Abstract
Neoadjuvant chemotherapy (NCT) is the standard treatment for locally advanced breast cancer (LABC). Pathological complete response (pCR) is commonly used as a valid predictor of NCT long-term outcomes. Blood-based tumor biomarkers have the potential to predict response to NCT at early stage non-invasively. We believed plasma CCL5 could be a potential marker to predict NCT of LABC. Its efficiency and possible mechanism was studied in this work. Human Cytokine Antibody Microarray was applied to screen different cytokine concentration in plasma between low histological regression (Low-R) and high histological regression (High-R) patients. LABC patients were divided into two groups according to pathological reactivity. The concentration of plasma CCL5 in different groups was determined by ELISA analysis. CCK8 assay was performed to analyze epirubicin susceptibility of breast cancer cells. Transwell assay was performed to determine the effect of CCL5 on breast cancer cells’ migration and invasion. qRT-PCR and western blot were used to verify the EMT (epithelial-mesenchymal transition) markers in CCL5-treated and epirubicin-treated breast cancer cells. The concentration of plasma CCL5 of Low-R group was higher than High-R group before NCT. The plasma levels of CCL5 were significantly reduced after NCT in the group of high histological regression (High-R). Epirubicin susceptibility decreased in the breast cancer cells treated by recombinant CCL5. Migration and invasion were significantly enhanced in breast cancer cells treated by recombinant CCL5. E-cadherin expression was decreased whereas vimentin increased significantly in CCL5-treated breast cancer cells. The phosphorylation of ezrin in Y-567 and its downstream protein cortactin increased significantly in CCL5-treated breast cancer cells. Plasma CCL5 level could be a promised candidate to predict chemotherapy response of breast cancer. Plasma CCL5 plays an important role in EMT process of breast cancer.
Introduction
Breast cancer is the most common cause of death among women worldwide [1]. Patients present with locally-advanced breast cancer (LABC) require the administration of systemic treatment prior to surgery. In these women, neoadjuvant chemotherapy (NCT) is often associated with a reduction in tumor volume that facilitates definitive local control. Numerous studies confirmed NCT as the standard treatment for LABC patients [2]. pCR implies the absence of residual invasive or in situ disease. In NCT, many trials have demonstrated pCR is an important independent prognostic indicator for disease-free survival (DFS) and overall survival (OS) [3, 4]. However, 10–35% breast cancer patients do not benefit from this clinical approach and only a minority of patients achieves a complete response [5]. LABC patients have heavy tumor burden. Chemotherapy failure would lead to tumor progression even losing operation chance. It is vital to predict the response and evaluate the efficacy of NCT as early as possible. Several studies have been conducted to identify markers predicting pCR after NCT [5]. But so far, there is still no ideal marker for predicting response to NCT in LABC patients.
Blood-based tumor biomarkers have potential to predict response to NCT at early time due to its feasibility and repeatability [6]. On the other hand, immune response is pivotal during chemotherapy. The concentration of multiple cytokines related to immunity and inflammation [7]. As a cornerstone of breast cancer chemotherapy, anthracycline is contained in most actively described chemotherapy regimens. In the present study, the therapeutic effect of LABC patients received anthracycline-based regimen NCT were evaluated. We used the RayBiotech Human Cytokine Antibody Array to screen the different concentration of cytokines in plasma from patients with different response after NCT. Chemokine CCL5 was found to be one of the most remarkable cytokines.
CCL5 was a CC chemokine that attracts T cells and monocytes. The function of CCL5 is relevant to induce proper immune responses against tumors. On the other hand, CCL5 may facilitate metastasis formation and contribute to disease progression [8]. In breast cancer, CCL5 serum levels are higher in lymph-node-positive patients, larger tumor size, the presence of lymphovascular invasion and multifocal tumors [9]. CCL5 also plays a role in Tamoxifen resistance in breast cancer [10]. CCL5 as a potential immunotherapeutic target in triple-negative breast cancer has been reported in a number of studies [11]. However, whether and how CCL5 play an important role in predicting NCT efficacy of LABC is still unclear. The present work aims to evaluate the predictive effect of CCL5 in NCT in breast cancer and reveal the possible mechanism.
