Abstract
Cholesterol, an essential component of higher eukaryotic membranes, was discovered more than two centuries ago. The development and progress of cholesterol research in the last 200 years has been truly fascinating, with elements of surprise, serendipity and intrigue. In this review, we trace this journey the way we see it, and follow it up with the role of membrane cholesterol in crucial areas of contemporary research (transbilayer domains, regulation of GPCR function and role in the entry of intracellular pathogens into host cells), with considerable footprint from our work. We believe that cholesterol will continue to surprise and fascinate future researchers, thereby justifying its evergreen nature.
Abbreviations
7-dehydrocholesterol
25-[ cholesterol recognition/interaction amino acid consensus dehydroergosterol (Δ5,7,9(11)22-ergostatetraen- 1,2-dipalmitoyl- fluorescence recovery after photobleaching G protein-coupled receptor 7-nitrobenz-2-oxa-1,3-diazol-4-yl red edge excitation shift Smith-Lemli-Opitz syndrome
Cholesterol: An ancient molecule with a remarkable history
Cholesterol was initially discovered as a component of human bile and gallstone sometime around 1760 AD by Poulletier de la Salle, a French physician and chemist (see Fig. 1) [13]. The naming of cholesterol was done by another French chemist Michel Chevreul in 1816. He named it cholesterine (
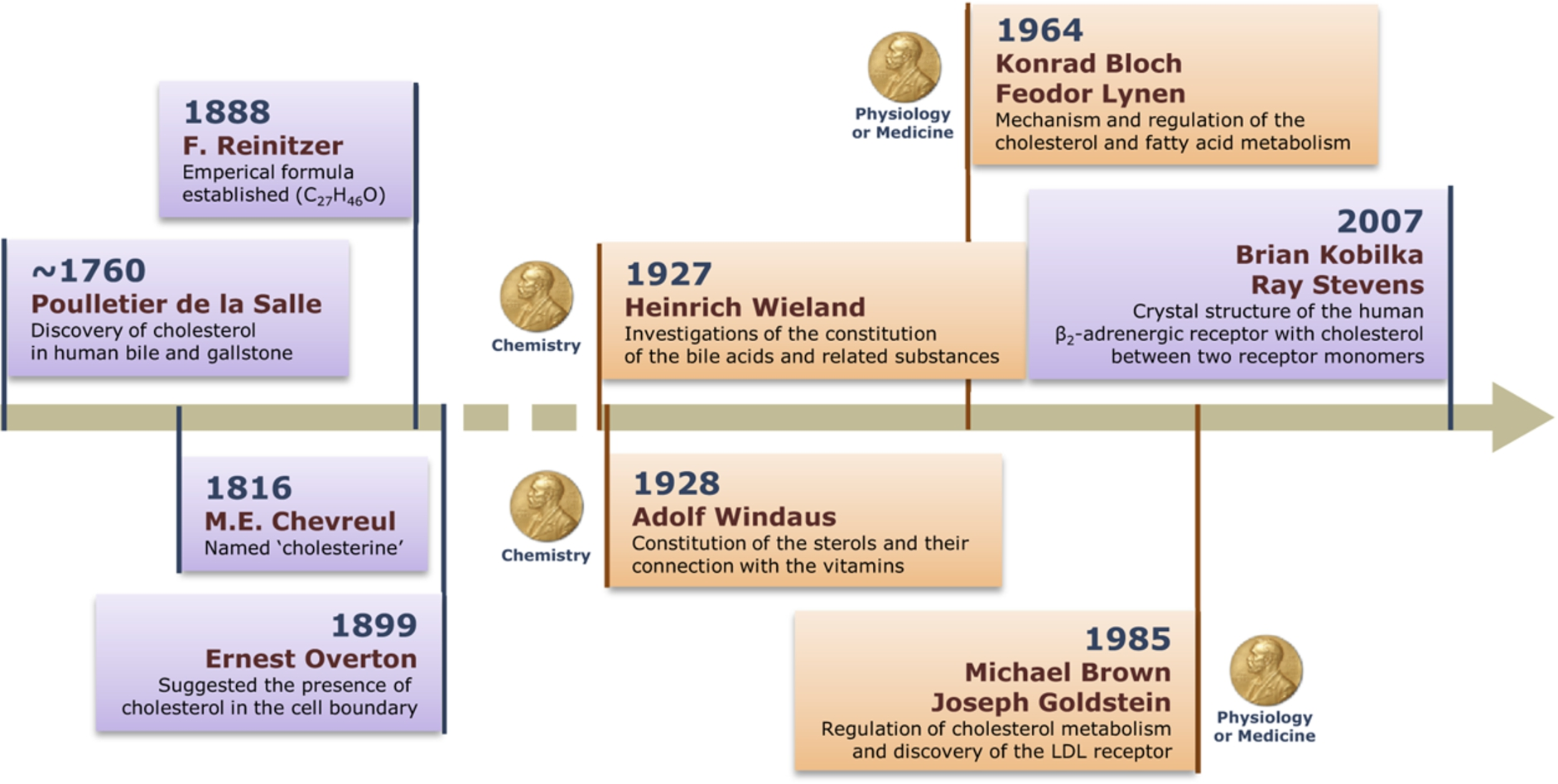
The journey of cholesterol: 200 years and beyond. A timeline depicting landmark discoveries associated with cholesterol, and its structure, function and metabolism. Importantly, discoveries related to cholesterol have been recognized by the award of several Nobel Prizes, due to its importance in cellular physiology, health and disease.
