Abstract
BACKGROUND:
Targeting protein for Xenopus kinesin-like protein 2 (TPX2) is a microtubule-associated proteinrequired for mitosis and spindle assembly. It has been revealed that TPX2 is overexpressedin various human cancers and promotes cancer progression.
METHODS:
The expression of TPX2 was examined in ovarian cancer (OC) tissues and by Western blotting, quantitative real-time reverse transcription PCR (qRT-PCR) and immunohistochemistry. The effects of TPX2 on proliferation and migration of two OC cell lines SKOV3and RMG1 were analyzed using the methylthiazol tetrazolium (MTT) assay, flow cytometry and transwell assay. The mechanisms underlying the effects of TPX2 on OC cells were explored by qRT-PCR and Western blot.
RESULTS:
In this study, we found that TPX2 was upregulated in OC tissues. We observed knockdown of TPX2 inhibited the expression of Polo-like kinase 1 (PLK1), which has an important role in the regulation of M phase of the cell cycle, and the activity of Cdc2, induced cell arrested at the G2/M phase and decreased proliferation. Moreover, our data revealed that the levels of PLK1,
CONCLUSIONS:
TPX2 promotes the proliferation and migration of human OC cells by regulating PLK1 expression.
Introduction
Ovarian cancer (OC) remains the leading cause of death among gynecological malignancies, owing to late detection, clinical chemoresistance and extraordinary heterogeneity [1]. The standard treatment options of OC consist of primary tumor surgery as well as platinum- and paclitaxel-based chemotherapy. However, most patients develop chemoresistance and eventually die from the disease [2, 3]. Despite optimization of surgical and chemotherapy protocols and the introduction of new therapies into standard treatment, such as angiogenesis inhibitors and PARP inhibitors, only modest gains have been achieved in prolonging survival in OC [4]. Therefore, the development of novel, effective therapies for OC is urgently needed.
As the hallmark of cancer revolves around cell-cycle deregulation, strategies targeting mitotic regulators are considered effective against the abnormal proliferation of transformed cells [5]. Several mitotic regulators have been demonstrated to play pivotal roles in tumor progression [6]. For example, altered expression of the mitotic checkpoint protein MAD2 lead to increased susceptibility to tumor development [7, 8]. Motor protein Eg5 is responsible for centrosome separation and bipolar spindle formation [9]. Overexpression of Eg5 causes disruption of normal spindle development and promotes the development of tumors [10]. The aurora kinases are also known as important mitotic regulators. Aberrant expression and activity of these kinases usually result in aneuploidy and tumorigenesis [11].
Formation of a microtubule-based structure known as the spindle is critical important for accurate chromosome segregation during cell division [12]. TPX2 is identified as a microtubule-associated protein, and its expression is tightly regulated by the cell cycle [13]. As a critical regulator of mitosis, TPX2 cooperates with Aurora-A kinase and Eg5 kinesin to control microtubule assembly, bipolar spindles formation and chromosome segregation [14]. It has been reported that TPX2 is highly expressed in multiple kinds of human tumors and promotes tumorigenesis, such as gastric cancer [15], bladder cancer [16], prostate cancer [17], breast cancer [18] and so on. A systematic study revealed TPX2 as a prognostic biomarker in serous epithelial OC [19]. However, the particular effect of TPX2 on OC cells and the underlying mechanisms remain largely unclear.
In the current study, we demonstrated the upregulation of TPX2 in OC tissues. Functional analyses disclosed that TPX2 promoted OC cell proliferation and migration in vitro. Mechanistic analyses revealed that knockdown of TPX2 suppress the expression of PLK1, which is a member of the polo-like kinase family and is known to function at mitosis, thus leading to the inhibition of OC cell proliferation and migration. Furthermore, the overexpression of PLK1 reversed TPX2 knockdown-induced anti-proliferation and anti-migration effects in OC SKOV3 cells. Taken together, our findings suggested that TPX2 could be targeted to develop potential therapeutic strategy for OC.