Methods
Patients
A total of 36 LABC patients from March 20, 2016 to December 20, 2017 who received NCT in Jiangsu Breast Disease Center, the Frist Affiliated Hospital with Nanjing Medical University and 12 healthy volunteers as control was enrolled in this study. All patients had core biopsy to determine pathological type [12]. All the samples (blood and tissues) were collected from First Affiliated Hospital with Nanjing Medical University after approval by Institutional Ethical Committee and written informed consent. Breast cancer patients received EC-T regimen (epirubicin 80 mg/m
Histological study
All breast cancer patients underwent surgery and were divided into two groups according to pathological reactivity. The classification of response to NCT refer to the Miller-Payne [13], and cases with G1
Plasma separation
The blood samples were collected at time of biopsy and after the last time of NCT. 3 ml blood samples were collected. The plasma samples were clarified by spinning at 2500 rpm for 10 min at 4
Cytokine antibody assay
The soluble proteins in the plasma of patients with breast cancer were measured using a biotin label-based human antibody array in accordance with the recommended protocols (Raybiotech Human Cytokine Antibody Array G series 2000; Raybiotech, Norcross GA, USA). The plasma samples were from 6 LABC patients and divided into High-R and Low-R group according to the results of pathology reports.
ELISA analysis of plasma and tissue CCL5
The concentration of plasma and tissue CCL5 were detected by enzyme-linked immunosorbent assay (ELISA). The concentration of plasma and tissue CCL5 was measured using commercially available kits (R&D System) following procedures recommended by the manufacturer. The range of sensitivity for human CCL5 was 31.3–1000 pg/ml.
Cell lines and culture
Human breast cancer cell lines (MCF-7 and ZR-75-1) were purchased from ATCC (Manassas, VA, USA) and cultured in Roswell Park Memorial Institute 1640 (RPMI-1640) supplemented with 10% fetal bovine serum (FBS), 1% penicillin, and 1% streptomycin. All cells were incubated at 37
Cytotoxicity assay
Cells (5
Cell migration and invasion assay
In vitro the cell invasion assay was conducted using a transwell chamber (8-
To carry out the migration assay, cells (3
The CCL5 levels in LABC patients and clinical characteristics
The CCL5 levels in LABC patients and clinical characteristics
Total RNA was extracted using Trizol total RNA isolation reagent (TaKaRa), and cDNA was synthesized using Primescript RT Reagent (TaKaRa) according to the manufacturer’s instructions. Specific primers from Invitrogen (Shanghai, China) were used for transcript detection. All PCR reactions were performed with SYBR Green I (Roche) for detection. Real-time quantitative PCR was performed on StepOne Plus Real-Time PCR system (Applied Biosystems, USA). The following PCR primers were used:
Vimentin forward, 5’-CAGATGCGTGAAATGGA AGA-3’ Vimentin reverse, 5’-CTCAATGTCAAGGGCCA TCT-3’ E-cadherin forward, 5’-CAGCACGTACACAGCC CTAA-3’ E-cadherin reverse, 5’-TGAGGCTTTGGATTCCT CTC-3’ Snail forward, 5’-CCTCCACGAGGTGTGACTA ACT-3’ Snail reverse, 5’-CCGACAAGTGACAGCCATTA -3’ Slug forward, 5’-CGCAATCAATGTTTACTCGA AC-3’ Slug reverse, 5’-TCTCAATCTAGCCATCAGCAA A-3’ GAPDH forward, 5’-GCTGCGAAGTGGAAACC ATC-3’ GAPDH reverse, 5’-CCTCCTTCTGCACACATT TGAA-3’
The average of three independent analyses for each gene and sample was calculated and normalized to the endogenous reference control gene GAPDH.
