Abstract
BACKGROUND AND OBJECTIVE:
Breast cancer is the leading cause of death in women worldwide. There are evidences that human cytomegalovirus (HCMV) infection is associated with several malignant tumors. This study aims to investigate the infection of human cytomegalovirus (HCMV) in a large sample of breast cancer patients, and conduct a correlation analysis of clinical and pathological factors, to provide evidence for whether HCMV infection is associated with breast cancer development, progression and metastasis.
MATERIALS AND METHODS:
A total of 438 tissue samples (including breast cancer tissue, paracancerous tissue and sentinel lymph node [SLN] tissue) obtained from 146 patients who were diagnosed with breast cancer and intraoperatively underwent unilateral axillary SLN biopsy at the Affiliated Hospital of Qingdao University from June 2013 to June 2014 were included into this study. These tissue samples were divided into two groups: SLN positive group and SLN negative group. The clinical information is collated and numbered. Normal breast tissues of 40 patients with cyclomastopathy were taken as controls. The expressions of HCMV immediate-early (IE) and late antigen (LA) proteins of breast cancer tissues, paracancerous tissues, SLN tissues and normal breast tissues were analyzed by immunohistochemistry, and HCMV infection of the samples was graded according to the percentage of the positive cells, and HCMV IE2 mRNA expression was detected by reverse transcription polymerase chain reaction. The clinical data were collated and statistically analyzed.
RESULTS:
HCMV IE and LA proteins were highly expressed in all breast cancer tissue samples. IE proteins were detected in 47.9% (70/146) of paracancerous tissue samples, and LA proteins were detected in 53.4% (78/146) of paracancerous tissue samples. IE and LA proteins were expressed in 92.6% of metastatic SLN samples (62/68) and in most of the tumor cells. Inflammatory cells in 60% (42/70) of non-metastatic samples were positive for HCMV. HCMV DNA was present in 100% of breast cancer tissue samples, 50% of paracancerous tissue samples, and 91% of metastatic SLN samples; but this was not present in HCMV negative and non-metastatic SLN samples. Differences in HCMV infection, and estrogen receptor-
CONCLUSION:
HCMV infection is common in breast cancer tissues, paracancerous tissues and SLN tissues. The severity of HCMV infection varied markedly with tissue type. HCMV infection might be associated with metastasis and invasion of breast cancer. The expression of HCMV IE2 was associated to breast cancer and lymph node metastasis. The expression level of estrogen receptor-
Introduction
Breast cancer is the most common malignancy in women worldwide, and has a very high mortality rate [1]. Presently known risk factors include age, high-dose estrogen use, and family history. However, 50–80% of patients do not have these known risk factors [2]. Hence, it is very important to explore these unknown risk factors.
Recent studies have revealed that breast cancer is associated with viral infections such as Epstein-Barr virus (EBV) [3], murine mammary tumor virus (MMTV) [4], human papillomavirus (HPV) [5],and the recently studied human cytomegalovirus(HCMV) [6]. Breast cancer patients who are positive for HCMV and human herpes virus-8 (HHV-8) are often associated with short disease-free survival and low overall survival rate.
As a member of the
HCMV protein and nucleic acid have been identified in a series of malignant tumors such as breast cancer, colon cancer, prostate cancer, glioblastoma in the salivary gland, mucoepidermoid carcinoma, medulloblastoma and rhabdomyosarcoma [9, 10, 11]. Until recently, HCMV was known to be involved in encoding approximately 180 proteins; and these proteins exhibit a variety of biological activities that interfere with the physiological functions of infected cells [12]. A latest study has revealed that this figure exceeds 750 [13]. This suggests that HCMV may be much more complex than previously thought. Most of these proteins are not required for viral replication, but may be associated with a variety of pathological process [12]. In Taher’s study HCMV IE and LA proteins were abundantly expressed in 100% of breast cancer specimens. In SLN specimens, 94% of samples with metastases were positive for HCMV IE and LA proteins, mostly confined to neoplastic cells while some inflammatory cells were HCMV positive in 60% of lymph nodes without metastases [14].
