Abstract
The applications of laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS) in biomedical research are progressively increasing in the last decade. The main advantage of this powerful analytical technique is the ability to perform multi-elemental analysis directly from biological samples, such as tissues, organs, and single cells, with minimal sample preparation. This feature is particularly important for studies regarding disease biomarkers and mechanism of action of drugs. In this mini-review, the recent advances in the use of LA-ICP-MS for the analysis of biological samples will be discussed, as well as representative biomedical applications.
Introduction
Laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS) is an analytical technique that enables direct multi-elemental analysis and isotope ratio measurements at the trace and ultra trace level in solid samples [3]. Nowadays, it is the most employed technique for mass spectrometry imaging (MSI) of metals, semimetals, and non-metals in biological samples, such as tissues and organs, with a spatial resolution usually in the micrometer range [32].
LA-ICP-MS employs a focused laser beam to ablate material from the surface of a solid sample with a thickness of 5–30
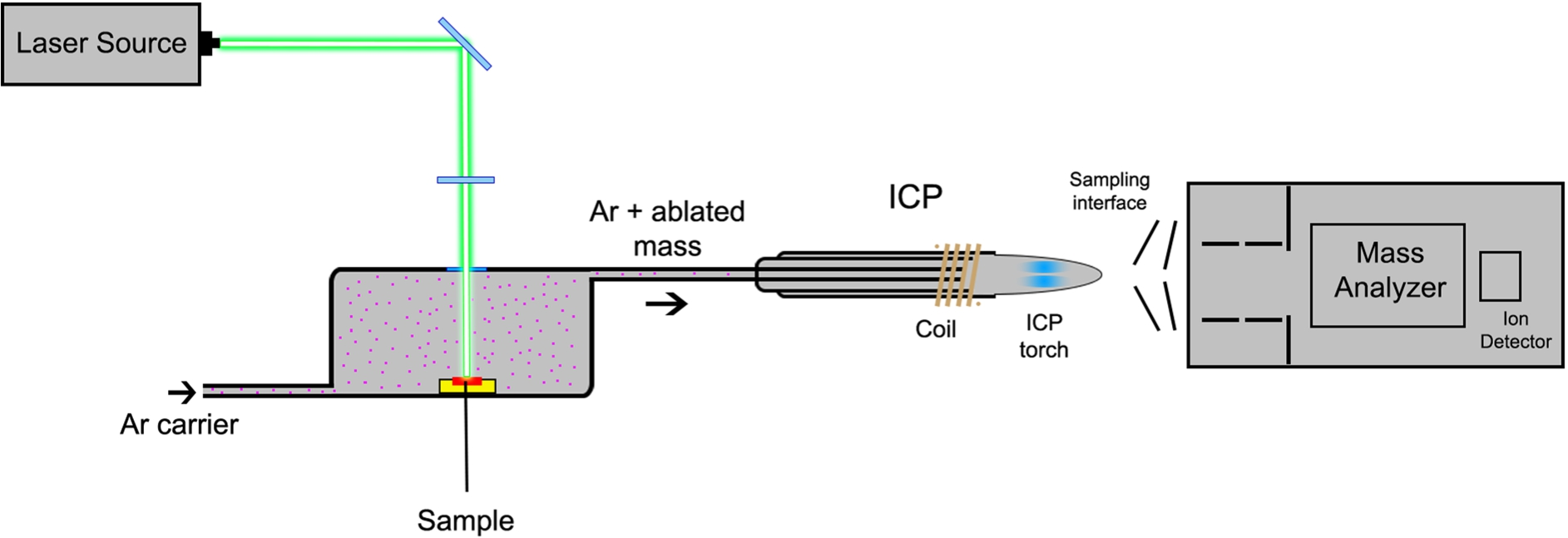
Schematic diagram of a LA-ICP-MS instrument.
The key parameters that have to be optimized in a LA-ICP-MS experiment are laser spot size, scan speed, and repetition frequency. These parameters influence the ablation efficiency and therefore the sensitivity of the measurements [32].
The precision and accuracy of LA-ICP-MS measurements are directly dependent on the calibration, standardization, and normalization steps. In the case of biological samples, certified reference standards are usually not commercially available, which requires the preparation of laboratory prepared matrix-matched standards (a homogenized tissue spiked with different concentrations of aqueous standards from the analytes) for calibration and standardization [35]. For signal normalization, the measurement of surrogate markers of slice thickness, such as carbon or sulfur, or internal standards is necessary. The most common normalization strategies are based on the measurement of one isotope, the total ion current (TIC), or selected isotopes that represent the ion current related to the sample matrix (extracted ion current, EIC) [17].
LA-ICP-MS can be operated in three different modes: imaging, line scan, and micro local analysis of selected spots [33]. When LA-ICP-MS is used in the imaging mode, the reconstruction of images for visualization of elemental distribution in the analyzed sample is also an important step that will guide the interpretation of the biological question under investigation. Different software tools can be used with this purpose, from commercial to open source ones, such as ELAI [36] and LA-iMageS [27].
