Abstract
Pancreatic cancers with poor prognosis are highly malignant, readily metastatic and of immune tolerance, mainly due to delayed detection. The metastatic progression and immune tolerance of pancreatic cancer is greatly attributed to the intercellular communication. However, exosomes are deemed to be the most important tool of intercellular communicators. Thus, we present a review of pancreatic cancer and exosomes in this article. We intensively summarize the progress of early pancreatic cancer and the relationship of the proliferation, progression and metastasis of pancreatic cancer and pancreatic cancer-derived exosomes, and propose new ideas of the study of pancreatic cancer.
Keywords
Introduction
Pancreatic cancer (PC), a common malignancy with metastasis, poor prognosis and aggressive disease, is associated with a high mortality rate. The major histological type of pancreatic cancer is pancreatic ductal adenocarcinoma (PDAC) accounting for
Pancreatic cancer often progresses asymptomatically in the early and presents in the late, resulting in only a minority being suitable for resection, because of symptoms of pancreatic cancer being quite non-specific and including nausea, anoxeria, jaundice, weight loss and abdominal pain [8]. Because the pancreas, being a retroperitoneal organ, is buried deep in the torso, there is no external lumps that can be palpated or external skin changes that can be seen during an annual routine physical examination [5]. By the time pancreatic cancer is diagnosed, 80–85% of patients present with advanced unresectable disease, and responds poorly to most chemotherapeutic agents [9]. For patients with advanced pancreatic cancer, the disease usually progresses rapidly, and few patients survive more than 1 year after diagnosis. Encouragingly, prognosis of patients with pancreatic cancer who receive surgery is better, surgical margins are free of disease, and perineural or lymphovascular invasion don’t appear, when the tumor is smaller [5]. Meanwhile, a report of pancreaticoduodenectomies for pancreatic cancer revealed that: for cancers
Pancreatic intraepithelial neoplasia
Precursor lesions of tumor can happen to pancreas, which is a powerful case for screening of early pancreatic cancer. Three precursor lesions, including pancreatic intraepithelial neoplasia (PanINs), intraductal papillary mucinous neoplasms (IPMNs), and mucinous cystic neoplasms (MCNs), have each been identified as distinct precursors to ductal adenocarcinomas of pancreas [5]. PanINs are noninvasive epithelial proliferations within the smaller pancreatic ducts, the progress of which is from PanIN-1 (low-grade) to PanIN-2 (intermediate-grade) to PanIN-3 (high-grade) [11, 12]. We assume the progression model of PanINs in pancreatic cancer, as shown in Fig. 1. PanINs are microscopic (diameter is