And Afsar Rahbar et al. reported that the negative correlation between high-grade expression HCMV-IE and hormone receptor expression suggests a role for HCMV in hormone receptor-negative breast cancer tumors, possibly by forcing breast cancer cells into a more aggressive phenotype [15].
Research objective
HCMV has been found in many primary tumors, but it is still unknown whether the virus is still active in the tumor cells. At present, there are few studies on this topic in the world, only a few experimental data are available, the reliability is low. The present study was designed to detect the infection rate of HCMV in 146 cases of breast cancer samples (matched with corresponding paracancerous tissues and sentinel lymph node [SLN]) tissues, the expressions of HCMV immediate-early (IE) and late antigen (LA) proteins in breast cancer tissues, paracancerous tissues, SLN tissues and normal breast tissues were detected by immunohistochemistry, and HCMV infection of the samples was graded according to the percentage of the positive cells. HCMV IE2 mRNA expression was detected by reverse transcription polymerase chain reaction (RT-PCR). The clinical data were collated and statistically analyzed.
Materials and methods
Main test instruments and equipment
Confocal laser scanning microscope (OLYMPUS BD, Japan, OLYMPUSOptical Instrument Co Ltd); Ultra clean bench (SW-CJ-2FD, Suzhou purification); Inverted microscope (XD-101 Jiangnan photoelectric); Cryogenic centrifuge (5702R, Eppendorf, Germany); Rotary microtome (Leica, RM2235, Germany Leica Company); Real time quantitative PCR (Bio-Rad);
Refrigerator (BCD-232, Qingdao, AUCMA); Vertical sterilizer (LMQ.R-3260B, Shandong,Xinhua); Electric constant temperature blower drying box (DHG-9240A type Shanghai Jing Hong); Model pipettes (Gilson, France); Full automatic micro camera system (BX50, Japan OLYMPUS Optical Instrument Co., Ltd.).
PrimeSCript RT reagent Kit with gDNA Eraser, kit (TaKaRa); Human cytomegalovirus (HCMV) monoclonal antibodies (CHEMICON, USA); SP ultra sensitivity Kit (Fuzhou Maixin Biotechnology Development Co., Ltd.); Pepsin (BioGenex, CA, USA); Equilibrium in citrate buffer (pH 6, BioGenex); Avidin/biotin blocking Kit (Dako, cytomation, Glostrup, Denmark); Fc receptor blockers (the Innovex, Sciences, Richmond, CA, USA); Anti IE (1:200, ChemiconTemecula, CA); Anti LA (1:100, Chemicon, Temecula, CA); Anti cytokeratin (1:100, Dakocytomation, Glostrup, Denmark); Monoclonal mice resistant to EBV (EBNA2) (Dako, cytomation, Glostrup, Denmark); Polyclonal rabbits were resistant to HSV-1, HSV-2 (Dako, cytomation, Glostrup, Denmark); Picopure DNA Extraction Kit (Applied, Biosystems, Branchburg, NJ); Ethanol hydration liquid (Apoteket, pharmaci); TBST buffer solution (0.9%Nacl, 0.1 M, Tris, pH7.5, Triton, X-100, 2–3 drops/liter); 4% neutral formalin buffer solution (Apoteket PharmaciLaboratorier AB); PCR primers were synthesized by Shanghai bio engineering company; Other agents were provided by the breast disease diagnosis and treatment center and the pathological diagnosis center of the Affiliated Hospital of Qingdao University.
All patients were diagnosed at the Affiliated Hospital of Qingdao University between June 2013 and June 2014, and underwent unilateral axillary SLN biopsy. All patients who participated into the study provided a signed informed consent. The Ethics Committee and the Breast Center Biological Database of the Affiliated Hospital of Qingdao University granted the investigators permission to use the samples. All samples were anonymously coded and analyzed, in order to make it impossible to connect the HCMV virus information to the patient, except for the clinicians.
