Abstract
The birth, maturation, and integration of new neurons in the adult hippocampus regulates specific learning and memory processes, responses to stress, and antidepressant treatment efficacy. This process of adult hippocampal neurogenesis is sensitive to environmental stimuli, including peripheral signals from certain cytokines, hormones, and metabolites, which can promote or hinder the production and survival of new hippocampal neurons. The trillions of microorganisms resident to the gastrointestinal tract, collectively known as the gut microbiota, also demonstrate the ability to modulate adult hippocampal neurogenesis. In doing so, the microbiota-gut-brain axis can influence brain functions regulated by adult hippocampal neurogenesis. Unlike the hippocampus, the gut microbiota is highly accessible to direct interventions, such as prebiotics, probiotics, and antibiotics, and can be manipulated by lifestyle choices including diet. Therefore, understanding the pathways by which the gut microbiota shapes hippocampal neurogenesis may reveal novel targets for non-invasive therapeutics to treat disorders in which alterations in hippocampal neurogenesis have been implicated. This review first outlines the factors which influence both the gut microbiome and adult hippocampal neurogenesis, with cognizance that these effects might happen either independently or due to microbiota-driven mechanisms. We then highlight approaches for investigating the regulation of adult hippocampal neurogenesis by the microbiota-gut-brain axis. Finally, we summarize the current evidence demonstrating the gut microbiota’s ability to influence adult hippocampal neurogenesis, including mechanisms driven through immune pathways, microbial metabolites, endocrine signalling, and the nervous system, and postulate implications for these effects in disease onset and treatment.
Keywords
Adult hippocampal neurogenesis
Introduction to adult hippocampal neurogenesis
The hippocampus is one of only two regions of the mammalian brain where new neurons are produced throughout life via a process called hippocampal neurogenesis which occurs in the neurogenic niche of the subgranular zone (SGZ) within the dentate gyrus (DG) of the hippocampus [1]. In 1965, by using thymidine-H3 tagging, Altman and Das discovered the existence of new-born granule cells in the adult rat dentate gyrus [2, 3]. Thymidine-H3 tagging was later combined with neuronal nuclear protein (NeuN), a marker of mature neurons, to confirm that neural stem cells proliferate and differentiate into mature granule neurons in the rat dentate gyrus [4], igniting interest in the functional relevance of these adult-born hippocampal neurons, and raising questions about whether adult hippocampal neurogenesis is also prevalent in humans.
By using a thymidine analogue called bromodeoxyuridine (BrdU), which incorporates into the DNA of all proliferating cells, and combining this with the neuronal markers NeuN, calbindin, and neuron specific enolase, a landmark paper in 1998 revealed evidence that adult hippocampal neurogenesis also occurs in the human dentate gyrus [1]. However, several publications in recent years have encouraged debate around the existence and importance of adult hippocampal neurogenesis in humans, and how it relates to brain disorders in humans [5–8].
Immunohistochemistry is currently the most widely used method to directly analyse hippocampal neurogenesis, as it allows for the identification and spatial understanding of specific proteins, or antigens, expressed during specific stages of the neurogenesis process. Various protein markers have been identified as being expressed at specific stages of neurogenesis such as during proliferation of neural stem cells, neuronal differentiation, maturation and survival, and integration into existing neuronal circuitry (Fig. 1) [9, 10].
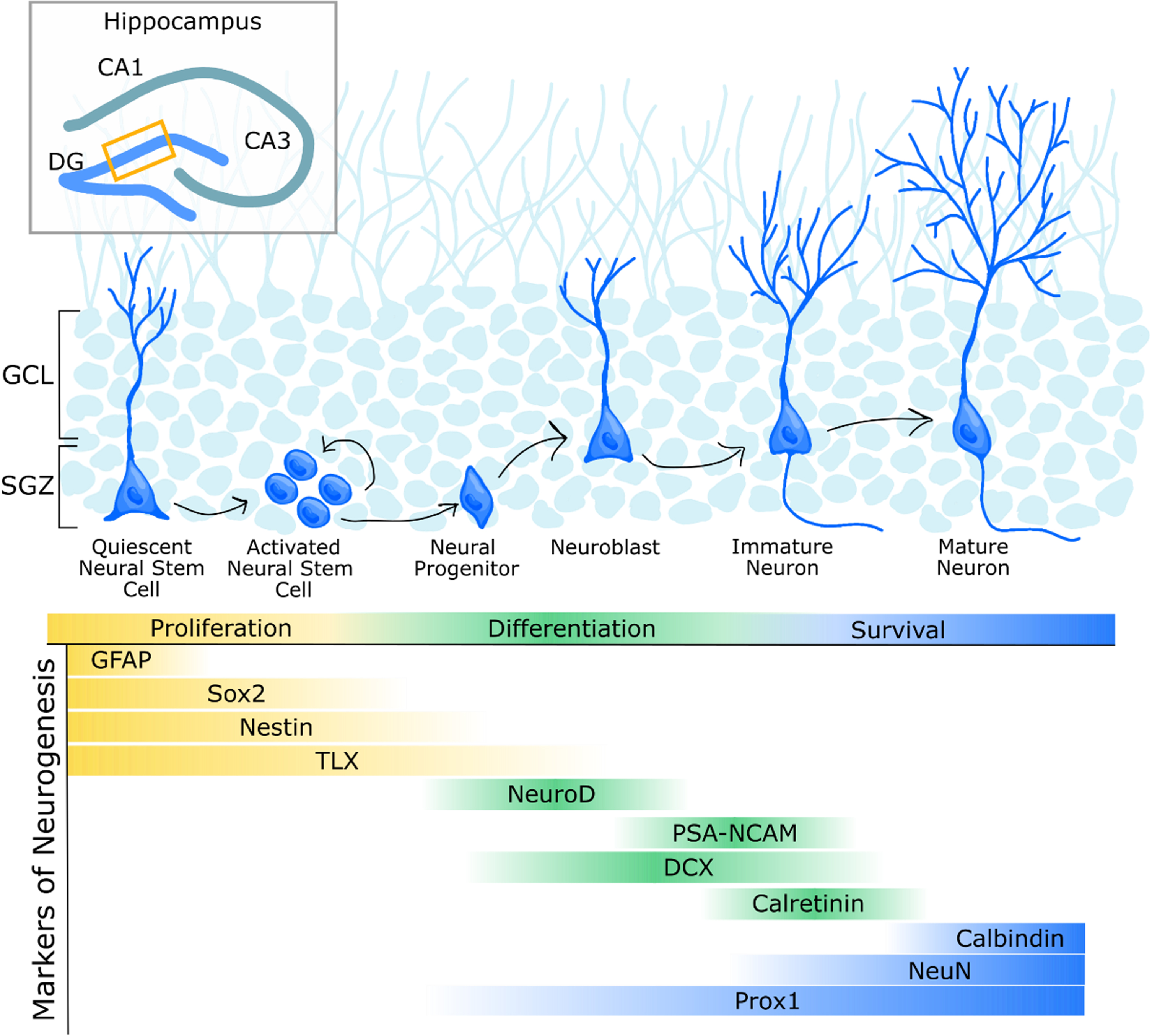
Common markers for various stages of hippocampal neurogenesis. Schematic representation of hippocampal neurogenesis in the mouse dentate gyrus (DG). Quiescent neural stem cells in the subgranular zone (SGZ) undergo activation yielding daughter cells that can self-renew the pool of neural stem cells or differentiate to generate neural progenitor cells and eventually neurons. Neuroblasts that survive subsequent phases of maturation will migrate into the granule cell layer (GCL) and develop into granule neurons. Finally, these cells will incorporate into hippocampal circuitry. Various commonly used markers expressed at different stages of hippocampal neurogenesis are indicated. In addition to these markers, endogenous cellular proliferation markers, such as Ki67, as well as administerable markers, such as BrdU, have become a widely used method to answer multiple questions around hippocampal neurogenesis. Upon administration via injection, BrdU incorporates into the DNA during the S-phase of the cell cycle, when cells are undergoing division [250]. Proliferation markers such as Ki67 and BrdU do not specifically label cells fated to become neurons, and thus must be used with at least one additional neuronal marker. Marker abbreviations: double cortin (DCX); glial fibrillary acidic protein (GFAP); neuronal nuclear protein (NeuN); neurogenic differentiation (NeuroD); prospero-related homeobox (Prox1); polysialylated neuronal cell adhesion molecule (PSA-NCAM); SRY-box transcription factor 2 (Sox2); homologue of the Drosophila tailless gene (TLX), also known as nuclear receptor subfamily 2 group E member 1 (NR2E1). Figure adapted from [10].
