Abstract
BACKGROUND AND OBJECTIVE:
Clusterin promotes cell proliferation, motility and invasiveness in human renal cell carcinoma (RCC) cells but the underlying molecular mechanisms of this action are largely unknown. The aim of this study was to investigate the effects of clusterin on cancer cell growth, invasion and S100A4 expression and to determine the effects of clusterin on in vitro cell proliferation and migration and in vivo tumour growth in RCC cells.
METHODS:
We have established stable transfectants of highly invasive Caki-1 human RCC cells with expression of clusterin shRNA targeting clusterin (Caki-1/clusterin shRNA). We also established stable transfectants of 786-O human RCC cells with expression of clusterin cDNA plaismid (786-O/clusterin cDNA). Clusterin and S100A4 expression was detected by reverse transcription (RT) PCR and western blot assay; Caki-1/clusterin shRNA and 786-O/clusterin cDNA clones were subjected to in vitro-invasion assays. Cell viability and cell growth was assessed in MTT and clonogenic assay. Specific small interfering RNA was employed to down-regulate S100A4. The expression plasmid for S100A4 (pCMV-S100A4) was used to upregulate S100A4. Caki-1/clusterin shRNA clones were injected subcutaneously in nude mice to determine tumour growth and cancer cell invasiveness in vivo. Xenograft tumour tissues were assessed by immunohistochemistry and frozen tissues were used for the detection of S100A4 and clusterin.
RESULTS:
Overexpression of clusterin increased cell invasiveness; and targeting clusterin reduced cell invasiveness in vitro. This increase in cell invasiveness was mediated by S100A4. Targeting clusterin decreased cell proliferation and down-regulated cellular S100A4 levels in Caki-1 cells; Overexpression of clusterin increased cell proliferation and up-regulated cellular S100A4 levels in 786-O cells; Stable Caki-1/clusterin shRNA transfectants produced smaller xenograft tumours containing reduced S100A4 protein levels in vivo. Stable 786-O/clusterin cDNA transfectants produced larger xenograft tumours containing increased S100A4 protein levels in vivo.
CONCLUSION:
Our results indicate that clusterin promotes growth and invasion in RCC cells in vitro and in vivo through upregulation of S100A4; And targeting clusterin confers growth inhibitory and anti-invasive properties in RCC cells in vitro and in vivo through a down-regulation of S100A4. These findings provide the rationale for future oncostatic strategies aimed at suppressing clusterin-mediated signal transduction pathways as a novel therapeutic approach in human RCC.
Introduction
Renal cell carcinoma (RCC) is a group of malignancies arising from the epithelium of renal tubules [1]. It is the most common type of kidney cancer in adults, accounting for 90%–95% of cases; Clear cell RCC (ccRCC) is the dominant histology, accounting for approximately 65% of reported cases, followed by papillary and chromophobe RCC, accounting for approximately 15–20 and 5%, respectively; and the remaining includes a variety of uncommon sporadic and familial carcinomas, some of which have been recently described, along with a group of unclassified carcinomas. One of these tumors is extremely rare and is known as tubulocystic renal cell carcinoma [2, 3]. Hereditary RCC, which accounts for around 4% of cases, has been a relatively dominant area of RCC genetics. By far, the most studied somatically mutated gene is VHL mutations in von Hippel-Lindau disease, which follows the classic tumor suppressor gene paradigm of a germline cancer susceptibility gene also manifesting as being somatically mutated in the sporadic form of cancer type [4, 5]. VHL is somatically mutated in up to 80% of ccRCC [6].
At presentation, a third of patients with RCC already have locally advanced or metastatic disease and a third of patients who undergo surgical resection for local disease will have a recurrence. When metastasis occurs in RCC, the most common spread is to the lungs, bones, lymph nodes, liver, adrenal glands and brain. The median survival of patients with metastatic RCC (mRCC) is approximately eight months [7]. Patients with metastatic RCC (pT4) have a median survival of around 13 months and the 5 year survival rate is under 10% [8]. RCC is insensitive to chemotherapy and radiotherapy. Currently, the targeted therapies for the RCC are mainly focused on VEGF pathway and mTOR inhibition, however, the efficacy of targeted treatment is also limited [9]. Therefore, the further insight into the molecular mechanisms of RCC tumorigenesis and metastasis is needed to identify the new potential therapy targets.
