Abstract
BACKGROUND:
One of the known risk factors for non-small cell lung cancer (NSCLC) is somatic mutation in the Kirsten rat sarcoma (KRAS) gene. The relationship with smoking is well known.
METHODS:
We retrospectively studied the data of 92 patients who underwent pulmonary resection January 2003 and June 2012 and were diagnosed as KRAS-mutated pathological stage I adenocarcinoma.
RESULTS:
Among them, 33 patients who were non to light smoker (NLS) (smoking index, 0 to 400) were compared with 59 middle to heavy smoker (MHS) (
CONCLUSIONS:
A non to light smoking habit might contribute to an improvement in prognosis that is equivalent to that of wild-type KRAS, and p53 mutation did not affect survival in smokers harboring KRAS codon 12.
Introduction
In most countries, mutations in the epidermal growth factor receptor (EGFR) and Kirsten rat sarcoma (KRAS) and the rearrangement of anaplastic lymphoma kinase (ALK) gene adenocarcinoma is the most common histologic type of non-small cell lung cancer (NSCLC). The presence of KRAS gene is lower among Asians than among Caucasian NSCLC patients [1]. A study reported that the cause of KRAS mutation has not been clear, except for its relationship with smoking habit [2]. However, Riely et al. reported that KRAS mutation was frequently detected in a significant proportion of never smoker NSCLC patients, with an incidence of up to 15% [3]. Dogan et al. reported that permanent DNA damage due to tobacco carcinogens acquired at the time of smoking was the major source of most KRAS-mutated lung adenocarcinoma; therefore, the likelihood of a KRAS mutation in a lung cancer patient has been determined by pack-years and did not significantly decrease over time after smoking cessation [4]. p53 plays a key role in the regulation and initiation of DNA repair and has been reported as unfavorable prognostic marker in NSCLC [5, 6].
The role of KRAS mutation as a prognostic factor in NSCLC remains controversial; however, the majority of accumulating evidence suggests that its presence is a significantly worse prognostic factor in NSCLC [7]. In resected early stage NSCLC, Graziano et al. demonstrated that the presence of KRAS mutation was not of prognostic significance [8]. In contrast, Slebos et al. reported that the presence of KRAS point mutations defined a subgroup of lung adenocarcinoma patients in whom the prognosis was very poor and disease-free survival (DFS) was not usually long despite radical resection and small tumor load [9]. Unfortunately, the clinical role of the variations of KRAS remains unsolved.
In the present study, we examined the following four questions: whether there was difference in perioperative clinicopathological factors in stage I NSCLC patients [2]; whether the degree of smoking habit correlated with prognosis in patients with KRAS mutation [3]; whether the distribution of amino acid substitution in codons 12 and 13 correlated with smoking habit; and [4] whether p53 in KRAS codon 12 mutation correlated with a higher risk of unfavorable survival.
Patients and methods
We reviewed all patients with pathologically confirmed stage I NSCLC who underwent surgical resection at Aichi Cancer Center between January 2003 and June 2012. The clinicopathological stage was assessed according to the seventh edition TNM classification for lung cancer [10]. Patient records were examined for age, gender, preoperative carcinoembryonic antigen (CEA) levels, and preoperative nodal status and tumor size on both mediastinal and lung window chest computed tomography (CT) images. Pathological examination was based on the 2004 World Health Organization classification [11]. The predominant pattern was defined as the morphological subtype present in the greatest proportion of the resected specimen. Tumors were classified according to the original IASLC/ATS/ERS classification [12]. KRAS and p53 mutations were detected by direct sequencing, as previously demonstrated [13]. The smoking index was calculated by multiplying the number of cigarettes smoked per day by the number of years.
Because individual patients were not identified, the institutional review board of Aichi Cancer Center approved this study without the requirement to obtain patient informed consent. The patient records and information were made anonymous prior to analysis.
The proportion of KRAS mutation status and histological types in the pathological stage I patients with pulmonary resection. Red: Adenocarcinoma with KRAS mutation (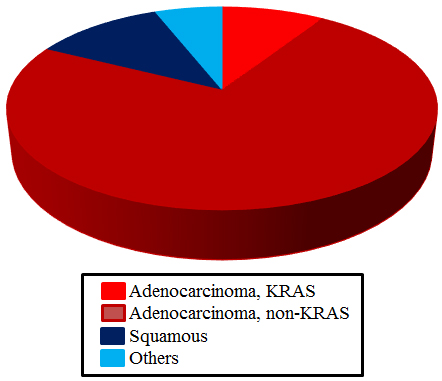
Demographic data and smoking habit of the study population
AD, adenocarcinoma; C, codon; CEA, Carcinoembryonic antigen; SD, standard deviation.
