Abstract
BACKGROUND:
New biomarkers may help us provide individualized prognosis and allow risk-stratified clinical decision making about radical treatment.
OBJECTIVES:
This study aimed to determine the tumor necrosis factor of receptor superfamily 19 (TROY) expression in urothelial carcinoma and its relationship to clinicopathological findings.
METHODS:
Immunohistochemical staining for TROY was carried out in 136 archival radical cystectomy specimens with immunoreactivity being stratified on a 0–9 scale. Expression scores for TROY were further stratified into negative (score 0) and positive (score 1 or greater). Median age was 65 years, and the median follow-up period was 50.7 months.
RESULTS:
Expression of TROY was significantly associated with the pathological stage (
CONCLUSIONS:
TROY may therefore be a new molecular marker to aid in identifying and selecting patients undergoing radical cystectomy who could potentially benefit from multimodal treatment.
Introduction
Urothelial carcinoma of the bladder (UCB) is the ninth most frequently diagnosed cancer in the world and the second most common cause of death among people with genitourinary tumors [1, 2]. At initial diagnosis, 70% of patients have non–muscle-invasive UCB and are treated with transurethral resection and intravesical therapy. Ranges from 50–70% of these tumors are recurrent and 10–15% of cases progress to muscle-invasive lesion [3]. For patients with muscle-invasive or non-muscle-invasive cancer resistant to intravesical therapy, radical cystectomy with bilateral pelvic node dissection is considered the standard treatment [4]. Following cystectomy, however, approximately 30% of patients with muscle-invasive cancer have distant metastasis or show local recurrence within 2–3 years [5]. The 5-year cancer-specific survival rate remains at only 50–60% [4]. While standard pathological findings such as stage and grade are used as parameters for cancer recurrence or progression, they are not sufficient to predict the biological aggressiveness of UCB. New biomarkers may help us provide individualized prognosis and allow risk-stratified clinical decision making about radical treatment.
Tumor necrosis factor receptor superfamily (TNFRSF) member 19 (TROY) is a type 1 transmembrane receptor located within the plasma membrane [6]. The human TROY gene (TNFRSF19) encodes a 423-amino acid isoform 1 (TNFRSF19.1) and a 417-amino acid isoform 2 (TNFRSF19.2) [7]. Recent research has shown that TROY plays an important role in the regulation of various biological activities, including cell proliferation, differentiation, survival, and apoptosis [8]. In addition, TROY expression has been reported in several tumors, including, for instance, melanoma, glioblastoma, and intestinal type gastric cancer [9, 10, 11], but not in UCB. The primary objectives of this study were to describe the expression pattern of TROY in UCB and to investigate the clinicopathological significance of the observed differences in patients with UCB who underwent radical cystectomy.
Materials and methods
Patient population
From 1990 to 2015, 161 consecutive patients who had undergone radical cystectomy with pelvic and iliac lymphadenectomy at Kitasato University Hospital were included in this retrospective study. In total, 10 patients who had histologic variants including squamous cell carcinoma, adenocarcinoma, and small cell carcinoma and 15 patients who had been previously treated with neoadjuvant chemotherapy were excluded. None of the remaining patients were treated preoperatively with either systemic chemotherapy or radiation therapy. The final study group consisted of 111 (81.6%) male and 25 (18.4%) female subjects, with a median age of 65 years (range, 40–82; mean, 64.1). The median follow-up was 50.7 months (range, 0.7–288.3; mean, 77.3) for patients who were alive at the last follow-up. When a patient died, the cause of death was determined by the patient’s physician, chart review supported by death certificate, or death certificate alone. In total, 33 patients (24.3%) received adjuvant chemotherapy after surgery for adverse pathological characteristics including regional lymph node metastases or extravesical involvement. A total of 34 patients (25.0%) received cisplatin-based treatment for disease recurrence. This study was approved by the ethics committee of Kitasato University School of Medicine and Hospital (B17-010).
Immunohistochemistry and scoring
Monoclonal antibodies specific for TROY were generated using the previously described hybridoma technology [12]. In brief, 300
Formalin-fixed, paraffin-embedded blocks representing the most invasive areas of each tumor were collected for further investigation. Normal urothelia were harvested from cystectomized specimens. The 2002 TNM classification was used for pathological staging, and the 1973 World Health Organization classification was used for pathological grading. Lymphovascular invasion (LVI) determined the presence of cancer cells within the endothelial space. Cancer cells that merely invaded a vascular lumen were considered negative [14].