Western blot
Cells were harvested using the Total protein extraction kit (KeyGEN BioTECH, Nanjing, China). Protein concentration were determined using the bicinchoninic acid (BCA) protein assay kit (Pierce). Protein samples were subjected to electrophoresis on SDS-polyacrylamide gradient gels, transferred to a PVDF membrane and blocked in 5% non-fat milk in TBST (phosphorylated proteins were blocked in 5% BSA in TBST) for 2 h at room temperature. Blots were incubated with primary antibodies to the following proteins: E-cadherin (CST), Vimentin (Abcam), Slug (CST), Snail (CST), ezrin (CST), phosphorylated ezrin at Y-567 (CST), cortactin (Abcam) and GAPDH (Beyotime). GAPDH was used on the same membrane as a loading control. The signal was detected after incubation with anti-rabbit or anti-mouse IgG secondary antibody (CST) coupled to peroxidase, using ECL (Millipore). Protein expression levels were evaluated by densitometric analysis.
Statistical analysis
Results were expressed as mean
The relative concentration of different cytokines before NCT in High-R and Low-R groups after NCT. The plasma concentration of EGF, CCL5, PDGF-BB, TACE, CEACAM-1, LAP, PDGF-AA, BDNF, SAA and XEDAR were increased in low histological regression (Low-R) patients than those in high histological regression (High-R) patients before NCT. The concentration of CCL5 in Low-R group was 3.973-fold increased than that in High-R group, 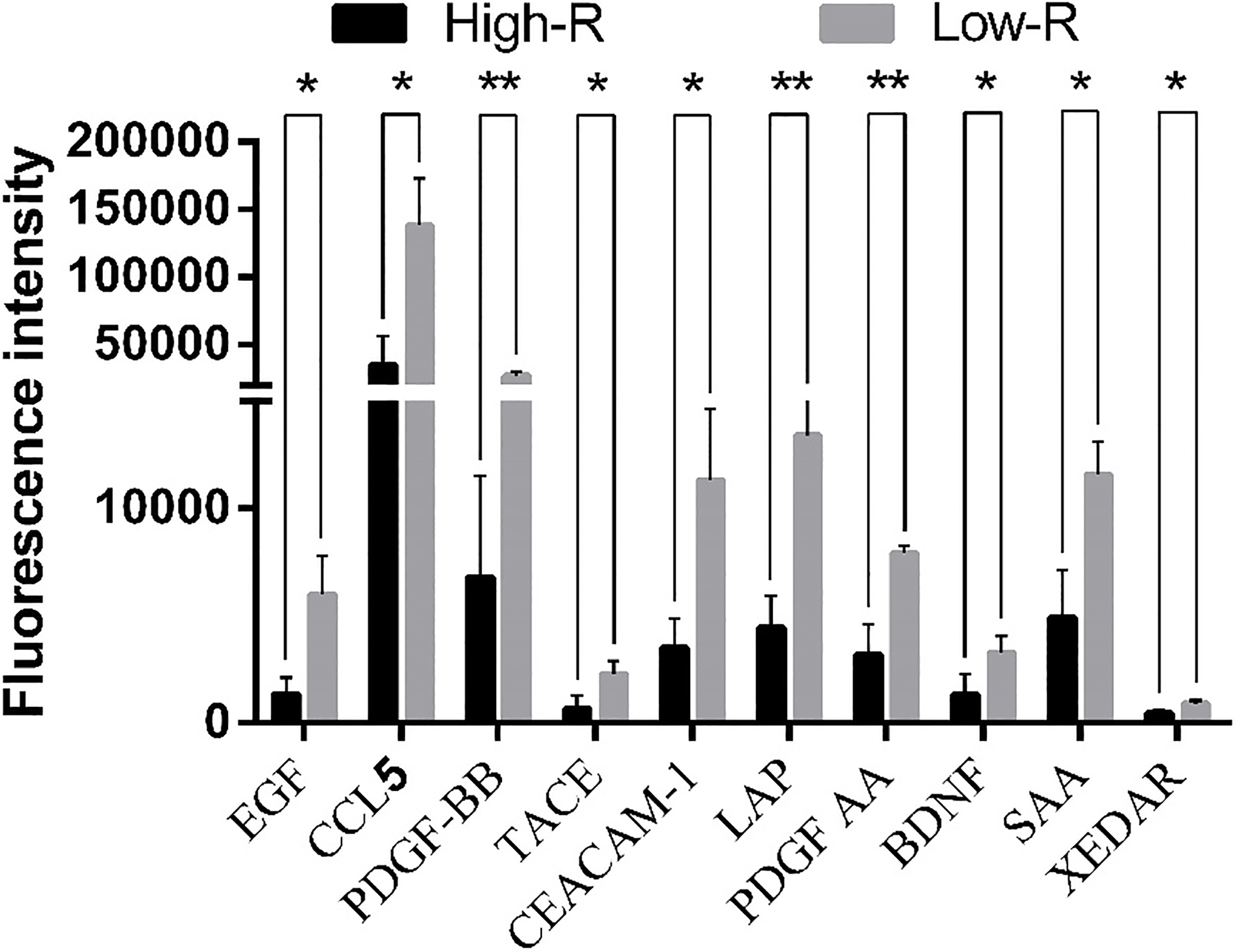
The plasma CCL5 level of healthy volunteers and breast cancer patients. (A) The concentration of plasma CCL5 in LABC patients (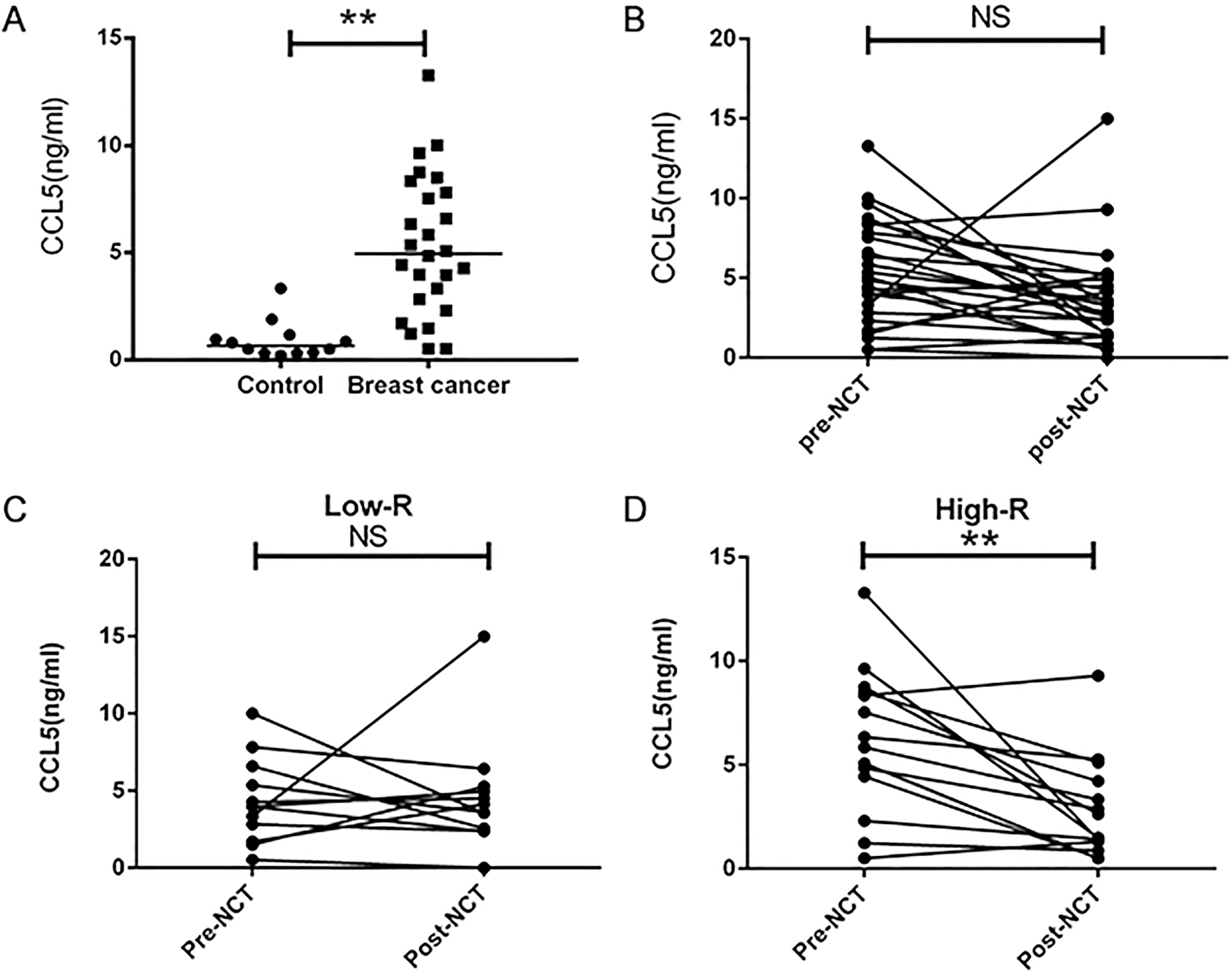
Human cytokine antibody array
The plasma cytokines array were performed in 6 patients, three of whom achieved high histological regression, and the other three developed progressive disease after NCT. Plasma concentration of