In this review, we will first highlight some unique structural features of cholesterol that are closely related to its functionality. This will be followed up with highlights of the role of cholesterol in three representative areas of membrane biology. These topics (transbilayer domains, regulation of G protein-coupled receptor (GPCR) function and role in the entry of intracellular pathogens into host cells) are chosen primarily because of our previous and ongoing work in these areas.
The molecular structure of cholesterol (see Fig. 2) is characterized by a number of distinct features, all of which combine to make cholesterol a unique molecule in functional terms. Cholesterol is a predominantly hydrophobic molecule comprising a near planar tetracyclic fused steroid ring and a flexible isooctyl hydrocarbon tail. The
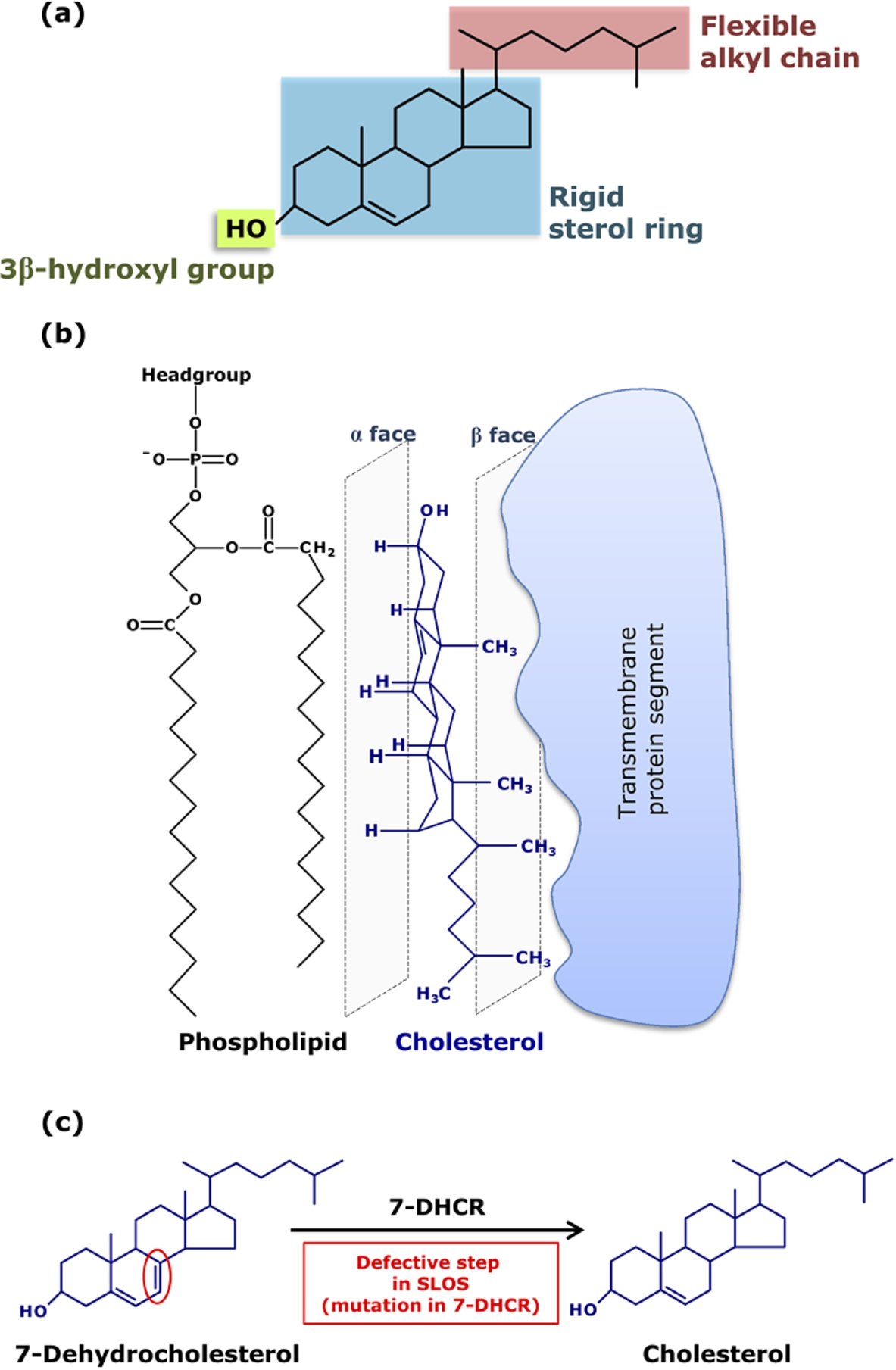
Key structural features of cholesterol and its interaction with membrane proteins and diseases resulting from defective cholesterol biosynthesis. (a) Chemical structure of cholesterol depicting its three structurally distinct regions, each outlined in a shaded box: a polar
The molecular structure of cholesterol is exceedingly fine-tuned over a very long time scale of natural Darwinian evolution. This is exemplified by a number of inherited disorders, such as the Smith-Lemli-Opitz syndrome (SLOS; see Fig. 2(c)), the molecular etiology of which lies in subtle defects in cholesterol biosynthesis [3,35,40,42]. SLOS is a congenital and developmental malformation syndrome associated with defective cholesterol biosynthesis. In SLOS, the immediate precursor in the Kandutsch-Russel pathway of cholesterol biosynthesis is 7-dehydrocholesterol (7-DHC), which differs with cholesterol
Cholesterol is an essential lipid in higher eukaryotic cellular membranes and is crucial in membrane organization, dynamics, function, and sorting [27,54]. A hallmark of the organization of membrane cholesterol is its nonrandom distribution in domains [30]. A unique property of cholesterol that contributes to its capacity to form membrane domains, when present at high concentrations, is its ability to form liquid-ordered-like phase in higher eukaryotic plasma membranes [26]. Many of these domains (sometimes termed as ‘lipid rafts’ [22]) are thought to be important for the maintenance of membrane structure and function, although characterizing the spatiotemporal resolution of these domains has proven to be challenging [53].