Previous studies have shown that patients with RCC clusterin mRNA is 3-fold overexpressed in tissue of the tumors compared with adjacent non-tumor tissue [10]. Overexpression of clusterin prolongs cell survival, resulting in the enhanced metastatic potential of renal cell carcinoma in vitro and vivo [11]. Furthermore, targeting clusterin inhibited invasion and enhanced the proapoptotic activity in the RCC cells and human RCC model [12, 13]. However, the underling mechanism is still not very clear.
S100A4, an important member of S100 family proteins, functions to increase the tumor progression and metastasis. The molecular mechanisms of S100A4 involving in the progression and metastasis are diverse in various malignant tumors [14]. Extracellular S100A4 is believed to aid in tumor metastasis by inducing the secretion of matrix metalloproteinases [15], promoting angiogenesis through the stimulation of endothelial cell motility [16], and by increasing the formation of plasmin through binding to the endothelial cell surface receptor Annexin II [17].
Recently, S100A4 gene expression was described as a marker for poor prognosis in clear cell renal cell carcinoma and as an independent prognostic factor for lymph node metastasis in clear cell renal cell carcinoma [18, 19]. In renal cell carcinoma cells, S100A4 was shown to mediate tumor growth and metastasis [20].
The aim of the present study was to investigate the effect of clusterin on in vitro cell migration and in vivo tumour growth in RCC model. We found in our study that clusterin induces enhanced expression of S100A4. Knockdown of clusterin blunts S100A4 expression and also decreased tumour growth, proliferation and migration activity of the cells.
Materials and methods
Cell culture
Renal cell carcinoma Caki-1, A-498, 786-O, Caki-2, 769-P cells were maintained in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal calf serum, 2 mmol/l L-glutamine, 1,000 U/ml penicillin, and 1 mg/ml streptomycin. All cells were incubated at 37
Plasmid constructs and stable transfection
Clusterin cDNA amplified from Caki-1 cells was inserted into the pcDNA3.1 vector (Thermo Scientific). These lentiviral vectors were then transfected into 293T cells with other packaging plasmids to generate viruses that were used to obtain 786-O cells expressing Clusterin. The stably transfected cells (786-O/ clusterin cDNA or 786-O/pcDNA3.1) were generated by hygromycin B selection. The plasmids were generated using standard techniques and verified by sequencing.
shRNA-mediated knockdown of endogenous clusterin
shRNA constructs targeting clusterin and scrambled control were purchased from Open Biosystems (Lafayette, CO, USA). Transfection of Caki-1 cells with lentiviral particles containing clusterin shRNA and verification of the percentage of knockdown were performed as the manufacture’s instruction. Briefly, bacterial glycerol stocks containing the lentiviral plasmid vector pLKO.1 with shRNA inserts against clusterin, an empty vector or a scrambled shRNA control were obtained from Sigma Aldrich (Poole, UK). Lentivirus transfected 293T cells were incubated at 37
Transient S100A4 siRNA or pCMV-S100A4 transfection
The 786-O/clusterin cDNA, 786-O/pcDNA3.1, Caki-1/clusterin shRNA and Caki-1/pLKO.1 cells were seeded at 1
RT-PCR
Total RNA was extracted from each sample of various cells using TRIzol reagent (Invitrogen). RT was carried out with Superscript III reverse transcriptase using an oligo (dT) primer at 55
Western blotting
The whole-cell extracts were lysed in lysis buffer [20 mmol/L Tris (pH 7.4), 250 mmol/L NaCl, 2 mmol/ L EDTA (pH 8.0), 0.1% Triton X-100, 0.01 mg/mL aprotinin, 0.005 mg/mL leupeptin, 0.4 mmol/L phenylmethylsulfonyl fluoride (PMSF), and 4 mmol/L NaVO
Cell viability assay
