Abstract
BACKGROUND:
The prevalence of nonalcoholic fatty liver disease (NAFLD) has been increasing worldwide. Pioglitazone is a pharmacologic agonist of peroxisome proliferators-activated receptor-
OBJECTIVE:
We aimed to evaluate the effects of pioglitazone in NAFLD and investigate the underlying mechanism by testing platelet derived growth factor (PDGF) and tissue inhibitory of metalloproteinase-2 (TIMP-2).
METHODS:
A total of C57BL/6 wild-type mice were randomized to three groups, control group (NC,
RESULTS:
Upon pioglitazone treatment, the PDGF and TIMP-2 expression levels were decreased compared with high-fat diet-fed mice devoid of drug stimulation. Analysis of liver histology showed pioglitazone treatment could reduce steatosis and inflammatory changes, which was helpful to inhibit hepatic fibrosis in NAFLD mice.
CONCLUSIONS:
The study showed pioglitazone might exert an inhibitory effect on hepatic inflammation and fibrosis in NAFLD. Moreover, this study provided novel evidence for the promising clinical application of pioglitazone in intervening NAFLD.
Background
Nonalcoholic fatty liver disease (NAFLD) is an umbrella term for a spectrum of conditions characterized by lipid deposition in hepatocytes. With the improvement of living standard, NAFLD is increasingly common worldwide and patients trend to suffer this disease at a younger age. In the United States, it is estimated to occur in one-third of the general population [1, 2, 3]. From a pathological point of view NAFLD may be classified into nonalcoholic simple fatty liver (NAFL, or steatosis), nonalcoholic steatohepatitis (NASH-fatty changes with inflammation and hepatocellular injury or fibrosis), and advanced cirrhosis. Although simple steatosis will usually follow a slow course, it is well documented that it can progress to NASH and fibrosis [4], and approximately 15% to 25% of patients with NASH progress to cirrhosis and its complications over 10 to 20 years. Patients with the latter two diseases are at a significant risk of increasing morbidity [5, 6, 7]. Although the clinical course of NAFLD is variable, the most prevalent causes of deaths in NAFLD patients are cardiovascular disorders and malignancy followed by liver-related deaths [8]. The pathogenesis of NAFLD is multifactorial and epidemiological associations strongly support the notion that NAFLD is closely associated with MetS and its individual components [1, 9]. Currently, NAFLD appears as a global public medical hotspot, where multiple insulin resistance (IR)-related complications are involved [2, 8]. There is a directional association between NAFLD and metabolic syndrome (MS), MS plays an crucial role for the onset and perpetuation of NAFLD and, in return, NAFLD may be also a cause of MS [10, 11].
The peroxisome proliferator-activated receptor (PPAR)-
There are several cytokines generated and participating in the progression of NAFLD. Upon the stimulation of reactive oxide species (ROS), liver mesenchymal cells secrete platelet derived growth factor (PDGF) and tissue inhibitory of metalloproteinase-2 (TIMP-2), two proteins which are proved to be potent hallmarks in wound healing and regeneration of periodontal and bone [20]. However, so far there is no report concerning an effective method by which we can detect the aberrant expression patterns of proteins in patients’s era to determine hepatic fibrosis. Meanwhile, the lack of large-scale trials limits the exploration of pioglitazone intervening NAFLD. Therefore, the present study aimed to evaluate the effects of pioglitazone on NAFLD and investigate the underlying mechanism.
Methods
NAFLD Model construction
A total of 180 male C57BL/6 wild-type mice weighting 200
Enzyme-linked immunosorbent assay
After treatment, the sera were collected from mice of three groups and subjected to ELISA. PDGF and TIMP-2 protein detection kits were bought from Nanjing Jiancheng Biotech Co., Ltd (Nanjing, Jiangsu, China). Standard ELISA method was performed to measure PDGF and TIMP-2 protein concentrations in serum by using detection kits according to the manufacturer’s instructions. All determinations were performed in triplicate and data are presented as the mean
Hematoxylin and Eosin staining
The same positions of livers in each group were fastened to 40 g/L formaldehyde buffer. Afterwards, the slices were stained with hematoxylin for 10 min, washed twice with distilled water, and then were dehydrated in an ethanol series (80%, 90%, 95%), and stained with eosin for 5 min, followed by twice 2-min rinse with anhydrous ethanol and then 20-min immerse in xylene. Liver histology samples were examined under light microscope.