Materials and methods
Cell culture, infection and transfection
The human ovarian cell lines SKOV3 and RMG1 were purchased from ShanghaiCell Bank of the Chinese Academy of Sciences (Shanghai, China). The cells were maintained at 37
Patients and tissue samples
A total of 32 paired tumor and adjacent non-tumor tissues with a median age of 48.2 years (20–65 years) were recruited from the First Affiliated Hospital of Xi’an Medical University and Shanxi Provincial People’s Hospital, between January 2012 and October 2016. The specimens included 19 samples of serous papillarysubtypes, 6 samples of mucinous subtypes, 4 samples of endometroid subtypes, and 3 samples of clear cell subtypes. Informed consent was obtained from all individual participants included in the study. This study was approved by the ethics committee of the hospital. The study conforms with The Code of Ethics of the World Medical Association (Declaration of Helsinki), printed in the British Medical Journal (18 July 1964). Histological diagnosis of all OC tissue samples was performed according to the WHO classification. All patients were histopathologically determined by 2 pathologists. Patients received chemotherapy, radiotherapy or immunotherapy have been removed.
RNA extraction and quantitative reverse transcription PCR
Total RNA was extracted by Trizol (Invitrogen Life Technologies, Carlsbad, CA, USA). Briefly, 1
Western blot analysis
The total protein was extracted from cells after transfection using RIPA buffer. The extract was centrifugated for 15 min at 4
Immunohistochemical staining (IHC)
To detect the expression of TPX2, IHC staining was performed using a standard streptavidin-biotin-peroxidase complex method. In brief, paraffin sections were deparaffinized and hydrated. Endogenousperoxidase activity was blocked with 3% hydrogen peroxide (H
Plasmid construction and transfection
The human TPX2 and PLK1 expression plasmids were constructed by amplifying the entire open reading frame (ORF) of the TPX2 and PLK1 genes with the polymerase chain reaction (PCR) and inserted into the pcDNA3.1/V5-His expression vector (Invitrogen, CA, USA). Then all the constructswere confirmed