Abstract
BACKGROUND:
The role of adopokines in adrenal tumors’ hormonal activity remains unclear. Obesity may induce arterial hypertension, disorders of carbohydrate metabolism, and is a risk factor of cardiovascular disease. In patients with subclinical hormone secretion by the adrenal cortex or medulla the risk of metabolic disease is increased.
OBJECTIVE:
Authors of this retrospective study selected 78 patients with subclinical hormone secretion out of all adrenal incidentaloma patients hospitalized in the Department of Endocrinology and Internal Medicine between 1995 and 2014.
METHODS:
The analyzed group comprised of 38 subclinical Cushing’s syndrome (SCS), 40 incidentally discovered pheochromocytoma (PHEO) and 42 patients operated due to an adrenal tumor without pathological hormonal activity. Expression of adiponectin (AdipoR1, AdipoR2) and leptin (Ob-R) receptors in adrenal tumors was assessed in relation to body mass index (BMI) and hormonal activity.
RESULTS:
We found statistically significant negative correlations between BMI and expression of all examined receptors in SCS patients (AdipoR1:
CONCLUSIONS:
Data obtained show that the most significant factor associated with the expression of AdipoR1, AdipoR2 and leptin Ob-R receptors in the adrenal tumor tissue is BMI, not their hormonal activity.
Introduction
Adipose tissue is considered the largest ‘endocrine gland’ of the human body. It synthesizes and secrets over 50 distinct hormones and cytokines, which play an important role in the metabolism, insulin sensitivity and cardiovascular disease [5, 21].
Age, sex and BMI of subclinical Cushing syndrome – SCS, incidentally discovered pheochromocytoma – PHEO patients and non – functioning adrenocortical tumors – CONT group
Age, sex and BMI of subclinical Cushing syndrome – SCS, incidentally discovered pheochromocytoma – PHEO patients and non – functioning adrenocortical tumors – CONT group
Normal weight: BMI (body mass index) 18.5–24.9 kg/m
Hormonal data of subclinical Cushing syndrome – SCS patients and non – functioning adrenocortical tumors (CONT)
The adrenal glands, which are surrounded by adipose tissue, produce both gluco- and mineralocorticoids as well as catecholamines that also influence the cardiovascular system and glucose metabolism [2, 19]. In patients with subclinical hormonal secretion of the adrenal cortex or medulla risk of metabolic diseases is increased [2, 7, 8, 18, 22].
So far, few reports have been published on adipone-ctin receptor expression in the adrenal cortex [4] and medulla [12] tumor tissues. Adiponectin receptors demonstrated in the adrenal cortex tissue may help elucidate the role of a functional connection between BMI, arterial hypertension, and adrenal function. It has been suggested adipose tissue may regulate the function of the human adrenal cortex [19].
The authors of the present study attempted to answer the question whether the overproduction of glucocorticoid hormones and catecholamines by adrenal tumors affects the expression of adiponectin Adipo R1, Adipo R2 and leptin Ob-R receptors in resected tumors.
The SCS group comprised of 38 patients; the PHEO group – 40 patients and the non-functioning adrenocortical tumors – control group (CONT) – 42 patients. Patients were aged 23 to 92 (mean age 53.3 years). In SCS group patients were aged 27 to 77; in PHEO group – 24 to 74 and CONT group patients were aged 25 to 75 (Table 1).
Both groups had comparable BMIs: mean BMI in SCS patients was 27.1 and in PHEO patients 25.8 (Table 1). Obtained results were compared with those adrenal tumor patients who did not present subclinical hormonal activity of the adrenal cortex or medulla. Mean BMI of patients with tumors without pathological hormonal activity of the adrenal glands were 26.8 (Table 1).
Urinary metanephrine and normetanephrine excretion in incidentally discovered PHEO patients and non-functioning adrenocortical tumors – CONT
Urinary metanephrine and normetanephrine excretion in incidentally discovered PHEO patients and non-functioning adrenocortical tumors – CONT
IQR – interquartile range.