The process of adult hippocampal neurogenesis is regulated by multiple endogenous factors. For instance, GABAergic excitation induces neuroblasts to develop into immature neurons [11]. Neurotrophic factors, such as brain-derived neurotrophic factor (BDNF), glia-derived nerve factor (GDNF), nerve growth factor (NGF) and insulin-like growth factor 1 (IGF-1) promote adult hippocampal neurogenesis [12–14]. Microglia, which are innate immune cells resident to the central nervous system, prune synapses of maturing neurons and can produce cytokines which can induce or hinder neuron development and survival [15].
Multiple exogenous factors also influence adult hippocampal neurogenesis (Fig. 2), including diet [16], stress [17–20], antidepressant treatment, exercise, and environmental stimuli [17, 21–23]. While the role of adult hippocampal neurogenesis in humans is not entirely clear, adult hippocampal neurogenesis has been shown to affect specific processes of learning and memory including spatial learning and memory [24–28], trace conditioning memory [29], pattern separation [30–32], and object recognition [33], and has been implicated in stress resilience [21, 34, 35], and antidepressant action [23, 36, 37] in rodents. While the existence of hippocampal neurogenesis during adulthood remains controversial in humans due to limitations such as tissue processing and the necessity to obtain brain tissue quickly
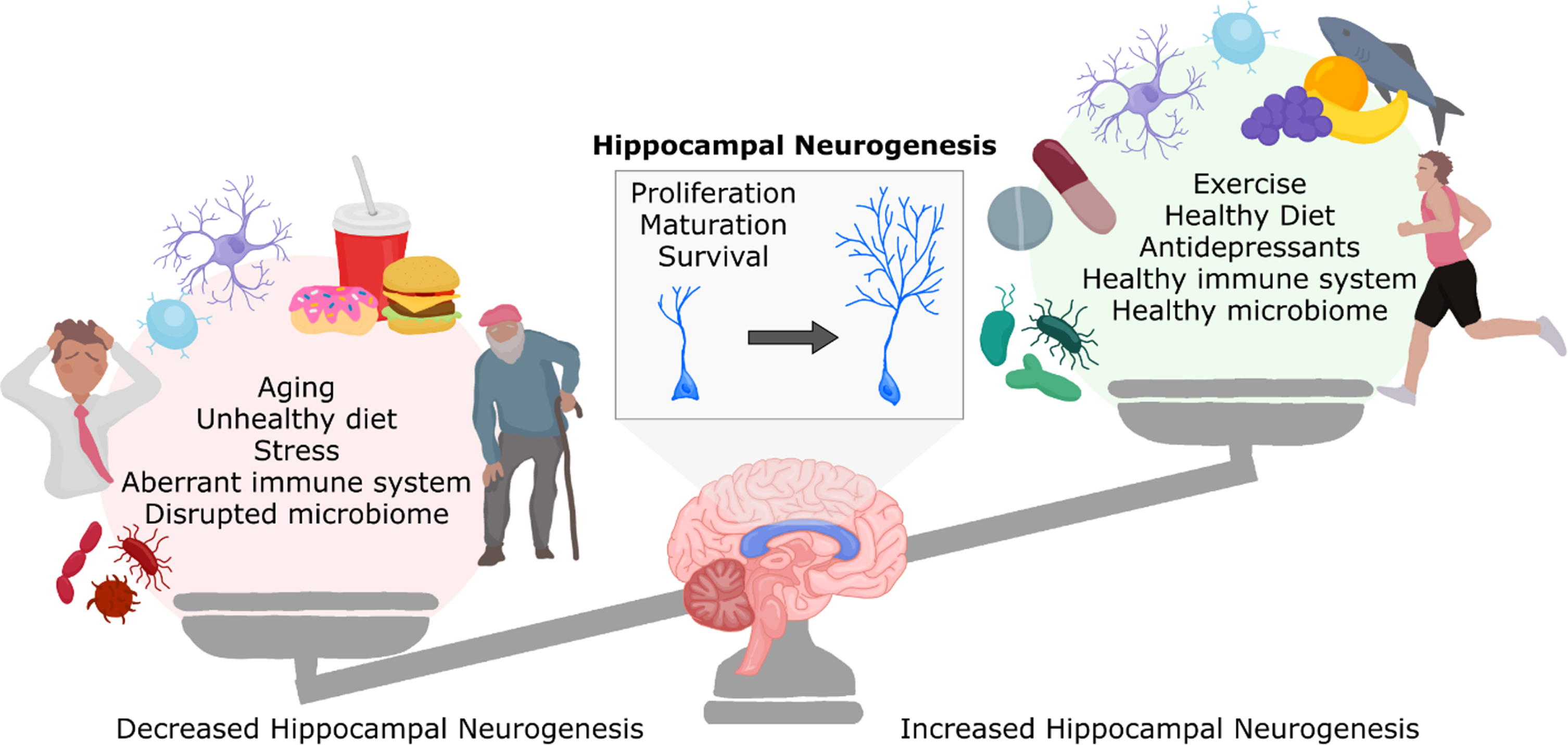
Factors influencing adult hippocampal neurogenesis. Adult hippocampal neurogenesis is increased following exercise, antidepressant drug consumption, a healthy diet, neurogenic immune signalling, and a healthy gut microbiome. However, other factors can be detrimental to adult hippocampal neurogenesis, including aging, diets high in fat and sugar, stress, an aberrant immune system, and disruption of the gut microbiome. These factors can influence the proliferation, maturation and survival of neurons in the adult hippocampus. Figure adapted from [41].