Multistep progression model of PanINs in pancreatic cancer.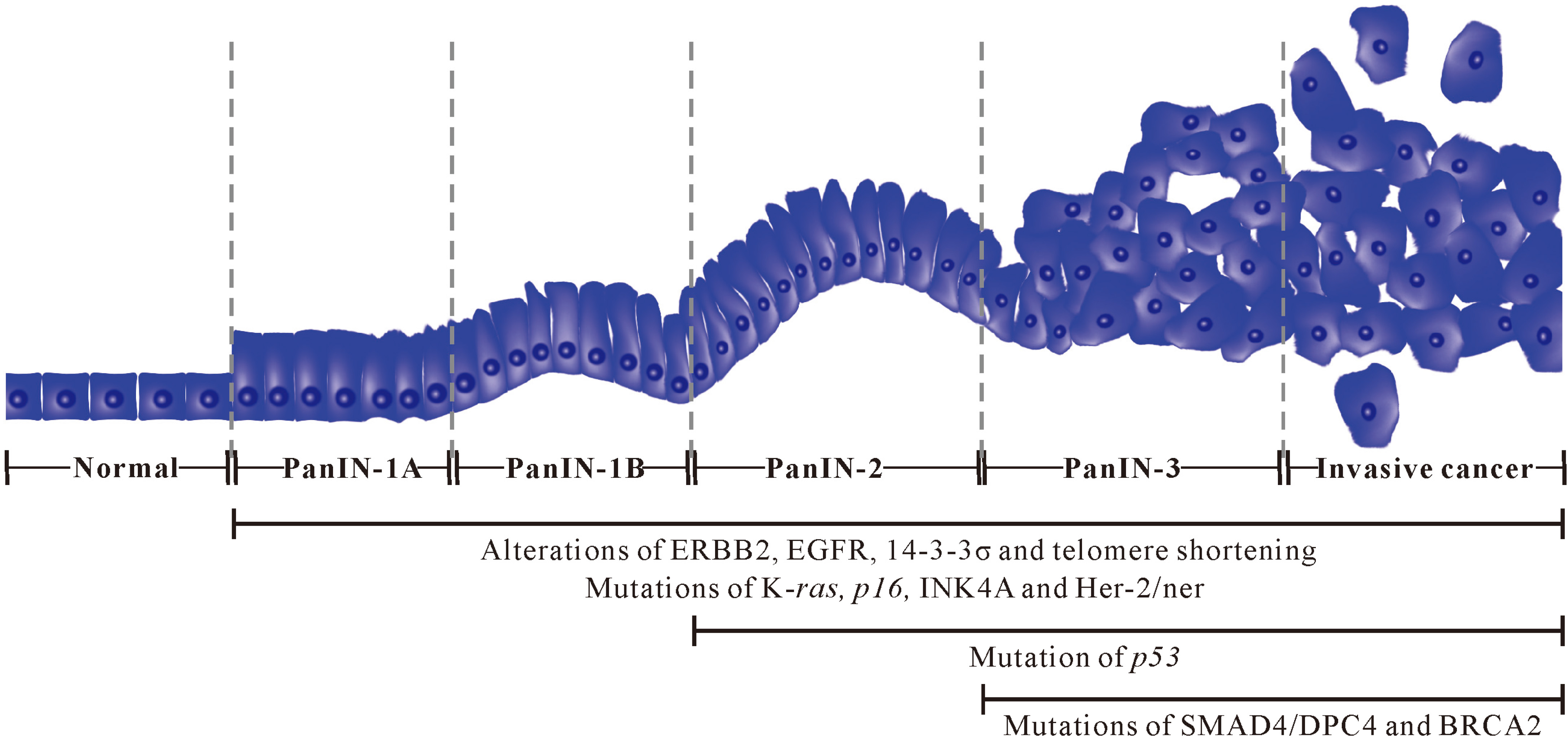
There are specific molecular alterations in the process from normal epithelium to PanIN-1 to PanIN-2, and then to high-grade PanIN-3 and invasive cancer. For example, alterations of ERBB2, EGFR, 14-3-3
At present, three main conventional methods of diagnosing pancreatic cancer are serum biomarkers, imageological diagnosis and pathological diagnosis. However, currently there are no biomarkers that accurately discriminate between early pancreatic cancer/high-grade PanIN-3 and normal/low-grade (PanIN-1, PanIN-2) lesions [17], as well as no noninvasive screening tools that detect lesions of early pancreatic cancer [2].
Serum biomarker
Cancer antigen 19-9 (CA 19-9) in serum, the most common biomarker in clinical routine, is the most extensively evaluated with pooled data from 2283 patients with pancreatic cancer, median sensitivity and median specificity of which is 79% (70
Imageological diagnosis
Modern non-operative techniques for tumor staging, including transabdominal ultrasound, computed tomography (CT), magnetic resonance imaging (MRI) and endoscopic ultrasound (EUS) are less invasive and less costly than surgery [24]. However, CT is short of sensitivity to detect small pancreatic lesions, such as PanINs, which don’t sensitively detect early pancreatic cancer [2]. EUS with high resolution detects lesions as small as 1 to 2 mm and allows biopsy and/or fine-needle aspiration cytology, which distinguished benign from malignant disease when focal lesions are present [1, 2]. However, pancreatic cancer is more common with increasing age and most frequently diagnosed among people older than 55 years [25]. Because of age [26], smoking [27], alcohol [28] and other factors, the pancreas of a high proportion of individuals older than 50 years appear significant changes, such as pancreatic fibrosis. The changes during EUS imaging could obscure early lesions, especially noninvasive PanIN-3 [2]. Hence, EUS for the detection of early pancreatic cancer has a certain limitation in the clinical.
Pathological diagnosis
Pathological diagnosis is also an important method for diagnosing pancreatic cancer. At present, needle biopsy is an effective means of obtaining pathologic diagnosis information. Image-guided fine needle aspiration (FNA) is a popular method for evaluating pancreatic lesions, sensitivity, specificity, positive predictive value and negative predictive value of which were 79.4, 99.0, 99.4 and 67.9, respectively [29]. However, for screening of early pancreatic cancer, this approach is in a weak position.