A total of 438 tissue samples were extracted from the 146 patients with breast cancer. These samples were fixed with formalin, and embedded by paraffin. Among these patients, 70 patients had SLN metastasis, and 76 patients had no SLN metastasis. The diagnosis of breast cancer was confirmed by histological evaluation through the Pathology Department of the Affiliated Hospital of Qingdao University. The age of these patients was 29–79 years old, with a median age of 57 years old.
Immunohistochemical analysis
Four-mm-thick paraffin-embedded tissue sections were sliced, dewaxed with xylene, and underwent hydration in a series of aqueous solutions with decreasing ethanol concentrations. After reaching equilibrium in Tris-Buffered Saline and Tween 20 (TBST), these sections were fixed in 4% formalin neutral buffer solution. The antigen was treated with pepsin at 37
In order to exclude the cross reaction between HCMV IE- and LA-specific antibodies and other herpesvirus proteins in this experiment, cells infected by EBV, herpes simplex virus type-1 and type-2 (HSV-1 and HSV-2, respectively), and HHV-8 were collected. Using the immunocytochemical method, the presence of the above viruses were confirmed on cell glass slides using monoclonal mouse anti-EBV (EBNA2), polyclonal rabbit anti HSV-1 and HSV-2, and rat monoclonal antibody directed against HHV-8 latent nuclear antigen (LNA-1/ORF73). Then, cell samples were collected and added with the HCMV specific antibodies used in this study to test their reactivity. Results revealed that there was no immune response.
By estimating the percentage of IE and LA positive cells, the investigators classified and evaluated the results of the HCMV staining: negative (the percentage of positive cells was 0%), grade I (the percentage of positive cells was
In order to verify the presence of HCMV nucleic acid in the lesion sites, 146 positive or negative HCMV tissue samples were extracted using a Picopure DNA extraction kit, according to manufacturer’s instructions. Briefly, the breast cancer sections, paracancerous tissues, or SLNs were dewaxed in xyelene, scraped with a sterile blade, washed with 99% alcohol, and air dried. Then, the samples were added with the DNA extraction buffer, digested with proteases K at 65
Analysis of immunohistochemistry results (IE)
Analysis of immunohistochemistry results (IE)
The positivity of HCMV was graded into four grades based on the estimated percentage of HCMV IE positive cells in tumors from breast and paired SLN: Grade I (
Analysis of immunohistochemistry results (LA)
The positivity of HCMV was graded into four grades based on the estimated percentage of HCMV IE positive cells in tumors from breast and paired SLN: Grade I (
Other herpesvirus HSV-1, HSV-2 and HHV-8 were detected using SLN positive samples (
Data were statistically analyzed using the statistical software SPSS17.0. The expression of HCMV IE and LA proteins in breast cancer tissues, SLN tissues and paracancerous tissues were analyzed by
Results
The expressions of HCMV IE and LA antigen proteins in breast cancer tissues, paracancerous tissues, SLN tissues and normal tissues
In order to determine whether there was HCMV antigen in the pathological tissues of each group, obtained section samples was detected using the monoclonal antibodies of HCMV IE and LA proteins by immunohistochemitry (IHC). According to the percentage of HCMV IE and LA positive cells in the immunohistochemical results, based on the grade of infection, all tissue samples were divided into five levels: negative and levels I, II, III and IV (Tables 1 and 2).