Fascinatingly, several of the factors that are known to influence adult hippocampal neurogenesis demonstrate the parallel ability to influence the composition or function of the gut microbiome, the trillions of microorganisms which reside in the gastrointestinal tract (Fig. 3) [42]. The gut microbiota has been increasingly shown to manipulate cell processes and biological functions in the brain, including adult hippocampal neurogenesis, through what is known as the microbiota-gut-brain axis (Fig. 4) [43].
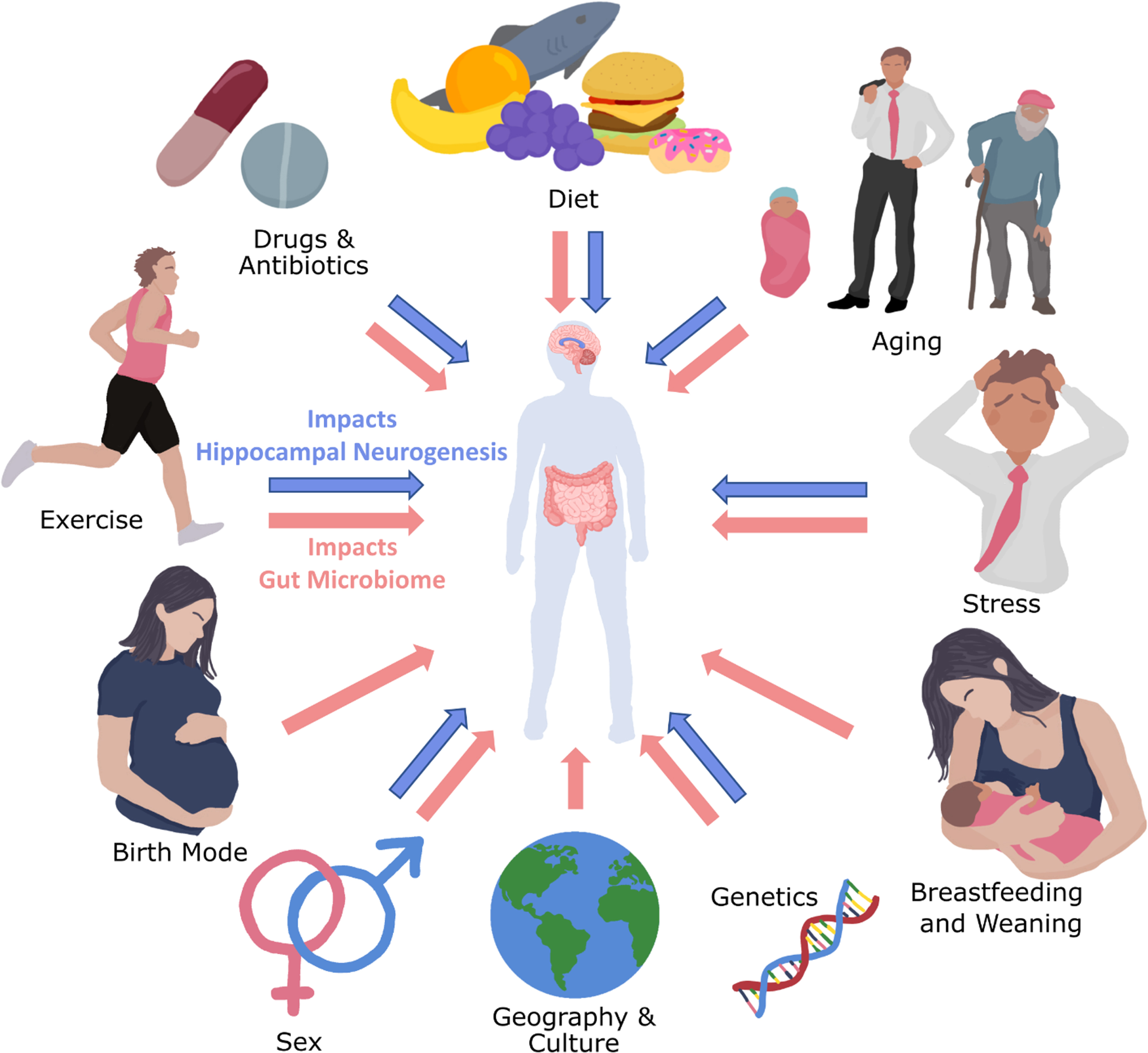
Factors infleuncing the gut microbiota and hippocampal neurogenesis. Blue arrows indicate that the factors have been shown to influence hippocampal neurogenesis, while pink arrows indicate the factors have been shown to influence the gut microbiome. Currently, there is no evidence that birth mode (vaginal vs caesarean section), geography/culture, nor breastfeeding impact adult hippocampal neurogenesis. Some evidence indicates that early weaning decreases hippocampal neurogenesis in mice [51], although it is impossible to untangle whether this response was due to stress caused by premature maternal separation or the temporally altered change in nutrition.