Cells in different groups were seeded onto each well of 96-well plates for 48 h. Cell viability was quantified by measuring the absorbance of the solution at 570 nm wavelength by a spectrophotometer. The percentage of viable cells for each treatment group was calculated by adjusting the untreated control group to 100%. Duplicate wells were assayed for each condition and repeated at least three times. The IC
Clonogenic assay
Caki-1/clusterin shRNA, 786-O/clusterin cDNA cells and its control cells were seeded in six-well plates at 500 cells per well and incubated at 37
Matrigel invasion assay
We used 6.5 mm-diameter Transwell inserts (Costar, Cambridge, MA) with the 8
Renal cell carcinoma mouse models
Subconfluent Caki-1, 786-O, Caki-1/clusterin shRNA, 786-O/clusterin cDNA, 786-O/pcDNA3.1 and Caki-1/pLKO.1 cells were harvested by trypsinization, and resuspended in RPMI 1640. Only single cell suspensions with
Histological analysis and immunohistologic staining
Tissues were fixed with formalin, embedded in paraffin, sectioned (5
Statistical analyses
Differences in continuous variables were analyzed using the Student’s
Results
Expression of clusterin in RCC cells
We first used RT-PCR to detect the mRNA expression levels of the clusterin in renal cell carcinoma Caki-1, A-498, 786-O, Caki-2, 769-P cells. Clusterin mRNA expression was the richest in the Caki-1 cells, and rare clusterin mRNA expression was found in the 786-O cells (Fig. 1A). Western blot assay has the same results as RT-PCR assay (Fig. 1B). In the present study, we used Caki-1 and 786-O cells for further study.
Silencing of clusterin expression by stable clusterin shRNA transfection downregulates S100A4 expression
To corroborate whether clusterin regulated S100A4 expression, we employed the clusterin-specific shRNA approach to silence clusterin expression. Caki-1 cells were stably transfected with either the control vector or the clusterin shRNA expression vector. Individual clones were selected to detect clusterin and S100A4 expression by RT-PCR (Fig. 1C) and Western blots (Fig. 1D). The results showed that stable expression of the clusterin shRNA blocked clusterin expression and accordingly downregulated S100A4 expression.
Overexpression of clusterin by stable clusterin cDNA transfection upregulates S100A4 expression
Next, we hypothesized that whether clusterin-mediated upregulation of clusterin involves increased S100A4 transcription. The results showed that ectopic expression of clusterin by stable transfection of pcDNA3.1/clusterin cDNA in the clusterin-negative 786-O cells upregulated S100A4 expression by RT-PCR (Fig. 1E) and Western blots (Fig. 1F). Although out of the main focus of this study, the molecular mechanism by which clusterin positively regulates S100A4 transcription warrants further investigation.
Clusterin regulates S100A4 expression in RCC cells. A, Clusterin mRNA expression was detected in RCC cells by RT-PCR assay; B, Clusterin protein expression was detected in RCC cells by western blot assay; C, clusterin and S100A4 mRNA expression levels in Caki-1 cells transfected with clusterin shRNA and control shRNA by RT-PCR assay; D, clusterin and S100A4 protein expression levels in Caki-1 cells transfected with clusterin shRNA and control-shRNA by western blot assay; E, S100A4 mRNA expression levels in 786-O cells transfected with clusterin cDNA or control-pcDNA3.1 by RT-PCR assay; F, clusterin and S100A4 protein expression levels in 786-O cells transfected with clusterin cDNA and control-pcDNA3.1 by western blot assay; G, S100A4 protein expression levels in Caki-1/clusterin shRNA cells transfected with S100A4 cDNA or control-pcDNA3.1 by western blot assay; H, clusterin S100A4 protein expression levels in 786-O/clusterin cDNA cells transfected with S100A4 siRNA or control siRNA by western blot assay; I, S100A4 mRNA expression levels in 786-O/clusterin cDNA cells transfected with S100A4 siRNA or control siRNA by RT-PCR assay.
