Abstract
BACKGROUND:
Cancer progression has been associated with altered immune cell function and activation. Neopterin, which is secreted by interferon-
OBJECTIVE:
The goal of this discovery study was to screen chitotriosidase for diagnostic capacity in detecting cancer and compare its operating characteristics with those of neopterin.
METHODS:
Serum from subjects with breast (
RESULTS:
Chitotriosidase activity was significantly higher in both cancer types compared with gender matched controls, though only in breast cancer was the diagnostic capacity significant (area under the ROC curve of 0.97
CONCLUSIONS:
The operating characteristics of serum chitotriosidase were different from neopterin and further analysis of chitotriosidase as a biomarker for breast cancer is warranted.
Introduction
Tumor growth and progression is associated with inflammation, altered immune function and activation [21]. The tumor microenvironment is characterized by the presence of host leucocytes in the tumor and its surrounding stroma [5]. Tumor-associated macrophages are a significant component of inflammatory infiltrates in malignant tumor tissues. They play an important role in cancer progression by stimulating tumor-cell proliferation, promoting angiogenesis, tumor invasion and metastasis [4, 5] and are associated with poor prognosis in a variety of human cancers [34]. Neutrophils are normally relatively short-lived rapid responders to inflammation. Tumor associated neutrophils have also been associated with tumor progression and poor prognosis [16, 28, 39].
Neopterin, a GTP metabolite, is a product of human monocyte-derived macrophages stimulated by activated T cell release of interferon-
Chitotriosidase, a member of the glycosyl hydrolase family, is a secreted enzyme that catalyzes the hydrolysis of both chitin and chitin-like substrates [30]. Chitotriosidase serves as a diagnostic marker of Gaucher disease where tissue resident macrophages are chronically activated and secrete high levels [14]. Chitotriosidase production was also found to be associated with pathogen-driven diseases, particularly with chronic fungal infection and with reticulo-endothelial activations, indicating its role as an innate immunity component of the macrophage-driven inflammatory process [20, 42]. Neutrophils can also be a potential source of serum chitotriosidase activity, releasing it from granules when stimulated with granulocyte-macrophage colony-stimulating factor [42]. The serum distribution of chitotriosidase has not been described in the setting of cancer.
We hypothesized that serum chitotriosidase activity would reflect a tumor extrinsic (host) contribution to modifying disease progression and, given its association with chronic inflammation, that chitotriosidase would exhibit greater sensitivity and specificity than neopterin for cancer detection. Hence, the current discovery study characterized a novel cancer biomarker, chitotriosidase, and compared it with the extensively studied innate immune biomarker neopterin. The bio- markers were characterized in terms of distribution and diagnostic capacity using serum from normal subjects and subjects with breast or prostate cancer. Breast and prostate cancer share a number of similarities including steroid hormone modulation, stromal cell/tumor microenvironment promotion of disease progression, and immune system changes that facilitate cellular transition through different oncogenic steps [31]. Biomarker associations with physiologic covariates (age, BMI, % body fat) and clinical measures were also determined.
Materials and methods
Sample selection
Serum from 66 subjects with breast cancer (
Clinical measures
Anthropometric measures included height and weight. Foot-to-foot bio-impedance analysis was conducted to estimate percentage of body fat (% fat) using a Tanita scale (Tanita Corporation of America; Arlington Heights, IL). The physiological covariates associated with each specimen included age, gender, BMI and % body fat. Clinical findings associated with the serum samples included Union for International Cancer Control (UICC) staging, estrogen receptor (ER), progesterone receptor (PR) and HER-2 neu expression status for subjects with breast cancer and prostate-specific antigen levels (PSA) for subjects with prostate cancer. The hormone sensitivity of the prostate tumors was not determined. The study was approved by Johns Hopkins Bayview Medical Center institutional review board.
Study subject characteristics
Study subject characteristics
Values for age, BMI and % body fat are mean
Serum specimens were obtained in a resting and fasting state in the morning. All venous samples were placed at 4
The lower limit of detection (LLOD) and lower limit of quantitation (LLOQ) were determined for the chitotriosidase assay following methodology proposed by the International Council for Harmonization [33]. LLOD was calculated using the formula LLOD
Statistical analysis
All statistical calculations were carried out using Prism and InStat software (GraphPad, Inc.). The comparison of variables or serum markers between groups were performed by using a Mann-Whitney U-test. The relationship between specificity and sensitivity for each marker was profiled by receiver operating characteristic (ROC) curves. Optimal cut off values for biomarkers were identified using Youden’s J statistic. Correlations between serum markers and individual physiologic covariates or clinical measures were assessed using Spearman rank correlations. There were limited numbers of subjects in some clinical stage, hence cancer samples were divided into two groups for comparison: those with localized disease and those with evidence of distant metastasis.