EGF, CCL5, PDGF-BB, TACE, CEACAM-1, LAP, PDGF-AA, BDNF, SAA and XEDAR were higher in High-R group than those in Low-R group before NCT. The concentration of CCL5 in Low-R group was 3.973-fold increased than that in High-R group (Fig. 1).
The plasma level of CCL5 in high-R group between pre- and post-NCT
In 26 LABC patients, plasma concentration of CCL5 were significantly higher than those in the 12 healthy volunteers (control 0.94
CCL5 level of tumor tissue and plasma
The CCL5 level of the other 10 patients tumor tissue and plasma samples were compared by ELISA. The CCL5 level of tumor tissue was significant positive related with the CCL5 level of plasma (Supplementary Fig. 2).
CCL5 reduced the response to epirubicin in breast cancer cells
The efficacy of anthracycline-based chemotherapy has been proven in multiple large cohort trials and meta-analyses [15, 16]. CCK-8 assay was used to analyze the effects of epirubicin (0.1–1.6
Epirubicin susceptibility decreased in the breast cancer cells treated by recombinant CCL5. Breast cancer cells were exposed to indicated concentration of epirubicin alone (EPI) or combined with CCL5 20 ng/mL (EPI 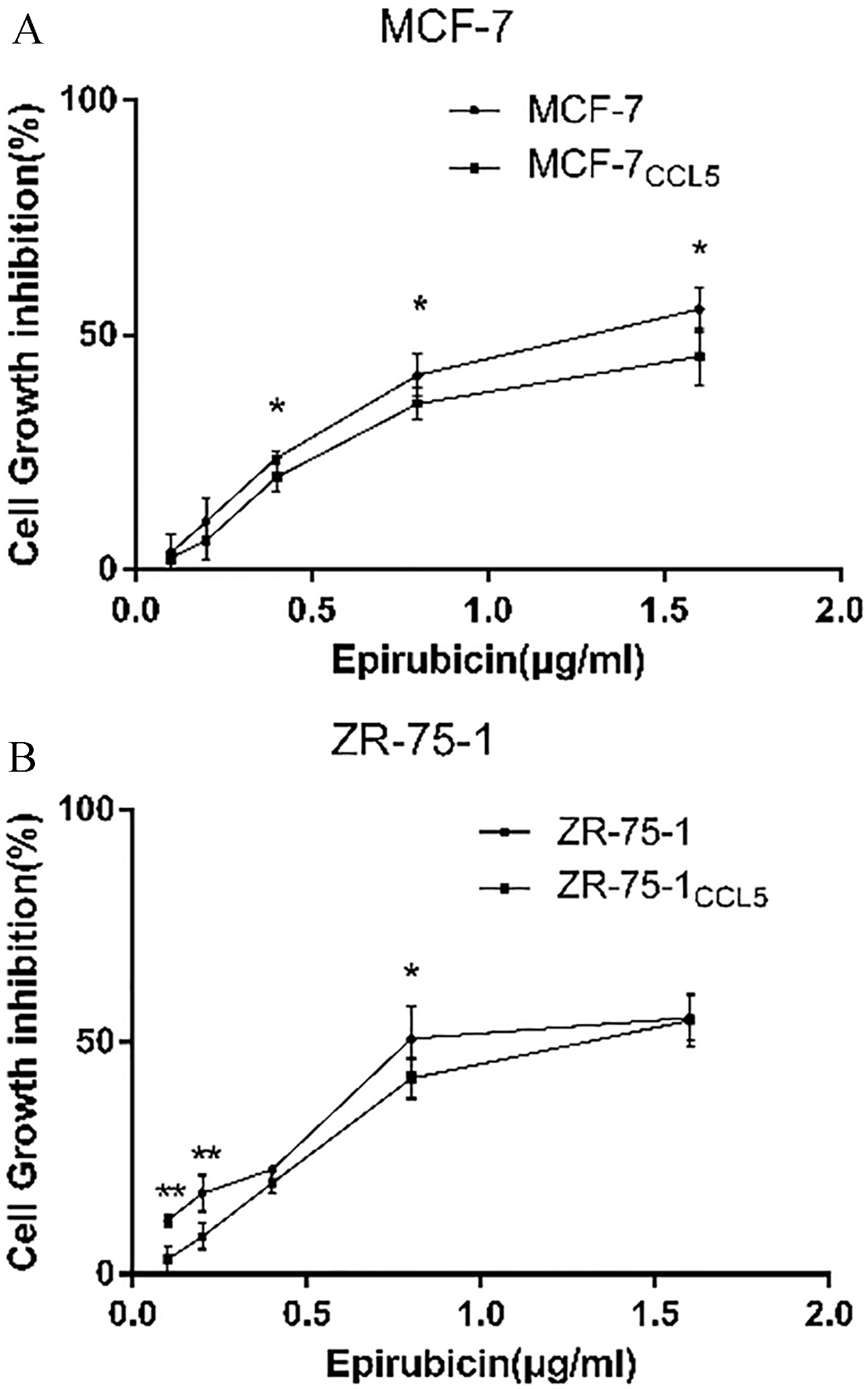
The treatment of CCL5 significantly increases the migratory and invasive potential of breast cancer cells. MCF-7 and ZR-75-1 cells treated with CCL5 were subsequently subjected to transwell filter migration assay and Matrigel invasion assay. Representative photographs (upper panel) and quantification (lower panel) are shown. Columns: average of three independent experiments, 
Treating with 20 ng/ml CCL5 for 1 week, breast cancer cells had a significantly increased ability to migrate and invade compared to the control group (Fig. 4). After the intervention of epirubicin, migration rate was too low to show differences between CCL5 treated group and the control group due to cell death.