We next conducted loss of clusterin function experiments to determine its contribution to Caki-1 cell growth. Anti-clusterin siRNA specific constructs were used to achieve knockdown; a non-targeting construct was utilized as control. Marked clusterin knockdown was noted 48 h after transfection by RT-PCR assay (Fig. 2A) and western blot assay (Fig. 2B). MTT assays demonstrated significant (
We next examined whether stable shRNA expression decreases cell growth. Colony formation assay showed that Caki-1/clusterin shRNA cells yielded a significantly few number of colonies relative to control cell lines (Fig. 2D).
We next examined whether stable shRNA expression decreases cell invasion. The Matrigel invasion assay revealed that clusterin depletion significantly suppressed the invasiveness of Caki-1 cells (Fig. 2E). The average count of cells that crossed a matrigel-coated membrane in five high power fields was 57.0
To determine if S100A4 downregulation is required for Caki-1/clusterin shRNA induced invasion and growth inhibition in Caki-1 cells, the Caki-1/clusterin shRNA were transfected with a control pCMV or pCMV-S100A4 for 48 h. The up-regulation of S100A4 in Caki-1/clusterin shRNA cells (Fig. 1G) reverted the growth (Fig. 2C), number of colonies (Fig. 2D) and invasion (Fig. 2E) in Caki-1 cells. Thus these in vitro studies indicate that targeting clusterin reduced growth and prometastatic activities in Caki-1 cells by downregulation of S100A4.
Effect of clusterin silencing on growth and invasion in Caki-1 cells in vitro. A, Caki-1 cells were transfected with clusterin shRNA or control shRNA for 24 h, clusterin mRNA expression was detected by RT-PCR assay; B, Caki-1 cells were transfected with clusterin shRNA or control shRNA for 48 h, clusterin protein expression was detected by western blot assay; C, Cell viability was detected by MTT assay after the Caki-1 cells were transfected with clusterin shRNA or/and S100A4 cDNA for 48 h; D, Colony formation assay; E, Matrigel invasion assay.
786-O cells were stably transfected with a control pcDNA3.1 or pcDNA3.1/clusterin cDNA. Western blotting confirmed clusterin overexpression in the 786-O cell lines (Fig. 1F). We then verified their increased migratory capacity in Transwell Migration Boyden chambers. Indeed, 786-O cells transfected with pcDNA3.1/clusterin cDNA demonstrated a
Next, we examined the effect of clusterin overexpression on the capacity of 786-O cells to form anchorage-independent colonies in soft agar. As shown in Fig. 3B, 786-O cells overexpressing clusterin yielded more numerous colonies than pcDNA3.1-transfected control cells.
To determine if S100A4 is required for clusterin-enhanced invasion and colony formation, control and clusterin-overexpressing 786-O cells were transfected with a control siRNA or siRNA-targeting S100A4 prior to seeding in Transwell insert or soft agar. Knockdown of S100A4 effectively reduced S100A expression (Fig. 1H–I) and reversed clusterin-enhanced invasion (Fig. 3A) and colony formation (Fig. 3B). Control siRNA did not have significant effect on clusterin-induced invasion and colony formation (data not shown). Thus these in vitro studies indicate that clusterin enhances growth and prometastatic activities in 786-O cells by upregulation of S100A4.
