Abstract
Long non-coding RNAs (lncRNA) constitute a significant percentage of RNAs with no translation to proteins. Their participation in fundamental aspects of cell physiology as well as their dysregulation in a number of pathologic conditions such as cancer have been documented. Among lncRNAs is actin filament associated protein 1 antisense RNA1 (AFAP1-AS1) whose elevated expression levels have been demonstrated in different cancers. In the in the present study we evaluated expression levels of AFAP1-AS1 and its antisense protein coding gene AFAP1 in breast cancer samples compare with adjacent non-cancerous tissues (ANCTs) as well as breast cancer cell lines with special focus on the assessment of the association between their transcript levels and patients’ clinicopathological data. AFAP1-AS1 has shown significant up-regulation in both MDA-MB-231 and MCF-7 compared with control sample. AFAP1-AS1 has been shown to be expressed in all of tumor tissues but 76% (39 out of 51) ANCTs. AFAP1 expression was not significantly different between tumor samples and ANCTs. AFAP1-AS1 has been demonstrated to be significantly up-regulated in tumor tissues compared with ANCTs (fold change
Introduction
Long non-coding RNAs (lncRNA) constitute a significant proportion of RNAs transcribed from human genome but not translated to proteins [1]. They exert functional roles in almost every aspect of cell physiology including DNA repair [2], cell cycle control and cell proliferation and differentiation [3]. Dysregulation of these prominent functions have been implicated in tumorigenesis process. Consequently, researchers have focused on expression analysis of lncRNAs in cancers in order to find biomarkers for cancer detection or prognosis in addition to therapeutic targets [4, 5, 6, 7]. Among lncRNAs is actin filament associated protein 1 antisense RNA1 (AFAP1-AS1) whose elevated expression levels have been detected in pancreatic ductal adenocarcinoma, esophageal adenocarcinoma, lung cancer and colorectal carcinoma which have been associated with poor prognosis [8]. AFAP1-AS1 is coded from the antisense strand of protein-coding gene AFAP1. AFAP1-AS1 exon 2 overlaps with exons 14, 15, and 16 of the mentioned protein coding gene. AFAP1 codes for a motor fiber-related protein, which constitutes a network for connecting to other proteins, such as Proto-Oncogene Tyrosine-Protein Kinase Src (SRC) and protein kinase C (PKC). Its interaction with these proteins alters actin filament integrity and consequently participate in processes such as cytophagy, cell movement, tumor invasion, and metastasis [9]. Consequently, this protein coding gene as well as its antisense non-coding partner might participate in the pathogenesis of breast cancer. Consequently, in the present study we aimed at evaluation of expression levels of AFAP1 and AFAP1-AS1 in breast cancer patients as well as cell lines with focus on the assessment of the association between their transcript levels and patients’ clinicopathological data.
Material and methods
Cell culture
The human breast cancer cell lines MDA-MB-231 and MCF-7 were acquired from Pasteur Institute of Iran and cultured in RPMI-1640 medium (Sigma Aldrich, St. Louis, MO, USA) supplemented with 10% fetal bovine serum, 100 U/ml penicillin, and 100
Patient samples
In the current study, 51 patients diagnosed with invasive ductal carcinoma of breast were included. Corresponding adjacent non-cancerous tissues (ANCTs) and breast cancer tissues were excised and then snap-frozen in liquid nitrogen instantly. No anticancer treatments have been administered before surgery. Written formal consent was also obtained from all the patients. The study protocol was approved by the ethical committee of Shahid Beheshti University of Medical Sciences (IR.SBMU.MSP.REC.1395.586).