Antibodies used for detection along with applied methodology
Expression of AdipoR1 receptors in adrenal cortical tumor (A), PHEO (B), magnification 20x.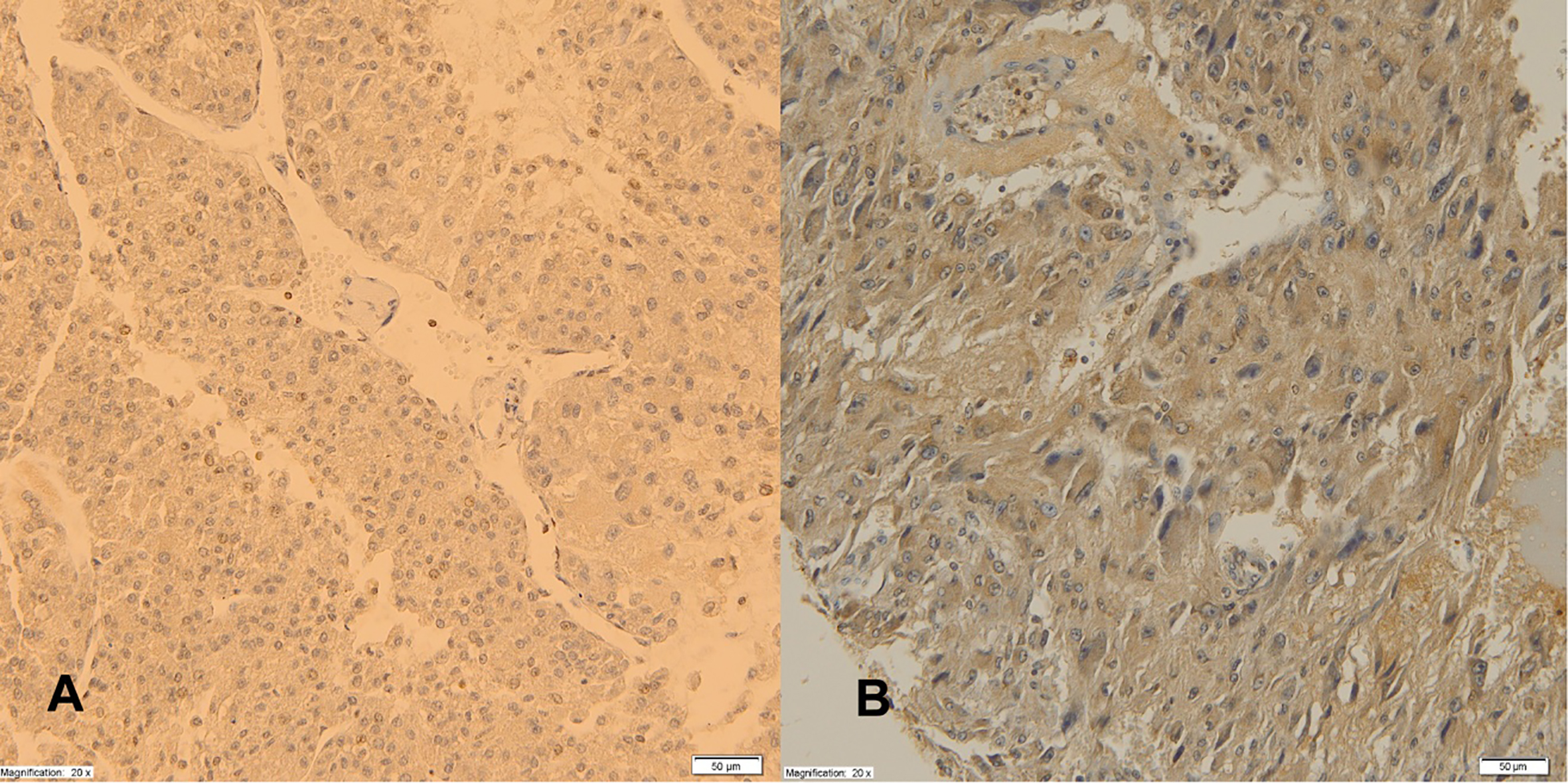
All patients were admitted to our Department. SCS was diagnosed in accordance with the definition in patients with an incidental adrenal mass found in an abdominal CT or MRI scan, who did not have typical physical features of Cushing Syndrome (CS) and exhibited endogenous hypercortisolemia in hormonal tests. Cortisol morning serum concentration of 140 nmol/L (5.04
Obtained data were compared with those of 42 adrenal tumor patients who were not diagnosed with SCS. Hormonal characteristics of study samples are shown in Table 2.
