Abstract
BACKGROUND:
Cisplatin-based chemotherapy and radiotherapy are the most commonly used treatments for small cell lung cancer (SCLC). However, despise initially dramatic response, the response duration of SCLC patients is variable and resistance to chemo- and radio-therapy inevitably develops.
OBJECTIVE:
The aim of the study is to investigate the role of Bcl-2 family proteins in predicting SCLC sensitivity to cisplatin treatment, and to identify the potential sensitizer of cisplatin or ratiation treatment in SCLC.
METHODS:
We collected cisplatin sensitivity data from public available database, and evaluated its possible association with mRNA or protein expression of Bcl-2 family members in SCLC cell lines.
RESULTS:
The IC
CONCLUSIONS:
Our study indicates that the ratio of Bcl-2/Bim could be a SCLC response predictor to cisplatin, and ABT-263 addition could be an effective strategy to improve the activity of chemo- or radio-therapy in SCLC.
Introduction
Small cell lung cancer (SCLC) is a clinically very aggressive lung cancer and less than 7% of SCLC patients survive more than 5 years [1]. Although SCLC patients have initially high response to chemo- and radio-therapy, the response duration is variable and relapse inevitably develops due to therapeutic resistance. SCLC patients who relapse
Pharmacogenomic strategy provides an approach to develop predictors of drug sensitivity by using integrative analysis of gene expression profiling dataset and drug sensitivity dataset [3]. Currently, the drug sensitivity and gene expression profiling of SCLC cell lines are available in the Genomics of Drug Sensitivity in Cancer (GDSC) and Cancer Cell Line Encyclopedia (CCLE) databases [4, 5]. The Bcl-2 family proteins are critical regulators of apoptosis induced by chemo- or radio-therapy. The Bcl-2 family anti-apoptotic proteins (Bcl-2, Bcl-xL and Mcl-1) are frequently over-expressed in cancer cells, and confer cellular chemo- and radio-resistance [6]. In SCLC, up-regulation of Bcl-2 increased resistance to cisplatin, a widely used chemotherapeutic drug for the treatment of SCLC [7]. Moreover, the Bcl-2 family pro-apoptotic BH3-only proteins (Bim, Bid, Puma and Noxa) are also critical for initiating apoptosis by activating Bax and Bak, but the induction of apoptosis can be impeded by the interactions between the Bcl-2 family anti-apoptotic proteins and pro-apoptotic proteins. Thus, the ratio between Bcl-2 family anti- and pro-apoptotic proteins is an important determinant of cell survival and cell death [8]. In the study, we firstly examined whether SCLC response to cisplatin was associated with the ratio of Bcl-2 family anti-apoptotic and pro-apoptotic proteins by mining public available databases.
Interestingly, we found that SCLC cell lines with higher ratio of Bcl-2/Bim were significantly correlated with the resistance to cisplatin. The data promoted us to test the hypothesis that a BH3-mimetic ABT-263, which disrupts the interaction of Bcl-2 and Bim, is able to enhance the antitumor activity of cisplatin or radiation in SCLC cells.
Materials and methods
Materials
ABT-263 and cisplatin were purchased from Selleck chemical (Shanghai, China). ABT-199 was purchased from MCE (Shanghai, China). ABT-263 was dissolved in DMSO (Sigma-Aldrich, Saint Louis, MO, USA) to prepare a 10 mM stock solution. Cisplatin was dissolved in PBS (Cellgro, Manassas, VA, USA) to prepare a 5 mM stock solution. Antibodies against PARP, Bcl-2, Mcl-1, Bim were from Cell Signaling Technology, Danvers, MA, USA.
Cell lines and cell culture
The human SCLC cell lines H82, H526, DMS79, H69, H1963, H446 and H196 were maintained in RPMI1640 media (Cellgro, Manassas, VA, USA) supplemented with 10% FBS (ExCell Bio, Shanghai, China) and 1% penicillin/streptomycin (Gibco, Life Technologies, Grand Island, NY, USA) in a humidified incubator at 37
X-ray irradiation
SCLC cells were exposed to radiation at a dose of 2 or 4 Gy using X-RAD 320 Biological Irradiator (Precision X-Ray, North Branford, CT, USA). For the combination treatment of radiation and ABT-263, SCLC cells were exposed to radiation after treated with ABT-263 for 2 hours.