For diagnosing early pancreatic cancer, the three conventional methods of seem to be insufficient and powerless. Therefore, there are currently many challenges for the detection of early pancreatic cancer. With the development of current science and technology, detection of early pancreatic cancer requires to screen asymptomatic subjects among high-risk groups through noninvasive tests. If a marker of early pancreatic cancer is available, some genetically susceptible but asymptomatic individuals could be screened and offered effective therapy in the early stages. Hence, screening early lesion of pancreas through noninvasive tests and molecular imaging will major breakthrough and significant change for the detection of early pancreatic cancer. Noninvasive detecting and screening absolutely depend on specific and sensitive biomarker in blood, saliva and urine, and accurate and distinct imaging technology. Noninvasive screening technology based on the two aspects is more suitable for the detecting and screening of a high proportion of individuals with susceptibility.
Ideal biomarkers of screening early pancreatic cancer should be universally present in curable-stage of pancreatic cancer, while absent in individuals with cancer-free. The biomarkers should be handily detected in readily obtainable biological samples, such as blood, saliva and urine. The detecting process should be rapid, inexpensive, widely distributable to maximize testing results, and the biomarkers must be highly sensitive and specific to accurately detect the critical target. Most recently, the detection of exosomes may offer diagnostic, predictive and prognostic markers of pancreatic cancer.
Exosomes
Exosomes are lipid bilayer membrane-enclosed nano-sized (30
Various effects of pancreatic cancer-derived exosomes
Exosomes are deemed to be the most important tool of intercellular communicators, including cancer cells and normal cells [53]. In terms of pancreatic cancer, exosomes deriving from pancreatic malignancies can be loaded with a great deal of substance associated with pancreatic cancer and produce some special physiological functions. We summarized the relative researches of pancreatic cancer and associated exosomes. The relevant principle, mechanism and details were as follows:
Pancreatic cancer-derived exosomes as vehicles of K-ras and p53 genes
Kahlert et al. identified that mutated
Pancreatic cancer-derived exosomes as biomarkers of noninvasive detection
Melo et al. identified that glypican-1 (GPC1) specifically enriched on cancer-cell-derived exosomes, and verified that GPC1
Lau et al. firstly developed a pancreatic cancer mouse model that yielded discriminatory salivary biomarkers by implanting the mouse pancreatic cancer cell line (Panc02) into the pancreas of the syngeneic host C57BL/6 mouse. The role of pancreatic cancer-derived exosomes in the development of discriminatory salivary biomarkers was then tested by engineering a Panc02 cell line that is suppressed for exosome bio-genesis, implanting into the C56BL/6 mouse, and examining whether the discriminatory salivary biomarker profile was ablated or disrupted. Suppression of exosome biogenesis leads to the ablation of development of discriminatory salivary biomarker [62]. This study supports that tumor-derived exosomes provided a mechanism in the development of discriminatory biomarkers in saliva and distal systemic diseases.
Pancreatic cancer-derived exosomes as platform of microRNA signature
MicroRNAs (miRNAs) are small non-coding RNAs (17–25 nucleotides) that regulate gene expression at post-transcriptional level, and play important roles in tumorigenesis, tumor-progression and tumor-metast-asis [63, 64]. And previous studies have shown that abnormal expression of miRNAs have the potential to identify various types of cancers, including gastric cancer [65], prostate cancer [66], breast cancer [67], lung cancer [68], pancreatic cancer [69], etc. For pancreatic cancer, miRNA-21, miRNA-155, miRNA-210, miRNA-221 and miRNA-222 were over-expressed in diseased tissues than in the control samples, whereas miRNA-31, miRNA-122, miRNA-145 and miRNA-146a were under-expressed [70]. Lai et al. reported that high exosomal levels of miRNA-10b, miRNA-21, miRNA-30c and miRNA-181a, and low exosomal levels of miRNA-let7a readily differentiated PDAC from normal control and chronic pancreatitis samples, and all 29 cases of PDAC exhibited significantly elevated exosomal miRNA-10b and miRNA-30c levels [71]. Madhavan et al. tested that miRNA-1246, miRNA-4644, miRNA-3976 and 4306 by qPT-PCR and discovered the four miRNAs were significantly upregulated in 83% of serum-exosomes from pancreatic cancer, but rarely in control groups [33]. Takikawa et al. found that pancreatic stellate cell–derived exosomes contained a variety of miRNAs including miRNA-21-5p, and several miRNAs such as miRNA-451a were enriched in exosomes compared to their source pancreatic stellate cells [72]. In addition, Ding et al. reported that pancreatic cancer-derived exosomes transfer miRNAs to dendritic cells and inhibit RFXAP expression via miRNA-212-3p, which decrease MHC II expression and induce immune tolerance of dendritic cells [73]. MiRNAs from pancreatic cancer-derived exosomes are superior to those from serum, and are able to more really reflect the development of pancreatic cancer, which provide a more promising signature for diagnosing pancreatic cancer.