Statistical analysis
All experiments were repeated in triplicate. The results of multiple experiments are presented as the mean
Informed consent and Statement of animal right
The materials and methods above have been reviewed and approved by the Research Review and Ethics Board (RREB) of the Yantaishan Hospital. We declare that the animal studies were performed according to National or Institutional Guide for the Care and Use of Laboratory Animals, and the ethical treatment of all experimental animals were observed carefully.
PDGF expression in NC, HF, and L groups.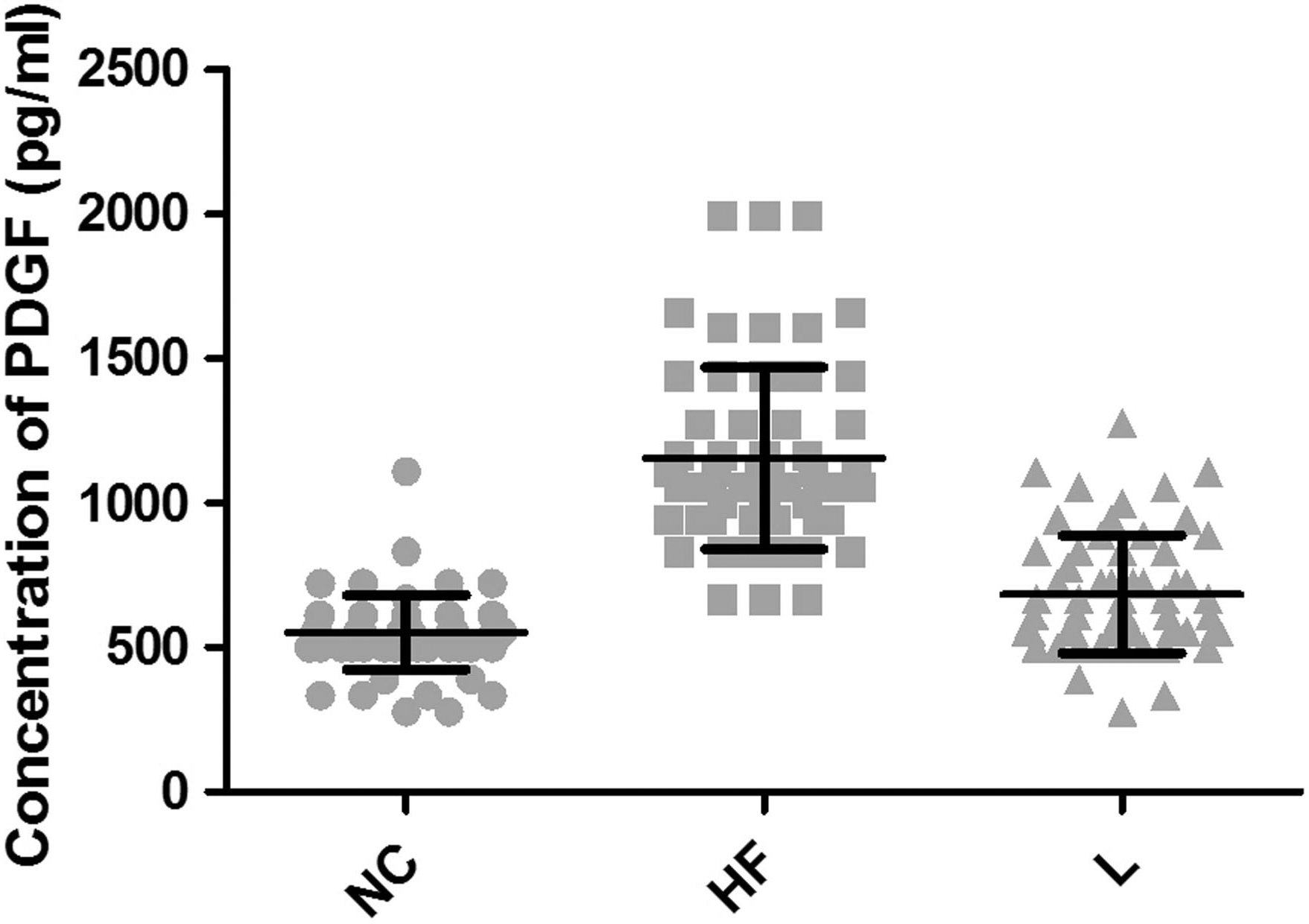
TIMP-2expression in NC, HF, and L groups.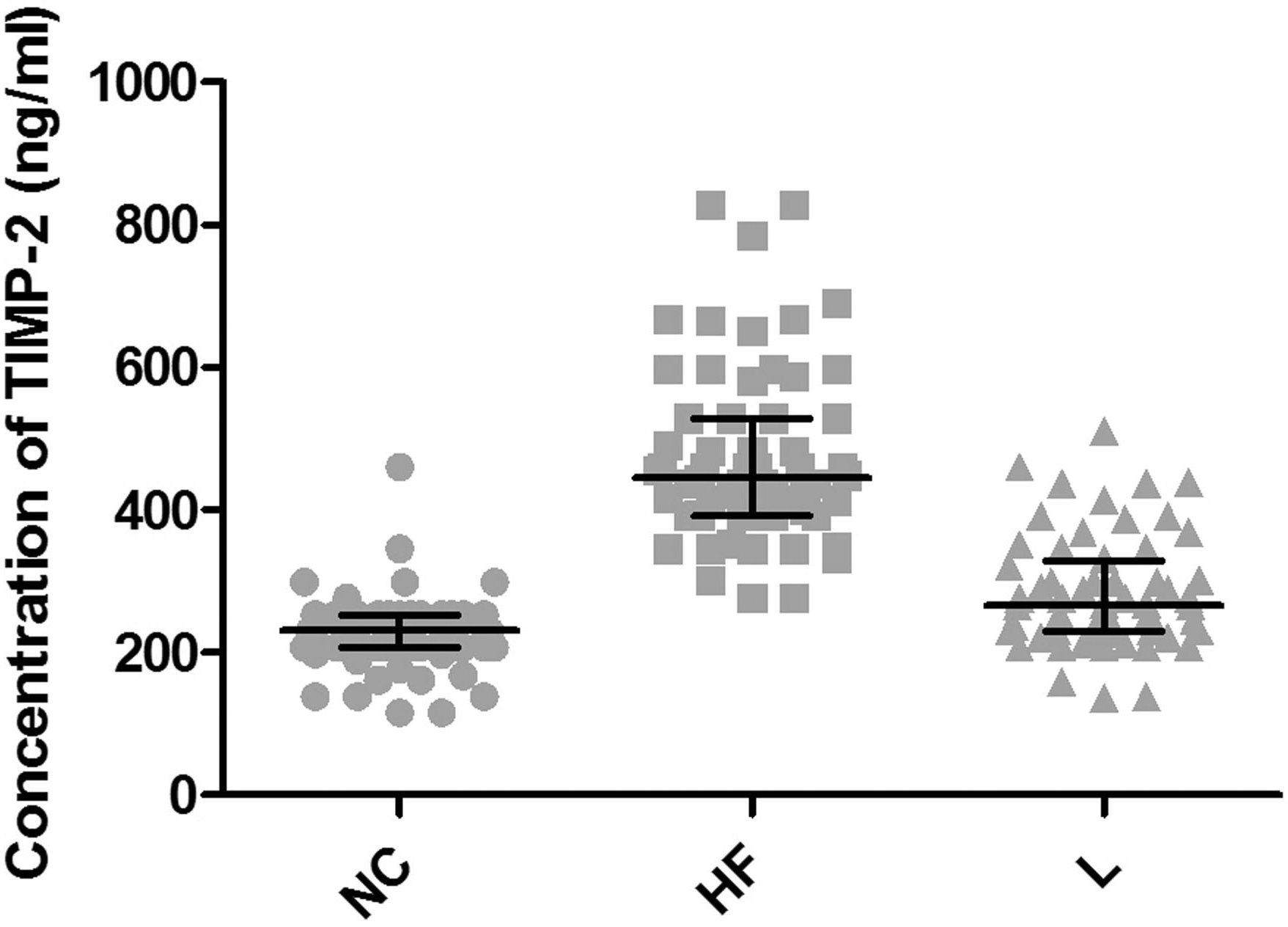
Evaluation of aberrant protein expression in serum of NAFLD mice
ELISA assay was performed to investigate the expression of PDGF and TIMP-2 in NAFLD mice. As shown in Fig. 1, a significant increase in the expression level of PDGF was observed when mice were exposed to high-fat diet, compared with the NC group. Of note, there was a remarkable decline in the PDGF expression level in these high-fat diet-fed mice upon pioglitazone treatment. Likewise, treatment of mice with pioglitazone dramatically compromised TIMP-2 accrual caused by the high-fat diet (Fig. 2). The decreased TIMP-2 expression in L group had no statistically significant difference in comparison to the NC group (