Cell proliferation assay
SCLC cells were plated in 96-well culture plates. After 24 hours, cells were subjected to the treatment of DMSO control, cisplatin, radiation, ABT-263, cisplatin/ABT-263 combination and radiation/ABT-263 combination for 72 hours. After treatment, cell viability was measured by using Cell Titer-Glo Luminescent assay (Promega, Madison, WI, USA) according to the manufacturer’s instructions. Luminescence was measured in a multi-label plate reader (Envision PerkinElmer, USA). Data were normalized to DMSO control and represented by the mean
Colony formation assay
For suspension cells (H526 and H69), cells were plated in 60 mm dishes and incubated for 24 hours. Cells were then treated with DMSO control, cisplatin, radiation, ABT-263, cisplatin/ABT-263 combination and radiation/ABT-263 combination for 24 hours. After treatment, 1.25
Annexin V apoptosis analysis
The treated cells were stained with FITC Annexin V and PI using a FITC Annexin V Apoptosis Detection Kit (BD Pharmingen, San Diago, CA, USA). The stained samples were subjected to flow cytometry analysis by FACS Calibur (BD Pharmingen, San Diago, CA, USA), and then analyzed by FlowJo software (BD Pharmingen, Ashland, OR, USA).
Western blot
After treatment, western blot was performed using specific antibodies following the procedure as described previously [9]. For quantification of western blot data, band intensities were quantified using ImageJ software.
Immunoprecipitation (IP)