Effect of clusterin overexpression on growth and invasion in 786-O cells in vitro. A. Transwell assay for cell invasion potential was performed. B. Soft agar colony formation assay was performed.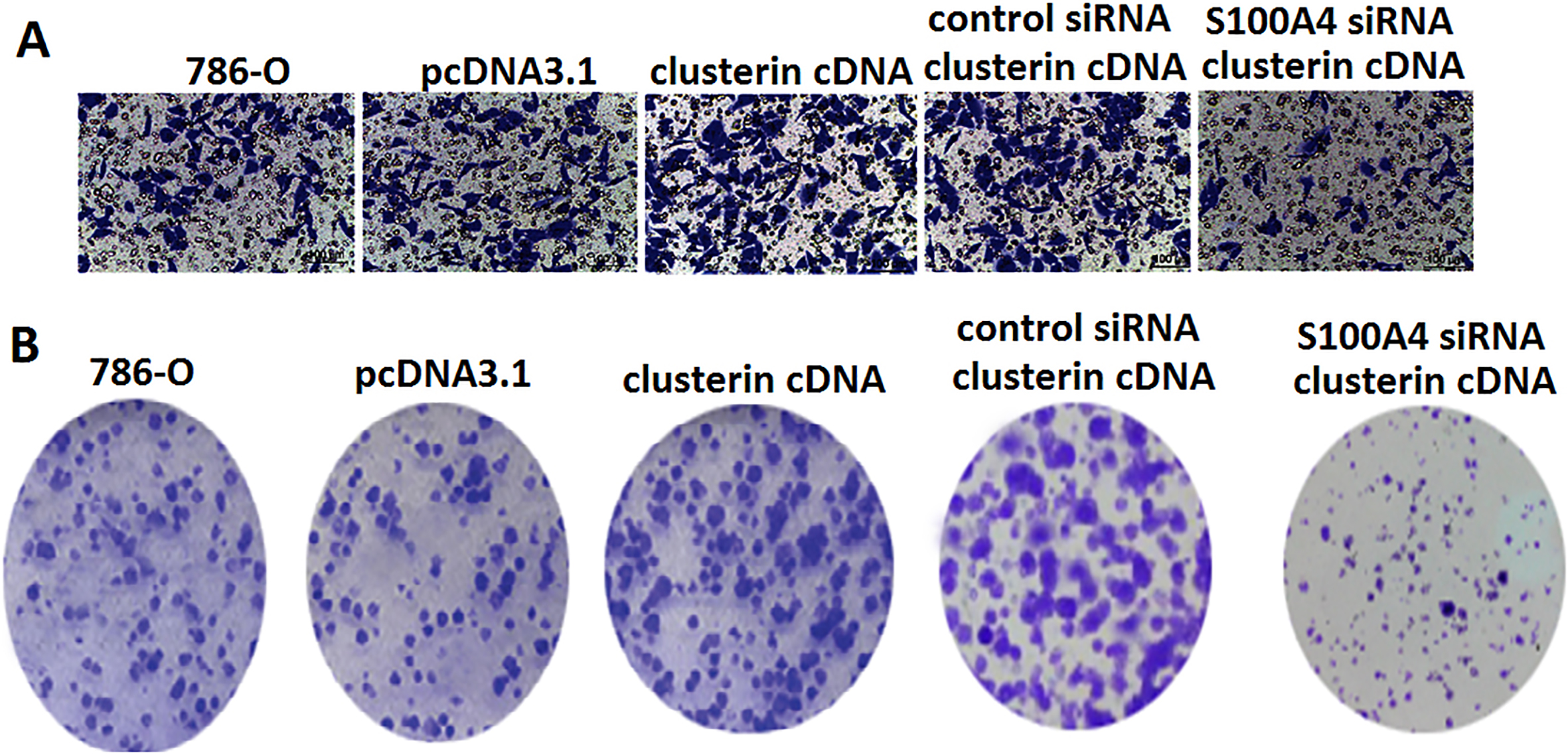
Subconfluent Caki-1, 786-O, Caki-1/clusterin shRNA, 786-O/clusterin cDNA, 786-O/pcDNA3.1 and Caki-1/pLKO.1 cells were harvested by trypsinization, and resuspended in RPMI 1640. Only single cell suspensions with
Targeting clusterin decreases tumor growth in vivo. A and B Established 786-O, 786-O/clusterin cDNA, 786-O/pcDNA3.1, Caki-1, Caki-1/pLKO.1 and Caki-1/clusterin shRNA tumour xenografts in athymic male nude mice for a total of 6 weeks. Tumour volume