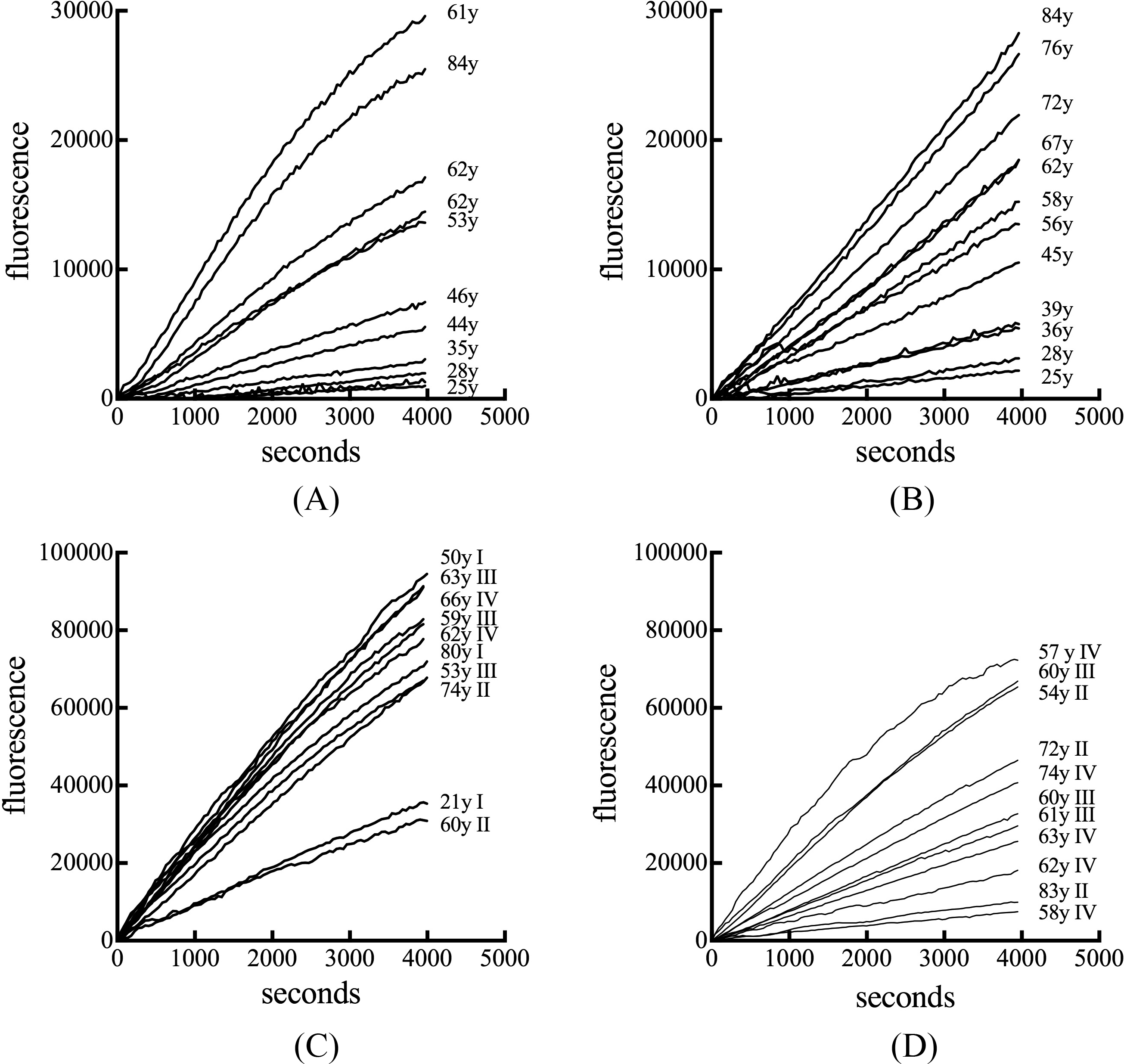
Chitotriosidase activity in human serum. Representative profiles of the evolution of fluorescent enzymatic product as a function of time are shown for serum obtained from normal female subjects (A), normal male subjects (B), subjects with breast cancer (C), and subjects with prostate cancer cancer (D). Age and cancer stage are indicated.