After treatment, cells were lysed with immunoprecipitation lysis buffer (250 mM NaCl, 50 mM Tris-HCl pH 7.4, 0.5% Triton X-100, 10% glycerol) and subject to IP analysis as describe previously [10].
Analysis of combination index
The additivity or synergy of drug combination was determined quantitatively using the combination index (CI) method of Chou and Talalay [11]. CI was calculated using the formula: CI
Bim siRNA treatment
H69 cells (2
Statistical analysis
All data were analyzed using PRISM5 Software (GraphPad Software, Inc., La Jolla, CA, USA). Statistical analysis was performed using an unpaired t-test.
Bcl-2/Bim expression ratio predicts SCLC response to cisplatin. (A) Scatter plot of cisplatin IC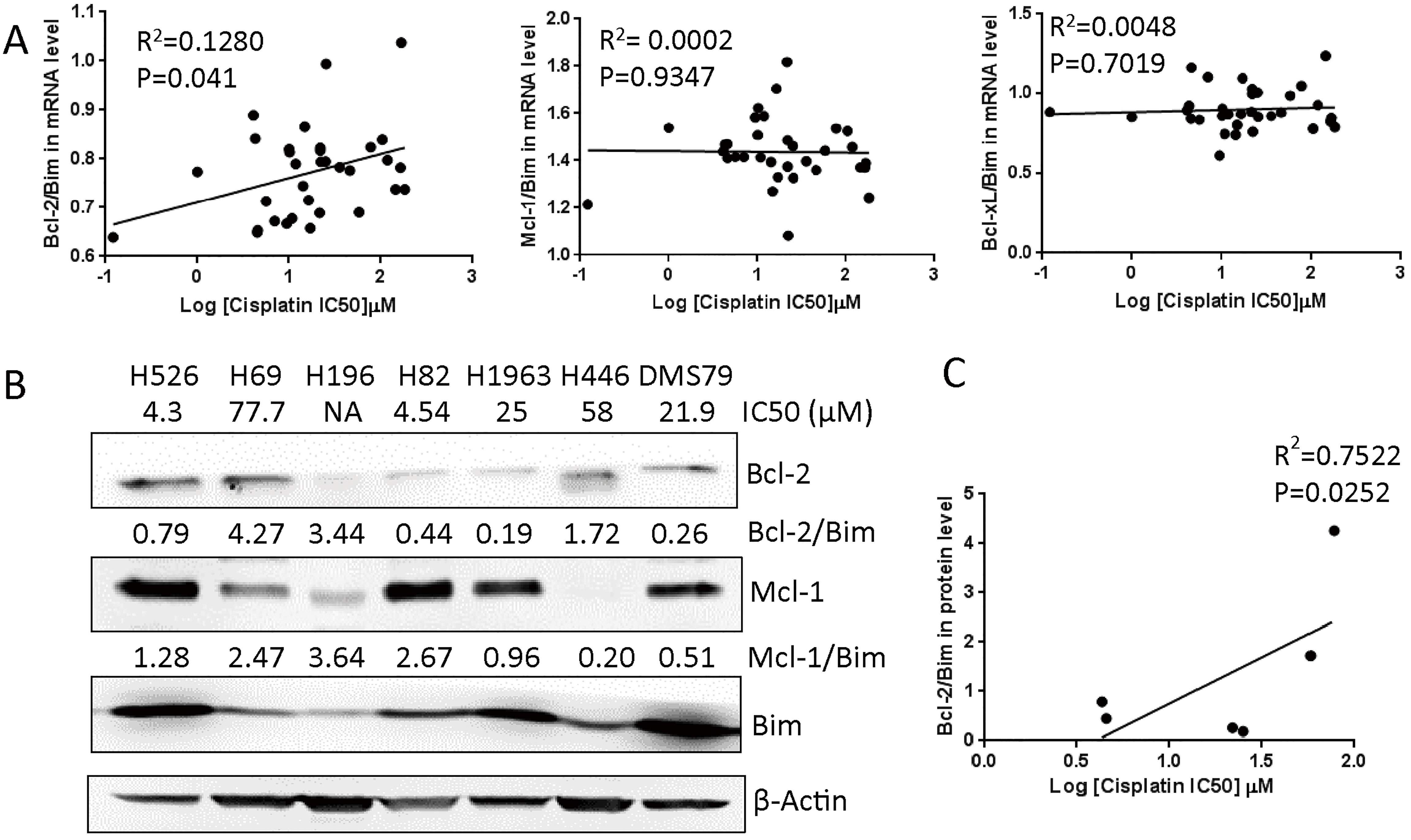
ABT-263 sensitizes SCLC cells to cisplatin or radiation. (A) Cell proliferation assay indicates ABT-263 addition enhances cisplatin or radiation-induced anti-proliferation activity in SCLC. SCLC cells (H526, H69 and H446) were treated with DMSO control, ABT-263, cisplatin, radiation or the combination for 72 hours. Growth inhibition was determined by CellTiter-Glo Luminescent assay after treatment (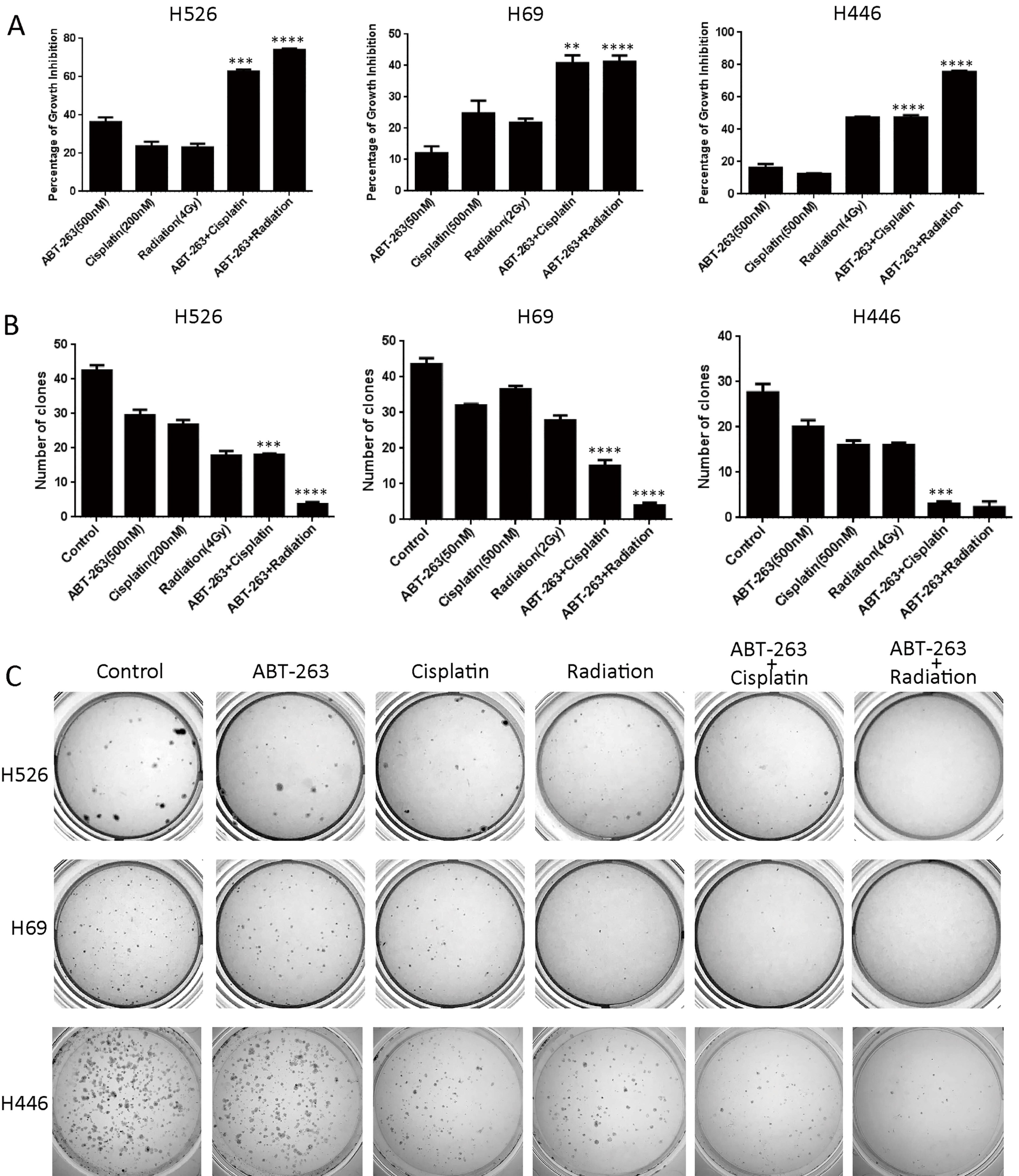
SCLC sensitization to cisplatin or radiation by ABT-263 was associated with the increased apoptosis. (A) Annexin V apoptotic assay indicates that the addition of ABT-263 enhances cisplatin- or radiation-induced apoptosis. SCLC H446 cells were treated with control, ABT-263 (500 nM), cisplatin (500 nM), radiation (4Gy) or their