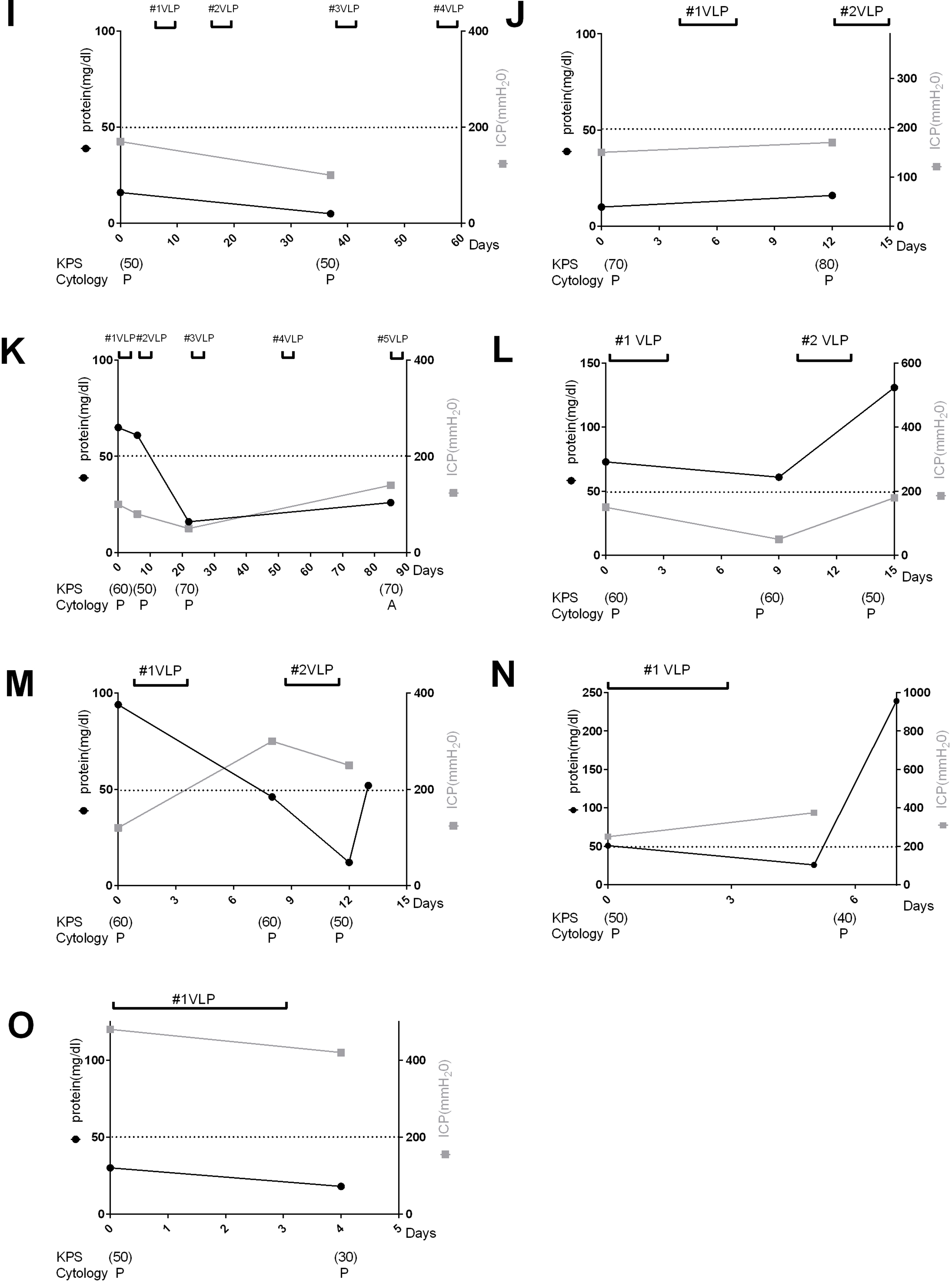
Different level of CSF CYFRA 21-1 using cut-off value at the individual time points during VLP chemotherapy according to the clinical or biological status
Abbreviation: VLP, ventriculo-lumbar perfusion, NS; not significant.
The comparison of therapeutic responses between groups with different level of CSF CYFRA 21-1 and CSF protein
Abbreviation: NS; not significant.