by DNA sequencing and transfected into cells using Lipofectamine 2000 reagent (Invitrogen, CA, USA). The sequence of siRNA targeting TPX2 was as follows: 5’-AUGAAAGUUUCUAACAACAAATT-3’. Cells were plated in 6-well plates andtransfected for 48 h. Transfected cells were used for further assays.
Cell viability assay
Cell proliferation status was determined at 24 h, 48 hand 72 h respectively after transfection. Cells at 2
Cell migration assay
A total of 1
Flow cytometry assay
Cell-cycle analysis was performed via the FACS approach. Briefly, 1
Statistical analysis
All the quantitative data were presented as means
The expression of TPX2 in human OC tissues. (A) Westernblot analysis was performed to assess TPX2 protein levels in five pairs of human primary OC tissuesand adjacent non-tumor tissues. (B) Quantitative RT-PCR analysis of TPX2 mRNA expression in 32 pairs of human primary OC tissues and adjacent non-tumor tissues. NT, non-tumor; T, tumor. 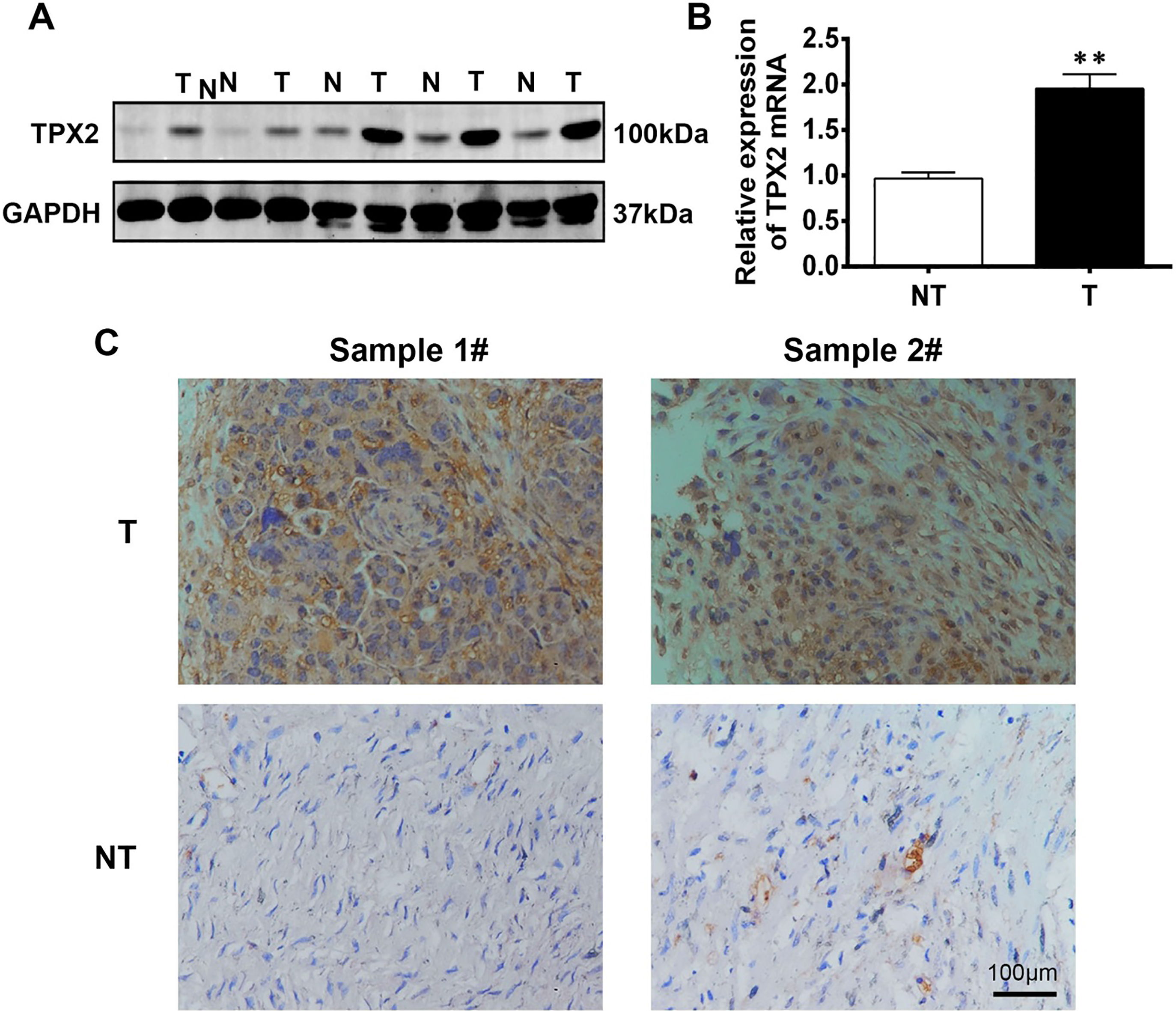