combination for 48 hours. After treatment, cells were stained with FITC Annexin V and PI, and then flow cytometry was performed. (B) The addition of ABT-263 enhances cisplatin- or radiation-induced PARP cleavage. SCLC cells (H526, H82 and H446) were treated with control, ABT-263 (500 nM), cisplatin (500 nM), radiation (4Gy) or their combination for 24 hours. After treatment, PARP and loading control 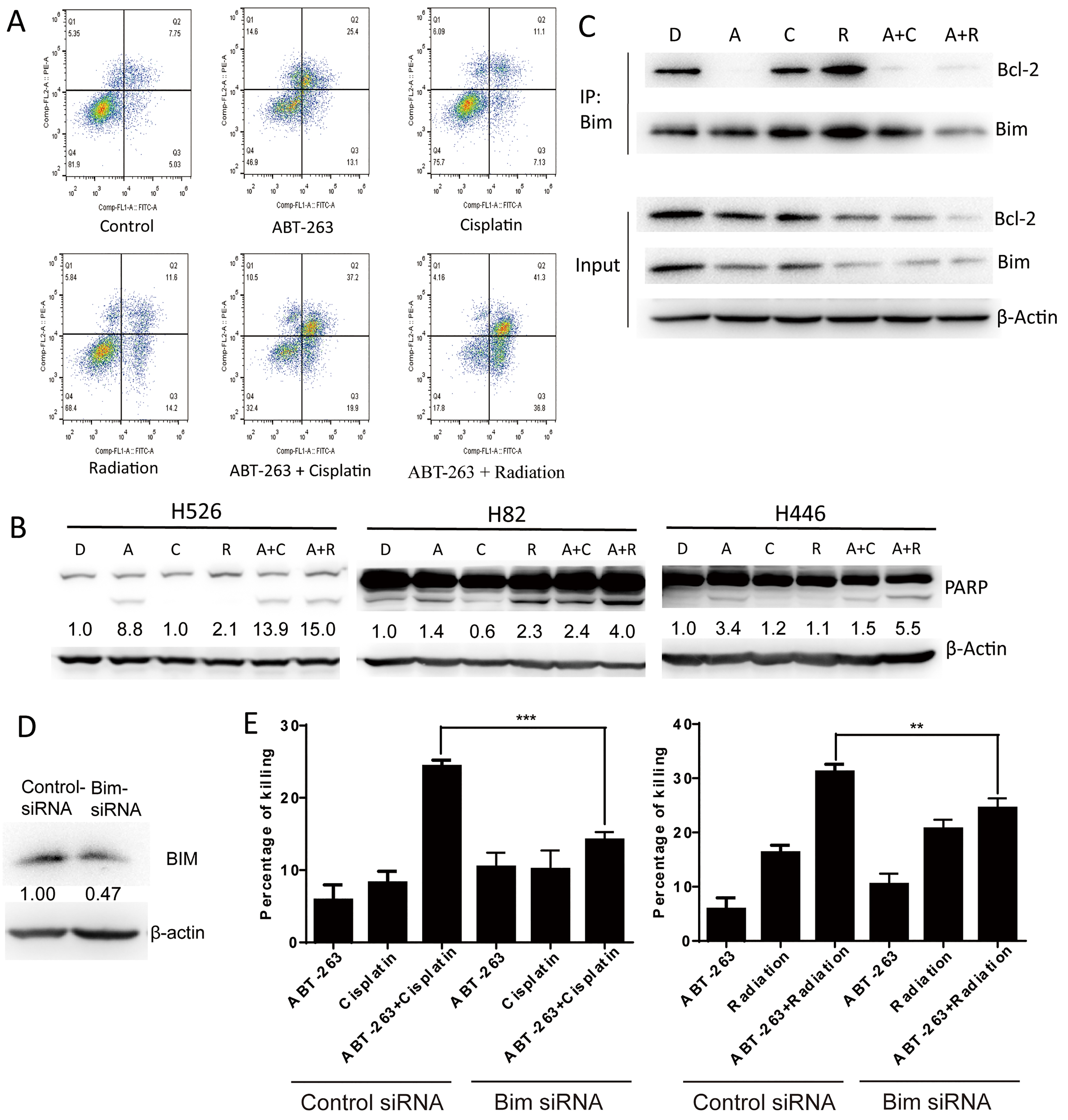
Ratio of Bcl-2/Bim predicts the response of SCLC cells to cisplatin
To identify the predictors of response to cisplatin in SCLC cells, we collected IC
We further examined whether the ratio of Bcl-2/Bim in protein level was correlated with SCLC response to cisplatin. By performing western blot, we detected the protein expression levels of Bcl-2, Mcl-1 and Bim in SCLC cell lines, and quantified band densities by densitometry (Fig. 1B). As shown in Fig. 1C, the ratios of Bcl-2/Bim protein expression were significantly correlated with the IC
ABT-263 sensitizes SCLC cells to cisplatin or radiation
The significant correlation between Bcl-2/Bim ratio and cisplatin sensitivity suggests that Bcl-2 and Bim may be mechanistically associated with the anti-tumor response of cisplatin in SCLC. Thus, we hypothesized that ABT-263, which disrupts the Bcl-2/Bim complex to release Bim for apoptotic induction, was able to potentiate the cytotoxic effect of cisplatin or radiation in SCLC cells. As shown in Fig. 2A, cell viability assay demonstrated that anti-SCLC