Expression of IE protein in breast cancer tissues. The expression rates of IE protein IV, III, and II (142/146, 97.3%) were significantly higher than those of grade I and negative (4/146, 2.7%), 
Expression of IE protein in paracancerous tissues. The expression rates of IE protein IV, III, and II (20/146, 13.7%) were significantly lower than those of grade I and negative (126/146, 86.3%), 
Expression of IE protein in sentinel lymph node carcinoma. The expression rates of IE protein IV, III, and II (58/68, 85.3%) were significantly higher than those of grade I and negative (10/68, 14.7%), 
Results revealed that, HCMV IE and LA proteins were presented in 100% of breast cancer tissue samples (
In comparison of the infection level, the overall expression of viral protein in tissue samples at levels II, III and IV was defined as high expression, the overall expression of viral protein in tissue samples at negative and level I was defined as low expression. Results revealed that, in cancer tissues, the high expression rate of IE protein (142/146, 97.3%) was significantly higher than low expression rate (4/146, 2.7%), and the difference was highly statistically significant (
Expression of IE protein in non cancerous tissues of sentinel lymph node. The expression rates of IE protein IV, III, and II (68/138, 49.3%) were not statistically significant with grade I and negative grade (70/138, 50.7%), 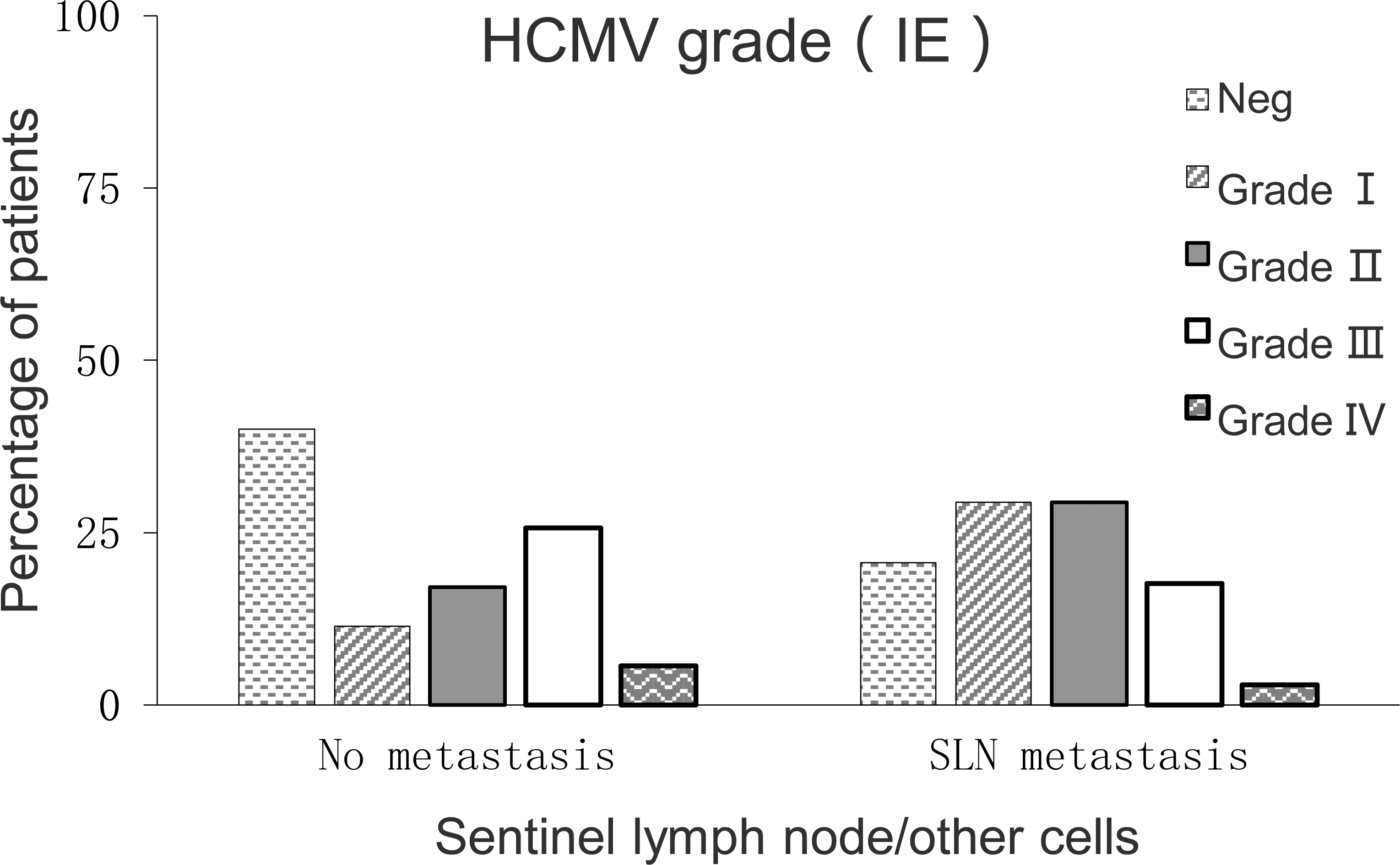
Expression of LA protein in breast cancer tissues. The expression rates of LA protein IV, III and II (120/146, 82.2%) were significantly higher than those of grade I and negative (26/146, 17.8%), 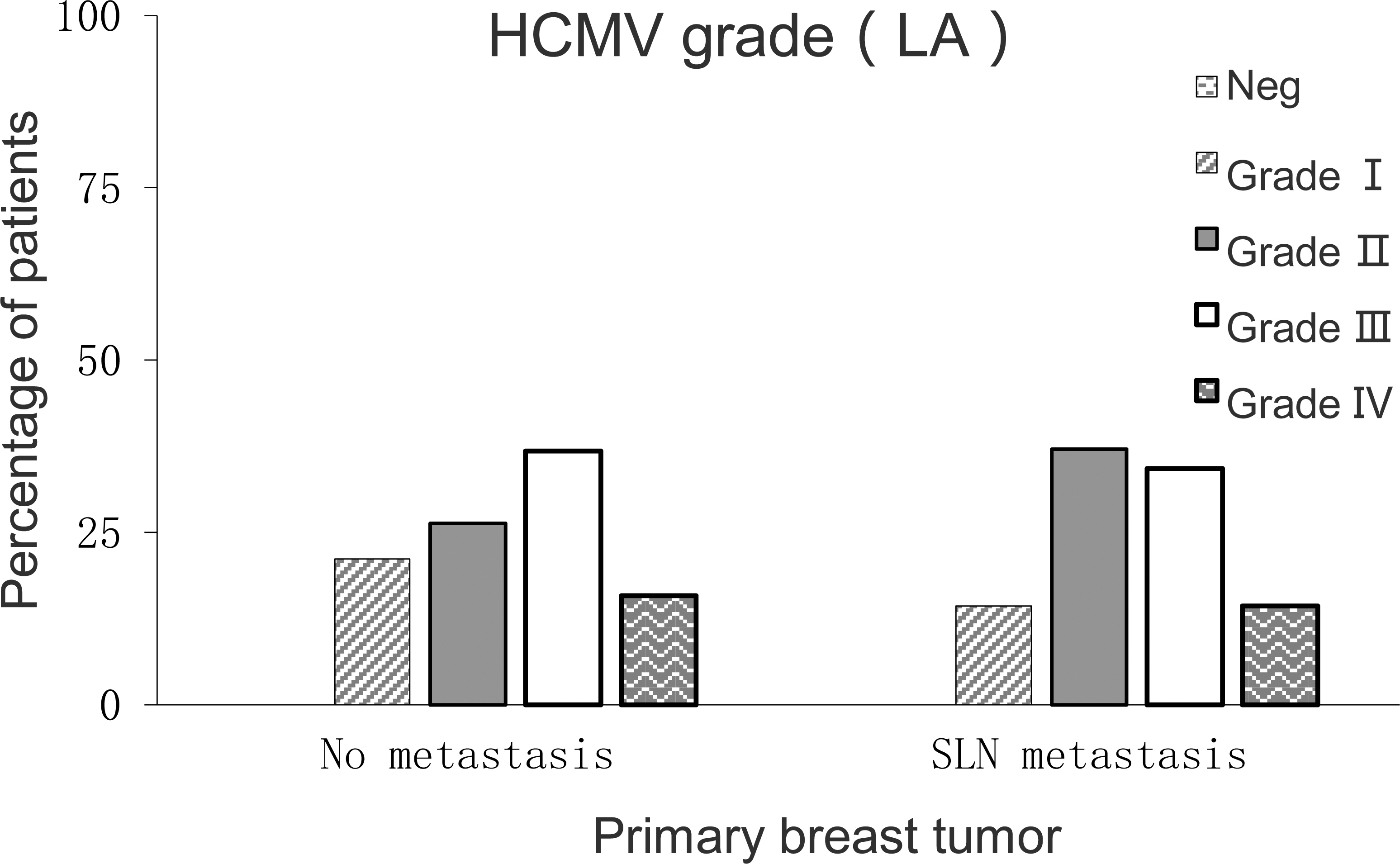