Interestingly, cholesterol is distributed heterogeneously among various intracellular membranes. The lowest cholesterol concentration is found in membranes of the endoplasmic reticulum, which is the site of cholesterol biosynthesis. Cholesterol concentration increases progressively along the
Evidence for specific organization of cholesterol in membranes at low concentrations was obtained from three independent studies carried out by various groups ([7,15,16,28]; reviewed in [10]). Harris et al. [16] monitored the cryoscopic depression of multilamellar vesicles of DPPC/cholesterol and explained their results on the basis of transbilayer dimer formation in the fluid phase at low concentrations. Differential scanning calorimetric studies showed that there was a sharpening of the phase transition (due to increase in the size of cooperative unit) by cholesterol at very low concentrations, instead of the broadening expected if chain melting was accompanied by continuously changing compositions of the two phases [16]. Further analysis of cholesterol-DPPC interaction, monitored by lowering of transition temperature as a function of increasing cholesterol concentration in DPPC membranes showed that the slope of the curve (of transition temperature
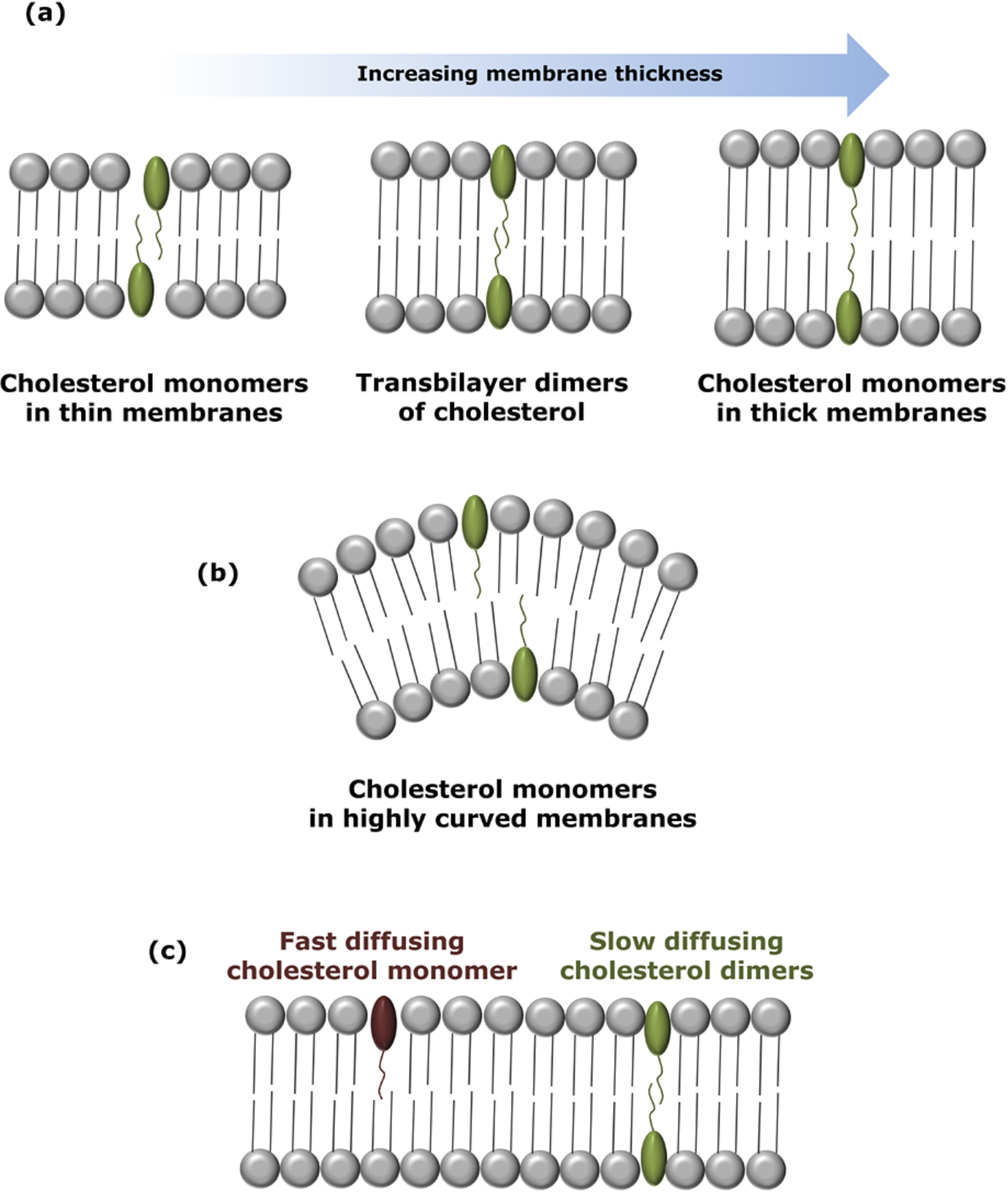
Organization and dynamics of transbilayer dimers of cholesterol at low concentrations. The schematic diagram shows the organization of cholesterol dimers in (a) membranes of varying thickness. The existence of cholesterol as transmembrane tail-to-tail dimers is stringently controlled within a narrow window of membrane thickness. Insights from our previous work suggest that cholesterol exists as monomers in relatively thin and thick membranes, while it forms transmembrane tail-to-tail dimers in membranes of intermediate (optimum) thickness. (b) Cholesterol dimers are unstable at high surface curvature. (c) Monomers of cholesterol show faster lateral dynamics in membranes relative to transbilayer tail-to-tail dimers of cholesterol (see [46] for details).