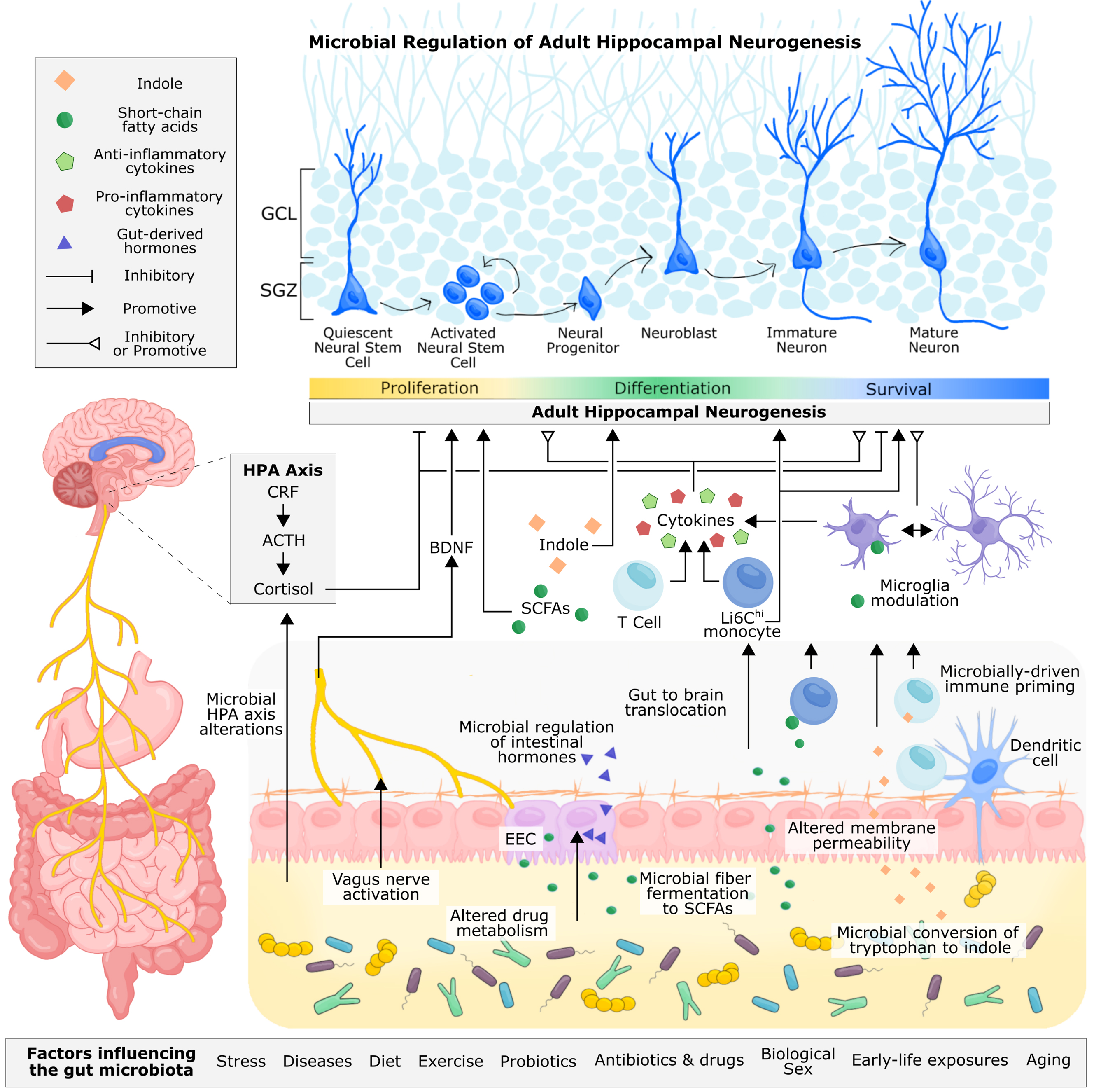
Proposed and established mechanisms of microbial regulation of adult hippocampal neurogenesis. Microbe-derived signals can be transmitted to the brain through neuronal, endocrine, metabolite-driven, and immune pathways. The vagus nerve allows for direct neuronal transmission of gut signals to the nucleus tractus solitarius in the brain, which can be relayed to the hippocampus via the medial septum. Through fecal microbiota transplant and probiotic supplementation, microbial signalling from the gut has been shown to alter levels of hippocampal BDNF and influence adult hippocampal neurogenesis. Immune cells circulating the gut, including monocytes and T cells, are sensitive to the gut microbiota, reacting to microbial cell wall components, such as lippopolysaccharide, and microbially-produced metabolites, such as short-chain fatty acids and indoles, that permeate the intestinal epithelium. These immune cells are also primed by the gut microbiota during early life, shaping their functionality in adulthood. T cells and monocytes can traffic to the brain, where they, along with resident microglia, can produce pro-inflammatory and anti-inflammatory cytokines, which have been shown to inhibit or promote hippocampal neurogenesis. Microglia are also sensitive to microbially-produced short-chain fatty acids, which can cross the intestinal epithelium and blood-brain barrier to directly modulate the functionality of microglia. In addition to cytokine production, microglia are responsible for pruning neuronal dendrites and supporting neuronal development and survival. The gut microbiota can metabolize tryptophan, a precursor to serotonin, kynurenine, and indole. Indole produced by the gut microbiota can directly bind to aryl hydrocarbon receptors (AhR) on neural progenitor cells, promoting neuronal differentiation. Immune cells are also sensitive to indoles through AhR receptors. Intestinal neurotransmitter and hormone production is mediated by enteroendocrine cells and under partial control by the gut microbiota, but whether microbial regulation of these metabolites directly influences adult hippocampal neurogenesis
Although it is currently largely unknown whether lifestyle factors that influence adult hippocampal neurogenesis and the gut microbiome are driven by alterations to the gut microbiome or whether these effects happen independently and in parallel, increasing evidence through microbiota transplant studies [44–46], germ-free (GF) rodents completely devoid of microbes [47, 48], and microbiota-targeted interventions (such as antibiotics or dietary supplementation) [49, 50] have revealed a clear importance of the gut microbiota in hippocampal neurogenesis, as discussed later.
Adult hippocampal neurogenesis is disrupted during stress [46, 52], and has emerged as a target of antidepressant action [52]; a hallmark of chronic antidepressant treatments is the ability to increase adult hippocampal neurogenesis [53–58], which is required for some of the behavioural effects of some antidepressants [36, 37, 59]. A direct causal link between disrupted adult hippocampal neurogenesis and depression has not yet been established in humans. Nonetheless, human
Both stress [62, 63] and antidepressants [64] also have a noticeable impact on the gut microbiome, and the gut microbiome has also been found to contribute to the stress response in rodents [65, 66]. Stress induces volatility in the gut microbiome [63], and different types of stress have varying effects on the composition of the gut microbiome. Stress during early life appears to reduce
Moreover, the antimicrobial effects of some antidepressants suggest their action may be, at least in part, mediated by their ability to restructure the gut microbiome. For instance, serotonin reuptake inhibitor antidepressants, such as fluoxetine and sertraline, can inhibit efflux pumps on bacterial cell walls, and display antimicrobial properties against some gram-positive bacteria, including
Meanwhile, germ-free (GF) mice, devoid of all microbes, exhibited an exaggerated hypothalamic-pituitary-adrenal (HPA) stress response compared to specific pathogen free (SPF) mice, and expressed less BDNF in the hippocampus compared to SPF mice [65]. The colonization of GF mice with a single strain of
Environmental enrichment and exercise
Environmental enrichment, including exercise and learning tasks, increases adult hippocampal neurogenesis in healthy adult rodents [26, 76–78]. Through the promotion of hippocampal neurogenesis, environmental enrichment has been reported to promote the extinction of submissive and depressive-like phenotypes following social defeat stress and chronic exposure to an aggressor mouse, which are forms of psychosocial stressors [79]. Exercise is a form of environmental enrichment that promotes the production and survival of neurons in the adult hippocampus and has antidepressant-like behavioural effects [80, 81]. Although the mechanisms by which exercise promotes hippocampal neurogenesis are not fully established, exercise-induced release of serotonin in the central nervous system is key for the promotion of neuronal proliferation [82]. Moreover, exercise has been reported to increase serum BDNF in humans [81].