proliferation activity of cisplatin or radiation was significantly enhanced by ABT-263 addition in H526, H69 and H446 SCLC cells. Furthermore, we performed the analysis of combination index (CI) to determine the synergistic or additive effect of cisplatin and ABT-263 combination. By calculating the combination index (CI), we found that ABT-263 and cisplatin co-treatment exerted synergistic anti-proliferative effect (CI
We further tested the chemo- and radio-sensitization of ABT-263 in SCLC cells by colony formation assay. As shown in Fig. 2B and C, co-treatment of ABT-263 and cisplatin dramatically suppressed colony formation ability of H526, H69 and H446 SCLC cells. Similarly, radiation combined with ABT-263 also strongly reduced colony formation of H526, H69 and H446 SCLC cells. In summary, the data indicate that ABT-263 sensitizes SCLC cells to cisplatin or radiation.
SCLC sensitization to cisplatin or radiation by ABT-263 was associated with the increased apoptosis
To determine the mechanism of ABT-263-mediated sensitization to cisplatin or radiation, we performed cell apoptotic assay of SCLC cells treated with ABT-263, cisplation, radiation or in combination. Annexin V apoptotic assay indicated that the apoptotic induction of SCLC cells by cisplatin or radiation was remarkablely enhanced by ABT-263 addition (Fig. 3A). Western blot also demonstrated that ABT-263 addition increased cisplatin or radiation-induced PARP cleavage (apoptotic marker) in SCLC cells (Fig. 3B).