Relative expression of AFAP1 and AFAP1-AS1 in breast cancer cell lines.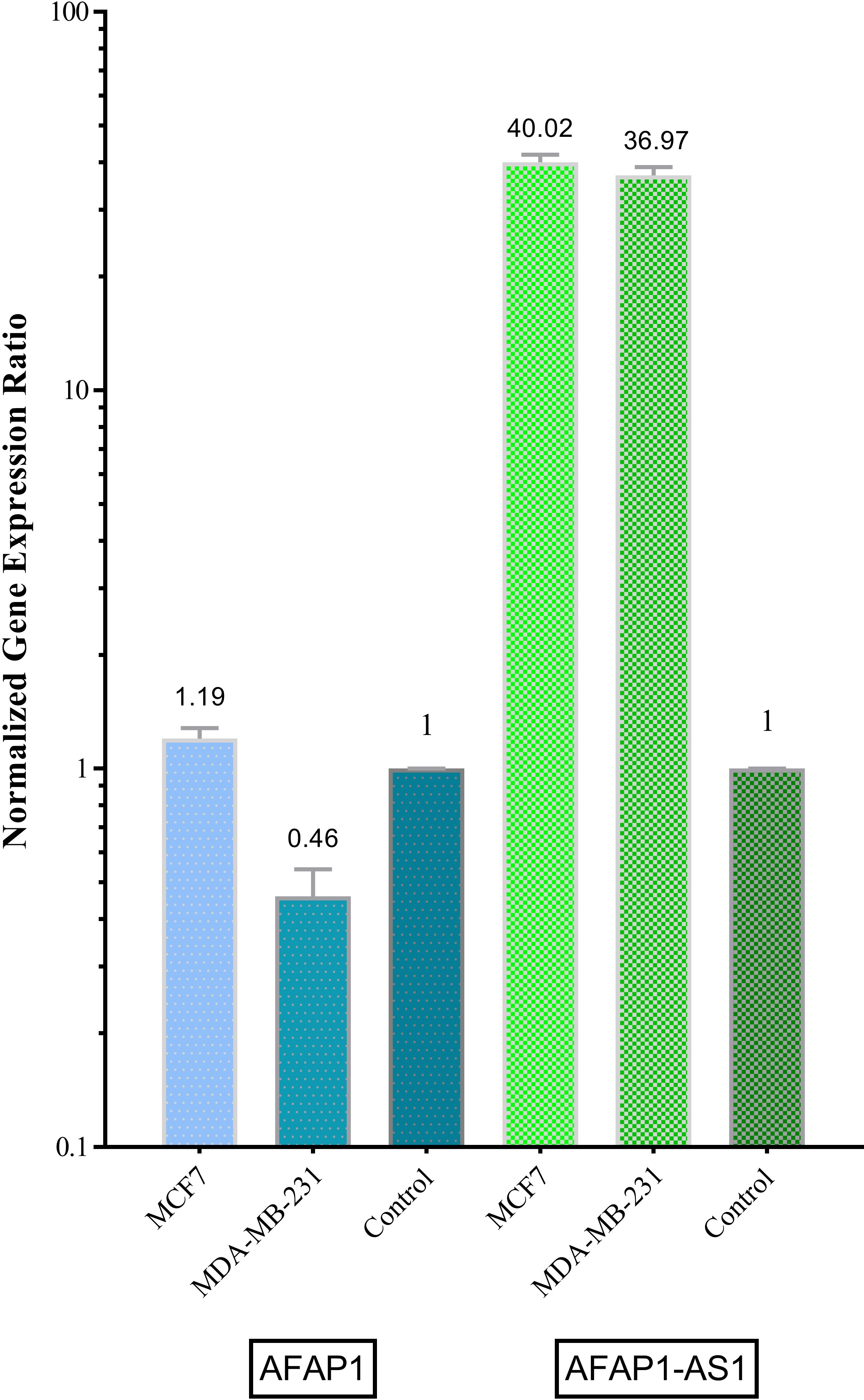
Total RNAs from tissues or cells were extracted using the AccuZol™ total RNA extraction solution (Bioneer, Korea) according to the manufacturer’s instructions. PrimeScript RT reagent kit (Takara Bio, Ohtsu, Japan) was used to synthesize cDNA from RNA. The mRNA expression levels of AFAP1-AS1 and AFAP1 were quantified by using SYBR
Demographic and clinical data of patients
Demographic and clinical data of patients
AFAP1 expression and its associations with patients’ clinical and demographic data
ER, PR, Her2/neu and Ki-67 status were extracted from the patients’ medical record. All experiments had been carried out by immunohistochemistry (IHC) assay. For ER and PR staining of
Statistical analysis
All statistical analyses were performed by using SPSS 22.0 software (IBM, Chicago, IL, USA). AFAP1 and AFAP1-AS expression difference between breast cancer tissues and ANCTs was analyzed using paired samples’
Relative expression of AFAP1 and AFAP1-AS1 in tumor tissues and adjacent non-cancerous tissues (ANCTs).