Effects of TPX2 over-expression on proliferation and migration of SKOV3 cells. (A) Over-expression of TPX2 significantly promotes cell proliferation in SKOV3 cells measured by MTT assays. 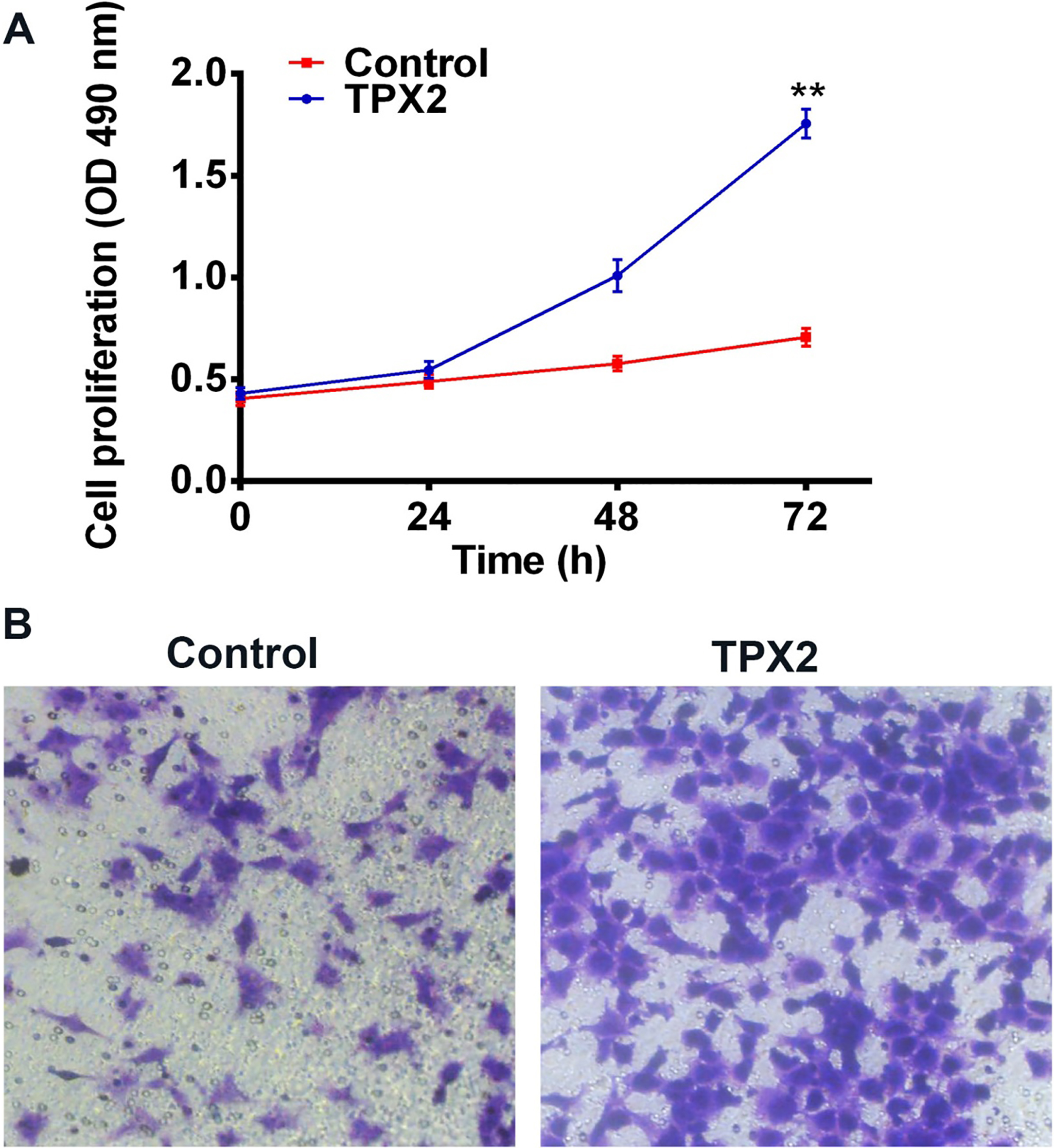
Effects ofTPX2 silencing on proliferation and migration of RMG1 cells. (A) Knockdown of TPX2 significantly inhibits cell proliferation in RMG1 cells measured by MTT assays. 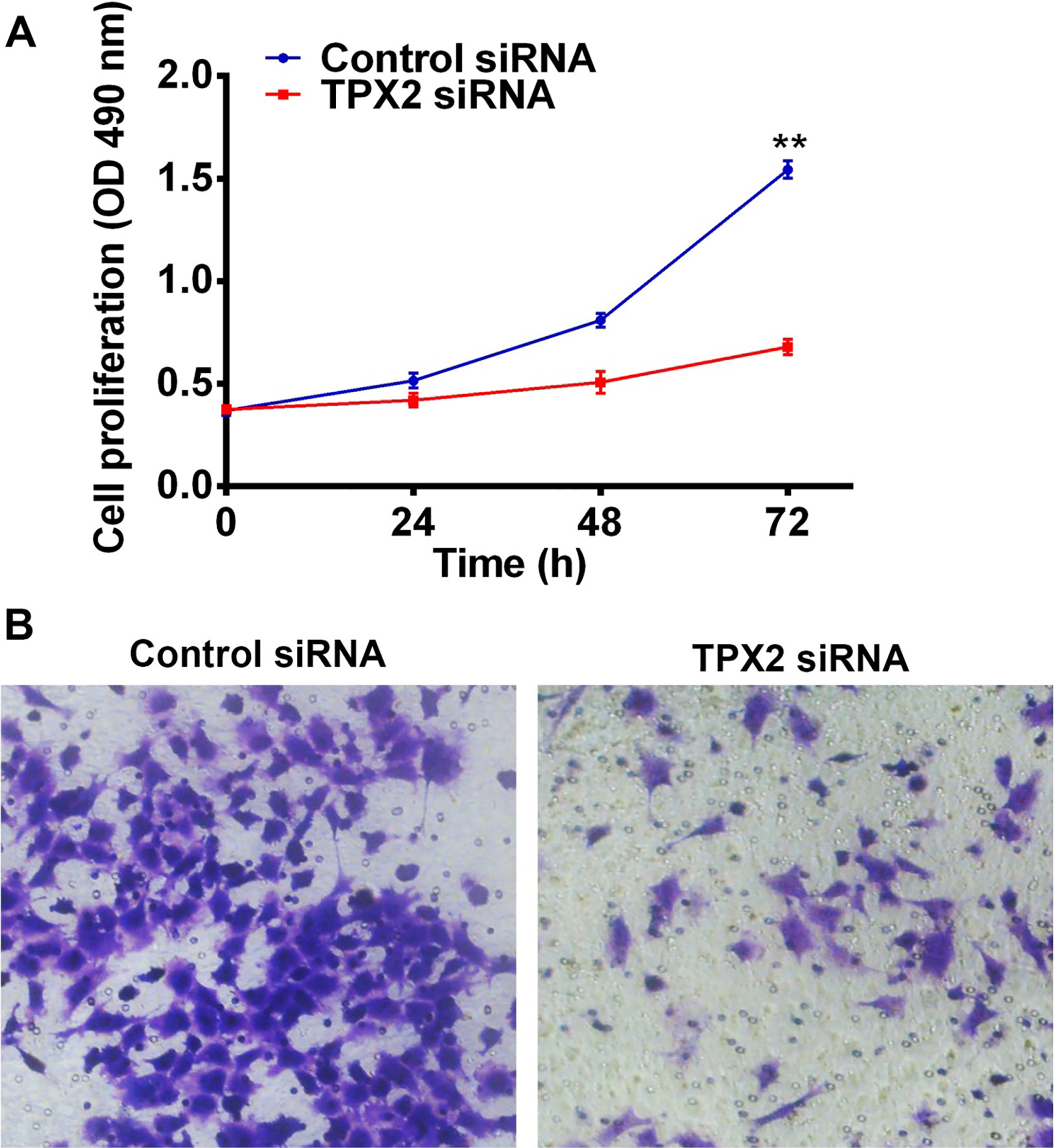
Effects ofTPX2 silencing on cell cycle distribution of RMG1 cells. (A) Representative images of the effects of TPX2 inhibition on the cell cycle distribution of RMG1 cells as determined by flow cytometry assays. (B) Quantification of the effectsof TPX2 inhibition on the cell cycle distribution of RMG1 cellsshown in (A). Knockdown of TPX2 induced G2/M phase arrest in RMG1 cells.
TPX2 silencing induced PLK1/Cdc2/Cyclin B activation. (A) Western plot analysis was performed to determine the effects of TPX2 inhibition onthe protein levels of PLK1, p-Cdc2 (Tyr15) and Cyclin B in RMG1 cells. (B) Quantitative RT-PCR analysis was employed to assess the effects of TPX2 inhibition on the mRNA levels of PLK1 and Cyclin B in RMG1 cells. 