The immunoreactivities of TROY in bladder cancer tissues (A): negative; (B): positive and normal urothelium (C).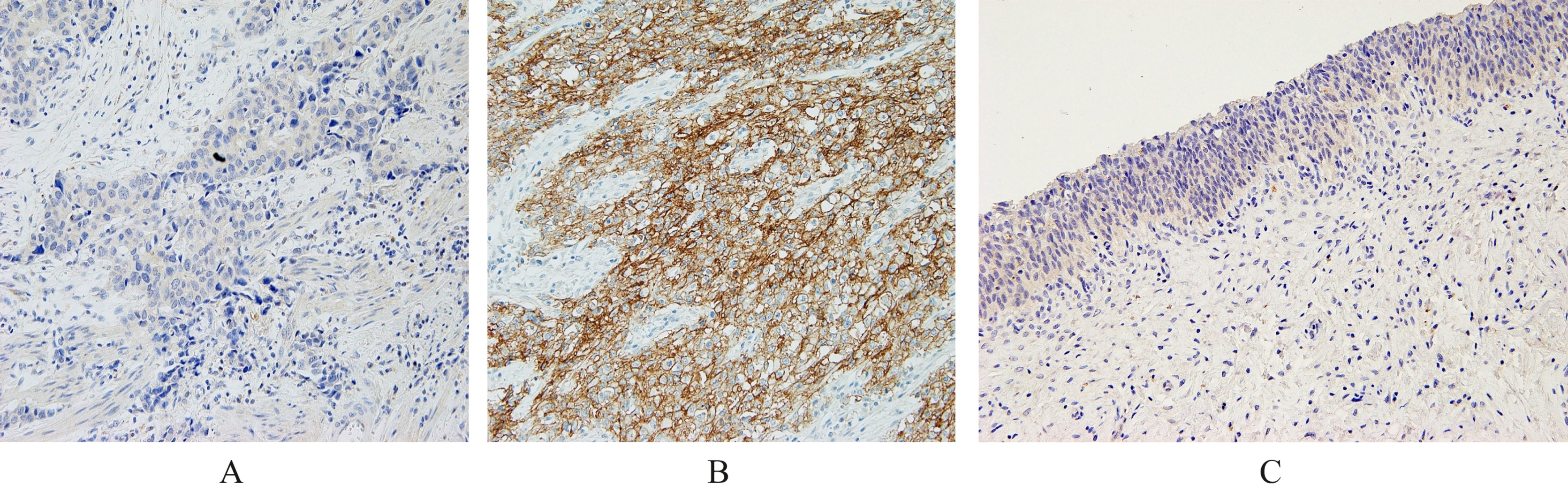
Three-
We also performed nestin (polyclonal antibody, IBL, Takasaki, Japan) immunohistochemical staining as previously described [16]. Nesting expression scores using the sum index score were stratified into negative (score 0) and positive (score 1 or greater).
For this analysis, age (
Association of TROY expression with clinical and pathological characteristics
Association of TROY expression with clinical and pathological characteristics
*Fisher’s exact test (2-sided). **Six patients had unknown pathologic status of the lymph nodes. ***Sixteen patients had no lymphovascular invasion status. ****Ninety-three patients were investigated in previous study.
Multivariate Cox proportional hazard regression analyses for prediction of recurrence and survival of bladder cancer
HR: hazard ratio; CI: confidence interval; LVI: lymphovascular invasion.