Mukherjee and Chattopadhyay [28] utilized aggregation-sensitive fluorescence characteristics of 25-NBD-cholesterol, a fluorescent analogue of cholesterol, to study the local organization of cholesterol in the membrane at very low concentrations. Lipids labeled with the NBD group have been extensively used as fluorescent probes to monitor membrane environment and dynamics due to a number of excellent photophysical properties of the NBD group [7,15]. By careful analysis of the emission spectral features of 25-NBD-cholesterol in DPPC membranes at low concentration, the possible presence of transbilayer tail-to-tail dimers (Fig. 3(a)) of cholesterol in such membranes was detected both in gel and fluid phases.
These results were further validated by Loura and Prieto [23] by utilizing the intrinsic fluorescence of DHE, a naturally occurring fluorescent cholesterol analogue found in yeast. Loura and Prieto [23] analyzed DHE concentration-dependent depolarization in membranes using a theoretical framework and explained their results on the basis of transbilayer tail-to-tail dimers of cholesterol, previously reported by us [28] and others [16]. These conclusions were further supported by measurements in which the change in peak intensity ratio of the vibronic bands of DHE was monitored as a function of DHE concentration [50]. In addition, it was indicated that cholesterol dimers are sensitive to membrane curvature [50].
The effect of membrane curvature stress on the transbilayer organization of cholesterol dimers in membranes was comprehensively addressed by us [50]. By monitoring the distinct spectral feature of 25-NBD-cholesterol in membranes of varying curvature, we showed that the transbilayer dimer arrangement is sensitive to membrane curvature and dimerization is not favored in highly curved membranes (see Fig. 3(b)). This was attributed to differential packing arrangements in the two leaflets of highly curved membranes. Interestingly, cholesterol dimerization was favored upon release of curvature stress [50]. Furthermore, the transbilayer cholesterol dimers exhibited sensitivity to membrane thickness (see Fig. 3(a)). Taken together, these results showed that the process