Kaplan-Meier plots of overall survival based on smoking habit in KRAS-mutated NSCLC and the distribution of tumor recurrence after pulmonary resection. (A) Overall survival. Black line: NLS and Green: MHS. (B) Cancer-specific overall survival. Black line: NLS and Green: MHS. (C) Disease-free survival. Black line: NLS and Green: MHS. (D) Overall survival compared with wild-type KRAS. Red line: NLS; Green: MHS; and Red: wild-type KRAS.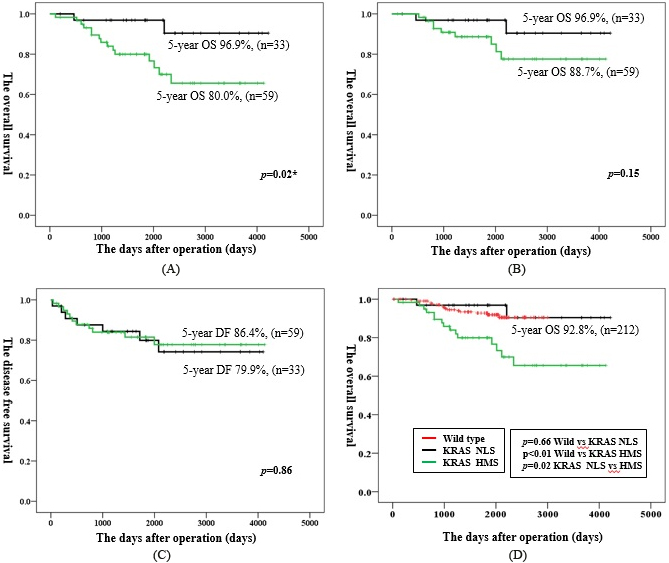
All data were analyzed using SPSS version 17.0 (SPSS Institute Inc., Chicago, IL, USA). Differences between two groups were calculated using the Mann-Whitney U test and Fisher’s Exact Tests. The analysis of survival rates was performed using the Kaplan-Meier method; survival rates between patient groups were compared using the log-rank test. A
Univariate and Multivariate analyses for overall survival
Univariate and Multivariate analyses for overall survival
OS: Overall survival; MHS: Middle to Heavy smoke, CEA: Carcinoembryonic antigen, HR: Hazard ratio; CI: Confidence intervals. *:
Association between clinicopathological characteristics and outcome
The schematic including histological and KRAS mutation status is presented in Fig. 1. The 87.6% (92/105) patient data is available from the medical records, and the clinicopathological characteristics are summarized in Table 1. The mean age was 66 years (range, 41–83 years) in the non to light smoker (NLS) group (
The 5-year overall survival (OS) rate was significantly better in the NLS group than in the MHS group (96.9% vs. 80.0%,
In NLS patients, the 5-year OS rate was the same between patients with KRAS mutation and those with wild-type KRAS (96.9% vs. 92.8%,
Univariate and multivariate analyses showed that Age
Postoperative distribution of histologic subtypes
When tumors were stratified by the percentage of histological subtypes, the most common subtypes in the NLS group were mucinous (65.6%), lepidic (56.3%), solid (25.0%), papillary (25.0%), and acinar (21.9%), whereas the most common subtypes in the MHS group were papillary (49.0%), solid (49.0%), lepidic (34.0%), mucinous (22.4%), and acinar (14.3%). Solid and papillary components were significantly more frequent in MHS patients than in NLS patients (
Distribution of oncogenic substitutions in KRAS-mutated NSCLC patients
Distribution of oncogenic substitutions in KRAS-mutated NSCLC patients
KRAS, Kirsten rat sarcoma; NSCLC, non-small cell lung cancer.