Probability of survival in patients with urothelial carcinoma in the bladder according to TROY expression estimated by the Kaplan-Meier method. (A) progression-free survival and (B) cancer-specific survival.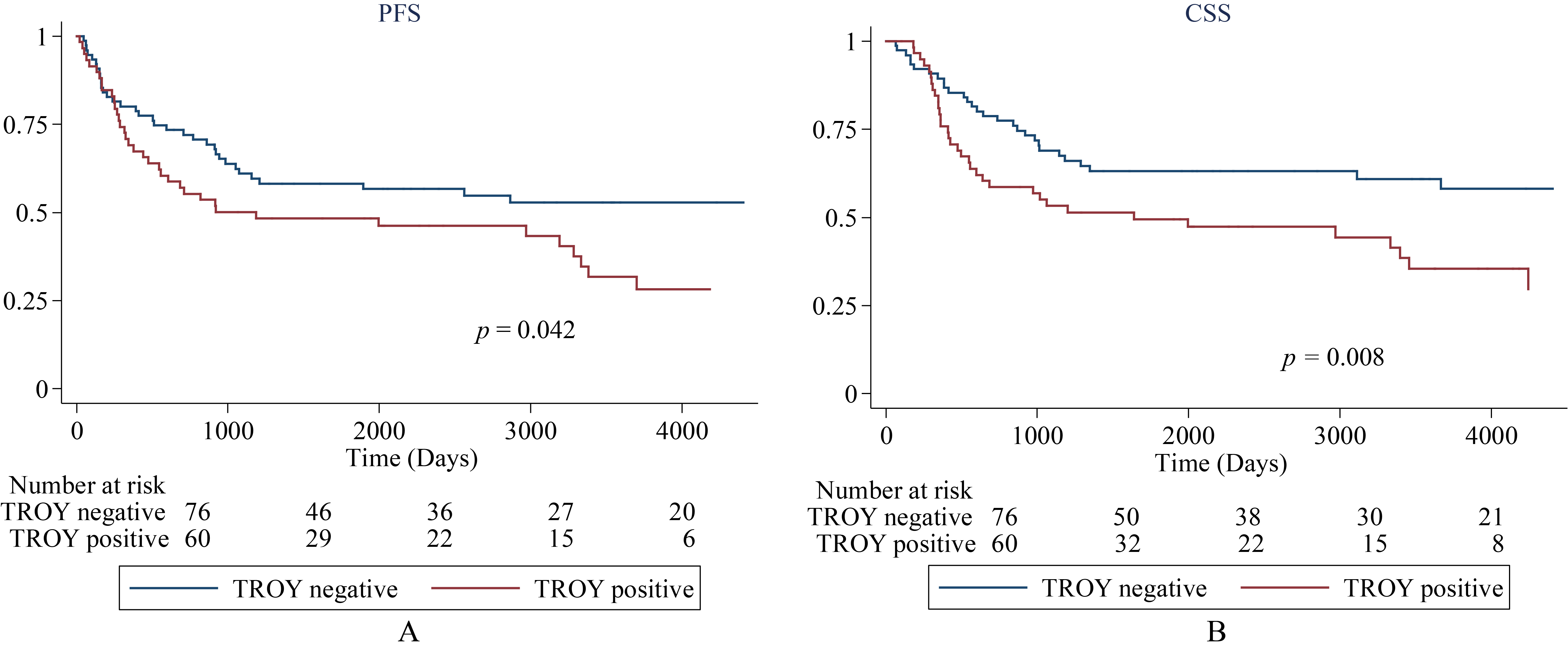
Association of TROY expression with clinicopathological characteristics
Representative cases of immunostaining for TROY in normal urothelium and UCB tissues are shown in Fig. 1. Positive UCB tissues demonstrated membranous immunoreactivity (Fig. 1B). Normal urothelium showed negative immunoreactivity for TROY (Fig. 1C).
The clinicopathological characteristics of the 136 patients and the association with TROY expression are shown in Table 1. Positive and negative expressions of TROY were noted in 60 (44.1%) and 76 (55.9%) patients, respectively. Positive expression of TROY was associated with muscle invasive disease and nestin expression (
Association of TROY expressions with prognosis
Disease progression developed in 71 patients (52.2%) at a median time of 13.7 months (range, 0.7–123.3; mean 26.3). Of all patients, 74 (54.4%) had died at the time of analysis at 21.4 months (range, 2.2–224.5; mean 39.7). The cause of death in 65 patients (87.8%) was identified as advanced or metastatic lesions, while 9 patients died of intercurrent causes without evidence of disease progression.
A Kaplan-Meier analysis indicated that positive expression of TROY was associated with increased probability of disease progression and cancer-specific death (
In the Cox proportional hazards regression analyses, only lymph node metastasis was found to be associated with disease progression and cancer-specific survival (Table 2). Expression of TROY was marginal in terms of cancer-specific survival (
Discussion
While current clinical and pathological variables are limited in their ability to predict tumor progression and survival, investigation of protein alterations in UCB is important to detect reliable biomarkers for predicting patient prognosis and to establish clinical follow-up protocols. Although nodal involvement was the sole independent prognostic factor for disease progression and cancer-specific survival in the present study, lymph node metastasis was already an established factor in many previous reports. We found that TROY expression in tumor cells was associated with muscle-invasive lesion, nestin expression, disease progression, and cancer-specific survival in patients with UCB.