Liver histology (H&E). In the absence of pioglitazone, large lipid vacuoles were seen representing a severe fatty degeneration, which was alleviated in the presence of pioglitazone.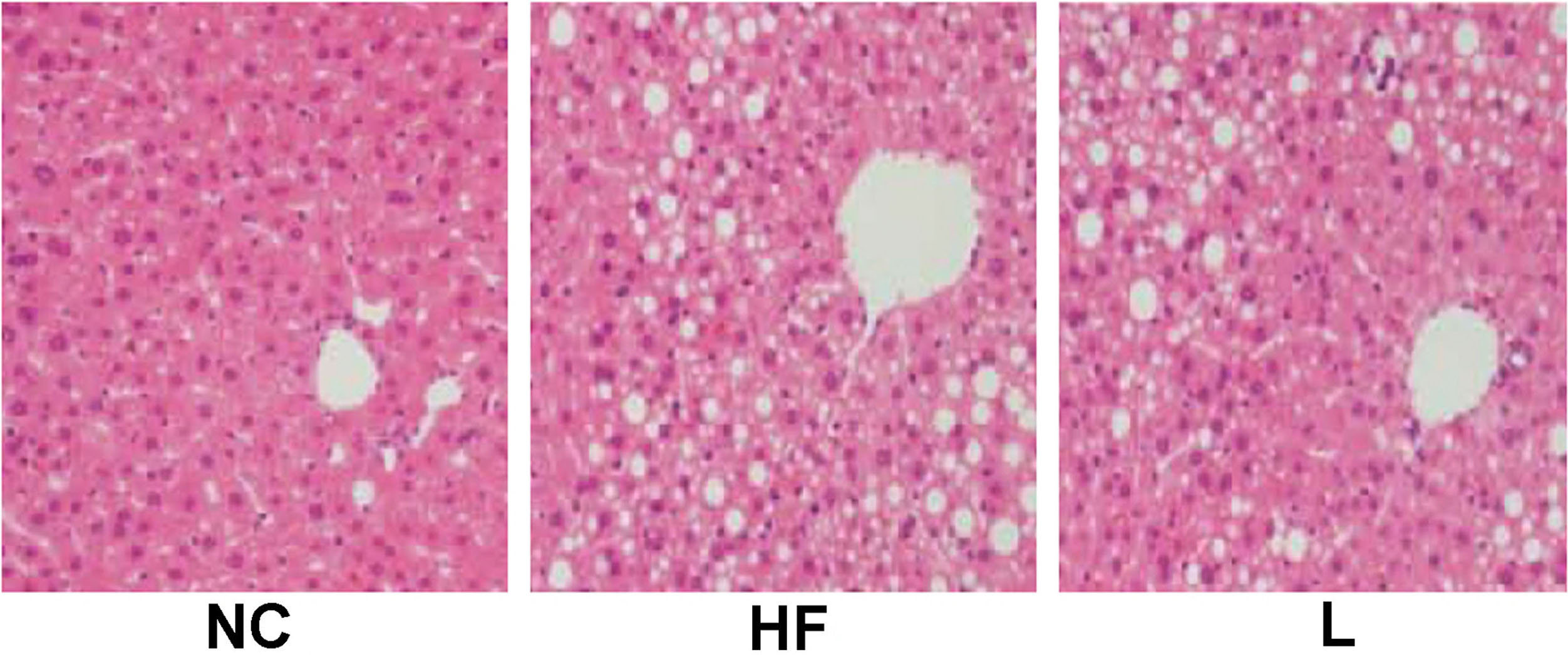
The histologic examination of liver biopsies was conducted and shown in Fig. 3. In NC group, the structure of hepatic lobule was complete. The hepatocytes arranged uniformly and radically, with round nucleus located in the center. No obvious inflammatory cell was detected. However, there was accumulation of triglyceride droplet vacuoles, known as fatty degeneration and accompanied with diffuse bullous, within the liver cells from mice in HF group, and these vacuoles occupied a large proportion of the cell’s volume and push the cytoplasm up against the cell membrane. Some hepatic tissues appeared with hepatic fibrosis together with hepatocyte necrosis and inflammatory cell infiltration. In the presence of pioglitazone, there were a small number of bullous fatty degeneration with no obvious hepatocyte necrosis or inflammatory cell infiltration. Therefore, it revealed that pioglitazone treatment could significantly all eviate steatosis and inflammatory changes, thereby suppressing the subsequent hepatic fibrosis in NAFLD mice.
Discussion
With the changes of dietary structure and life style, NAFLD has gained increasing attention as a common cause of chronic liver disease worldwide over the last decade [21, 22]. NAFLD is closely related to MetS and its individual features [9, 23].