TPX2 silencing induced PLK1/
TPX2 modulates Cdc2/Cyclin B and 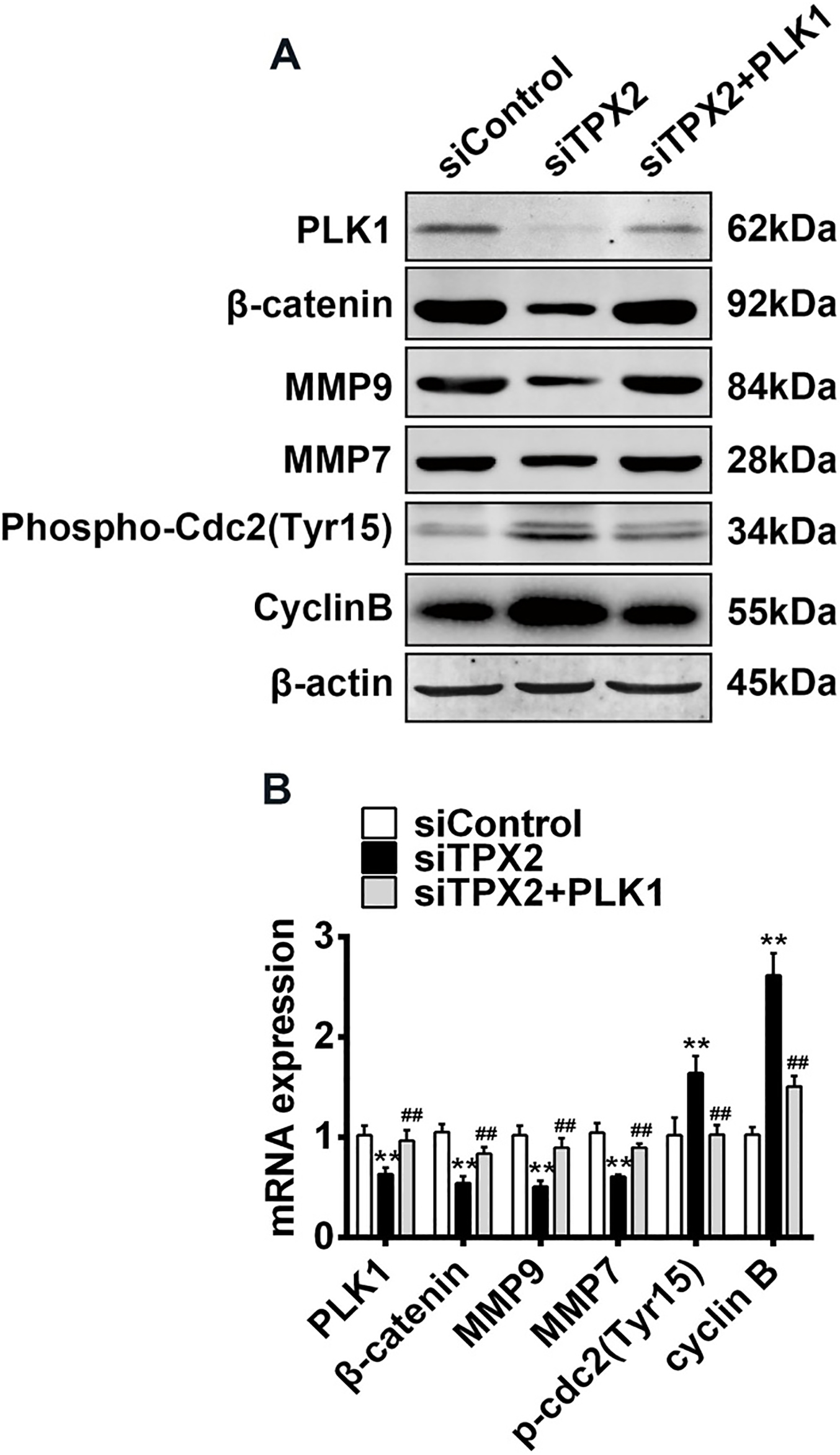
Aberrant over-expression of TPX2 in OC tissues
To characterize the role of TPX2 in OC, we first profiled the protein level of TPX2 in five paired OC tissuesand corresponding adjacent tissues.Our results showedthat the protein expression of TPX2 was significantly markedly elevated in OC tissues comparedwith paired normal tissues (Fig. 1A). We then performed quantitative PCR analysis toanalyze the expression level of TPX2 mRNA in 32 paired OC tissuesand corresponding adjacent tissues. TPX2 expression in OC tissueswas substantially increased compared to the pair-matched adjacent tissues (
Over-expression of TPX2 promotes proliferation and migration of OC cells
The significantly increased expression of TPX2 in OC tissues prompted us to exploreits biological role in OC cells. The OC cell line SKOV3, with a relative low endogenous expression level of TPX2, was transfected with pcDNA-TPX2 orpcDNA3.1 as control. Quantitative PCR assay was employed to examine the over-expression effect of TPX2. Cells were then subjected to MTT assay to measure the impact of TPX2 on the proliferation of OC cells. The results showed that over-expression of TPX2 caused a marked increase inthe viability of the SKOV3 cells compared with thecontrol group (
TPX2 silencing inhibits proliferation and migration of OC cells
To confirm the findings by using exotic expression of TPX2 in OC cells, we introduced a small interfering RNAspecifically targeting TPX2 inRMG1 cells, which has a relative high endogenous expression level of TPX2. Quantitative PCR assay was performed to confirm the successful inhibition of TPX2 in RMG1 cells. In the contrast to the findings evidenced by over-expression of TPX2 in SKOV3 cells, knockdown of TPX2 significantly decreased the proliferation of RMG1 cells assayed by MTT (
TPX2 silencing induces G2/M arrest