Pancreatic cancer-derived exosomes as emerging drivers of pre-metastatic niche formation in liver
Primary tumor cells orchestrate pre-metastatic niche formation in secondary organs through secretion of a variety of cytokines and growth factors that promote mobilization and recruitment of bone marrow derived cells to future metastatic sites, and tumor-derived exosomes can serve as drivers of pre-metastatic niche formation [74]. Costa-Silva et al. demonstrated that exosomes from PDAC induced liver pre-metastatic niche formation in naive mice and consequently increase liver metastatic burden, and found that macrophage migration inhibitory factor (MIF) was highly expressed in PDAC-derived exosomes, but its blockade prevented liver pre-metastatic niche formation and metastasis [75]. They found that the PDAC-derived exosomes selectively taken up by liver Kupffer cells caused transforming growth factor
Pancreatic cancer-derived exosomes as coordinator of pancreatic tumor immune tolerance
Zhou et al. found that pancreatic cancer-derived exosomes negatively regulated toll-like receptor 4 (TLR4) in dendritic cells (DCs), and also down-regulated the expression of tumor necrosis factor-
Various effect of exosomes in pancreatic cancer.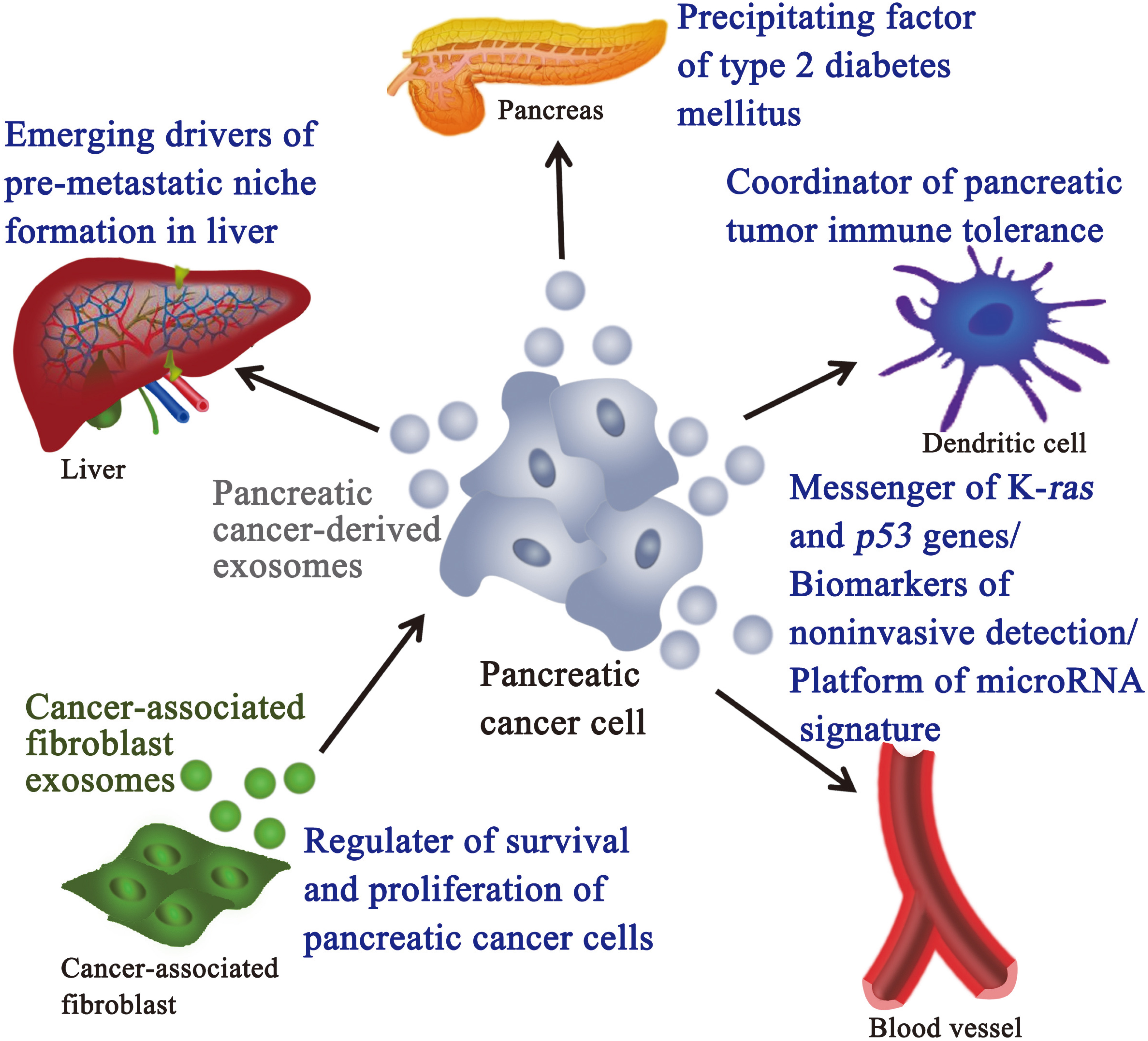
Javeed et al. found that pancreatic cancer-derived exosomes contained adrenomedullin and CA19-9, readily entered
Exosomes of cancer-associated fibroblast as regulator of survival and proliferation of pancreatic cancer cells
Cancer-associated fibroblasts (CAFs) comprise the majority of the tumor bulk of PDAC. Richards et al. found that CAFs exposed to gemcitabine dramatically increased the release of exosomes. These exosomes increased expression of chemoresistance-inducing factor, Snail, in recipient epithelial cells, and promoted survival, proliferation and drug resistance of cancer cells [88]. If gemcitabine-exposed CAFs
Exosomes derived from pancreatic stellate cells as communicator of intercellular communication