in Caki-1/clusterin shRNA mice was significantly smaller than that in Caki-1 and Caki-1/pLKO.1 groups (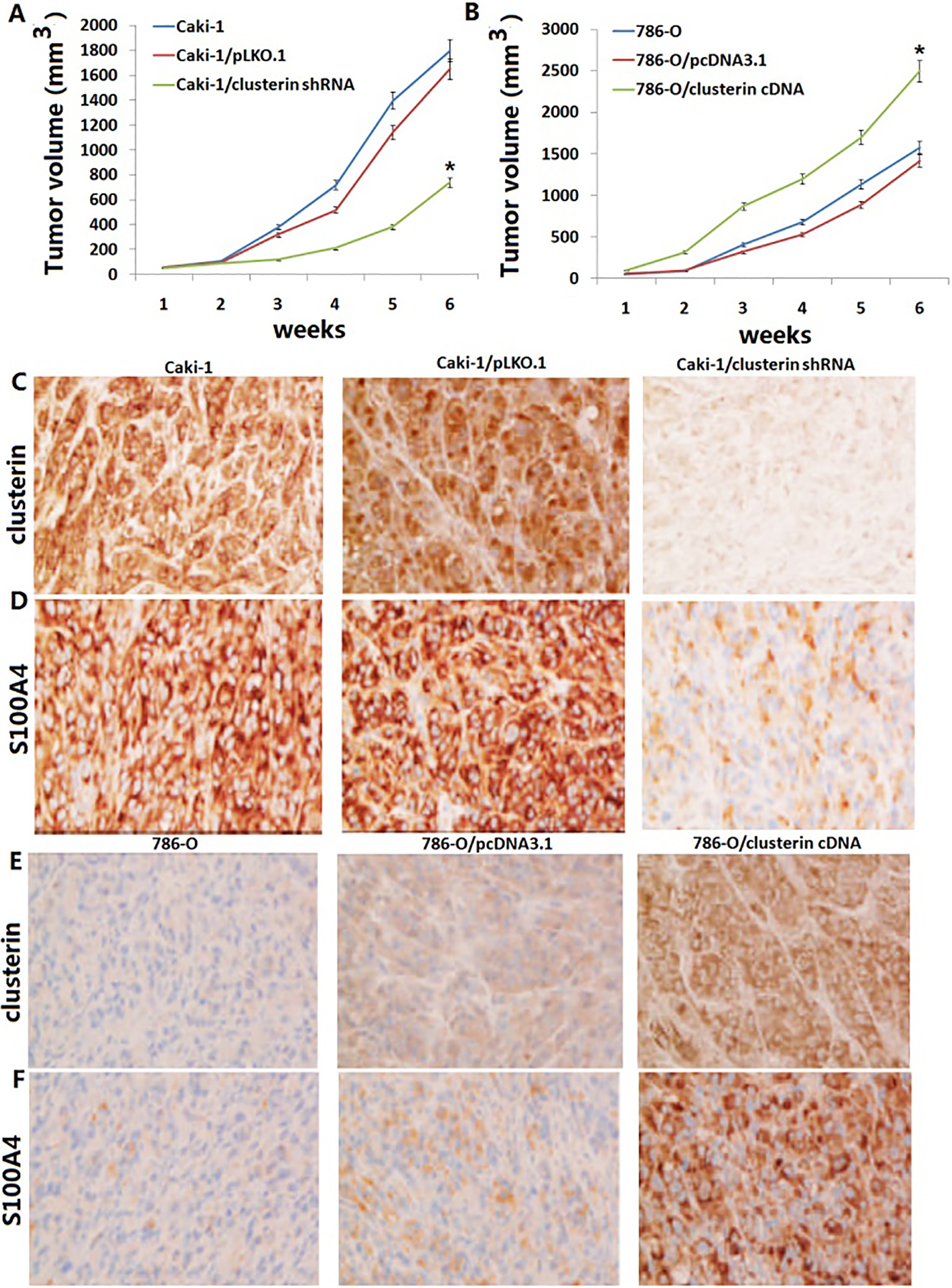
Clusterin is increased in human carcinoma and is associated with increased migratory capacity of carcinoma cells of the renal cell carcinoma [11, 12], nasopharyngeal carcinoma [21] and hepatocellular carcinoma [22]. Furthermore, targeting clusterin inhibited invasion and metastasis of cells in lung adenocarcinoma [23], breast cancer [24] and ovarian cancer [25]. In addition, clusterin could promote cell survival in hepatocellular carcinoma cells [26], and targeting clusterin could inhibit cell survival and tumor growth in breast cancer [27].