Tumourigenesis is the result of uncontrolled cellular proliferation, which mainly caused by cell cycle disorganisation. Therefore, we next examined the impact of TPX2 silencing on cell cycle distribution measured by flow cytometry. Compared to control group, knockdown of TPX2 in RMG1 cells gradually reduced the G1 phase cells proportion, while increased the G2/M and Sphase cells proportion (Fig. 4A and B). These result indicated that TPX2 silencing inhibited the proliferation of RMG1 cells through entailing cell cycle arrest in G2/M phase.
TPX2 silencing induces inactivation of PLK1/Cdc2/cyclin B signaling
Aurora-A and PLK1are centrosomal kinases involved in centrosome maturation and spindle assembly. TPX2 is a MT-binding protein involved in spindlepole formation and is the best characterized RanGTP-dependent spindle activator. TPX2 is required for Aurora-A binding to spindle MTs and its binding to Aurora-A holdsthe latter in an active conformation. Recently, afunctional interplay between Aurora-A, PLK1 and TPX2 at spindle poles. To identify the underlyingmechanisms by which TPX2 promotes OC cell proliferation and migration, we evaluated the effect of TPX2 silencing on PLK1. We examined the protein and mRNA levels of PLK1 after knockdown of TPX2 in RMG1 cells by Western blot and qRT-PCR assays, respectively. The result showed that TPX2 silencing inhibited both the protein and mRNA levels of PLK1 (Fig. 5A and B).
PLK1 have been shown to be important contributors to several other cell-cycle events, especially through inducing inactivation of the mitotic cyclin-dependent kinase complex consisting of a Cdc2 catalytic subunit and a cyclin B regulatory subunit. To confirm that TPX2 functions as a cell cycle modulator via Cdc2/CyclinB, we also determined the regulation of TPX2 on this complex. As shown in Fig. 5A, knockdown of TPX2 significantly activated p-Cdc2 and Cyclin B determined by Western blot in RMG1 cells. Consistently, up-regulation of Cyclin B mRNA level was also found in TPX2-depleted RMG1 cells (Fig. 5B). Collectively, TPX2 may promote cell proliferation through modulating the PLK1/Cdc2/CyclinB signaling in OC cells.