CCL5 up-regulates EMT indicator expression
The mRNA levels of CDH1, Vimentin, Snail and Slug were verified in MCF-7, ZR-75-1. The mRNA of CDH1 was decreased in ZR-75-1
Treatment of CCL5 promotes EMT in MCF-7 and ZR-75-1 cells. (A) The treatment of CCL5 increased the expression level of Vimentin in MCF-7 and ZR-75-1, decreased the expression level of E-cadherin in ZR-75-1, but not in MCF-7, by qRT-PCR, 
In MCF-7 and ZR-75-1 cell lines, the expression levels of epithelial marker (E-cadherin) and mesenchymal marker (Vimentin) were also texted with western blot. Compare to the control, Vimentin was increased in MCF-7
In our previous studies, we discovered ezrin, based mainly on its phosphorylation state, could collaborate with cortactin to promote EMT process in breast cancer [18]. So we estimate this EMT mechanism in the CCL5 interposed epirubicin treating breast cancer cells. Ezrin did not be influenced by epirubicin or CCL5 in MCF-7 or ZR-75-1 cells. The phosphorylation of ezrin (Y567) increased significantly in MCF-7
Treatment of CCL5 promotes the phosphorylation of ezrin at position Y-567 and the expression of cortactin in MCF-7 and ZR-75-1 cells. The expression levels of ezrin did not change in MCF-7 and ZR-75-1 after treatment of CCL5 and intervention of epirubicin by western blot. The phosphorylation of ezrin at position Y-567 and the expression of cortactin were increased in MCF-7 and ZR-75-1 intervened by CCL5 or epirubicin. When the two factors work together, the phosphorylation of ezrin at position Y-567 and the expression of cortactin were significantly higher than any other group. (A) MCF-7 and MCF-7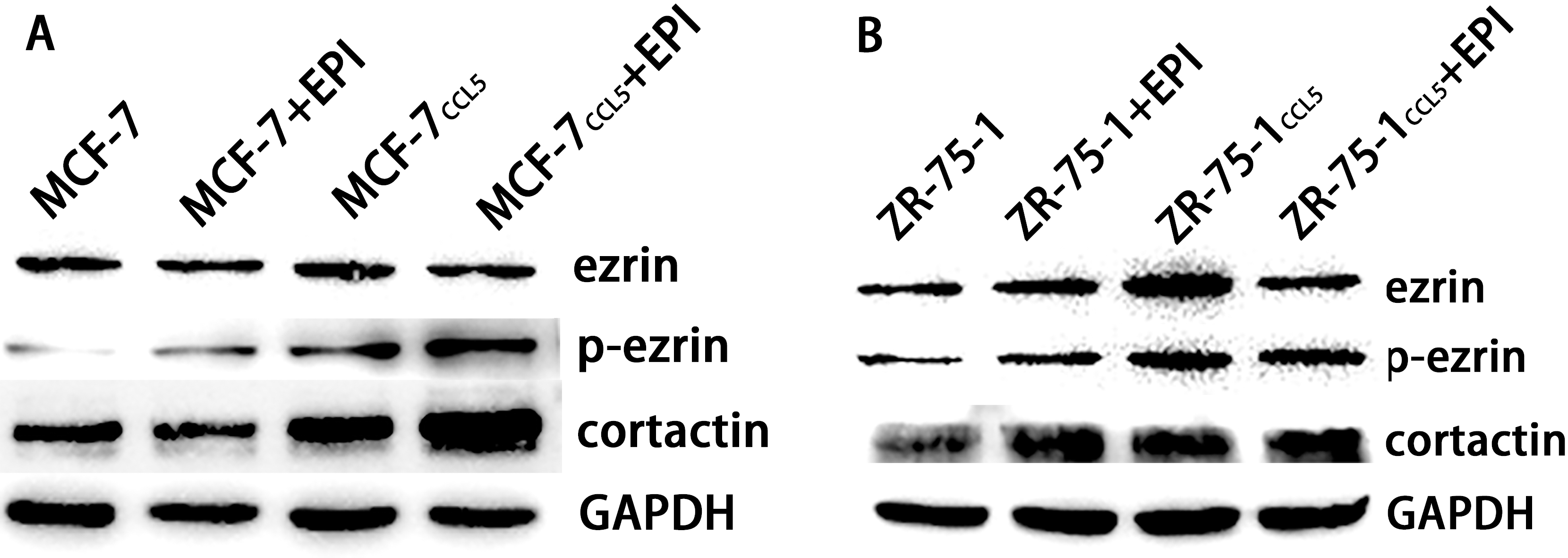
NCT curative prediction after 2 or 3 cycles of initial NCT regimens greatly influences the decision-making of the follow treatment. At present, magnetic resonance imaging (MRI) is the most effective techniques to evaluate the residual cancer burden, but changes in lesion on MRI usually are not detectible until 6 weeks following chemotherapy [19]. Core needle biopsy and pathological detection is timely effective and accurate, but frequently invasive examination is not appropriate for clinical practice.
