Abstract
BACKGROUND:
Intratumoral up-regulation of genes coding for drug transporters and metabolizing enzymes, such as MDR1 and CYP3A4, after chemotherapy are linked to cancer drug resistance. However their expression in primary soft tissue sarcomas (STS) prior to drug treatment and their role in innate resistance remain unclear.
OBJECTIVE:
The aim of this study was characterize MDR1 and CYP3A4 expression pattern before to chemotherapy and its clinical implication in pediatric STS.
METHODS:
In this prospective study we analyzed MDR1 and CYP3A4 mRNA expression in both normal and tumor tissues from 28 newly diagnosed STS pediatric and then compared with patients’ clinical-pathological data, including chemotherapy response.
RESULTS:
Our data showed that the expression of the MDR1 gene was significantly higher in malignant tissue than in the normal tissues of patients with STS. In addition, high MDR1 expression was significantly associated with local advances, as well as poor response to treatment. In contrast, CYP3A4 expression level was negligible in both tumoral and non-tumoral tissues.
CONCLUSIONS:
These results suggest that a significant mRNA level of MDR1 gene was intrinsically present in STS before exposure to chemotherapeutic drugs, suggesting that MDR1 may be important contributors of innate chemoresistance of this tumor type.
Introduction
Soft tissue sarcomas (STS) are a heterogeneous group of malignant tumors that originate from primitive mesenchymal tissue and account for approximately 7% of all tumors in children and adolescents [1]. Rhabdomyosarcomas (RMS) constitute about half of all soft tissue sarcomas, and the remainder are referred to as non-RMS soft tissue sarcomas (NRSTS), a designation that includes more than 50 different histological subtypes [2]. This heterogeneity makes therapy particularly difficult because each of these histological subtypes represents a unique disease with distinct biologic behavior and varying sensitivity to chemotherapy [3]. While chemotherapy is the only available treatment option for many patients with advanced, unresectable or metastatic STS, it has demonstrated moderate activity with a low response rate and an overall survival at 5 years of less than 10% [4]. Thus, there is a clear need to identify the prognostic factors that are common to the different subtypes of STS and the patients who can benefit from adjuvant or neoadjuvant chemotherapy.
Drug resistance is one of the major obstacles limiting the efficacy of cancer chemotherapy. Clinically, some tumors share the ability of being intrinsically resistant to first-line chemotherapy drugs prior to the start of therapy, standing in contrast to acquired resistance, which arises during treatment [5, 6]. Drug resistance has been correlated by the existence of a myriad of complex mechanisms, including metabolic factors that reduce the intracellular concentration of active drugs in tumors, such as the expression of drug-transporting proteins or drug-metabolizing enzymes, primarily by P-glycoprotein (P-gp) and cytochrome P450 3A4 (CYP3A4), respectively [7, 8]. P-gp, a member of the ATP-binding cassette family of drug transporters, is encoded by the multidrug-resistance-1 (MDR1) gene, which extrudes chemotherapy drugs out of the cells, thereby lowering their intracellular concentration [8, 9]. Increased expression levels of P-gp or its RNA transcript have been found in many tumor cells including breast, colon, gastric, kidney, leukemic, liver and pancreatic cells [10, 11, 12]. The chemotherapy substrates of P-gp include taxanes, vinca alkaloids, anthracyclines and epipodophyllotoxins, which are involved in the regimen of chemotherapy for STS [12].
CYP3A4 is a member of the cytochrome P450 family of oxidizing enzymes and is responsible for the phase I metabolism of approximately 50% of drugs that are currently in use [13]. CYP3A4 in tumors may be a relevant clinical factor in drug sensitivity because it may inactivate drugs, e.g., doxorubicin, epirubicin, etoposide, vincristine, and vinblastine (also substrates of MDR1/P-gp), or activate them, e.g., cyclophosphamide and ifosphamide [14, 15].
The functional synergism between P-gp/MDR1 and CYP3A4 in cancer cells to acquired drug resistance has been extensively studied both in vitro and in vivo [16, 17]. It is now known that these proteins, in addition to sharing numerous anticancer drug substrates, also have the same regulatory machinery governing their transcriptional regulation [18]. Furthermore, the close proximity of their genes on the same chromosome (7q21.1 to MDR1 and 7q22.1 to CYP3A4) emphasizes this relationship [19]. However, intratumoral characterization of MDR1 and CYP3A4 in STS prior to chemotherapy has not yet been fully investigated.
