Abstract
BACKGROUND:
Recurrence following conventional therapies in patients with breast cancer is a major cause of morbidity and mortality. This study aimed to investigate potential predictive biomarkers for breast cancer recurrence especially microRNAs (miRNAs).
METHODS:
The primary breast cancer patients who were scheduled to undergo curative resection in our hospital from May 2007 to May 2012 were recruited in this study. Clinical and pathological characteristics were compared in patients with or without recurrence. The expressions of tissue miRNAs by quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR) were also analyzed in patients with recurrence or not. Multivariate Cox analysis was fit to evaluate potential predicative factors for breast cancer recurrence. Kaplan-Meier plots was conducted to further evaluate the association between miR-17-5p expression and recurrence-free survival (RFS).
RESULTS:
A total of 133 consecutive patients were enrolled into the final analysis and 29 of them have suffered recurrence within 5 years after the operation. Our results revealed tissue miR-17-5p expression as an independent risk factor for breast cancer recurrence (HR: 4.45; 95% CI: 1.58–11.53,
CONCLUSIONS:
This study suggested that miR-17-5p might be a useful predictive factor for breast cancer recurrence.
Introduction
Breast cancer is the most frequently diagnosed cancer and the leading cause of cancer death in women worldwide [1, 2]. In recent years, the mortality rate of breast cancer has declined due to the development of screening tools and effective adjuvant systemic therapies [3]. However, cancer recurrence following conventional therapies in patients with breast cancer is a major cause of morbidity and mortality [4]. Making more accurate predictions about clinical outcomes and recurrence is the great challenge for breast cancer management [5]. Until now, the molecular mechanisms underlying breast cancer recurrence have remained unclear likely due to clinical and molecular heterogeneity. An investigation of potential predicative biomarkers for breast cancer recurrence is critical.
MicroRNAs (miRNAs), small non-coding RNAmolecules that are 19 to 25 nucleotides in length, are well-known regulators of gene expression and many other fundamental cellular processes including cell differentiation, proliferation and apoptosis [6]. Recent studies have indicated the important role of miRNAs in tumor initiation, proliferation, progression and metastasis [7]. Previous data have also indicated that miRNAs are potential predicative biomarkers for prognosis and therapy response in cancer [8]. A recent study conducted with miRNA PCR array containing primers for 384 human mature miRNAs has revealed that miR-340-5p, miR-17-5p, miR-130a-3p, and miR-93-5p were significantly associated with breast cancer recurrence by logistic regression analysis [9]. In addition, another study conducted in young breast cancer patients identified that three candidate miRNAs (miR-183-5p, miR-194-5p, and miR-1285-5p) could possibly be used separately or in combination as prognostic biomarkers [10]. However, the predicative value of these candidate miRNAs for breast cancer recurrence remains unclear.
In this study, we performed a comprehensive analysis of the seven candidate miRNAs mentioned above and the clinicopathologic characteristics across breast cancer patients with or without recurrence.
Material and methods
Patients
This retrospective study protocol was approved by the Medical Institutional Ethics Committee of Zhejiang province. The primary breast cancer patients who were scheduled to undergo curative resection by the same operation team in Ningbo No. 2 Hospital from May 2007 to May 2012 were recruited in this study. The exclusion criteria was described as follows: (1) receiving neoadjuvant treatment before the operation; (2) having a prior history of cancer; (3) missing follow-up data. Until May 2017, a total of 133 consecutive patients were enrolled for inclusion in the final analysis. Twenty-nine of these 133 patients have suffered recurrence within 5 years after the operation, which is an incidence of 21.8%.
Clinical and pathological characteristics
Adjuvant treatment was assigned to each patient according to their clinical and pathological characteristics after the operation. Radiation therapy was routinely administered to those patients who underwent breast-conserving surgery. The pathological data including the positive status of estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2), and axillary lymph node metastasis (pN) were evaluated by the same experienced pathologist. The ER, PR and HER2 positive statuses was determined by immunohistochemistry (IHC). The Ki-67 expression was assessed by measuring the percentage of Ki-67-positive tumor cells using the IHC method. The mean value of Ki-67 expression was accepted as the cut-off value for high versus low Ki-67 status.