Expression of LA protein in paracancerous tissues. The expression rates of LA protein IV, III, and II (20/146, 13.7%) were significantly lower than those of grade I and negative (126/146, 86.3%), 
Expression of LA protein in sentinel lymph node carcinoma. The expression rates of LA protein IV, III, and II (50/68, 73.5%) were significantly higher than those of grade I and negative (18/68, 26.5%), 
Expression of LA protein in non cancerous tissues of sentinel lymph node. The expression rates of LA protein IV, III, and II (38/138, 27.5%) were significantly lower than those of grade I and negative (100/138, 72.5%), 
In the SLN samples, viral proteins are mainly expressed in nucleus of metastatic tumor cells. However, the present study also revealed that, inflammatory cells infected by HCMV were detected in 79.4% of positive SLN samples, while a small number of these cells were also detected in 60% of the negative SLN samples (Tables 1 and 2). No cytokeratin was detected in one patient with negative HCMV in the positive SLN group, this indicated that the sample has no tumor cells.
The levels of viral infection were generally higher in breast cancer than in metastatic SLN tissues. For IE protein,
Expression of HCMV IE2 mRNA in breast cancer tissues, paracancerous tissues and normal tissues
*The expression of IE2 and mRNA in breast cancer tissues is higher than that in adjacent tissues and normal tissues, and the expression in paracancerous tissues is higher than that in normal tissues (
Expression of HCMV IE2 mRNA in sentinel lymph nodes
*The expression of IE2 and mRNA in metastatic sentinel lymph node carcinoma tissues was higher than that in non cancerous tissues and non metastasis tissues (