In parallel, it has been reported that exercise also impacts the gut microbiome [83, 84]. One study comparing endurance athletes to individuals with a high body mass index found that 48 taxa were increased in elite rugby players including increased
Aging
Aging involves the gradual deterioration of homeostatic functions coinciding with an increased incidence of disease [88]. The characteristics of aging can be seen at a genetic and cellular level, resulting in immunosenescence, an imbalance of chemical messengers (including a reduction in growth hormones, disequilibrium of neurotransmitters) and disrupted hormonal and nutrient sensing [88]. This whole-body degeneration is also evident in the physiology and function of the gastrointestinal tract, with marked changes in gastric motility, declining gastrointestinal epithelial barrier integrity [89], complications in digestion (such as hypochlorhydria), and deterioration of the ENS contributing to striking alterations of the gut microbiota [90].
Hippocampal neurogenesis is substantially reduced during physiological aging in rodents, primates, and humans [4]. Aged rodents (20–24 months) have a substantial loss in dentate gyrus progenitor proliferation, leading to a reduction in the production of new-born granule cells [91, 92]. Furthermore, senescence of the hippocampal neurogenic niche reduces the proportion of neural stem cells that are actively dividing [93, 94], perhaps due to a depletion in the quantity of neural stem cells [95]. This reduction in neurogenesis is noticeable in mice as young as 6 months of age and correlates to impaired cognition in rodents [96] as well as the progression of cognitive decline in Alzheimer’s Disease in rodent models and humans [9, 40]. Rejuvenating the aged neurogenic niche through diet, exercise, or factors in the blood has been shown to rescue aging-associated declines in hippocampal neurogenesis and cognitive deficits, as reviewed extensively elsewhere [97], further implicating hippocampal neurogenic processes with age-related cognitive decline. Furthermore, lifestyle factors such as exercise and cognitive stimulation promote hippocampal neurogenesis and may be protective against aging-related neuropathologies including cognitive decline [78, 98, 99]. This evidence suggests that targeting the aging-associated decline in hippocampal neurogenesis may be an important strategy for developing novel therapeutics for treating declining cognition in aging.
In addition to declining hippocampal neurogenesis, aging triggers an increase in intestinal microbial diversity which is correlated to frailty, longevity [100–103], and other markers of health in aging [104, 105]. Studies involving centenarians have implicated the gut microbiota in longevity and extreme aging, revealing specific taxa such as
Diet
Preclinical dietary manipulations have revealed the importance of diet in promoting and maintaining adult hippocampal neurogenesis. Diets high in fat and sugar decrease hippocampal cell proliferation, the survival of newly born neurons, and hippocampal-dependent behaviour in mice and rats [50, 115, 116], potentially by influencing neuronal mitochondria biogenesis [50] and/or the increase in circulating corticosterone [116]. Meanwhile, long-term running exercise was able to counteract the blunting consequences of a high fat diet on the survival of newly born hippocampal neurons and promote spatial learning and reversal learning performance in the Morris water maze task in mice [117]. On the other hand, dietary patterns which can influence the gut microbiome, including caloric restriction and intermittent fasting, promote longevity and improve memory [118–127] potentially through the upregulation of brain-derived neurotrophic factor [128, 129].
Some dietary compounds have been deemed prebiotics since they are selectively utilized by the gut microbiome, such as fermentable fibres and phenolics [130]. Diets rich in prebiotics appear to ameliorate aging-related cognitive and immune deficits [131, 132] and have also been shown to alter hippocampal neurogenesis. For instance, higher consumption of certain prebiotic polyphenols can promote hippocampal neuron proliferation and survival [133–135]. Dietary supplementation with specific bacteria can directly alter adult hippocampal neurogenesis [46, 49, 110, 136, 137], as discussed in section 3.1.4.
The gut microbiota regulates adult hippocampal neurogenesis
The parallel between factors that influence both adult hippocampal neurogenesis and the gut microbiome make it less surprising that the gut microbiota can orchestrate alterations to neurogenesis in the hippocampus. While the factors outlined above could exert independent effects on gut microbiota and hippocampal neurogenesis, there is emerging evidence that the gut microbiota can regulate adult hippocampal neurogenesis. Manipulations of the gut microbiota, including germ-free rodents, antibiotics, microbiota transfers, and prebiotic dietary interventions, as well as studies interrogating potential mechanisms of action through the vagus nerve, immune system, and metabolite-driven routes, have laid solid preclinical evidence that there are multiple pathways by which the gut microbiota can regulate adult hippocampal neurogenesis, summarized in Fig. 4.
Evidence from gut microbiota manipulation studies
Germ-free rodents
As discussed previously, GF animals are completely sterile from all microorganisms, and therefore act as a clean slate for understanding how the microbiota shapes host biology. In addition to having distinctly aberrant immune profiles, gut abnormalities, and behavioural abnormalities including hyperactivity, cognitive deficits, and altered anxiety-like and depressive-like behaviour, GF rodents have unique disruptions within the brain, culminating in impaired blood-brain barrier function, immature microglia, increased myelination, and altered hippocampal neurogenesis [138]. In 2015, Ogbonnaya and colleagues found that adult GF mice had increased numbers of BrdU+/NeuN+ cells in the hippocampus [47]. Moreover, colonizing 3-week-old GF Swiss Webster mice with microbiota was unable to rescue these effects [47]. Others have since observed alterations in hippocampal neurogenesis in GF mice seems to be sex-, age-, and potentially strain-sensitive. Specifically, male C57BL/6J GF mice were shown to have fewer DCX+ maturing neurons in their hippocampus compared to conventional C57BL/6J mice at 4 weeks old –a transient effect which was not apparent at 8 or 12 weeks of age in this study [48], though the reduction in DCX+ cells in the hippocampus of adult male C57BL/6J GF mice compared to SPF mice was still significantly apparent in another study [139]. Meanwhile, the density of maturing neurons in 4-week-old female C57BL/6J mice did not significantly diverge from their conventional counterparts, and instead had a greater density of hippocampal DCX+ cells at 8 weeks of age, but not at 12 weeks of age [48]. Similar findings were observed in the proliferation and death of cells in the dentate gyrus [48]. Moreover, hippocampal serotonergic signalling [140] and serotonin biosynthesis [141] are disrupted in GF rodents, both of which are important regulators of hippocampal neurogenesis.
Antibiotics
While commonly administered to treat infections clinically, oral antibiotics also act as a useful tool for perturbing the gut microbiota and can therefore be useful for understanding the relationship between the gut microbiota and hippocampal neurogenesis. In mice, an orally administered cocktail of antibiotics consisting of ampicillin, vancomycin, ciprofloxacin, imipenem, and metronidazole for 7 weeks decreased hippocampal cell proliferation (BrdU), neuron maturation (BrdU/DCX/NeuN) and neuron survival (BrdU/NeuN), and also impaired novel object recognition [49]. Interestingly, this effect was prevented when mice were allowed free access to a running wheel or administered a faecal transplant from health SPF mice [49]. Moreover, through adoptive transfer or elimination of Ly6Chi monocytes, Möhle and colleagues demonstrated that gut microbiota signaling to trafficking Ly6Chi monocytes drive alterations to adult hippocampal neurogenesis [49]. Antibiotic use has been clinically associated with neurotoxicity in at-risk patients, including elderly and juvenile individuals [142], and chronic antibiotic use in middle-aged women was associated with decreased cognitive ability after a roughly 7-year follow-up [143]. Whether these clinical effects are mediated by the gut microbiota or are rather off-target direct consequences of antibiotics has not been confirmed.