The clinical and pathological stages were determined according to the Union for International Cancer Control (UICC) stage classification system. The postsurgical therapy including endocrine therapy, radiation therapy or chemotherapy varied for each patient according to their pathological characteristics.
Follow-up
Enrolled patients were regularly followed on an outpatient or inpatient basis. The primary end-point of this study was 5-year recurrence-free survival (RFS). Recurrence (including locoregional or distant metastasis) was evaluated by tumor biomarkers and imaging examination.
Tissue samples and RNA extraction
All enrolled participants should offer signed informed consent. The formalin-fixed, paraffin-embedded (FFPE) tumor samples tissues were obtained formiRNA detection and postoperative histopathologic diagnosis. The FFPE cancer tissue was separated by manual microdissection and then total RNA extraction was conducted with the Qiagen miRNeasy kit (Qiagen, MD, USA). A NanoDrop spectrophotometer (Thermo Scientific, MA, USA) was used for the determination of RNA concentration and quality.
MiRNA measurement by quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR)
The expressions of these candidate miRNAs were identified by the method of qRT-PCR. The SYBR based qRT-PCR was conducted in 96-well plates with the Real-time PCR system (Applied Biosystems, Foster City, CA, USA). MicroRNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) was used for the miRNA reverse transcription according to the manufacturer’s guidelines. Fluorescent dye detection method was performed with SYBR green Master Mix as described by previous publication [11]. The primer sequences were as follows: miR-16-5p (forward: 5’-ACACTCCAGCTGGGTAGCAGCACGTAAATA-3’, reverse: 