In order to investigate the infection of HCMV in normal breast tissue, the breast tissues from 40 patients with cyclomastopathy were detected using immunohistochemistry. Results revealed that, HCMV IE antigen was expressed in 7.5% of the samples (3/40), and LA antigen was expressed in 5% of the samples (2/40). This indicated that, HCMV infection rate is extremely low in normal human breast tissues. Compared to the infection of HCMV in the samples of breast cancer patients, this reveals from the side that there is a correlation between viral infection and breast cancer.
Correlation analysis revealed that, in breast cancer tissue, with the increase in the expression level of IE protein, LA protein also increased, there was a positive correlation between the two (
Expression of HCMV, IE2, and mRNA in breast cancer.
RT-PCR results revealed that, the expression of IE2 mRNA was higher in breast cancer tissues than in paracancerous tissues and normal tissues (
Immunohistochemical results of pathological indexes
Immunohistochemical results of pathological indexes
*The difference of ER
Relationship between pathological features and IE grade in breast cancer
*The expression of ER was negatively correlated with the level of HCMV IE infection (
Expression of HCMV, IE2, and mRNA in breast cancer tissues, paracancerous tissues and normal tissues. *The expression of IE2 and mRNA in breast cancer tissues and paracancerous tissues was higher than that in normal controls, and the cancer tissues were higher than those in adjacent tissues (
The expression of HCMV IE2 mRNA in sentinel lymph node tissue.