of transbilayer dimerization is stringently controlled by a narrow window of membrane thickness and curvature (see Fig. 3(a), (b)). These results assume relevance in the context of increasing membrane thickness from Golgi to the plasma membrane due to the gradient in cholesterol concentration [5]. We later showed, using wavelength-selective FRAP [46] and REES [29] that the dynamics of the cholesterol monomer and dimer population is considerably different. The monomer population exhibited faster lateral dynamics and was found to be less ordered than the dimer (Fig. 3(c)).
GPCRs are the largest class of molecules involved in signal transduction across membranes, and represent major targets in the development of novel drug candidates in all clinical areas [8,39,49]. Since GPCRs are integral membrane proteins, interaction of membrane lipids with them constitutes an important area of research in GPCR biology. In particular, membrane cholesterol has been reported to have a modulatory role in the function of a number of GPCRs [14,17,31,34,44,52]. Extensive work has been carried out in case of GPCRs such as the serotonin1A receptor, the
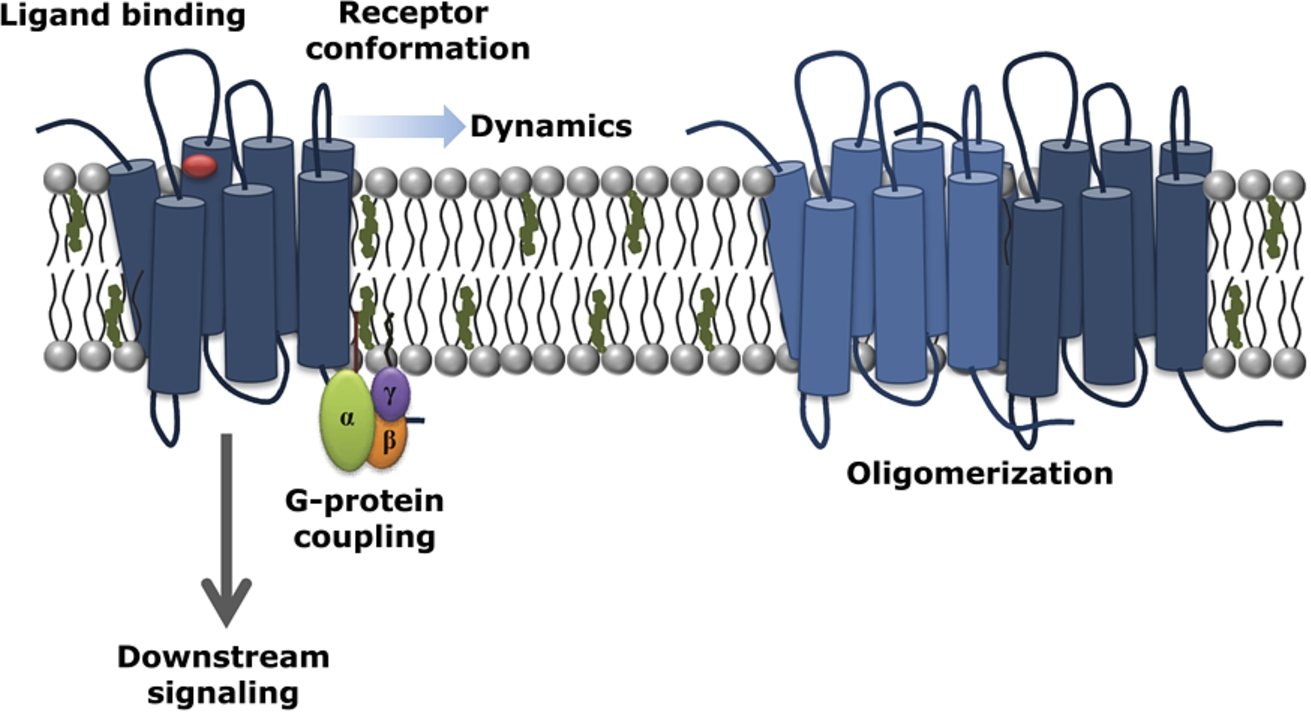
The multifaceted interaction of cholesterol with GPCRs. A schematic representation highlighting the consequences of modulation of membrane cholesterol (shown in green) in GPCR structure, function and organization. Cholesterol has been shown to modulate early events associated with GPCR function such as ligand binding and G-protein coupling, leading to consequences in downstream signaling. Experimental and computational evidences show that membrane cholesterol modulates the organization, dynamics and oligomerization of GPCRs. Such an intimate interplay between GPCRs and cholesterol could arise either due to specific interaction of cholesterol with hotspots on GPCRs where cholesterol enjoys higher occupancy, or due to modulation of membrane physical properties, or a combination of these effects (see [14,17,31,34,44,52] for more details).