LA-ICP-MS has been applied to obtain quantitative images of regionally specific element distributions in sections of different biological tissues, such as mouse brain, kidneys, among others [28,34] as reviewed in the literature [5,6,32]. In the following sections, the recent developments in LA-ICP-MS technique and selected biomedical applications will be discussed.
Multimodal imaging, combining LA-ICP-MS with other imaging techniques, is an analytical strategy of growing interest, since it combines the elemental distribution information with structural features, thus improving data that support unraveling complex biological processes.
Recently, Ackerman
Direct biofluid analysis by LA-ICP-MS is also a topic being continuously explored. A recent approach for serum analysis by LA-ICP-MS using dried serum spot (DSS) sampling, using only 20
In terms of instrumentation, recent improvements in the development of low dispersion ablation cells have significantly enhanced throughput and sensitivity for high-resolution LA-ICP-MS imaging, thus increasing the scope of LA-ICP-MS applications, mainly for single cell analysis and sub-cellular imaging [37,38]. The development of these LA cells combined with the use of time-of-flight (TOF) mass analyzers, instead of the common quadrupole, are indeed relevant for the advance of high-resolution LA-ICP-MS imaging [8].
Nowadays, the current hot topic in the use of LA-ICP-MS in biomedical applications is the analysis of single cells, which will be detailed in the following section.
Single cell analysis
LA-ICP-MS is being proposed as an elemental microscopy technique, since it has quantitative features when matrix-matched calibration standards are used, besides the ease of sample preparation and multi-elemental detection capabilities with high sensitivity and spatial resolution. For co-localization of cells and analytes, LA-ICP-MS profiles are usually correlated with a bright field image of the cell sample [20] or other imaging approaches, such as fluorescence confocal microscopy [37]; thus, multimodal imaging is widely applied for single cell elemental analysis.
The elemental analysis of single cells can be performed using the cells in solution (SC-ICP-MS); however, this type of analysis normally requires at least thousands of single cells in a sample, which is different from the analysis of single cells by LA-ICP-MS. Nevertheless, LA-ICP-MS presents a limitation due to the insufficient analytical speed of quadrupole mass analyzers, which separate the ions with basis on the sequential scanning of isotopes, thus impeding the determination of more than two isotopes during a transient signal from a single cell [7].
Currently, microscopic targets can be
A limiting point of using LA-ICP-MS at micrometer scale is the reduced laser spot size, which leads to a reduction of ablated sample material and therefore to a decreased number of detectable ions in the analyzed area, thus resulting in a compromise between sensitivity and spot size [20]. Another limitation of the single cell LA-ICP-MS imaging is the depth resolution, since the laser beam penetrates through the entire cell, and the signal from the cytosol is monitored together with the nucleus [39].
In order to overcome this issue, Herrmann
Recently, van Malderen
Precision, accuracy, and efficiency of current calibration strategies are also an important point to be considered in a single cell LA-ICP-MS experiment [21]. Van Malderen
Single cell analysis by LA-ICP-MS is being widely used for studies involving the exposure to metal nanoparticles. Zheng
Applications to biomedicine
Table 1 summarizes biomedical applications of LA-ICP-MS technique in the last 5 years. The authors would like to apologize for any relevant work not included in this mini-review. As can be noticed from this table, an important number of applications involves neurodegenerative disorders, the distribution of metallodrugs and exposure to nanoparticles. Therefore, some of these applications will be discussed in the following sections.
Recent LA-ICP-MS biomedical applications
Recent LA-ICP-MS biomedical applications
Wilson’s disease (WD) is a rare genetic disorder where copper concentration in body tissues, particularly in liver, brain and corneas, becomes excessive. Some common symptoms associated to this disorder include vomiting, weakness and yellowish skin [2]. The treatment of this disorder includes medications like penicillamine (Cuprimine), which remove copper from the body (chelation therapy) by urinary excretion and zinc salts, which prevent the gut absorbing copper from the diet.
LA-ICP-MS has been applied successfully in studies about Wilson’s disease. Hachmöller
Gadolinium distribution
Gadolinium-based contrast agents are widely used for magnetic resonance imaging (MRI) examinations and are recognized for being rapidly excreted and not metabolized. However, recent studies showed that Gd traces persists in human brain. Fingerhut
Other approaches for the study of Gd deposition were developed by Löhofer
Conclusion
LA-ICP-MS has been extensively used in biomedical research, providing multi-elemental information in a diversity of biological samples. Due to the improvement in the instrumentation, notably in the newly developed low dispersion LA cells and the use of TOF or QqQ as mass analyzers, LA-ICP-MS is able to enhance the information regarding the mechanisms of diseases, as well as the biological effects of metallodrugs and metal nanoparticles in a variety of tissues and also in single cells. Multimodal imaging, direct biofluid analysis, and single cell analysis by LA-ICP-MS have been improved in the last few years, which is particularly interesting for biomedical studies.
Footnotes
Acknowledgements
A.S. would like to thank FAPESP (grant number 2018/01525-3) for financial support.
Conflict of interest
The authors have no conflict of interest to report.