AFAP1-AS1 expression and its associations with patients’ clinical and demographic data
Expression of AFAP1 and AFAP1-AS1 in breast cancer cell lines
Both genes have been shown to be expressed in MCF-7 and MDA-MB-231 cell lines. AFAP1-AS1 has shown significant up-regulation in both MDA-MB-231 and MCF-7 compared with control sample (
Correlation between AFAP1 and AFAP1-AS1 expression level in tumoral tissues.
Correlation between AFAP1 and AFAP1-AS1 expression level in adjacent non-cancerous tissues.
A total of 51 female breast cancer patients with the mean age of 50.78
Expression of AFAP1 and AFAP1-AS1 in patients’ samples
AFAP1 expression has been detected in all tumor samples and ANCTs. AFAP1-AS1 has been shown to be expressed in all of tumor tissues but 76% (39 out of 51) ANCTs. AFAP1 expression was not significantly different between tumor samples and ANCTs (
Correlation between AFAP1 and AFAP1-AS1 expression levels
With the purpose of assessment of the correlation between the expressions of these two genes, the relative expression of these genes was compared in each set of samples. No significant correlation has been detected between the levels of transcripts in tumor tissues (R
Discussion
Previous studies have demonstrated the role of AFAP1-AS in the pathogenesis of various cancers. Knock-in and knock-down studies have confirmed its promoting role in induction of cell proliferation, migration, and invasion. In addition, there are preliminary evidences originated from studies in lung cancer, nasopharyngeal carcinoma, and hepatocellular carcinoma which demonstrate the regulatory role of AFAP1-AS1 on the expression of AFAP1 gene, leading to alteration of Rho/Rac signaling pathway and changing cytoskeleton remodeling, thus enhancing tumor cell invasion and metastasis [9]. However, the role of this lncRNA has not been evaluated in breast cancer previously. In the present study, we evaluated expression of AFAP1-AS and its correlation with expression of its anti-sense protein coding gene in clinical samples of breast cancer as well as breast cancer cell lines. We detected significant up-regulation of AFAP1-AS1 in breast cancer samples compared with ANCTs. The mechanism by which AFAP1-AS1 exert its oncogenic roles need to be elucidated. Rho/Rac signaling pathway as well as some apoptosis-related proteins such as BCL-2 and Bax are among AFAP1-AS1 target genes [9] whose dysregulation has been detected in breast cancer [10, 11]. Consequently, they might be involved in AFAP1-AS1 mediated tumorigenic process in breast cancer. Although we could not find any significant association between the levels of transcripts and clinicopathological characteristics of patients, AFAP1-AS1 has been down-regulated in all of Ki-67 negative samples. Considering the role of Ki-67 as a nuclear antigen which is expressed from G
Although preliminary researches have implied the regulatory role of AFAP1-AS1 on AFAP1 [9], we could not find any associations between the levels of these transcripts either in tumoral tissues or in ANCTs. Such discrepancy between our results and the previous in vitro studies might be due to the complex network of interactions between these genes and other partners especially in the context of a pathological condition such as tumor which preclude identification of a simple linear correlation. Consequently, the results of the current study do not rule out the possible regulatory function of AFAP1-AS1 on AFAP1 expression. However, consistent with our observations, Wu et al. have shown that AFAP1-AS1 had restricted influence on AFAP1 expression, but alternatively exerts its role in a virtually AFAP1-independent manner [14]. On the other hand, Han et al. have shown that AFAP1-AS1 knock-down could promote the expression level of AFAP1 protein with no effect on the level of AFAP1 transcripts [8]. So to better elucidate the correlation between AFAP1 and AFAP1-AS1 expressions, AFAP1 protein expression should be assessed in patients’ samples.
In brief, we have detected significant elevated expression of AFAP1-AS1 in breast cancer cell lines and patients’ samples compared with normal control and ANCTs respectively which potentiates it as a putative oncogene in breast cancer. Future studies are needed to elaborate its exact role in this pathogenic condition.
Footnotes
Acknowledgments
This article has been extracted from the thesis written by Ali Dianatpour in School of Medicine, Shahid Beheshti University of Medical Sciences (Registration No: 258M).