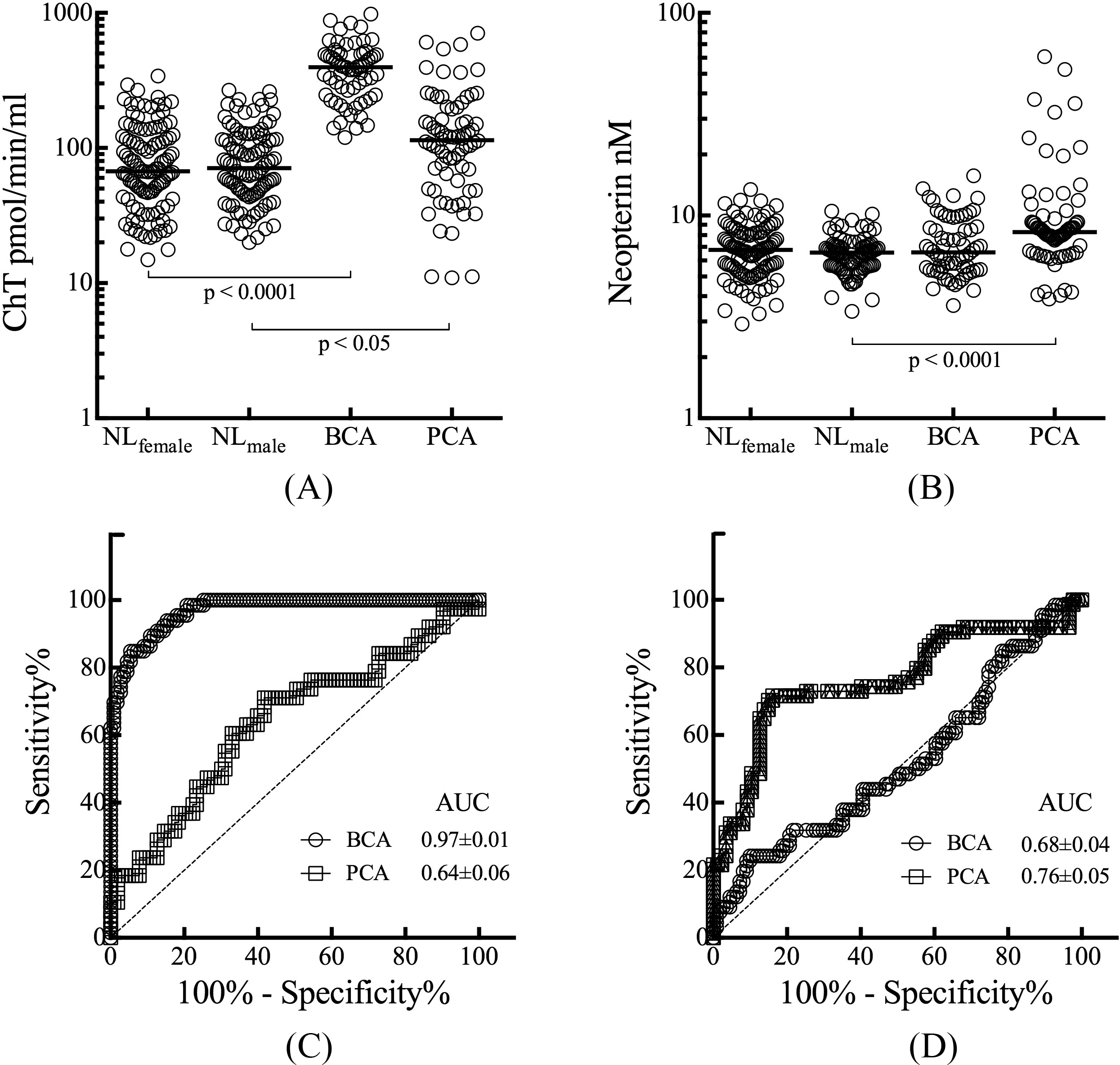
Distribution and discriminatory capacity serum markers cancer. Levels of chitotriosidase (A) and neopterin (B) were compared between normal females (NL
REMARK
The characteristics of normal and cancer subjects are listed in Table 1. Age was significantly higher in breast cancer subjects when compared to the normal female serum donors (unpaired t-test
An enzymatic assay was employed to quantify the amount of chitotriosidase present in serum. An age-dependent difference in the rate of substrate conversion into product was apparent in serum from normal female and male subjects (Figs 1A and 1B). Serum from subjects with breast cancer exhibited elevated enzyme activity rates independent of age (Fig. 1C). Prostate cancer serum also exhibited no age-associated change in enzyme activity and their rates of substrate conversion were less then in breast cancer serum (Fig. 1D). The median chitotriosidase activity for serum from normal female donors was 67.3 (range 14.8–340.8) pmol/min/ml which was not significantly different from the median value of 70.2 (range 20.0 to 267.1) pmol/min/ml for serum from normal males (Fig. 2A). Serum from subjects with breast cancer had a median chitotriosidase activity of 396.4 (range 119.7 to 978.4) pmol/min/ml,
The median value of neopterin was 6.77 (range 2.92 to 13.43) nM in serum from normal female donors and 6.56 (range 3.37 to 10.50) nM in serum from normal male donors, not significantly different (Fig. 2B). The median value of serum neopterin was 6.58 (range 3.60 to 15.69) nM in breast cancer subjects, no different from that in gender matched normal serum. A median neopterin value of 8.28 (range 3.88 to 60.74) nM in prostate cancer subjects was significantly higher from that in normal male serum,
The discriminatory capacity of serum levels of chitotriosidase to detect cancers was profiled by ROC curve analysis (Fig. 2C). For distinguishing between breast cancer subjects and disease-free females, the area under the curve (AUC) was significant for chitotriosidase with an AUC of 0.97 (95% confidence interval 0.95–0.99), (
The capacity of serum levels of neopterin to detect cancer was also profiled by ROC curve analysis (Fig. 2D). The AUCs were 0.68 (95% confidence interval 0.42 to 0.59,
Associations between the two biomarkers and physiologic covariates were investigated next. Significant correlations were observed in normal serum between age and chitotriosidase with a Spearman
In contrast, neopterin was significantly correlated with age in serum from normal females (Spearman