All hormone concentrations were determined in the same laboratory using freely available kits. The lab kits were not changed during the evaluated period. ACTH concentration was determined with a solid-phase, two-site sequential chemiluminescent immunometric assay Immulite 1000
To evaluate metabolic disorders, body mass index (BMI) was calculated in all patients based on weight and height measurements. In the SCS subgroup 33 patients were either overweight or obese (Table 1).
The diagnosis of incidentally discovered PHEO was based on elevated 24-h urinary metanephrine and normetanephrines excretion, positive imaging study and lack of clinical symptoms indicative of a PHEO tumor (labile arterial hypertension, tachycardia, diaphoresis). Imaging phenotype of PHEO means: increased attenuation on nonenhanced CT
The 24-h urinary metanephrine and normetanephr-ines excretion determined by means of high-perfor-mance liquid chromatography – HPLC on Bio-Rad. All medications that may interfere with urinary metan-ephrine and normetanephrine results were excluded. Hormonal characteristics of PHEO patients are shown in Table 3. Twenty seven patients with PHEO were either overweight or obese (Table 1).
Expression of AdipoR2 receptors in adrenal cortical tumor (A), PHEO (B), magnification 20x.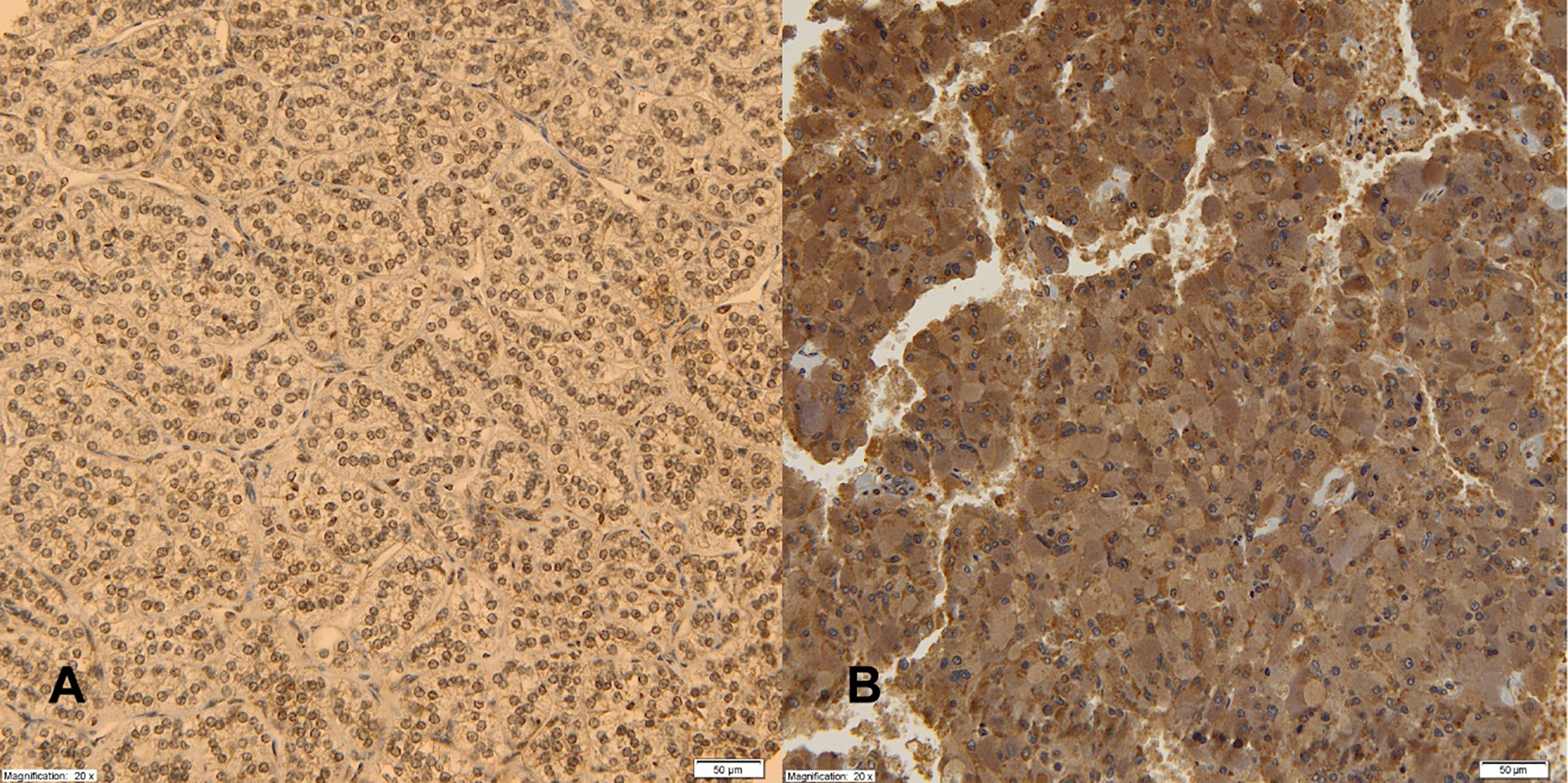
Expression of leptin Ob-R receptors in adrenal cortical tumor (A), PHEO (B), magnification 20x.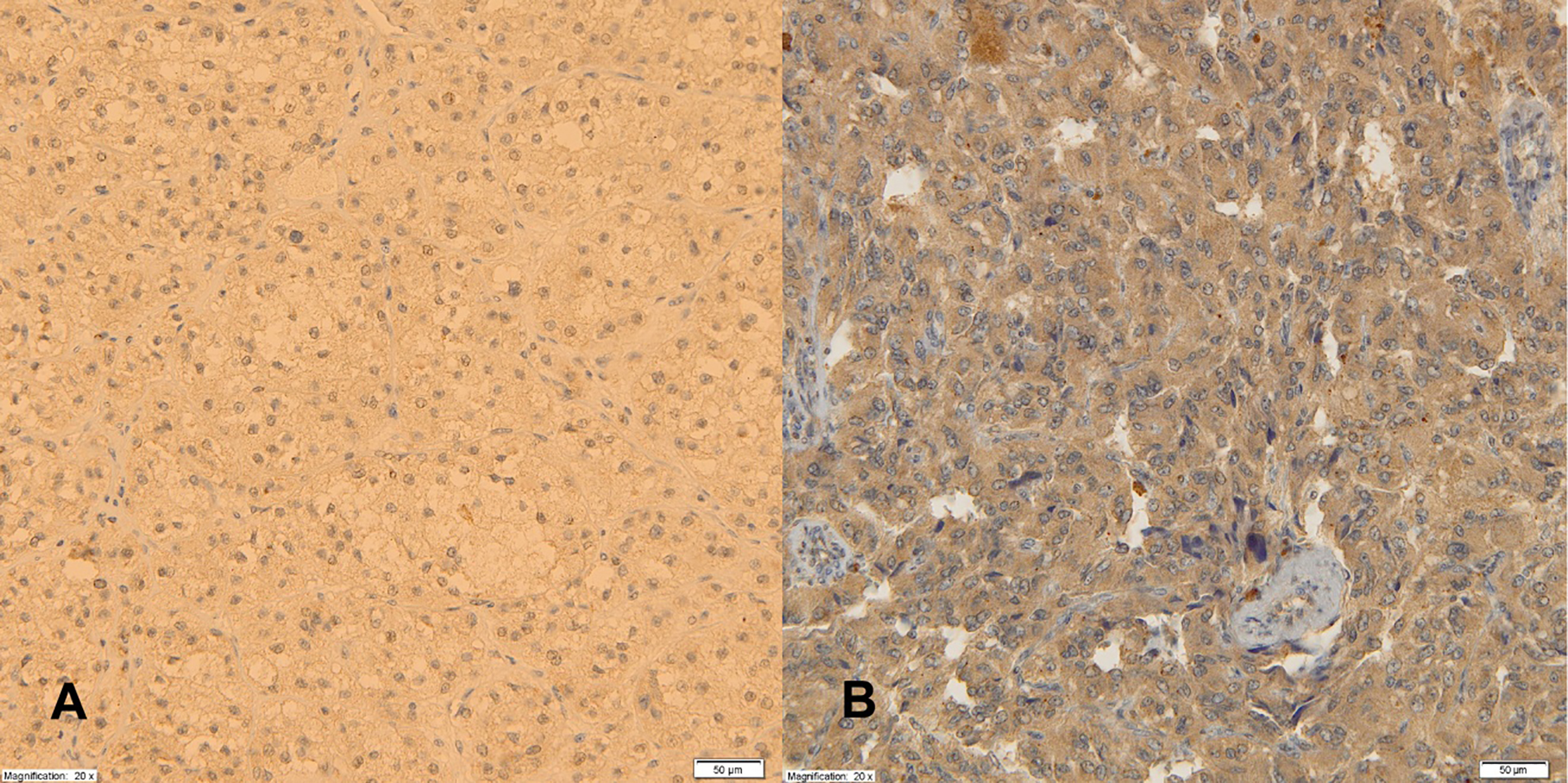
Due to subclinical hormonal activity all patients with SCS and incidentally discovered PHEOs underwent adrenalectomy. To eliminate possible BMI changes while waiting for surgery, BMI was evaluated 2–3 days before adrenalectomy. In the SCS group 23 patients were diagnosed with an adrenal cortex adenoma and 15 with nodular hyperplasia. In the incidentally found PHEO group, PHEO was in result confirmed histopathologically in all cases.
In the group of patients without pathological hormonal activity (CONT) indication for surgery was the size of tumor over 4 cm or suspicion of malignancy in MRI or CT. The lack of hormonal activity means the exclusion of SCS and overproduction of metanephrine or normetanephrine, but also the absence of testosterone, androstenedione, DHEAS or hydroxyprogesterone secretion. The adrenal adenoma was found in 16 cases and nodular hyperplasia in 26 in that control group.
Expression of adiponectin and leptin receptors was determined in the excised tumor tissues of the adrenal cortex or medulla.
Tumor tissue was preserved according to standard procedures (fixed for approximately 48 hours in 10% formaldehyde solution, later dehydrated using appropriate alcohol and xylene solutions, and, finally, embedded in low melting point paraffin). Tissue specimens of analyzed cases were re-assessed in routinely prepared hematoxin-eozin (H&E) stains to verify the histopathological diagnosis. From the studied specimens areas most representative for neoplasm were chosen; these areas contained neither necrotic lesions nor thermal damage artefacts.
Correlation coefficients between BMI, adiponectin and leptin receptors expression
Spearman rank correlation coefficients. Statistically significant correlations were indicated:
Immunochistochemical stains were made on microtome sections of 4 micrometer thickness. They were placed on Superfrost PLUS slides (by Surgipath) and later incubated overnight at 37
The expression of adiponectin and leptin receptors were supplemented with serum adiponectin and leptin concentration in 12 cases. Plasma adiponectin assay was performed using a Human HMW Quantikine Elisa Kit (Biokom). Serum leptin concentration were determined by commercial sandwich enzyme immunoassay EIA kit (DRG MedTek).