Pancreatic stellate cells (PSC), analogous to a resident stellate cell population in the liver or other organs, are initially quiescent, but with appropriate stimuli PSC can convert to an activated state with characteristics of myofibroblasts [89]. Activated PSCs are present in the stromal reaction in pancreatic cancers and are responsible for the production of stromal collagen [90]. Takikawa et al. clarified the microRNA expression profile in exosomes derived from PSC and presented that PSC-derived exosomes might play a pivotal role in the interactions between PSCs and pancreatic cancer cells [72]. And then PSCs dynamically interact with other cell types to constitute the cancer-conditioned microenvironment and promote the progression of pancreatic cancer through interactions with pancreatic cancer cells [91, 92]. So, further elucidation of the roles of PSC-derived exosomes in pancreatic cancer would lead to the identification of novel therapeutic targets in the intractable disease.
Comparison between conventional diagnostic methods and exosomes
Comparison between conventional diagnostic methods and exosomes
The conventional diagnostic methods, including serum biomarkers, imageological diagnosis and pathological diagnosis, are clinically used to diagnose pancreatic cancer in, but it seems to be powerless for the screening of early pancreatic cancer. Exosomes, as specific vesicles provide diversified biomarker information about pancreatic cancer, including mutated
Moreover, exosomes are related with formation of pre-metastatic niche in liver, immune tolerance of pancreatic tumor, occurrence of type 2 diabetes mellitus, and survival and proliferation of pancreatic cancer cells, which are like an Almighty to exhibit a variety of biological functions. Hence, exosomes provide some new ideas for the study of early metastasis, immune tolerance, proliferation and treatment of pancreatic cancer. In order to comprehensively understand the biological function of exosomes, we summarize a relative figure, as shown in Fig. 2.
Conflict of interest
The authors declare no conflicts of interest.
Footnotes
Acknowledgments
We are thankful for the financial support of the Natural Science Foundation of Fujian Province (2017J01547) the Science and Technology Project of the Education Department of Fujian Province (JA15445) and the National Natural Science Foundation of China (81571613, 81572442 and 21405016).