We next examined the effect of ABT-263 on the interaction of Bcl-2 and Bim in SCLC cells. Immunoprecipitation assay indicated that the binding of Bcl-2 and Bim was markedly disrupted by ABT-263 treatment alone as well as its combination with cisplatin or radiation (Fig. 3C). To demonstrate whether Bim contributes to apoptosis induced by the combination treatments, we knocked down Bim expression by Bim siRNA (Fig. 3D). Upon Bim knockdown, the inhibition of cell growth by ABT-263 combined with cisplatin or radiation was significantly reduced in H69 cells (vs. control siRNA transfection) (Fig. 3E). The data suggest that ABT-263 disrupts the interaction of Bcl-2 and Bim, and the release of Bim contributes to the increased apoptosis by the combination of ABT-263 with cisplatin or radiation.
To further demonstrate that the disruption of Bcl-2/Bim interaction by BH3 mimetic sensitizes SCLC cells to cisplatin or radiation, we tested ABT-199, a highly potent and selective BCL-2 inhibitor [12]. As shown in Supplementary Fig. S3, cell viability assay demonstrated that the inhibition of cell viability by cisplatin or radiation was significantly enhanced by ABT-199 addition in H526 and H69 cells. Annexin V apoptotic assay indicated that the apoptotic induction of SCLC cells by cisplatin or radiation was also enhanced by ABT-199 addition (Supplementary Fig. S4).
Discussion
In the current study, we have used in vitro drug sensitivity data, coupled with gene expression data from public available databases, to develop gene expression predictor reflecting SCLC sensitivity to cisplatin. We identified the ratio of Bcl-2/Bim as a predictor of SCLC sensitivity to cisplatin. It could be potentially applied to SCLC patients to predict who may benefit from cisplatin-based therapy. Despite initially high sensitivity to chemo- and radio-therapy, relapse inevitably develops within months due to the therapeutic resistance. In addition,
ABT-263, a BH3 death domain mimetic, directly binds Bcl-2 to block its interaction with Bim, thereby enabling Bim-mediated induction of apoptosis [14]. The close correlation between Bcl-2/Bim ratio and SCLC response to cisplatin promoted us to examine whether ABT-263 was able to sensitize SCLC cells to cisplatin or radiation. Both cisplatin and radiation induce apoptosis by targeting DNA damage. The interaction between Bcl-2 family anti- and pro-apoptotic proteins determines the cellular sensitivity to DNA damage-induced apoptosis [15]. Indeed, our study found that ABT-263 enhanced cisplatin- or radiation-induced anti-tumor activity through the increased apoptosis in SCLC cells. Our study is consistent with previous studies. Loriot et al. reported that a novel Bcl-2 and Bcl-xL inhibitor S44563, which disrupted the interaction of Bcl-2/Puma interaction, was able to sensitize SCLC cells to radiation [16]. BH3 mimetics (ABT-263 and ABT-737) have been found to be synergized with chemotherapy in various cancer models, including non-small cell lung cancer [17, 18], head and neck cancer [19] and osteosarcoma [20]. Lochmann et al. reported that ABT-199, a BH3 mimetic which only targets Bcl-2 inhibitor, was effective to kill SCLC cells with high Bcl-2 expression [21]. Like ABT-263, our study indicated that ABT-199 enhanced cisplatin or radiation-induced cell death in SCLC cells. In summary, to our knowledge, it is the first study to report a BH3 mimetic sensitizes SCLC cells to chemo-therapy.
In summary, the present study identified a new potential predictor, the ratio of Bcl-2/Bim expression, which might predict SCLC sensitivity to cisplatin. The combination of ABT-263 with cisplatin or radiation in SCLC can lead to marked cellular apoptosis and therefore may improve the efficacy of chemo- and radio-therapy for the treatment of SCLC.
Supplementary data
The supplementary files are available to download from
sj-docx-1-cbm-10.3233_CBM-181692.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-181692.docx
Footnotes
Acknowledgments
This study was supported by National Natural Science Foundation of China (grant numbers: 81872438, 81672647, 81502632 and 81372214), Natural Science Foundation of Anhui Province (grant number: 1608085MH179), Key Technologies R and D Program of Anhui Province (grant number: 1604a0802103), The Major/Innovative Program of Development Foundation of Hefei Center for Physical Science and Technology (grant number: 2016FXCX006) and the 100-Talent Program of Chinese Academy of Sciences.
Conflict of interest
Authors have no financial/commercial conflicts of interest regarding the study.