Faecal microbiota transplant
Faecal microbiota transplant (FMT) involves the transfer of an entire microbiota from one organism into another organism and is therefore a valuable technique for interrogating how specific naturally existing microbial communities influence their host. FMT has been popularized medically for its 90% success rate in treating recurrent
In 2016, FMT from depressed patients into naïve rats was shown to induce anxiety-like and depressive-like phenotypes in the recipient rats, a phenomenon which did not occur when rats were given FMT from healthy people, however adult hippocampal neurogenesis was not investigated in this study [66]. Since then, FMT from depressed patients, as well as chronically stressed mice, has been shown to decrease the quantity of DCX+ immature neurons in the dentate gyrus of recipient naïve mice and to also induce depressive-like behaviour [46, 137], confirming that the gut microbiota is an active regulator of hippocampal neurogenesis, and also indicating that the gut microbiota may play an active role in the progression of diseases in which disrupted hippocampal neurogenesis has been implicated.
In addition to stress-related conditions, the gut microbiota has also been shown to play a causative role in the deficit in cognitive ability that occurs with aging. For instance, transplanting microbiota from aged mice into young mice impaired spatial memory and learning ability and altered synaptic plasticity-related protein expression [146]. Counterintuitively, despite the decreased hippocampal neurogenesis occurring in aged mice, when microbiota from aged mice where transplanted into young germ-free mice, the hippocampus of young recipient mice had increased DCX+ immature neurons in their dentate gyrus compared to their counterparts which received microbiota from young mice [44]. Meanwhile, FMT from 18-month old C57BL/6 mice into old germ-free mice did not induce differences in the density of hippocampal DCX+ cells [44], which indicates that, while the gut microbiota can confer alterations to hippocampal neurogenesis, the innate biology of the recipient is also critical for achieving specific microbiota-driven effects. Separately, adult hippocampal neurogenesis and memory function were impaired in 8-week-old male C57Bl/6 mice who were administered FMT from 9-month old 5xFAD mice (a mouse model of Alzheimer’s disease), but not from aged-matched C57BL/6 mice [147], potentially implicating the gut microbiome in the pathology of Alzheimer’s disease. There appears to be limitations for the ability of the gut microbiota to restore the aging-related reduction in hippocampal neurogenesis; a study investigating the impacts of faecal microbiota transfer from young mice to aged mice on the aging brain did not observe improvements in the survival of newly born hippocampal neurons, although some aspects of neurogenesis-linked spatial learning and memory were rescued following this microbiota transfer [148]. In addition to aging and stress related disruptions to hippocampal neurogenesis, FMT has also been utilized to demonstrate that the drug-induced restructuring of the gut microbiota also reshapes its ability to alter hippocampal neurogenesis. Mice that consumed a high-fat diet and also administered metformin, a drug commonly prescribed to treat diabetes, did not suffer from high-fat diet-induced deficits to hippocampal neurogenesis and cognitive ability. Moreover, when microbiota from these metformin-treated mice were transferred into mice currently on a high-fat diet but otherwise drug-naïve, FMT was sufficient to increase the number of proliferating cells in the hippocampus and improve novel object recognition [149].
Individual bacteria and consortia administration
While FMT is a valuable tool for understanding how complex communities of microorganisms impact their host, it does not allow for the granularity in deciphering whether specific microbes within the community are driving the observed effects. Moreover, FMT is an invasive procedure when conducted clinically, and carries a risk for transferring pathogenic bacteria. Therefore, administration of single bacterial strains or consortia of bacteria are useful approaches to tease out mechanistic details of how the gut microbiota impacts adult hippocampal neurogenesis. The discovery of individual probiotic strains or consortia of bacteria has historically been driven by correlative evidence showing a decreased relative abundance of specific bacteria in diseased populations and remains largely explorative. Nonetheless, through correlative approaches, a handful of bacteria have been shown to regulate hippocampal neurogenesis. For instance, administration of
While some bacteria appear to harness the ability to increase adult hippocampal neurogenesis, other strains have been shown to disrupt it. For instance, one study found that administration of
Prebiotics and diet
Diets including various fruits and vegetables, which are high in prebiotic polyphenols [152], have been clinically associated with improvements in depressive symptomology [153], and yields antidepressant and anti-anxiolytic effects in various rodent models [154–156]. Meanwhile, a Mediterranean-style diet, which is high in fish oils, was sufficient to improve mental health scores in individuals with depression [157] and improve cognitive performance in aging humans [158]. Omega-3 fatty acids, which are prevalent in high-fish diets, have been shown to promote hippocampal neurogenesis in mice [159] and lobsters [160]. Separately, omega-3 fatty acids were able to act directly on cultured human hippocampal progenitor cells, therein preventing cortisol induced reductions in neurogenesis [161]. Nonetheless, the mechanisms of action for how specific diets and dietary compounds, such as polyphenols and fibres, exert their effects on the brain remains unknown, though their ability to influence the gut microbiota may allow for their potential effects on hippocampal neuroplasticity-related behaviours [132, 162, 163].
As defined earlier, prebiotics are substrates selectively utilized by the gut microbiome, such as fermentable fibres and phenolics [130]. Fermentable fibres can be digested by bacteria into short-chain fatty acids including acetate, butyrate, and propionate, which have immunomodulatory and histone deacetylase inhibitory properties, including directly in the brain [164, 165]. When administered orally to pigs for 4 weeks, sodium butyrate increased hippocampal neurogenesis and hippocampal granular cell layer volume [166]. Oral sodium butyrate has also been shown to ameliorate cognitive deficits and increase the expression of neurotrophic factors in rats subjected to early life stress [167], and decrease hippocampal neuroinflammation in aging mice [168]. The soluble dietary fibres fructo-oligosaccharides (FOS) and galacto-oligosaccharides (GOS) have been shown to regulate BDNF and synaptic protein expression in rodents [169, 170], though whether they can directly alter hippocampal neurogenesis has not yet been demonstrated.
In addition to fibre, various flavonols exert neurotrophic effects, although it is largely unclear whether these effects require the gut microbiome or simply restructure the gut microbiota independently from their effects in the hippocampus. For example, the polyphenol quercetin can enhance learning and memory, alleviate anxiety-like behaviour, increase neural stem cell and progenitor cell proliferation, promote neurotrophic factors including BDNF, and increase the quantity of DCX+ dentate gyrus cells in rats [162, 171]. Meanwhile, a citrus flavonoid, 3,5,6,7,8,3′,4′-Heptamethoxyflavone, increases adult hippocampal neurogenesis in mice that were exposed to chronic unpredictable mild stress [172]. Moreover, the peel of the
Potential mechanisms by which the gut microbiota influences adult hippocampal neurogenesis
There are several proposed mechanisms by which the gut microbiota may exert influence on adult hippocampal neurogenesis, including immune, neuronal, endocrine, and metabolite-driven pathways (Fig. 4).