Statistical analysis
Standard descriptive statistics were calculated. Correlations were assessed using Spearman’s method. Multivariate analysis was performed using linear regression with robust standard errors. Some independent variables were log-transformed. p values lower than 0.05 were considered statistically significant. All calculations were performed using a PC computer and STATA v13.1 statistical software package (StataCorp LP, Texas, USA).
Results
Adiponectin and leptin receptors expression in adrenal cortex tumor tissue and subclinical hormonal activity
In univariate analysis no significant differences in AdipoR1 receptor expression were found between patients with and without SCS (
We analyzed adipokine receptors expression and cortisol levels after 1 mg dexamethasone suppression test in SCS patients.
In univariate analysis cortisol correlated with AdipoR1 (
Adiponectin and leptin receptors expression in incidentally discovered PHEO tissue.
In univariate analysis correlations were tested between expression of adipokine receptors and 24-h urinary metanephrine and normetanephrine excretion. A statistically significant, positive correlation was found only between AdipoR1 expression and metanephrine excretion (
A positive correlation between leptin Ob-R receptor expression and 24-h urinary metanephrine excretion was found (
In multivariate analysis, similarly to adrenal cortex tumors, the relationships were not statistically significant after taking into account patients’ BMIs (
AdipoR1, AdipoR2 and Leptin Ob-R receptors expression and BMI
Due to the confounding effect described above associations between BMI and adipokine receptor expressions were tested. Results were presented in Table 5.
In adrenal cortex tumors statistically significant, negative correlations between BMI and expression of all examined receptors were found (AdipoR1:
In PHEO, BMI correlated negatively with AdipoR2 (
Serum adiponectin and leptin secretion and AdipoR1, AdipoR2 and leptin Ob-R receptors correlation
Only 12 cases have been evaluated for serum adip-onectin and leptin concentration. The obtained results were correlated with AdipoR1, AdipoR2 and leptin Ob-R receptors expression in human adrenal tumors. The authors demonstrated statistically significant positive correlations between serum adiponectin secretion and adrenal AdipoR1 expression only (
Due to retrospective nature of the study, a small group of patients is significant limitation of obtained results.
Discussion
Interrelationship between human adipose tissue and the adrenal glands has lately been a subject of interest. Adipose tissue is the source of several adipokines and cytokines, which influence metabolism and neoformation [1, 5, 11, 14].
Obesity as a social problem may induce arterial hypertension, carbohydrate metabolism disorders and constitutes a risk factor for cardiovascular disease [5, 6, 20, 21].
Adiponectin is the best known adipokine which acts through two different receptors – AdipoR1 and AdipoR2. AdipoR1 is abundantly expressed in skeletal muscles and AdipoR2 predominates in the liver. Adiponectin stimulates phosphorylation and activation of AMP kinase, thus suppressing gluconeogenesis in liver and promoting fatty-acid oxidation and glucose uptake in muscle [9, 10]. Obesity decreases expression levels of AdipoR1 and AdipoR2, thereby reducing adiponectin sensitivity [9, 10, 12].
Concentration of circulating adiponectin is proportional to the amount of adipose tissue and is lower in patients with diabetes mellitus and/or cardiovascular diseases [5, 6, 8, 12, 11, 13, 16, 23]. Obesity is connected with reduced adiponectin levels even in healthy individuals [8, 12].
In patients with subclinical hormonal activity of the adrenal cortex or medulla the risk of metabolic disease is higher compared to patients with adrenal tumors without pathological hormonal activity [2, 18, 22, 24, 25].