5’-TGGTGTCGTGGAGTCG-3’),miR-340-5p (forward: 5’-ACACTCCAGCTGGGTT ATAAAGCAATGAGA-3’, reverse: 5’-TGGTGTCG TGGAGTCG-3’), miR-17-5p (forward: 5’-ACACTC CAGCTGGGCAAAGTGCTTACAGTGC-3’, reverse: 5’-T GGTGTCGTGGAGTCG-3’), miR-130a-3p (forward: 5’-ACACTCCAGCTGGGCAGTGCAATGTTA AAA-3’, reverse: 5’-TGGTGTCGTGGAGTCG-3’),miR-93-5p (forward: 5’-ACACTCCAGCTGGGCAA AGTGCTGTTCGTGC-3’, reverse: 5’-TGGTGTCGT GGAGTCG-3’), miR-183-5p (forward: 5’-ACACTC CAGCTGGGTATGGCACTGGTAGAA-3’, reverse,5’-TGGTGTCGTGGAGTCG-3’), miR-194-5p (forward: 5’-ACACTCCAGCTGGGTGTAACAGCAACTCCA-3’, reverse: 5’-TGGTGTCGTGGAGTCG-3’),miR-1285-5p (forward: 5’-ACACTCCAGCTGGGGA TCTCACTTTGTTG-3’, reverse: 5’-TGGTGTCGTG GAGTCG-3’). In brief, the thermal cycling and real-time fluorescence measurements were performed using a 7300 Fast Real-time PCR System (Applied Biosystems) with SYBR Green PCR Master Mix (Applied Biosystems). The PCR cycle protocol was as follows: 95
Statistical analysis
SPSS 21.0 (SPSS Inc., Chicago, IL, USA) and GraphPad 5.0 (GraphPad Inc., San Diego, CA, USA) were utilized for statistical analysis in this current study. Chi-square or Fisher exact test was used to compare categorical variables as appropriate. Student t test or Mann-Whitney U test was used to compare continuous data. Multivariate Cox proportional hazard analysis was fit to evaluate potential predicative factors for breast cancer recurrence. For survival analysis by Kaplan-Meier plots, the median
Characteristics of breast cancer patients with recurrence or not
Characteristics of breast cancer patients with recurrence or not
ASA, American Society of Anesthesiologists; BMI, Body Mass Index; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2
Patient characteristics