Immune system and neuroinflammation
The gut microbiota regulates several aspects of the host immune system, including in the brain, potentially allowing the gut microbiota to influence hippocampal neurogenesis via immune signaling pathways. For instance, germ-free mice have defective hippocampal microglia, and a higher density of microglia overall [177]. The regulation of microglia by the gut microbiota appears to be at least partially controlled by microbially produced acetate, which can translocate from the gut to influence the maturity and function of microglia in the brain [165]. Microglia contribute to the balance of neuronal cell birth and death in the subgranular zone through apoptosis-coupled phagocytosis [178], allowing them to clear debris from destroyed cells. In response to phagocytosis, the microglia secretome is altered towards substrates that have been shown to support new neuron proliferation, such as VEGF and FGF2 [178, 179]. Furthermore, upon activation, microglia produce various cytokines to induce an immune response to dangerous threats. Neuroinflammation, mediated by proinflammatory cytokines including interleukin (IL)-1β, tumour necrosis factor α (TNFα), and IL-6, has detrimental consequences to the proliferation and development of newly born hippocampal neuronal cells [180–183], and immune responses, including cytokine release, are blunted in GF mice [184]. By regulating the function and maturity of hippocampal microglia, the gut microbiota may be able to reshape hippocampal neurogenesis, although this direct mechanistic link is yet to be established.
In addition to impacting the function of microglia, the gut microbiota contributes to the systemic priming of immune cells that may traffic to the brain. Through the use of genetic knockout mice and antibody depletion, circulating Ly6Chi monocytes were shown to play a pivotal role in promoting hippocampal neurogenesis [49]. Ly6Chi monocytes traffic throughout the body and are highly sensitive to gut microbiota-derived signals. Following antibiotic treatment, mice had lower numbers of Ly6Chi monocytes in the brain, blood, and bone marrow, and displayed deficits in hippocampal neurogenesis and behaviour [49]. Adaptive transfer of Ly6Chi monocytes from the bone marrow of C57BL/6 mice to antibiotic-treated mice was able to restore the proliferation and survival of newly born neurons in the dentate gyrus [49]. Fascinatingly, supplementation with an 8-strain probiotic cocktail was also able to rescue antibiotic-induced deficits in hippocampal neurogenesis and Ly6Chi monocyte levels, providing further evidence that the gut microbiota can regulate hippocampal neurogenesis through Ly6Chi monocytes [49].
The gut microbiome has been shown to modulate the function of various types of T cells, including CD8+ T cells [185, 186], CD4+ T cells [187, 188], and Th17 cells [189, 190], suggesting potential T cell-dependent mechanisms by which the gut microbiota might influence hippocampal neurogenesis. T cells are essential regulators of hippocampal neurogenesis [191]. For instance, CD8+ T cells are required for the beneficial effects of environmental enrichment on hippocampal neurogenesis, synaptic plasticity, and behaviour including anxiety-like behaviour and spatial learning in mice [192]. Meanwhile, depletion of CD4+ T lymphocytes significantly reduced hippocampal neurogenesis, while neuronal precursor cell proliferation was increased in Rag2-/- mice, mice which produce no mature T cells or B cells, following repopulation of CD4+ cells [193]. Another subset of T cells, T helper 17 (Th17) cells, can also produce IL-17 [194], which inhibits adult hippocampal neurogenesis in mice [195], and can promote susceptibility to depressive-like behaviour in mice [196].
By microbial crosstalk with microglia, monocytes, and T cells, the gut microbiota demonstrates the ability to influence hippocampal neurogenesis in adult rodents. More research needs to be conducted however to fully understand the cellular pathways and scope of capability of microbiota-immune signalling on influencing hippocampal neurogenesis.
Vagus nerve signalling
The vagus nerve innervates the gastrointestinal tract, allowing for bidirectional communication between the gut and the brain. Through virus-based tracing techniques, it was shown that vagal signals derived from the gut are relayed from the medial nucleus tractus solitarius through the medial septum and to dorsal hippocampal glutamatergic neurons, allowing gut-derived signals transmitted through the vagus nerve to control hippocampus-dependent episodic and spatial memory, and reduced the expression of neurogenic and neurotrophic markers (DCX and BDNF) in the dorsal hippocampus [197] though whether these signals were derived specifically from microbiota-vagus nerve interactions was not determined. Nonetheless, specific gut bacteria, such as
Independent from the gut microbiota, stimulation of the vagus nerve has been reported to trigger increased progenitor cell proliferation, numbers of maturing neurons, and increased protein expression of the neurotrophic factor BDNF in the hippocampus of rodents [200–202]. Chronic simulation of the vagus nerve also promotes dendritic branching on immature hippocampal neurons [200]. Meanwhile, severing the vagus nerve via subdiaphragmatic vagotomy was sufficient to reduce hippocampal cell proliferation, neuronal differentiation, and BDNF mRNA expression in the adult hippocampus [197, 203].
Microbial Metabolites
Due to complexities in tracing the origins of metabolites within a living organism, it is difficult to decipher whether specific gut microbiota-derived molecules can translocate from the gut to the brain, where they have the potential to directly hinder or support neuronal birth, maturation, and survival. As such, few radioactive tracing studies exist that examine gut microbiota-derived metabolites within the brain. Recently, one study utilizing radioactive labelling revealed that the microbe-derived short chain fatty acid acetate translocated from the gut to the brain of mice, where it influenced hippocampal microglia metabolism and function [165]. While it is possible to speculate that alteration in the functionality of microglia would be able to alter hippocampal neurogenesis, this study did not examine hippocampal neurogenesis. Nonetheless, short chain fatty acids, including acetate, butyrate, and propionate, have been shown to directly promote neuronal differentiation in neural stem cells
As discussed extensively elsewhere [208], the gut microbiota can influence the bioavailability of hormones and neurotransmitters including within the tryptophan-kynurenine- serotonin pathways by synthesizing or degrading components of this pathway. These hormones have been strongly implicated in depression [208]. Of interest, tryptophan and its metabolites are able to be modulated by the gut microbiota, and can impact host cognitive function and mood [209], as well as hippocampal neurogenesis [139]. Recent research has found that indoles that are produced by the gut microbiota-driven metabolism of tryptophan can modulate hippocampal neuroplasticity and promote the neuronal differentiation of hippocampal neural progenitor cells through the activation of the aryl hydrocarbon receptor [139]. The gut microbiota can also regulate the production of serotonin in the gut [210]. Serotonin is integral for neuronal differentiation, neurogenesis, and the
The hypothalamic-pituitary-adrenal (HPA) axis and hormonal signalling
The hypothalamic-pituitary-adrenal (HPA) axis is a key endocrine pathway regulating the physiological response to stress. HPA axis activation triggers the release of glucocorticoids (including cortisol, or corticosterone in rodents) which can directly bind to receptors on neural stem and progenitor cells and hinder their proliferation and differentiation
Several peptide hormones that regulate appetite and food intake also demonstrate the ability to influence adult hippocampal neurogenesis such as ghrelin [217–220], leptin [221–224], cholecystokinin [225–227], neuropeptide Y [227, 228], insulin-like growth factor 1 [229–232], gastric inhibitory polypeptide [233], and orexin-A [234]. Moreover, the gut microbiome has been linked to the regulation of several of these hormones including ghrelin [235, 236], leptin [236, 237], cholecystokinin [236, 238, 239], neuropeptide Y [237], although the mechanisms underlying these relationships remain largely elusive [240]. By targeting some of these molecules, there have been improvements in aging-associated impairments in hippocampal neurogenesis. For instance, by restoring insulin-like growth factor 1 levels in mice, researchers rescued the age-related decline in adult hippocampal neurogenesis [241, 242]. Mechanistic research needs to be conducted to better understand the involvement of the gut microbiota in regulating adult hippocampal neurogenesis through hormone-driven pathways.