The relationship between the two biomarkers and clinical measures were assessed. For chitotriosidase in breast cancer, there were no significant associations with receptor status. The median chitotriosidase activity in localized breast cancer was 387.4 (range 119.7 to 978.4) pmol/min/ml versus 443.1 (range 191.8 to 786.1) pmol/min/ml for metastatic breast cancer. The values were not significantly different. In prostate cancer, serum chitotriosidase did not correlate with PSA levels. The median chitotriosidase activity in localized prostate cancer was 93.8 (range 11.2 to 583.8) pmol/min/ml while metastatic prostate cancer had a median chitotriosidase activity of 130.6 (range 37.1 to 606.9) pmol/min/ml, not significantly different.
Neopterin also had no significant associations with receptor status in breast cancer. In contrast to chitotriosidase, neopterin exhibited a number of associations with other clinical measures. The median value of neopterin in localized breast cancer was 6.34 (range 3.60 to 15.69) nM versus 10.02 (range 5.54–15.69) nM for metastatic breast cancer,
Discussion
Based on 2010–2012 data, at some point during their lifetime approximately 12% of women will be diagnosed with breast cancer and 14% of men will be diagnosed with prostate cancer [15]. Age-adjusted annual death rates per 100,000 for 2012 were 21.9 for breast cancer and 21.4 for prostate cancer [1]. Cancer progression and metastases are the cause of 90% of human cancer deaths [22]. Chronic immune activation is believed to contribute to tumor growth and metastasis [9, 32]. Validated tumor markers in breast cancer include tissue based ER, PR, and HER-2, status [36]. In the current study, there was no association between ER, PR and HER-2 status with neopterin or chitotriosidase levels. This is consistent with hormone and growth factor receptors being tumor intrinsic markers. whereas chitotriosidase and neopterin reflect the tumor extrinsic host immune response. Serum tumor markers that are clinically used for breast cancer are CA 15-3, CA 27-29, and CEA (carcinoembryonic antigen). However, due to their low sensitivity and specificity, the American Society of Clinical Oncology expert panel in breast cancer recommends using those serum tumor markers in metastatic disease only [13]. For prostate cancer, PSA has been used for screening purposes though it does not have ideal specificity as it can be elevated in certain benign conditions such as acute prostatitis and benign prostatic hyperplasia and approximately 15% of PCA cases occur in men with very low serum PSA values [3, 41]. The identification of novel candidate diagnostic biomarkers for both cancer types remains a priority.