Clinical factors analysis
In order to verify whether the level of HCMV infection is related to the known relevant clinical factors of breast cancer, such as SLN status, age, Elston level, estrogen receptor-
Prognostic factors analysis
In this study, only 9.5% (7/73) of the patients died of breast cancer. This made us unable to conduct a deeper statistical analysis of the association between breast cancer mortality and HCMV infection, and draw a survival curve of patients. We noticed that, in seven defuncts (
the relationship between ER alpha grade and sentinel lymph node metastasis. There was a statistically significant difference between the ER alpha class and the sentinel lymph node metastasis (
The relationship between the ER alpha class and the HCMV IE level. The expression of ER was negatively correlated with the level of HCMV and IE infection (
In order to exclude the cross reaction between HCMV IE and LA specific antibodies and other herpesvirus proteins in this study, we collected cells infected by epstein-barr virus (EBV), herpes simplex virus (HSV)-1, HSV-2 and human herpesvirus 8 (HHV-8). We did not found any reaction between these cells and HCMV-specific antibodies used in this study. Furthermore, from both the positive SLN group and negative SLN group four samples of breast cancer tissues and SLN tissues were obtained, and DNA analysis targeted above virus was performed. Except EBV was detected in one individual in the negative SLN group, no evidence for the presence of HSV-1, HSV-2, HHV-8 or EBV mRNA was found in tissue samples. This precludes the possibility that any other virus could interfere with the experiment.
The present study investigated the expression of HCMV infection in breast cancer and related tissues based on a large sample size for the first time, the universal expression of this virus in breast cancer patients was verified at the phenomenal level, thus the association between the level of HCMV infection and different tissue types in breast cancer patients was determined.
In this experiment, both immunohistochemical and RT-PCR results confirmed that, the expression of HCMV was extremely low in normal breast tissues. HCMV cannot easily infect normal breast tissue, the reason may be related to the neurotropiam of the virus. This ensures the relative simplicity of the experimental variables in subsequent experiments. Meanwhile, the negative results of EBV, HSV-1, HSV-2 and HHV-8 excluded the possibility of cross reaction between HCMV IE and LA specific antibodies and other herpesvirus proteins in this experiment, making the experimental data highly credible.
In the present study, with SLN metastasis as the primary grouping principle, and HCMV IE and LA as observation indexes, HCMV infection in breast cancer tissues, paracancerous tissues and SLN tissues was detected, a series of meaningful results were obtained. HCMV IE and LA were detected as positive in 100% of primary breast cancer tissue samples. In metastatic SLN tissues, we found that, HCMV antigen proteins could be expressed in most tumor cells. In normal breast tissues or lymph tissues, HCMV was not expressed or extremely lowly expressed, the difference were statistically significant. These findings suggest that, HCMV proteins could be expressed continuously in most tumor cells. The virus can somehow enter the SLN, and continues to replicate there. The results of this study are consistent with those of Harkins et al., who recently demonstrated the high expression of HCMV protein in breast cancer.
The present study revealed that, the virus was mainly confined in the primary tumor and metastatic cancer cells, and virus expression was also found in a small amount of inflammatory cells. More important is that, 92.6% of metastatic SLN were positive for HCMV. In the majority of breast cancer patients with SLN metastasis, the primary tumor infection level was IV. This was also common in breast cancer tissues without SLN metastasis. This reveals that there is a certain echo between the level of HCMV infection and the degree of breast cancer cell aggregation. For breast cancer tissue with the highest level of HCMV infection, the possible explanation is that, the virus enters the mammary epithelial cells through a short cut, and promotes it to release more viruses to migrate into the SLN.