TPX2 silencing induces inactivation of PLK1/
-catenin/MMPs signaling
Wnt/
TPX2 silencing induces inactivation of Cdc2/CyclinB and
-catenin/MMPs via PLK1
Since TPX2 plays as an oncogenic role in OC cellsand enhanced PLK1 and its downstream effectors Cdc2 and
Discussion
Over the past two decades, significant improvement has undoubtedly achieved in the treatment of OC as a result of better multi-modality care [20, 21, 22, 23, 24]. However, major challenges remain in the treatment of OC. Understanding more about the molecular abnormalities involved in OC will be significant for molecular targeted therapy and improvement of clinical outcome.
Chromosomal instability and subsequent aneuploidy play a causative part in a substantial proportion of malignancies [25]. The TPX2 gene first identified by Heidebrecht in 1977 [26] is located on human chromosome 20q11.2 and is revealed as a proliferation-related nuclear protein [27, 28]. Further researches uncover that TPX2 regulates bipolar spindle assembly through recruiting Aurora-A kinase to microtubules and driving the activation of this kinase during mitosis. The binding of TPX2-Aurora-A is dependent on Ran-GTP signaling pathway [29]. Expression levels of TPX2 were found to correlate with chromosomal instability [30]. TPX2 overexpression induces the amplification of centrosomes and leads to aneuploidy [31]. Accumulating evidences show that decreasing TPX2 levels may represent an efficient approach for cancer treatment. For example, TPX2 knockdown has demonstrated to attenuate the proliferation of breast [18], esophageal [32], and prostate [17] cancer cells. In this study, we showed that TPX2 was upregulated in human ovarian cancer tissues compared to adjacent normal tissues, indicating that TPX2 might be a promising target for ovarian cancer therapy. Future studies focusing on expression levels of TPX2 in samples from ovarian cancer patients in different disease stages would provide us more information about the association between TPX2 expression levels and ovarian cancer progression. More importantly, we uncovered that in TPX2 remarkably promoted the proliferation and migration of human ovarian cancer cells. In conclusion, our findings proposed TPX2 as a potential drug target in ovarian cancer.
PLK1 is a member of polo-like kinase (PLK). Of the five known human PLKs, PLK1 is the most extensively studied because of its crucial role in cell-cycle regulation [33]. Similar to TPX2, PLK1 regulates cell-cycle progression through its effects on bipolar spindle formation and chromosome segregation [34]. PLK1 is also involved in the development of various kinds of cancers since its role in the cell-cycle and is considered as a drug target for many cancers [35]. When we explored the mechanisms underlying the effects of TPX2 on OC cells, we found that TPX2 knockdown inhibited the expression of PLK1. Our results from flow cytometry indicated that TPX2 knockdown significantly increased the number of OC cells in the G2. M phase. The cell cycle progression at the G2/M phase may be triggered by the checkpoint such as Cdc2 and cyclin B [36]. When the influence of TPX2 knockdown on the regulatory proteins involved in G2/M arrest in OC cells were analyzed, we found that the activity of Cdc2 and the expression level of cyclin B were enhanced following TPX2 knockdown in OC cells. Moreover, overexpression of PLK1 obviously reversed the anti-proliferation and anti-migration effects of TPX2 knockdown on OC cells. These results suggest that TPX2 promotes OC cell proliferation and migration through PLK. We didn’t examine the expression level of PLK1 in OC tissues, cause a previous study has shown that PLK1 is highly expressed in OC tissues and is considered as a prognostic factor in ovarian carcinoma [37].
Conclusion
This study provided the first evidence that TPX2 promotes the proliferation and migration of OC cells through regulation of PLK1, elucidating the underlying mechanism of TPX2 as a prognostic biomarker in OC. Therefore, TPX2 played an essential crucial role in the development and progression of OC, providing a potential therapeutic strategy for OC.
Footnotes
Conflict of interest
The authors declare no conflict of interest.