During cancer progression, activated tumor associated macrophages and neutrophils can promote genetic instability, tumor growth, angiogenesis and invasive behavior [12]. In the current study, markers associated with macrophage and neutrophil activation were correlated with physiological and clinical correlates in breast and prostate cancer. The earliest stage of breast cancer was associated with significant elevation of chitotriosidase, while neopterin was significantly elevated in late stage metastatic disease. The observation of elevated neopterin levels in subjects with metastatic disease are in agreement with previous studies demonstrating that higher serum levels of neopterin were associated with poor prognosis and the presence of distant metastasis in breast and prostate cancer [10, 43]. The age- and BMI-associated contributions to biomarker levels observed in the current study are consistent with observations in other populations. Chitotriosidase was reported to exhibit an age-associated increase in serum levels in normal subjects [29]. The biomarker neopterin has been reported to be influenced by both age and BMI in serum from normal and obese subjects and subjects with sleep apnea [27, 35].
Breast and prostate cancer share similar biological (sex hormone sensitive and insensitive states) and epidemiological characteristics (incidence and mortality). For both breast and prostate cancer, a high neutrophil to lymphocyte ratio (a marker of inflammation) was prognostic for worse overall survival and disease-free survival [8, 11]. Despite these similarities, differences in underlying pathophysiology have been observed. Chronic inflammation was higher in biopsies from men with a benign prostate versus those with prostate cancer and inversely correlated with tumor volume in a two year repeat biopsy in men who had a negative baseline biopsy [17, 23]. In the current study, elevated chitotriosidase activity associated with breast but not prostate cancer may be indicative of a further difference between the two cancer types. Chitotriosidase is a marker of the chronic activation of the dedicated phagocytic cells - macrophages and neutrophils, hence there may be an organ-specific difference in how these immune cells interact with the tumor microenvironment.
Limitations to this study include that it involved relatively small discovery groups, the serum samples were cross-sectional and considerable overlap between normal and cancer values were evident in some cases. Measuring circulating levels of a biomarker does not necessarily identify the secreting cells, so it is unclear whether macrophages or neutrophils or some combination may be contributing to altered levels of chitotriosidase in breast cancer. Additionally, as potential biomarkers for cancer, chitotriosidase and neopterin may lack disease specificity in that they are induced in a number of other pathological states. Neopterin serum levels are increased in viral infections, various malignant disorders, autoimmune diseases, liver, kidney and heart disease [6, 26]. Serum levels of chitotriosidase are elevated in Gaucher disease [7] and in other disorders associated with chronic macrophage activation including Wegener’s granulomatosis [19], and sarcoidosis [40]. A large case – control validation study of chitotriosidase as a marker of early stage breast cancer would address a number of these limitations and is being planned.
Conclusion
Chitotriosidase exhibited a significant ability to distinguish between disease-free subjects and subjects with diagnosed breast cancer. Neopterin, though elevated in metastatic disease, did not exhibit diagnostic capacity in breast or prostate cancer. Further studies should be done to validate chitotriosidase as a biomarker and to investigate the biology of macrophage and neutrophil activation in breast cancer progression.
Footnotes
Acknowledgments
This research was supported by Department of Defense, CDMRP grant BC010478 (N.S.F.), T35 AG026 758 (R.R. and A.K.). and NIH T32 training grant 5T32AG000120 (M.S.T.). The authors declare that they have no conflict of interest. Drs. Thein, Kohli, and Fedarko participated in the conception and design of the study. Drs. Ram and Jain and Ms. Ingaramo developed the methodology to measure chitotriosidase in serum. Data was acquired by Ms. Ingaramo and Drs. Thein, Kohli, and Ram. Drs. Thein, Kohli, Jain and Fedarko performed the analysis and interpretation of data. The study was supervised by Drs. Thein and Fedarko. Writing and reviewing of the manuscript was carried out by all authors.
Conflict of interest
None.