Therefore, in order to determine the relationships between the expression of the MDR1 and CYP3A4 genes and the intrinsic clinical chemoresistance of pediatric STS, we examined these genes by RT-PCR analysis in both tumor and adjacent normal samples obtained at diagnosis from 28 pediatric patients with STS and then were compared with the patients’ clinical-pathological data and chemotherapy response.
Materials and methods
Patients and samples
At the time of diagnoses, biopsies from primary tumors and matched adjacent non-tumor tissues were obtained prospectively from 28 patients with STS prior to any therapeutic procedures at the National Institute of Pediatrics, Mexico between 2010 and 2016.
Tumor samples were obtained from the malign mass, whereas adjacent normal tissue samples (normal) were obtained from the surrounding tissues that showed no sign of cancer by visual inspection, localized within 2 to 5 cm of the boundary of the cancer site. All normal and tumor specimens were histophatologically confirmed according to standard diagnostic procedures. The freshly obtained samples were immediately submerged in RNA Later solution (Ambion, Courtaboeuf, France) to avoid RNA degradation and were stored at
The Research and Ethics Committee approved the study, and patients (or patients’ parents or guardians on behalf of the children) signed an informed consent to participate in this research.
RNA extraction and cDNA synthesis
Total RNA was isolated from tissue biopsy specimens using the Trizol (Invitrogen Life Technologies) extraction method, according to the manufacturer’s instructions. Briefly, 50–100 mg of frozen tissue sections were ground in liquid nitrogen, disrupted with 1 mL cold Trizol with a rotor-stator homogenizer (Fisherbrand), and incubated at room temperature for 5 min. After chloroform extraction and precipitation with isopropanol, RNA pellets were washed with 75% ethanol, air-dried, and re-suspended in 30 to 50
Real-time quantitative PCR (RT-qPCR)
Real-time quantitative PCR (TaqMan PCR) was performed using the ABI Prism 7700 Real-Time PCR System (Applied Biosystems, USA), according to the manufacturer’s protocol. We employed commercially available TaqMan Gene Expression Assay probes (Applied Biosystems) for MDR1 (ID: Hs01070651_m1), CYP3A4 (ID: Hs00430021_m1), and
The data were collected, and the results were expressed in values of CT (cycle threshold). The 2
Patient follow-up
First-line chemotherapy regimens for treatment- naïve, NRSTS patients usually consisted of doxorubicin or epirubicin plus ifosfamide, and RMS cases frequently used a cocktail of cyclophosphamida, vincristine and actinomycin-D. Subsequently, we evaluated the clinical response after the completion of first-line chemotherapy. The patients were considered to be clinically resistant when they had a steady disease, progressive disease, or recurrent disease within six months after the completion of first-line chemotherapy. The patients were defined as clinically sensitive when they had complete remission or experienced relapse later than six months after the completion of first-line chemotherapy. Overall survival (OS) was defined as the time from cancer onset until death or by censoring at the last follow-up date.
Statistical analyses
Clinical data are expressed as the mean and standard deviations for the continuous variables. The Mann-Whitney U-test was used to perform statistical analyses of both gene expression (relative quantification) and the relationship between gene expression and the clinicopathological parameters and chemotherapy response. Survival was analyzed by the Kaplan-Meier method for the high- and low-expression groups (classified based on the median gene expression value) and compared via the log-rank test. All P-values were two-tailed. P-values
Results
Demographic and clinicopathological characteristics
During the study period, a total of 28 patients with a confirmed diagnosis of STS were included. Demographic and clinicophatological characteristics are summarized in Table 1. The sex distribution was 11 females and 17 males, with a median age at biopsy of 9 years (range: 0.5–17 years). The primary tumors included 14 RMS (4 embryonal- and 10 alveolar-subtype) and 14 NRSTS (5 synovial-, 5 malignant peripheral nerve sheet tumors- and 4 extraosseous Ewing’s-subtype) histopathological types. In respect to staging, there was 2 patient diagnosed at stage I (7.1%), 4 diagnosed in stage II (14.3%) and most of them (
Baseline demographic and clinicopathological characteristics in pediatric STS
Baseline demographic and clinicopathological characteristics in pediatric STS
RMS, rhabdomyosarcoma type; NRSTS, non-rhabdomyosarcoma soft tissue sarcomas type.