To investigate the potential clinicopathological and molecular factors specific to breast cancer recurrence, enrolled 133 patients were divided into two groups, 29 in the recurrent group and 104 in the non-recurrent group. Patients’ characteristics were detailed summarized in Table 1. No significant difference was observed in clinicopathological factors including tumor size, clinical T stage, nodal, ER, PR, HER2 and Ki-67 status between patients with recurrence or not (
Relative expressions of miRNAs in tumor tissues of patients with recurrence or not using miR-16-5p as the internal control. The middle line indicates the median and the box indicates the 10–90 percentile range. 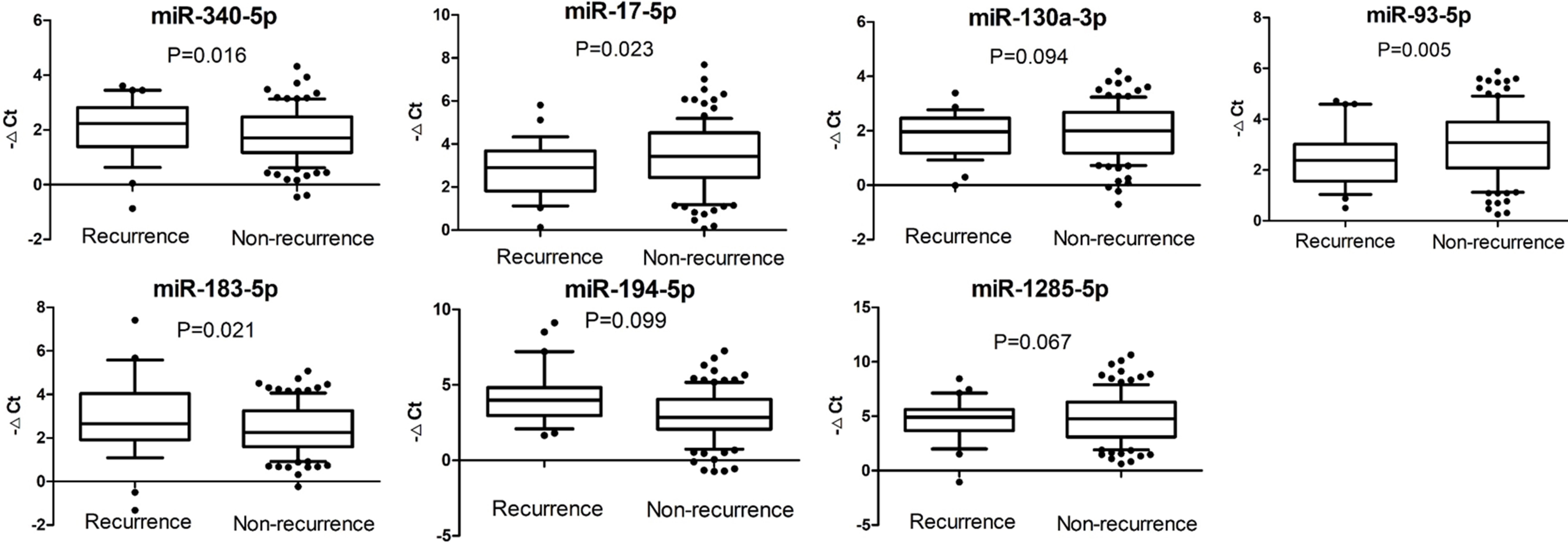
Risk factors for breast cancer recurrence by multiple Cox proportional hazards analysis
ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; CI, Confidence Interval; HR, Hazard Ratio. *
Kaplan-Meier survival curve analysis for recurrence-free survival between patients with high and low miR-17-5p expressions. Patients with a higher miR-17-5p expression (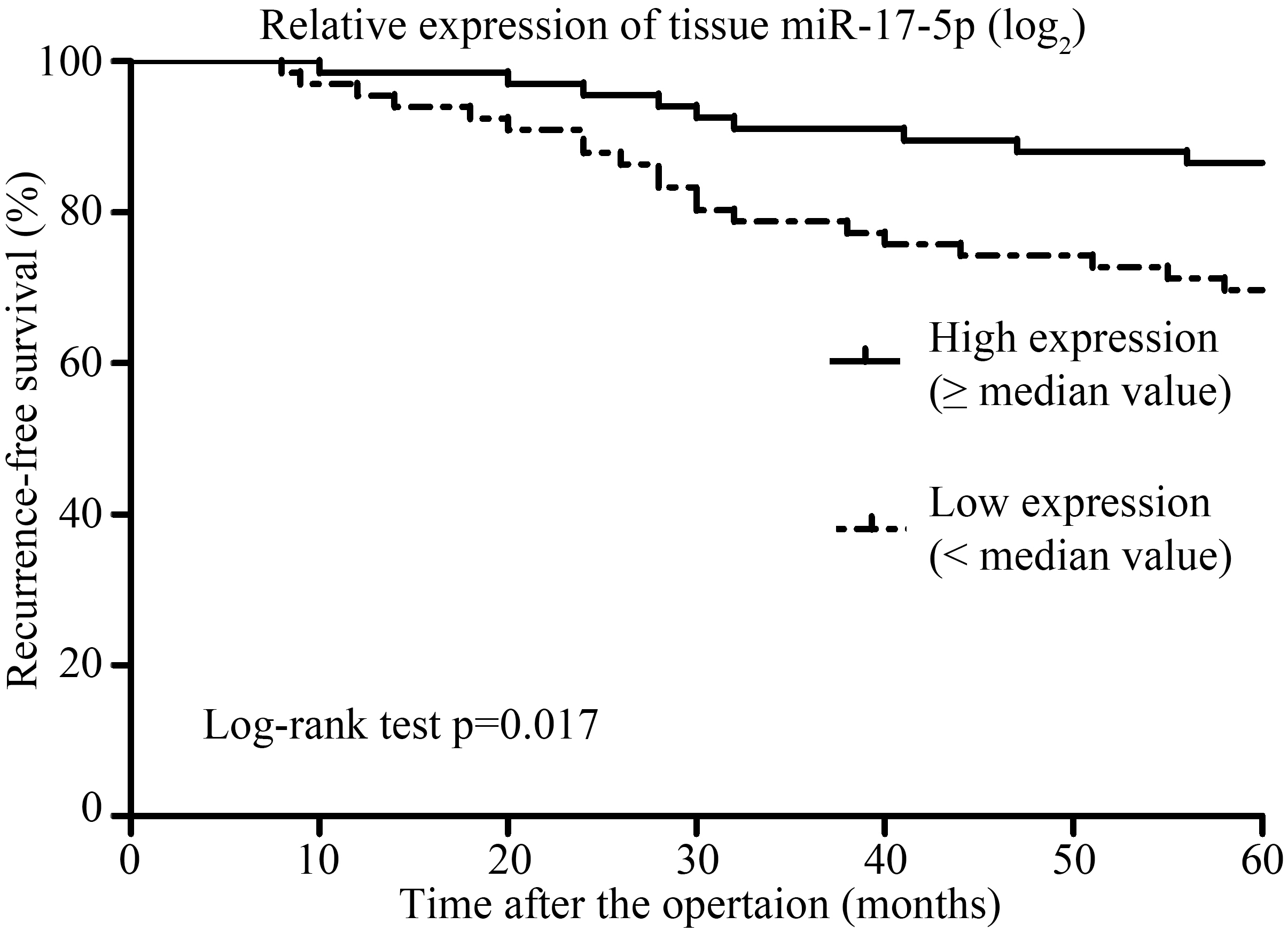
To investigate the potential associations between miRNA concentrations in cancer tissues and breast cancer recurrence, we selected seven candidate miRNAs from previous reports including miR-340-5p, miR-17-5p, miR-130a-3p, miR-93-5p, miR-183-5p, miR-194-5p and miR-1285-5p. The relative miRNA expressions normalized to MiR-16-5p by qRT-PCR were shown in Fig. 1 (raw data in supplementary file). Of these 7 miRNAs, miR-340-5p and miR-183-5p were expressed at significantly higher levels in recurrent group in comparison with non-recurrent group. In contrast, those patients with recurrence demonstrated significantly lower miR-17-5p and miR-93-5p expressions, which was consistent with previous reports [9].