The pathological features of NAFLD are fatty and necro-inflammatory changes, hepatocyte ballooning and hepatic fibrosis. Among them, fibrosis is deemed as the end stage result of chronic liver disease owing to various etiologies [24], which often progresses to cirrhosis. PDGF is the predominant stimulator for the proliferation of hepatic stellate cells (HSCs). Previous report suggested that PDGF-B and its receptor played an important role in the promotion of hepatic fibrosis of mice model [5, 6, 25] and, for this reason, we chose PDGF as a detection index for liver fibrosis in NAFLD. Figure 1 shows that in the HF group with a high-fat diet for 16 weeks, the PDGF expression was significantly increased in comparison with NC group, indicating that liver tissues in HF group appeared to be more susceptible to fibrosis. Of note, there was a remarkable decline in the PDGF expression level in these high-fat diet-fed mice upon pioglitazone treatment, consistent with results of the histological examination (Fig. 3). These findings suggest that, once NAFLD develops, there is an inherent risk in fibrosis progression. The mechanism underlying how pioglitazone restrains the process of hepatic fibrosis might be attributed to suppression PDGF expression in mesenchymal cells, followed by the inhibition of HSCs activation and related signal transduction pathways.
TIMPs are a family of secreted glycolproteins that can suppress the activity of almost all metalloproteinases (MMPs). Previous reports indicated that the expression of TIMP-1 and TIMP-2 was significantly higher in animal models of hepatic fibrosis induced by a variety of chronic liver diseases [26, 27]. Particularly, TIMP-2 suppresses the apoptosis of HSCs and promoted their proliferation. Figure 2 shows that pioglitazone treatment reduced the expression of TIMP-2 and further compromised its inhibitory effect on MMPs, which are involved in the degradation of collagen fibers in our mouse model of NAFLD.
This study evaluated the effects of pioglitazone on NAFLD and attempted to explore the molecular mechanisms for the first time. Taken collectively, our data support the notion that pioglitazone participated in the down-regulation of PDGF and TIMP-2, which sequentially inhibited the activation of HSCs and promoted HSCs apoptosis. However, this speculation remains to be verified by in vitro study, where the viability and apoptosis of HSCs would be assessed upon pioglitazone treatment. Moreover, the roles that PDGF and TIMP-2 genes play in the anti-fibrotic effects induced by pioglitazone require further investigation.
The PPAR-
Conclusions
We conclude that our study, in agreement with a previous pilot research [29] prompts for further evaluation of PPAR-