Evaluation of the proportion of postoperative histological subtypes in KRAS-mutated NSCLC. Comparison of the proportion of postoperative histological subtypes (NLS vs. MHS).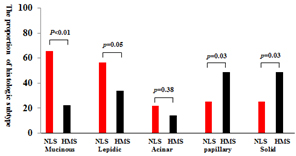
Distribution of p53 mutation in KRAS-mutated NSCLC according to smoking habit and the Kaplan-Meier curves for overall survival based on p53 mutation in KRAS codon 12-mutated NSCLC. (A) Red: p53 mutation negative in NLS; Dark red: p53 positive in NLS; Blue: p53 positive in MHS; and Light blue: p53 negative in NLS. (B) The overall survival between p53 negative in NLS (Blue line), p53 negative in MHS (Red), and p53 positive in MHS (Black). KRAS, Kirsten rat sarcoma; NSCLC, non-small cell lung cancer; NLS, non to light smoker; MHS, middle to heavy smoker.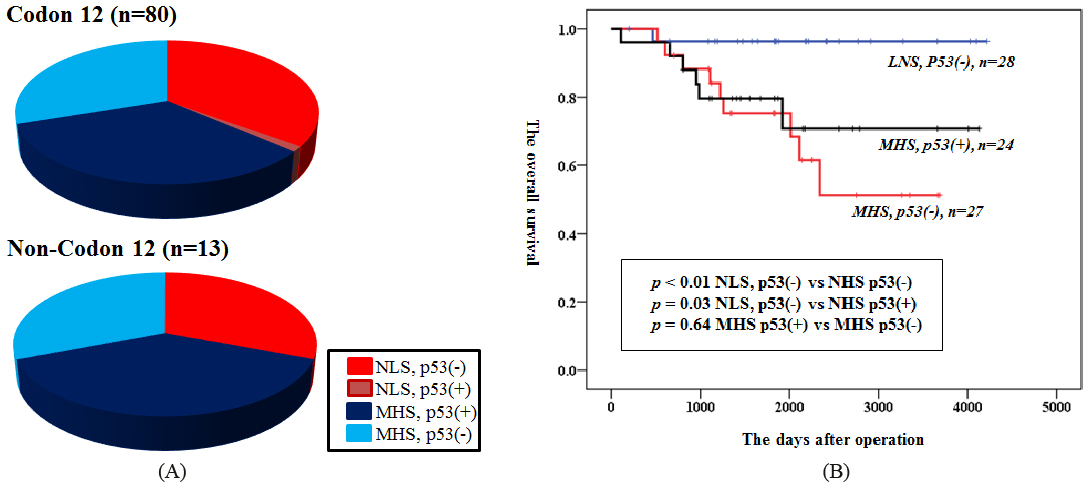
In KRAS-mutated NSCLC, the most common oncogenic substitutions were identified in G12C (33.3%), G12V (23.5%), and G12D (23.5%) in NLS patients and in G12D (31.0%), G12V (24.1%), and G12C (24.1%) in NLS patients (Table 3). However, the frequency of G12C, G12D, and G12D were not significantly different between NLS and MHS.
We additionally explored the distribution of p53 and prognosis by smoking habit in patients with KRAS codon 12 mutation of which occupied the great majority in oncogenic substitutions. Figure 4A illustrates the frequency of KRAS (codon 12 or non-codon 12) and p53 mutations in relation to smoking history. The p53 was found in only 3.4% (1/29) and 0% (0/4) of NLS patients with KRAS codon 12- and codon 13- mutated NSCLC, respectively (Fig. 3A); however, this proportion was notably lower than that in a relatively smaller study. In addition, the proportion of p53-positive cases in KRAS codon 12-mutated NSCLC was significantly higher in MHS than in NLS patients (
Finally, we exploited the prognostic influence of p53 status in patients with KRAS codon 12 mutation. The 5-year OS rate for p53-negative NLS patients was significantly better (
Discussion
In the present study, we addressed four issues in patients with stage I KRAS-mutated lung adenocarcinoma: (1) smoking habit affected the clinicopathological characteristics, including preoperative imaging, CEA expression, and histological subtype distribution; (2) NLS patients had more favorable outcomes than MHS patients but similar to patients with wild-type KRAS; (3) KRAS amino acid substitutions were different between G12 and G13 mutations; and (4) p53 mutation correlated with smoking habit in patients with KRAS mutation but did not affect the prognosis of patients with KRAS codon 12 mutation.
A novel association between KRAS mutation and solid growth pattern was reported in a recent study [14]; in our study, this association was significantly higher in MHS patients than in NLS patients. Mutation status has been well-known and clearly established to be related to ethnicity. Zhang et al. reported that KRAS mutation in mucinous adenocarcinoma was detected more frequently in never smoker Asian females [15], results that were compatible with our data. In addition, Finberg et al. reported that in bronchoalveolar carcinoma (BAC)/adenocarcinoma with bronchoalveolar features (AWBF), mucinous differentiation was significantly correlated with the absence of EGFR mutation and presence of KRAS mutation, suggesting that mucinous BACs/AWBFs are unlikely to respond to tyrosine kinase inhibitors (TKIs) [16]. In our study, the frequency of lepidic pattern was significantly higher in NLS patients than in MHS patients, albeit borderline.