The concentrations of CSF CYFRA 21-1 were determined using a commercially available electrochemiluminescence immunoassay (Elecsys CYFRA 21-1, Roche Diagnostics, Penzberg, Germany) according to the manufacturer’s instructions [18]. CYFRA 21-1 levels were estimated from 42 preserved CSF samples that were collected from the lumbar subarachnoid space and stored at
Therapeutic outcomes were evaluated based on clinical improvement in the KPS score during treatment, and decreasing ICP as a main biological indicator of treatment response in LMC patients undergoing VLP chemotherapy [15, 16]. We analyzed positive (
Fisher’s exact test was performed to compare different status of CSF CYFRA 21-1 levels at individual time points according to the clinical and biological status. Mann-Whitney test or Student’s
Demographics
Among the 15 patients with LMC, the male-to-female ratio was 1:2, and the mean (
Assessment of CSF CYFRA 21-1 levels during VLP chemotherapy: Association with clinical, biological, and cytological responses
Decreasing CSF CYFRA 21-1 levels was associated with improvement of KPS scores in all but 2 cases (Fig 1C and I), and with decreasing ICP in all but 5 cases (Fig. 1C, G, J, M and O). Analysis of CSF protein levels, estimated 1 to 3 days after each CSF CYFRA 21-1 assessment, revealed a modest association with KPS score or ICP (Fig. 2B–E, M and N). By contrast, cytological responses were randomly associated with KPS scores and ICP at the individual time points (Figs 1 and 2).
Comparison of KPS score and ICP between groups with different CSF CYFRA 21-1 and CSF protein levels
Table 1 shows the high (
Significant differences in the therapeutic responses defined by KPS (median: 60 vs. 70,
Discussions
We observed that the CSF CYFRA 21-1 levels at individual time points were associated with therapeutic responses reflected in the KPS scores and ICP as a major end point of clinical trials involving LMC patients undergoing VLP chemotherapy [15, 16]. When we compared the KPS scores and ICP of patients with high and low CSF CYFRA 21-1 levels, significant differences were observed. On the contrary, neither association of CSF protein levels and therapeutic outcomes nor difference in therapeutic outcomes between groups with high and low CSF protein levels, showed statistical significance. Further, cytological responses were randomly associated with therapeutic responses. These results suggest that CSF CYFRA 21-1 may serve as a therapeutic monitoring biomarker, and may facilitate the design of individualized therapeutic strategies for LMC patients treated with VLP chemotherapy.
As a biomarker, CSF CYFRA 21-1 can provide objective quantitative data for therapeutic monitoring in LMC patients, whereas CSF cytology yields only qualitative information. Furthermore, CYFRA 21-1 levels can be easily measured using preserved CSF samples and smaller CSF volumes are required compared to those used in CSF cytology testing [18, 19]. Additionally, through additional analysis of CSF protein levels at slightly delayed time points of CSF CYFRA 21-1 assessment, it appears that CSF protein is less immediately sensitive to therapeutic responses than CSF CYFRA 21-1. Therefore, CSF CYFRA 21-1 is likely to be a more well-timed biomarker than CSF protein.
VLP chemotherapy is administered by a passage from the intra-ventricular reservoir to the lumbar drainage site [15]. CSF could be a more specific source of biomarkers in patients with LMC than other body fluids, such as blood; however it is not always suitable for longitudinal therapeutic monitoring due to relative limitations in accessibility. Nevertheless, in patients undergoing VLP chemotherapy, longitudinal CSF samples can be easily obtained from the intra-ventricular reservoir or lumbar drainage site. In contrast to our previous study, which included CSF samples from the lumbar subarachnoid or intraventricular space [9], only samples from the lumbar subarachnoid space were evaluated in this study. By removing the potential influence of sampling site/source in analyzing CYFRA 21-1 [20], more consistent results could be obtained.
In this study, the prognostic cut-off value (2.94 ng/mL) for CSF CYFRA 21-1 was based on overall survival as an outcome in a previous study [9]. The use of cut-off values could be more convenient in clinical practice than absolute values. Using this cut-off value, we observed significant difference in the therapeutic responses of patients with high and low CSF CYFRA 21-1 levels. In addition, the median KPS score of patients with low CSF CYFRA 21-1 levels was 70, and KPS score
Methodological limitations of the current study include the enrollment of LMC patients from a single referral center and its retrospective design, which may have resulted in unintentional selection bias and estimation of CSF CYFRA 21-1 levels at irregular time intervals. Addressing these issues warrant further prospective multicenter studies with longitudinal assessments of CSF CYFRA 21-1 levels at regular time points.
Conclusion
In conclusion, CSF CYFRA 21-1 appears to be a potential therapeutic monitoring biomarker as a complement to other clinical and biological parameters in patients with LMC undergoing VLP chemotherapy.
Footnotes
Acknowledgments
This study was supported by the National Cancer Center (1741580-1) and the National Research Foundation of Korea (NRF-2018R1A5A2023127).
Conflict of interest
SHS, KSY, GHS, YH report no disclosures. HJW has received grants from the National Cancer Center and National Research Foundation of Korea. KSH has lectured, consulted, and received honoraria from Bayer Schering Pharma, Biogen, Genzyme, Merck Serono, and UCB and received a grant from the National Research Foundation of Korea. Dr. Kim received a grant from the National Research Foundation of Korea; received consultancy/speaker fees from Alexion, Celltrion, Eisai, HanAll BioPharma, Merck Serono, Novartis, Sanofi Genzyme, Teva-Handok, and Viela Bio; serves on a steering committee for MedImmune/Viela Bio; is a co-editor for the Multiple Sclerosis Journal and an associated editor for the Journal of Clinical Neurology.