Elevated clusterin expression leads to more malignant and aggressive phenotypes of tumor cells and therefore is closely associated with poor outcome of human cancer patients, implying that targeting of clusterin expression or activities may provide a novel strategy to combat metastatic cancer, improve prognosis and enhance survival of cancer patients. Since an antisense oligonucleotide technology (ASO) that inhibits CLU (Custirsen, OGX-011) is currently in phase III clinical trials for castration-resistant prostate cancer patients, and in phase II clinical trials for lung and breast cancer patients [28], we wished to investigate the roles played by clusterin in RCC cell line.
In the present study, we found that the human RCC cell line 786-O express very low endogenous levels of clusterin mRNA and protein. Thus, 786-O cells is a suitable model to study the effects of clusterin overexpression on cell invasion and survival. We show here that adenoviral-mediated expression of human clusterin enhance in vitro invasiveness, cell growth and promote cell colony formation in 786-O cells. However, targeting clusterin in Caki-1 cells, which expressed rich endogenous levels of clusterin mRNA and protein, inhibit in vitro cell invasiveness, growth and colony formation. However, the cellular mechanisms mediating this effect in cell invasion and cell growth are not fully understood.
S100A4 has been shown to be overexpressed during prostate cancer progression in humans and in transgenic adenocarcinoma of the mouse prostate [29]. The knockdown of S100A4 expression is found to inhibit prostate cancer cell proliferation, migration, and invasion [30]. Further, it is reported that S100A4 protein promotes prostate cancer cell invasion, and malignant phenotypes partially through the transcriptional activation of MMP-9 [31].
Here, we confirmed in vitro that clusterin induces S100A4 gene activity and increases protein content in human 786-O cells. Tatgeting S100A4 using siRNA reversed S100A4 expression and decreased clusterin-induced invasion and growth in 786-O cells. However, in Caki-1 cells, targeting clusterin reduced S100A4 gene expression. Restoration of S100A4 expression by pCMV-S100A4 transfection reversed clusterin siRNA-induced invasion and growth in Caki-1 cells. We therefore suggested that clusterin regulated growth and invasion of clear cell renal carcinoma cell in vitro by regulation of S100A4 expression, and S100A4 is a clusterin target molecule in vitro in human RCC cells.
It has previously reported that plasmids-carrying clusterin shRNA also significantly decreased the formation of liver metastases of CRC cell xenografts in mice [32]. Thus, application of RNAi-based therapeutics for the knockdown of clusterin expression in tumor cells might be a practical approach to effectively and efficiently suppress cancer metastasis and prolong the disease-free survival of cancer patients. We employed stable Caki-1/clusterin shRNA and 786-O/clusterin cDNA transfectants to investigate the long-term effects of clusterin in vivo. When injected subcutaneously in nude mice, all xenograft tumours derived from Caki-1/clusterin shRNA transfectants contained reduced or almost undetectable levels of S100A4 protein when compared with the Caki-1/ pLKO.1 tumour tissues. In addition, caki-1/clusterin shRNA transfectants showed a markedly reduced tumour size of xenografts. However, all xenograft tumours derived from 786-O/clusterin cDNA transfectants contained increased levels of S100A4 protein when compared with the 786-O/pcDNA3.1 tumour tissues. Furthermore, 786-O/clusterin cDNA transfectants showed a markedly increased tumour size of xenografts. This demonstrated for the first time that S100A4 is a clusterin target molecule in vivo in human RCC cells.
In summary, clusterin clearly promoted in vitro and in vivo tumour invasion and growth, and targeting clusterin inhibited these effect in RCC cells. The in vitro and in vivo regulation of S100A4 identifies a new and potentially clinically relevant property of clusterin in human RCC. These findings provide the rationale for future oncostatic strategies aimed at suppressing clusterin-mediated signal transduction pathways as a novel therapeutic approach in human RCC.
Footnotes
Conflict of interest
No.