We will instead briefly focus on the current understanding of the mechanistic basis of GPCR-cholesterol interaction. It appears that specific effects in terms of cholesterol binding to certain regions (sequences) of the receptor plays a role in these changes [33,37]. Some of these regions (such as the CRAC motif) have been identified [18]. The involvement of these regions in regulation of GPCRs by membrane cholesterol is being investigated for various types of GPCRs by experimental [32] and simulation approaches [51]. Detailed molecular dynamics simulations have revealed that cholesterol binding on GPCRs is weak yet dynamic with an occupancy time varying between ns and
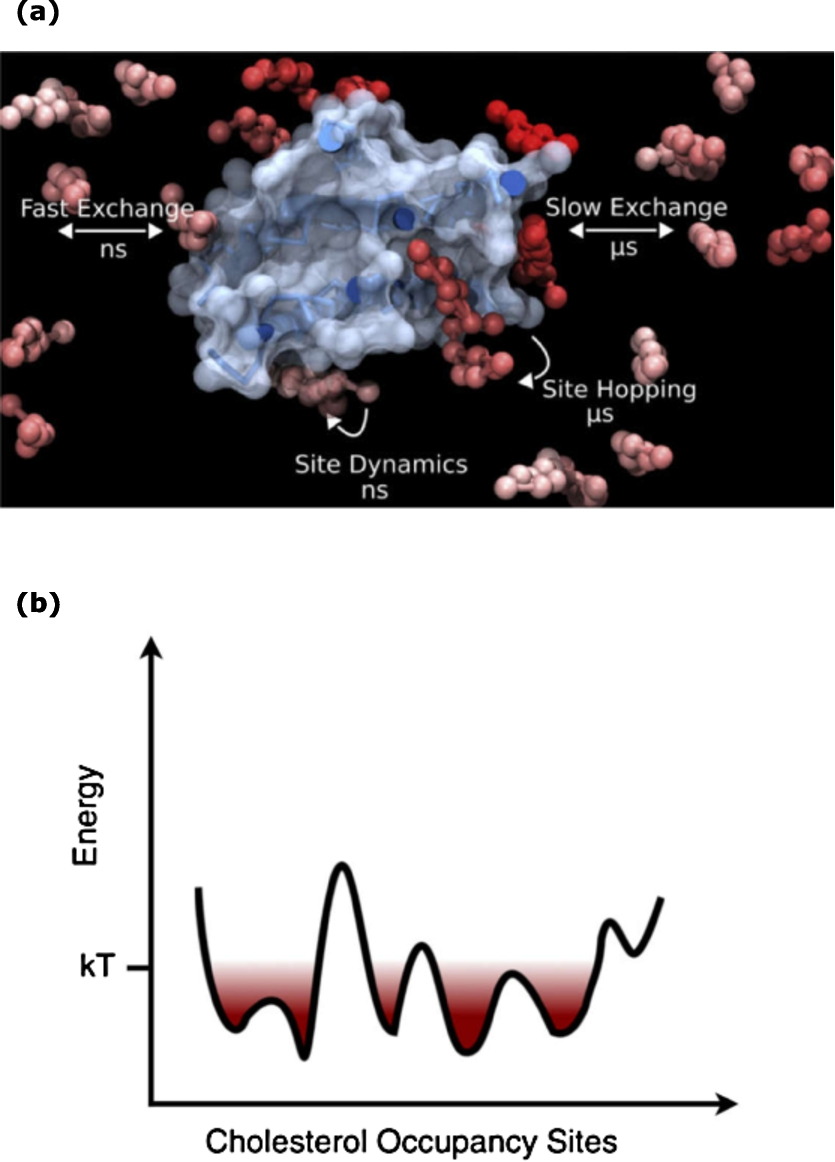
Interaction of cholesterol with GPCRs: dynamic and diverse. (a) The interaction of cholesterol with GPCRs exhibits diversity in terms of its occupancy at the GPCR surface. The interaction is weak but crucial. Cholesterol molecules interacting with receptors exhibit varying timescales of exchange with bulk lipids. (b) The energy landscape corresponding to the interaction of cholesterol with specific sites on the receptor surface involves a series of shallow minima connected by low energy barriers of the order of kT at room temperature. Taken from ref. [52].
The cellular plasma membrane serves as a gateway for the entry of intracellular pathogens. An essential step for an intracellular pathogen to enter into a host cell therefore is to be able to cross the cell membrane. A clever strategy to regulate the entry of the intracellular pathogen (thereby reducing infection) is to change host membrane composition such that pathogen entry is reduced. An effective way to achieve this is to reduce (or block) host membrane cholesterol. This approach has been successfully tried for entry of pathogens such as
We have recently proposed a working mechanism for this approach (see Fig. 6). The crux of the mechanism is that the conformation(s) of the host membrane receptors, responsible for pathogen entry, display cholesterol sensitivity. An optimum level of host membrane cholesterol is found to be necessary for supporting receptor conformation(s) that allow pathogen entry. Either depletion or enrichment of membrane cholesterol results in receptor conformation(s) that do not support the entry of pathogens into host cells (for details, see ref. [20]).