In this study the majority of patients were treated with systemic chemotherapy in combination with surgery and radiation therapy (64.4%,
The expression levels of the examined genes in 28 paired STS and non-tumor tissues from the same individuals were compared. Measurable levels of MDR1 mRNA were found in 22 of the 28 adjacent normal samples and 24 of the 28 tumor samples. Significantly higher MDR1 was detected in the tumor specimens compared with the normal adjacent tissues (Fig. 1A,
MDR1 and CYP3A4 mRNA expression in STS tissues. Relative expression of MDR1 (A) and CYP3A4 (B) genes were quantified by qRT-PCR in both normal and tumor tissues and its expression is normalized with 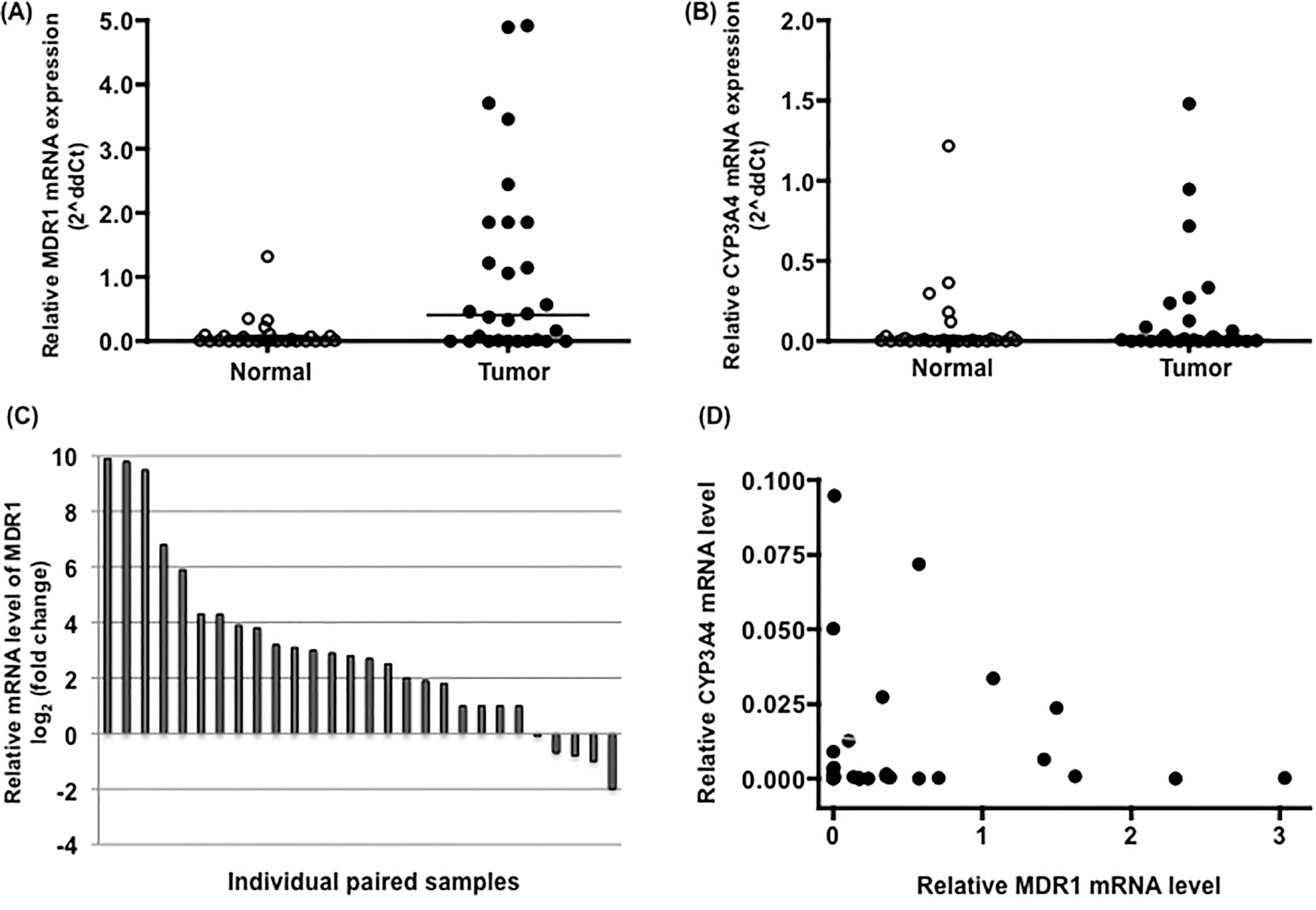
To examine the relationship between MDR1 tumor expression and the clinicopathological features, we classified the training cohort into subgroups according to the clinicopathological parameters, such as age at diagnosis, gender, histology (RMS or NRSTS), tumor stage (TNM stage; we grouped the TNM I
Correlation between MDR1 mRNA expression level and clinicopathological characteristics and outcomes of chemotherapy in STS patients
Correlation between MDR1 mRNA expression level and clinicopathological characteristics and outcomes of chemotherapy in STS patients
Data are expressed as means