It is previously believed that the chemotherapy of breast cancer on the basis of anthracyclines changes the immunoreaction profoundly. Comparing the cytokines in the plasma between High-R and Low-R patients after NCT may discover novel effective indicators in plasma to predict the NCT response. In increased-cytokine panel, EGF was proved to promote the progression of breast cancer [20, 21]. It implied that the increased CCL5 level in NCT LABC patients may also predict poor response. CCL5 may act in a paracrine or autocrine fashion on CCR5-positive tumor cells to promote their progression [8]. Therefore, it was reasonable that plasma concentration of CCL5 might be correlating with the NCT efficacy in LABC. We compared the plasma level of CCL5 in patients with different pathological response to NCT. The concentration of plasma CCL5 has no difference before or after NCT. However, a significant reduction was discovered in High-R group, but not in Low-R group. It is a powerful evidence to demonstrate CCL5 is an adverse factor in LABC patients received NCT. Inflammation is key mechanism in cancer pathogenesis, contributing to multiple hallmark capabilities by supplying bioactive molecules to the tumor microenvironment [22]. Recent reports suggested that the inflammatory reaction in breast cancer affects tumor growth and progression. CCL5 plays an active role in recruiting a variety of leukocytes into inflammatory sites, which increased in breast cancer tissue [23]. Interestingly, the concentration of CCL5 decreased in Her2+ patients after the NCT, but not in Her2- group. It has been shown that CCL5 signaling is preferentially active in the Her2 subtypes. The immune reaction between Her2+ and Her2- LABC patients received NCT deserve discussion in a larger sample size study.
It is important to reveal how CCL5 could induce epirubicin-resistance in breast cancer. There was significantly positive correlation between the CCL5 level of tumor tissue and plasma, so the breast cancer cells were treated by recombinant CCL5 in vitro. Clinic evidence suggest that hormone receptor positive patients have low pCR rate [24, 25]. We used ER positive breast cancer cell lines MCF-7 and ZR-75-1 to perform the mechanism study. The susceptibility to epirubicin was declined in the cells treated by CCL5 for one week in vitro. In addition, CCL5 was proven to promote the migration of breast cancer cells. Further more, the classic EMT markers changed significantly after the treatment of CCL5 in MCF-7 and ZR-75-1. The down-regulation of the epithelial marker and the up-regulation of the mesenchymal marker were founded in ZR-75-1 cell. But in MCF-7, only the up-regulation of the mesenchymal marker was founded. The EMT-like changes were more pronounced in ZR-75-1 treated by CCL5. CCL5 activity is mainly mediated through its binding to CCR5. CCR5 is high expression in breast cancer cell with Her2+ (ZR-75-1) [26]. Studies have demonstrated several opposite effects of chemotherapy, including contributing to tumor metastasis by EMT [17, 27]. In this study, MCF-7 and ZR-75-1 cell lines underwent EMT-like change after the intervention of epirubicin. Base on this, if there were constant stimulation of CCL5 in tumor microenvironment, more obvious EMT-like phenotype was observed in MCF-7
Based on the results, high concentration of plasma CCL5 may promote EMT of breast cancer cells. Plasma CCL5 could be a promised candidate to predict chemotherapy response in NCT of LABC.
Footnotes
Acknowledgments
This study was funded by the Natural Science Foundation of China (81202077, 81572607, 81172502, 81272916, 81572595, 81472703) “Qinglan” and “Six talent peaks” projects to Tiansong Xia, a project Funded by the Priority Academic Program Development of Jiangsu higher Education Institutions (PAPD).
Conflict of interest
The authors declare that they have no competing interests.
Abbreviations
Supplementary data
The pre- to post-NCT changes in the concentration of CCL5 in Her2+ and Her2- patients. The plasma levels of CCL5 were significantly reduced following NCT in Her2+ group, but not in the Her2- group. (NS no significance, 
Relationship between CCL5 level in tumor tissue and plasma. A significant correlations were observed between CCL5 level in tumor tissue and plasma. (Person correlation coefficient 