Risk factors for breast cancer recurrence by multiple Cox proportional hazard analysis
As summarized in Table 2, we conducted an exploratory analysis to investigate potential risk factors for breast cancer recurrence. The risk factors including age, clinicopathological factors and some miRNAs expressions were involved into the final multivariate Cox proportional hazards model. Our results revealed tissue miR-17-5p expression as an independent risk factor for breast cancer recurrence (HR: 4.45; 95% CI: 1.58–11.53,
Association between tissue miR-17-5p expression and 5-year RFS
To further evaluate the association between miR-17-5p expression and RFS, Kaplan-Meier plots was conducted. As illustrated in Fig. 2, patients with a higher miR-17-5p expression (
Discussion
To investigate potential prognostic biomarkers for the recurrence of breast cancer, we performed qRT-PCR analysis of some candidate miRNAs, which identified 2 over-expressed miRNAs (miR-340-5p and miR-183-5p) and 2 under-expressed miRNAs (miR-17-5p and miR-93-5p) in recurrent participants. These four miRNAs combined with other clinicopathological factors were validated by multivariate Cox proportional hazard analysis, Kaplan-Meier analysis revealed that miR-17-5p was closely associated with 5-year RFS. According to our results, we propose that miR-17-5p could serve as a potential prognostic biomarker for recurrence in breast cancer patients.
Aberrantly expressed miRNAs are widely observed in cancers, indicating their oncogenic or suppressive activities [12]. MiR-17-5p, also known as miR-17 and the main effector of the miR-17-92 cluster components, has been accepted as a solid cancer miRNA biomarker [13]. Recent data have indicated that miR-17-5p is involved in tumor proliferation through the modulation of the PI3K/Akt/mTOR pathway [14]. Overexpressed miR-17-5p has also been observed in triple negative breast cancer, and this miRNA could have a role in inhibiting ribosomal translation of tumor suppressor genes [15]. Previous studies conducted in high-invasive MDA-MB-231 breast cancer cells have indicated that miR-17-5p is involved in breast cancer cell invasion and migration by targeting the HBP1/Wnt/b-catenin pathway [16]. MiR-17-5p may serve as a tumor suppressor by suppressing the proliferative activity of E2F1 [17], which has both oncogenic andsuppressor roles [18]. In addition, miR-17-5p has also been recognized as a key oncogenic factor by targeting BIM, p21 and PTEN [19]. The results mentioned above might be possible explanations for the predictive role of miR-17-5p in breast cancer recurrence in the present study.
Another study conducted in patients with melanoma showed a close association between up-regulatedplasma miR-17-5p expression and advanced clinical stage [20]. Furthermore, there is evidence that altered miR-17-5p expression plays an important role in the development and progression of hepatocellular carcinoma (HCC) [21]. Significantly up-regulated miR-17-5p expression is observed in HCC tissues compared to paracarcinomatous liver tissues, and elevated serum miR-17-5p concentration is closely associated with poor prognosis [22]. Recent data have indicated the pivotal role of miR-17-5p in the proliferation and migration regulation of hepatocellular carcinoma cells by targeting the p38/MAPK/HSP27 pathway [23] and PTEN [24]. The abnormal expression of miRNAs in cancer tissues has attracted a great deal of attention. A recent study has suggested that circulating exosomal miR-17-5p may be a promising noninvasive prognostic biomarker for primary and metastatic colorectal cancer [25]. Another study conducted in non-small cell lung cancer has also reported that miR-17-5p is a key regulator for cell proliferation and invasion [26]. Furthermore, miR-17-5p is also reported to promote pancreatic cancer proliferation by altering cell cycle profiles through the modulation of RBL2/E2F4-repressing complexes [27].
Our results have offered new insight into the potential prognostic role of miRNA concentrations in breast cancer recurrence. However, whether miR-17-5p could serve as a predictive biomarker for breast cancer recurrence and the mechanisms involved still remain unclear and require further research. This current study also had several limitations. First, this was a retrospective study protocol conducted in a single center. Second, the sample size of our patient cohort was relatively small. Finally, no comparison of these miRNAs between cancer and normal tissues was available.
Footnotes
Conflict of interest
None.