Kaplan-Meier analyses for survival based on MDR1 expression levels in 28 pediatric soft-tissue sarcomas. Survival curves from the time of diagnosis for STS patients with either high or low expression of MDR1. The overall survival was defined as the interval death or the last follow-up examination. 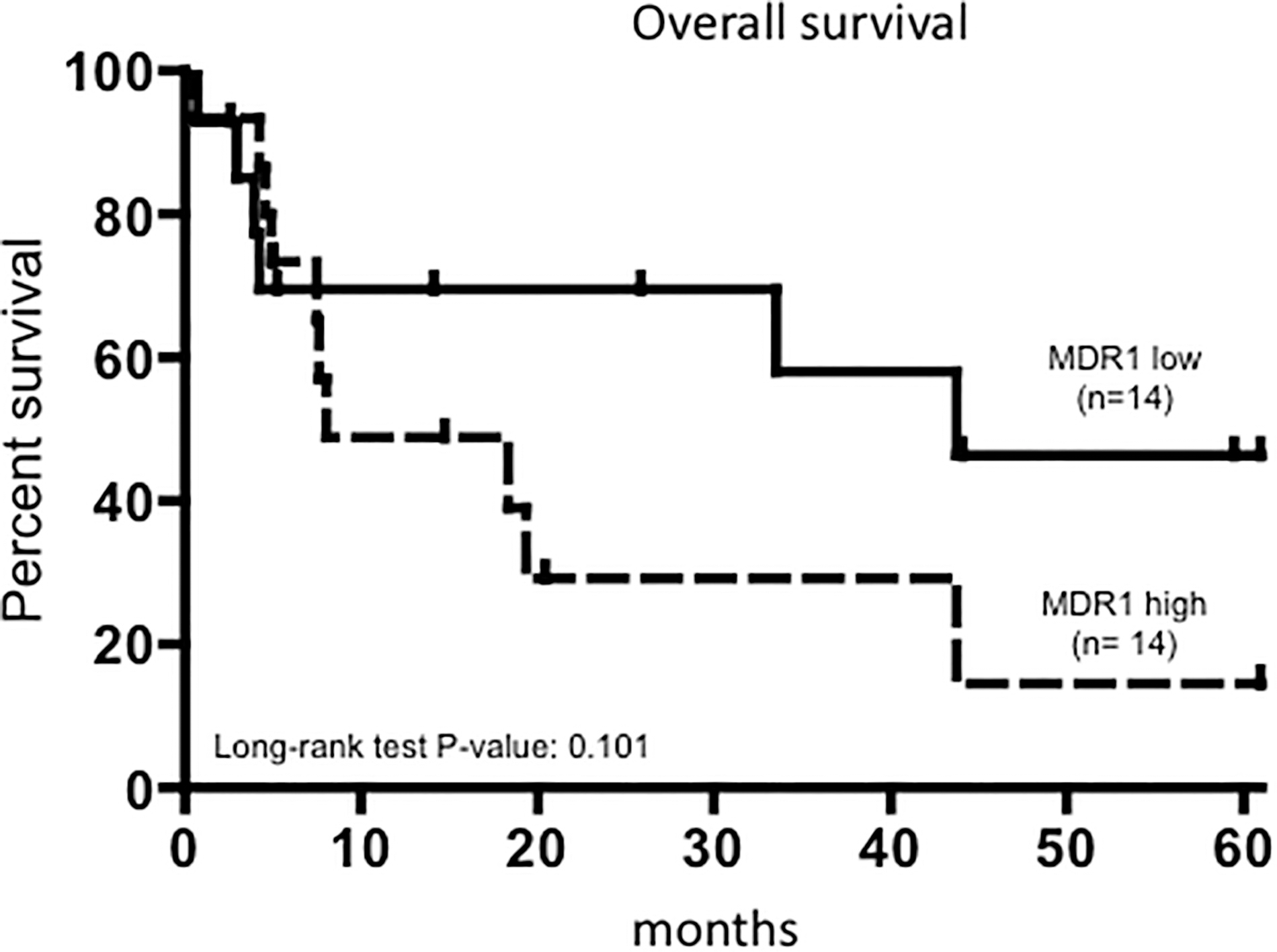
To calculate the potential prognostic impact of MDR1 in STS, the patients were assigned into two groups differing by either higher or lower gene expression than the median of all samples. Using the Kaplan-Meier survival analysis, we observed that STS patients with a high expression level of MDR1 tended to be associated with a shorter overall survival compared with those with a low expression; however, this association did not reach statistical significance probably due to the small cohort size (log-rank test,
Discussion
The emergence of intrinsic drug resistance is a persistent problem during the management of pediatric patients with STS, mainly in patients with advanced disease. Several alterations in many molecular pathways have been associated with acquired tumor resistance, including tumor alterations in genes coding for drug transporters and metabolizing enzymes, such as MDR1 and CYP3A4 [21, 22], which together may control the bioavailability of tumor cells to drugs used in cancer therapy, thus dictating their efficacy. Their expression in primary STS prior to drug treatment and, thus, the clinical implication, however, have not been well explored.