In summary, the experimental results of this study revealed that, HCMV infection is ubiquitous in breast cancer tissues and metastatic SLN tissues, and infection level was closely related to the type of samples. We will further investigate the possible mechanism of HCMV infection on breast cancer on the basis of the phenomenon in this study.
Discussion
More and more evidences have revealed that HCMV proteins and nucleic acids are highly prevalent in malignant tumors derived from a variety of tissues. The results of this study are consistent with the study conducted by Harkins et al., recently demonstrating the high expression of HCMV protein in breast cancer. In this study, HCMV was detected positive in 100% primary breast cancer tissue samples. Furthermore, this study also reports that virus proteins could be expressed in metastatic SLN tissues in most tumor cells. These findings suggest that most tumor cells continue to express HCMV protein. Hence, it is very important to explore the possible mechanism of HCMV in the formation and metastasis of breast cancer. However, the role of this virus remains to be further evaluated.
A literature has pointed out that virus infection is associated with more than 20% of all malignancies in the world [16]. Recent studies have involved the relationship between virus infections and breast cancer, such as EBV, MMTV [4], HPV [17] and HCMV [6]. Some scholars have speculated that the infections of common viruses in adulthood such as HCMV may be a potential risk factor for breast cancer [18]. In fact, HCMV IgG levels increase prior to breast cancer development [19]. In addition, Tsai et al. revealed that HCMV and HHV-8 are associated with shorter disease-free survival and lower overall survival rate in breast cancer patients [5]. This further suggests a close relationship between HCMV and tumor formation.
HCMV is present in human milk, saliva, urine, cervical secretions and semen. This suggests that HCMV will continue to infect epithelial cells [20]. The breast milk of more than 90% of lactating women contains HCMV, and the breast augmentation of 67% of patients is positive for HCMV [6]. These findings suggest that HCMV can be found in normal breast tissue. Furthermore, this study revealed that the virus is mainly confined to the primary tumor and in metastatic cancer cells, and virus expression was also found in a small amount of inflammatory cells. In addition, this study revealed that HCMV protein can be found in all breast cancer tissue samples (either metastatic or non-metastatic). More important is that 92.6% of metastatic lymph nodes were positive for HCMV. For the majority of breast cancer patients with SLN metastasis, primary tumor infection grade was IV. This was also common in breast cancer patients without SLN metastasis.
Many studies have revealed that inflammation is associated with cancer [21, 22]. Inflammatory mediator cyclooxygenase-2 (COX-2) and its metabolite prostaglandin have been determined as tumor cell growth factors [23]. Many tumors, including breast cancer, are COX-2 positive; and the high expression of COX-2 is closely associated with poor prognosis of tumors [24]. Selective and non-selective COX inhibitors can reduce cancer incidence, and has a certain anti-cancer behavior (refer to [25]). Interestingly, HCMV can induce inflammation and escape immune recognition through complex mechanisms [26, 27]. HCMV protein US28 is a constitutively active chemokine receptor homolog that can induce COX-2 expression, cause the phosphorylation of STAT3, increase the production of VEGF and IL-6, and induce tumor formation in the body [27]. Recent study confirmed that HCMV protein expression was found in the medulloblastoma, and virus infection led to COX-2 expression [30]. Aspirin and specific COX-2 inhibitor celecoxib can effectively inhibit HCMV replication in vitro, inhibit PGE2 production, and significantly decrease in vivo and in vitro tumor cell growth [29]. In patients with glioblastoma, Rahbar et al. found that low grade HCMV infection in tumors was closely correlated to prolonged survival [30]. These findings suggest that HCMV may be involved in tumor development. HCMV-targeted therapy may provide an additional treatment option for patients with HCMV positive tumors.
SLN status is currently the most valuable indicator for the measurement of breast cancer patient survival, and greatly influences the choice of adjuvant therapies [31, 32]. The downregulation of ER