Glucocorticoids participate in control of caloric intake and adipogenesis. Glucocorticoids stimulate food intake, synthesis and activity of adipose tissue lipoprotein lipase, adipocyte differentiation and distribution. Some of those actions are in synergy with insulin [24]. Glucocorticoids also participate in epinephrine synthesis in the adrenal medulla, both epinephrine and insulin influence energy metabolism [18].
Recent reports prove presence of adiponectin receptors in normal adrenal as well as adrenal tumor tissue [19]. In some studies, the authors have shown that adiponectin receptor expression is significantly higher in hormonally active adrenal tumors compared to normal adrenal tissue suggests that adipose tissue may regulate human adrenal cortex [8, 19]. Recent reports suggest also an active role of the fat depot surrounding the adrenal tumors with local secretion of adiponectin and leptin [15].
In the context of above-mentioned literature data, the current study analysed associations between adipo-nectin and leptin receptors expression in adrenal tumors and subclinical hypercortisolemia (SCS). Taking into account confounding, we showed that BMI increase was associated with decreased expression of AdipoR1, AdipoR2 and leptin Ob-R receptors in adrenal cortex tumor tissue. Subclinical glucocorticoid overproduction did not influence this expression. Higher risk of metabolic diseases in subclinical hypercorisolemic patients is most probably the result of higher incidence of overweight and obesity, and, in consequence, lower serum adiponectin concentrations present in these patients.
PHEO patients are characterized by decreased glucose tolerance and diabetes mellitus is present in one third of them [7]. The relationship between diabetes and PHEO involves suppression of insulin secretion by catecholamines and induction of insulin resistance [7]. Thus far, available data indicate that AdipoR1 and AdipoR2 receptors are also present in normal adrenal medulla tissue [12]. Isobe et al. reported that in examined patients adiponectin AdipoR1 receptor expression correlated with catecholamine oversynthesis by adrenal tumor tissue and an increase in the risk of diabetes [12]. In our research we showed that BMI was the only factor that negatively correlated with the expression of the AdipoR2 receptors in PHEOs.
In normal adrenal tissue the presence of leptin Ob-R receptor expression has also been demonstrated; however, its role is unclear [17]. Leptin inhibits adrenal steroid synthesis by influencing the cortex, and, enhances catecholamine release by the medulla. These observations allow putting forward a hypothesis that leptin has an activatory effect on the sympathetic-adrenal system [17]. Bottner and co-workers assessed serum leptin concentrations in PHEO patients. Despite catecholamine oversynthesis in PHEO adiponectin and leptin receptors’ expression was suppressed in this group of patients with increasing BMI [3].
Authors of the current study, after taking into account the confounding effect of BMI, found neither an association between catecholamine overproduction by adrenal medulla tumors and adiponectin nor leptin receptors’ expression. BMI was the only factor that negatively correlated with the expression of the AdipoR2 receptors in adrenal medulla tumors. This indicates that adiponectin and leptin receptors in PHEO are not under the control of inhibitory influence of the adrenergic system in patients with chronic hypercatecholaminemia. The decisive influence on their expression is mediated through BMI.
In the presented retrospective study, only 12 cases of serum adiponectin and leptin were evaluated. The authors demonstrated that higher serum adiponectin correlated positively with its AdipoR2 receptor in adrenal tumor tissue. Due to the small and heterogenous group of patients, the results could not be correlated with their hormonal activity or BMI. However, our research may suggest that adiponectin receptors mediate the effects of adiponectin in adrenal tumors.
Conclusions
Overweight and obesity are key in regulating hormonal activity of the adipose tissue. Our data suggest that adiponectin and leptin receptors expression in human adrenal tumors is mainly associated with BMI. Associations between serum concentrations of various adipokines, their receptors’ expression in the adrenal glands, and the influence of adipose tissue, including that one surrounding the adrenal glands, require further prospective research.
Footnotes
Acknowledgments
This research was supported with the funds of the Medical University of Gdańsk grant number: ST-81.
Conflict of interest
The authors declare that they have no conflicting interests.