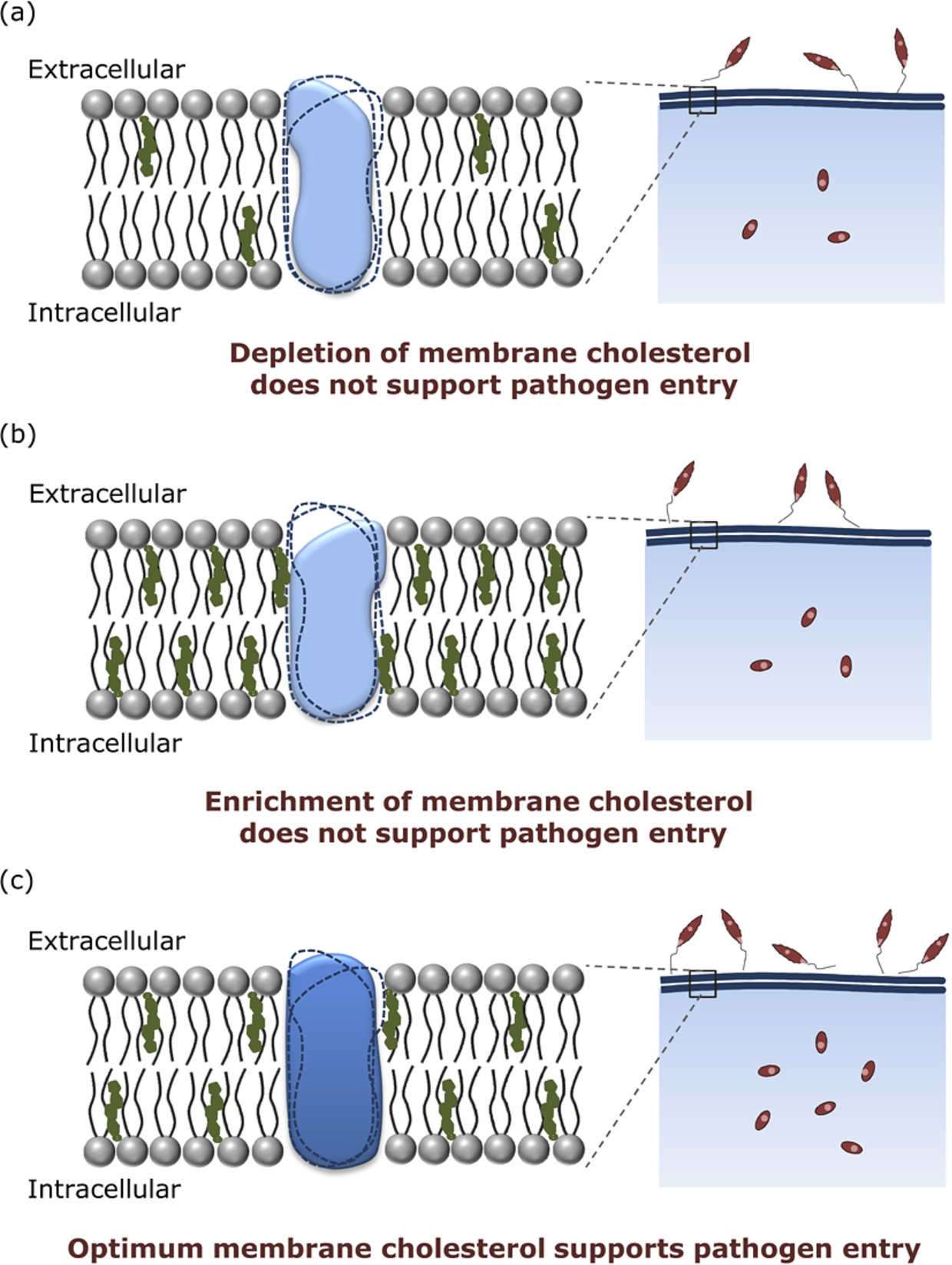
Regulation of pathogen entry by host membrane cholesterol. Host membrane cholesterol modulates the entry of intracellular pathogens (such as
The examples discussed above represent chosen areas where cholesterol has a unique role in membrane organization, function and pathogenicity. The interesting and fascinating aspect of cholesterol in biology is that we are yet to comprehensively understand the whole gamut of biological roles it could play and the crosstalk between pathways influenced by cholesterol. Every once in a while, reports appear in the literature that make us re-think our existing models about cholesterol in biology. For example, cholesterol has been implicated in maturation and signaling of hedgehog proteins involved in embryonic development in
Footnotes
Acknowledgements
Work in A.C.’s laboratory was supported by the Council of Scientific and Industrial Research, Department of Science and Technology, and Department of Biotechnology (Govt. of India). A.C. gratefully acknowledges the support of J.C. Bose Fellowship from the Department of Science and Technology. G.A.K. thanks the Council of Scientific and Industrial Research for the award of a Senior Research Fellowship. A.C. is an Adjunct Professor of Tata Institute of Fundamental Research (Mumbai), RMIT University (Melbourne, Australia), and Indian Institute of Technology (Kanpur). Some of the work described in this article was carried out by present and former members of A.C.’s research group whose contributions are gratefully acknowledged. We thank Md. Jafurulla for help during the preparation of the manuscript and members of the Chattopadhyay laboratory for their comments and discussions.