The most prominent result of our study was that survival after pulmonary resection was significantly poorer in MHS patients than in NLS patients. Interestingly, prognosis in the latter was equivalent to that in wild-type KRAS. The activity of EGFR-TKIs has been reported to differ according to the type of EGFR mutation (e.g., 19 deletion vs. Ex.21 L858R), which is one of the most frequently targeted key driver mutation in NSCLC [23]. Similarly, the potency and efficacy of the targeted drug for KRAS mutation may be affected by smoking habit. The role of KRAS mutation status as a potential prognostic factor for both early stage and advanced NSCLC remains controversial. Several authors have demonstrated that KRAS-mutated NSCLC was associated with poor prognosis [24, 25], even if the diagnosis was stage I [26]. Furthermore, Tsao et al. reported a poor response to chemotherapy in patients with KRAS mutation [27]. In contrast, in their pooled analysis of multiple adjuvant chemotherapy trials, Shepherd et al. demonstrated that the prognostic factor was not KRAS mutation status but rather KRAS codon 13 mutation, which was possibly predictive of a decreased response to chemotherapy [28]. In addition, in a study of NSCLC patients who received adjuvant chemotherapy in the JBR10 study, KRAS mutation was not a prognostic factor and did not contribute to poorer response to cisplatin-based chemotherapy [27]. Several studies reported that a never smoker status contributed to a better prognosis than a current or ex-smoker status [29]; this result may be due to the different distribution of oncogenic drivers, with KRAS being more common in smokers and EGFR mutation and ALK rearrangement being more common in never smokers [30]. It is important to note that increased survival was most likely due to better overall condition and lack of smoking related co-morbidities in never smokers [31]. In the present study that was limited to cancer-specific mortality, there was no significant difference in OS rates between NLS and MHS patients; however, that was found in OS including comorbidities related deaths. Sonobe et al. reported that the KRAS and EGFR gene mutation status of a tumor was not associated with DFS after the complete resection of lung adenocarcinoma, but patients with EGFR mutations had better prognosis than those with KRAS mutations [32]. Our cohort showed that DFS after pulmonary resection was the same between NLS and MHS patients, but locoregional recurrence was more common in NLS patients; therefore, many NLS patients were eligible to undergo second resection or to receive local control therapies.
Smoking habit has been reported to commonly correlate with G12C mutation [4]. In contrast, a study from Hungary identified an association between G12D mutation and never smokers [33]. In our study, there was no significant correlation identified between mutation and smoking habit, but there was a similar tendency. The presence of p53 mutation, which is the most frequent molecular abnormality in NSCLC, was associated with a higher incidence of invasive lung adenocarcinoma [34]. The proportion of p53 positivity in KRAS codon 12 mutation was similar to that in codon 13 in MHS patients. There were few p53 mutations in NLS patients.
Furthermore, p53 overexpression was reported to be a significant prognostic marker of shortened survival and predictor of greater response from adjuvant chemotherapy in completely resected NSCLC [27]. A p53 encodes a tumor-suppressor protein that contains transcriptional activation, DNA binding, and oligomerization domains, which all respond to diverse cellular stress to regulate the expression of target genes. In the present study, patients with p53-negative mutations in KRAS codon 12 mutation did not have significant survival benefit (
There were several limitations to the present study. First, it was a retrospective design with a small sample size at a single institution; therefore, the strength of our study to determine the prognostic factors was limited. Second, the study population was a heterogeneous group in terms of chemotherapeutic regimen or local treatment after recurrence. However, given the number of stage I NSCLC cases with KRAS mutation, this cohort represented a relatively large study group in whom p53 and amino acid substitutions in KRAS mutation were examined entirely.
In conclusion, prognosis was significantly poorer in KRASmutation than in wild-type pathological stage I NSCLC. However, non to light smoking habit might contribute to the improvement in prognosis, with an outcome equivalent to that of wild-type KRAS. We considered that differences in the pattern of recurrence, genetic subtype, and clinical parameters might be one of the factors for favorable prognosis.
Footnotes
Conflict of interest
The authors have no financial or commercial conflict of interest.