To the best of our knowledge, this study is the first to quantitatively assess MDR1 and CYP3A4 gene expression at diagnosis in paired tumor and adjacent normal tissue samples from 28 pediatric patients with STS and correlate these differences with clinical-pathologic features.
However, this approach has an important limitation that needs to be acknowledged. This is the lack of ideal control group since non-tumoral adjacent tissue may not represent truly the soft tissue components of the tumor nor its origin. Sarcomas are traditionally classified according to the morphology and type of tissue that they resemble. However, it is now known that sarcomas with high-grade lesions may lose gradually resemblance with the tissue of derivation. Moreover in numerous sarcomas, including synovial sarcomas, their origin is unknown and therefore they do not have normal counterparts [23].
Nevertheless, the study of tumor biology irrespective of approach requires controls; therefore we used adjacent tumor free tissue that is commonly used as a control in cancer studies [24, 25, 26]. Our study has also provided evidence that adjacent histological normal tissue control group reveals important deregulated MDR1 gene when compared to tumor samples and this may have predicted to drug resistance of some sarcomas to conventional chemotherapy.
Here, we demonstrated clearly that MDR1 gene expression was usually higher in the tumor tissues than in neighboring histologically normal samples. In contrast, low levels of CYP3A4 gene expression were observed in both the tumor and normal tissues. Our analysis also showed that there was no correlation between MDR1 and CYP3A4 mRNA levels in this cohort of patients. These data suggest that the intrinsic expression of MDR1 and CYP3A4 is regulated independently and that a significant mRNA level of the MDR1 gene was intrinsically present in STS even before exposure to chemotherapeutic drugs.
Specifically, the overexpression of the MDR1 gene at the time of clinical detection has been reported in some primary malignant tumors [27, 28, 29], including certain histological subtypes of STS [30], and in some cases, it has been correlated with chemoresistance in the clinical setting, but not in others [31, 32]. In this study, we observed that a high level of MDR1 transcript was significantly correlated with poor response to first-line chemotherapy, particularly in patients with tumors in advanced stages (III–IV stages), and a tendency to shorter survival times when compared with tumors with low MDR1 expression. It is worth mentioning that although we also observed a significant correlation between elevated levels of MDR1 and the RMS histological subtype, this may be because the vast majority of RMS tumors (13 from 14) presented at advanced stages of disease at diagnosis (III and IV group, data not shown).
In contrast, the intrinsically low levels of CYP3A4 mRNA found in the majority of both the tumor and adjacent matched normal tissues at diagnosis suggest that it is unlikely to carry out local activation or detoxification on the cytotoxic drugs used in the first-line chemotherapy treatments. These results are in agreement with a previous study from our group in which we demonstrated, in a small cohort of primary RMS, a negligible expression of CYP3A4 in paired normal and tumor samples [33]. Additionally, in a study of 37 patients with osteosarcoma, Trujillo-Paolillo et al. demonstrated that tumor and normal adjacent biopsy specimens taken before chemotherapy presented with lower CYP3A4 gene expression, while surgery tumor specimens taken from these same patients after chemotherapy showed significantly elevated CYP3A4 [34]. These data show the low presence of CYP3A4 levels in STS tumors before chemotherapy, suggesting that its local metabolic activity and participation in innate resistance of fist-line chemotherapy is insignificant.
In conclusion, although the sample analyzed in this study is small and heterogeneous, our results indicate that a significant level of MDR1 mRNA, but not CYP3A4 mRNA, is intrinsically present before chemotherapy in advanced STS tumors. Additionally, a poor response to first-line chemotherapy was found in patients with tumors with high MDR1 expression, suggesting that it may play an important part in generating an innate chemoresistance that is present in this childhood disease. Therefore, intrinsic MDR1 gene expression can not only be used as a prognostic marker of response to chemotherapy but also highlights MDR1 as a potential molecular target for the development of inhibitors that may be useful in reversing the innate drug resistance of pediatric patients with STS.
Footnotes
Acknowledgments
This study was supported in part by the Consejo Nacional de Ciencia y Tecnología, México Grant 262423, and Instituto Nacional de Pediatría.