Conclusions and Future Perspectives
In this review, we aimed to provide an overview of the current literature investigating the gut microbiota and adult hippocampal neurogenesis, including the impacts of specific microbes and mechanisms by which the gut microbiota has been shown to influence the proliferation, development, and survival of new-born neurons in the adult hippocampus. We first discussed overlaps in the factors that are known to influence adult hippocampal neurogenesis and the gut microbiome, postulating that these effects might be occurring independently and unrelatedly to one another, but that they also might be linked through microbiota-driven alterations to gut-brain dialogue. Nevertheless, there is a growing body of evidence that suggests that the gut microbiota regulates adult hippocampal neurogenesis. Therefore, we then provided the current state of evidence demonstrating that the gut microbiota plays an active role in shaping neurogenesis in the adult dentate gyrus. In doing so, we have outlined influential factors and biological pathways underlying the regulation of adult hippocampal neurogenesis by the gut microbiota.
The gut microbiota harbours the ability to utilize a variety of pathways to promote or hinder adult hippocampal neurogenesis, including direct communication through the nervous system, or indirect communications via microbial metabolites, the immune system, and hormones. Still, some potential avenues of the microbiota-gut-brain axis have yet to be investigated in the context of hippocampal neurogenesis. For instance, the gut microbiota contributes to the secretion of hormones by intestinal cells, such as ghrelin [240]. Meanwhile, intraperitoneal injection of ghrelin increases BrdU+/DCX+ cells in the dentate gyrus [217]. However, whether the microbial control of adult hippocampal neurogenesis is mediated by ghrelin, or other intestinal peptides, has not yet been demonstrated and should be explored. Moreover, it is currently unclear how an individual’s mode of birth and weaning timeframe impact hippocampal neurogenesis. Mouse pups born by caesarean section demonstrated heightened anxiety-like behaviour and altered neurochemistry that persists into adulthood –phenotypes which appear reversible by targeting the early life gut microbiota [243]. Moreover, the weaning timeframe is highly critical for gut microbiota-mediated early life immune system development, and substantially impacts regulatory T cells [244]. Therefore, it is plausible that early life events, including caesarean section birth and altered weaning timeframes may impact adult hippocampal neurogenesis in a microbiota-dependent manner.
Alterations to the gut microbiota are present in numerous brain disorders, including depression [66], Alzheimer’s Disease [245], and epilepsy [246], conditions in which adult hippocampal neurogenesis has been observed to be disrupted clinically or in relevant animal models [10, 246, 247]. Given the strong preclinical evidence demonstrating the ability of the gut microbiota to influence various aspects of adult hippocampal neurogenesis, the gut microbiota may be a viable therapeutic target for treating and preventing these diseases through its ability to alter adult hippocampal neurogenesis. The gut microbiota is highly accessible to interventions, including via probiotics, antibiotics, and lifestyle choices such as diet, which presents multiple opportunities to remediate abnormal hippocampal neurogenesis which may contribute to some brain disorders.
Currently, clinical conclusions about the mechanistic ability of the gut microbiota to influence adult hippocampal neurogenesis are highly limited by the technical challenges of assessing neurogenesis in living humans. Therefore, evidence demonstrating the role of the gut microbiota in modulating adult hippocampal neurogenesis, as well as its impact on behaviour and disease progression, is largely dependent on preclinical interrogations. While there are vast biological similarities between mouse and humans, evidence collected in rodent models might not fully reflect clinical biological scenarios. Importantly, the murine and human gut microbiomes are distinctly unique, and, although biological functionality appears relatively conserved between these species, differences in anatomy and physiology between mice and humans may mean that preclinically established mechanisms might not be capable of translating to humans [248, 249]. Therefore, it is difficult to confirm whether the gut microbiota utilizes the same pathways observed in preclinical studies to impact human adult hippocampal neurogenesis. Nonetheless, largescale clinical trials involving microbiota interventions will be incredibly useful for understanding how the gut microbiota may sculpt favourable effects on the brain and related behaviour and identifying clinical biomarkers of microbiota-driven health benefits to potentially reveal novel therapeutic options. Ultimately, understanding the pathways by which the gut microbiota influence hippocampal neurogenesis may allow for the development of novel therapeutics to ameliorate hippocampal neurogenesis dysfunction, which could provide crucial treatment options for improving cognition and mood in brain disorders.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
Funding
KEG was supported by an Irish Research Council (IRC) Government of Ireland Postgraduate Scholarship (GOIPG/2019/3198). JFC is funded by Science Foundation Ireland SFI/12/RC/2273_P2, the Saks Kavanaugh Foundation, EU H2020 project DLV-848228 DIS-COvERIE, and Swiss National Science Foundation project CRSII5_186346/NMS2068. OFO is funded by Science Foundation Ireland 20/FFP-P/8758, the Biostime Institute Nutrition and Care (BINC) –Geneva Funding Programs, and is a Funded Investigator at APC Microbiome Ireland, a research Institute funded by Science Foundation Ireland (SFI/12/RC/2273_P2).
CONFLICTS OF INTEREST
JFC has received research funding from 4D Pharma, Cremo, Dupont, Mead Johnson, Nutricia, and Pharmavite; has been an invited speaker at meetings organized by Alimentary Health, Alkermes, Ordesa, and Yakult; and has served as a consultant for Alkermes and Nestle. This support neither influenced not constrained the contents of this article. OFO has received funding for unrelated contract research from Alkermes plc. and has received renumeration as an invited speaker at a meeting organised by Janssen OFO is currently conducting unrelated collaborative research with Marigot Ltd. This support neither influenced not constrained the contents of this article. KEG declares no conflicts of interest.