TROY is a type I transmembrane glycoprotein with an extracellular ligand-binding domain, a single membrane-spanning region, and a cytoplasmic region [17]. Because TROY is capable of inducing apoptosis by a caspase-independent mechanism, it is thought to play an essential role in embryonic development [18]. During embryogenesis, TROY mRNA is exclusively expressed in the epithelium of many developing tissues, but after birth the expression is restricted. For example, low or lesser levels were seen in the heart, thymus, lungs, liver, uterus, skeletal muscle, colon, testes, kidney, and peripheral blood lymphocytes, but highly expression of TROY was detected in the brain, prostate, and hair follicles [9]. In the present study, normal urothelium was found to show negative immunoreactivity for TROY.
On the other hand, TROY expression was reported to be associated with several cancers in terms of cancer biology and clinical outcomes [9, 10, 11]. TROY mRNA was highly expressed in melanoma tissue and regulated melanoma growth [9]. In glioblastoma specimens, TROY was overexpressed. TROY mRNA expression was up-regulated in the invasive cells and knockdown of TROY expression in primary glioblastoma showed prolonged survival in vivo [10]. In gastric cancer, TROY was observed in the desmoplastic stroma and tumor cells using immunohistochemistry [11]. TROY was significantly common in intestinal compared to diffuse gastric cancer and correlated inversely with histological grade and lymph node metastasis. Loss expression of TROY was correlated with poor overall survival in intestinal type gastric cancer. In terms of urothelial carcinoma in the present study, TROY expression was associated with advanced stage and poor prognosis. Once TROY reacted in tumor cells, it had biologically aggressive potential.
Nestin is a class VI intermediate filament protein that was defined as a neuronal stem cell marker during central nervous system (CNS) development [19]. Expression of nestin has been shown in various tumor cells, including CNS tumor, gastrointestinal stromal tumor, pancreatic cancer, prostate cancer, breast cancer, malignant melanoma, dermatofibrosarcoma protuberance, and thyroid tumor [20]. In addition, nestin was reported to correlate with poor prognosis in patients with bladder cancers [16, 21]. T1 high grade bladder cancer patients with positive nestin expression showed significant recurrence compared to those with negative nestin expression [21]. Our previous study clarified that nestin expression in tumor cells was associated with poor progression-free, cancer-specific, and overall survival in UCB patients treated with radical cystectomy [16]. Although the roles of nestin in cancer cells have not been well demonstrated, nestin was expressed in proliferating cells during the developmental periods in a variety of embryonic and fetal tissues, and was reported as one of putative markers in cancer stem cells (CSCs) [16, 22].
CSCs were identified in malignant blood tumors and in solid cancerous lesions including UCB [23, 24, 25]. CSCs have the ability to self renew and differentiate into hierarchical tumor cells, thereby contributing to tumor heterogeneity [26]. We found the association between TROY and nestin expression in UCB specimens. Taken together, TROY would associate with the CSCs and contribute to tumorigenesis, disease progression, and other complex behaviors. CSCs also play an important role in resistance to chemotherapy and radiotherapy [27]. We speculate that determining the level of TROY expression in UCB tissues may help to choose the appropriate treatment modality and avoid ineffective therapy in patients who require optimized treatments like molecular targeting therapy and, possibly, immune-checkpoint inhibitors. Further study is warranted.
The current study has the limitations inherent to a retrospective study design. The relatively small sample size may have limited us to detecting small differences. Despite these limitations, this study reveals that positive TROY expression is associated with poor prognosis in patients with UCB after radical cystectomy. Future studies could validate this novel biological marker using a prospectively large cohort containing information on multimodal treatments.
Conclusions
We found that TROY is associated with muscle-invasive lesion, nestin expression, and poor prognosis. Although TROY was a marginal prognostic factor for cancer-specific survival, its prognostic significance still requires confirmation with larger patient populations.
Footnotes
Acknowledgments
This study was supported in part by JSPS KAKENHI Grant Number JP15K10607 and JP18K09206.
