Abstract
BACKGROUND:
Deep neck flexor muscle atrophy and increased superficial neck muscle activation are associated with disability and pain intensity in individuals with neck pain. There is a lack of evidence to support direct assessment of deep neck flexor muscles in a non-invasive way during exercise performance to help determine the effectiveness of different neck strengthening exercises.
OBJECTIVE:
Compare longus colli (LC) and sternocleidomastoid (SCM) activation between individuals with and without neck pain using real time ultrasound (RTUS) during a series of craniocervical exercises.
METHODS:
This cross-sectional cohort study recruited 10 control and 10 neck pain participants to complete four deep neck flexor activation activities involving varying levels of craniocervical flexion. Dimensions of the LC and SCM were measured using RTUS at rest and during exercise. Independent t-tests assessed baseline differences and analysis of variance examined activation changes.
RESULTS:
At rest, the neck pain group had significantly smaller cross-sectional area and thickness of the LC compared to the control group (
CONCLUSIONS:
Despite atrophy, individuals with neck pain can activate their deep neck flexor muscles appropriately without activating their superficial neck flexor muscles in a supine series of craniocervical flexion exercise as measured by non-invasive ultrasound imaging.
Introduction
Mechanical neck pain is a significant musculoskeletal disorder affecting adults with an estimated prevalence ranging from 16.7% to 75.1% [1, 2, 3]. Between 50% and 85% of those who experience neck pain will report neck pain again 1 to 5 years later [4]. Individuals with chronic neck pain have demonstrated atrophy in specific muscles of the neck that assist with cervical stabilization [5, 6, 7, 8]. Specific to the neck flexors, individuals with chronic neck pain have demonstrated atrophy in the deep neck flexor muscles including the longus colli (LC) compared to individuals without neck pain [7, 8, 9, 10]. It has been suggested that weakness in these muscles may cause unwanted rotary forces on the vertebral joints in this region due to compensatory muscle activation from extensor muscles [11, 12]. This deep neck flexor weakness paired with increased superficial neck muscle compensation has been shown to be negatively associated with self-reported disability and intensity of pain [9].
To prevent injury to cervical structures and the development of unwanted compensatory muscle tension, one rehabilitation target of intervention in individuals with neck pain is improvement in the stabilization of the spine by engaging muscle activity. This has been investigated in cadaveric specimens [13] and in vivo for both neck flexor and extensor muscles [14, 15, 16]. Exercises designed to target the deep neck flexor muscles have shown favorable responses in individuals with neck dysfunction to improve muscle function, pain, and disability [15, 16, 17]. During normal neck flexion function, there is an expected coordination of muscle activity between the deep neck flexors to stabilize the vertebra, and the superficial neck flexor muscles [sternocleidomastoid (SCM) and anterior scalenes] to produce force with which to allow the neck to move [18, 19, 20]. Compared to the superficial neck flexor muscles, the biomechanical contributions of the longus colli and capitis to force production at the neck is small [21]. Research has shown that in individuals with chronic neck pain, neck muscles can be weaker [22], activation of the deep neck flexor muscles is significantly reduced, and cervical feed forward neural activity is delayed compared to individuals without neck pain [23, 24]. Accompanying this reduction in deep neck flexor muscle activity is a compensatory increased activation in the superficial neck flexor muscles [25, 26]. Increased activity of superficial neck flexor muscles may contribute to the development of and persistence of neck pain in some individuals.
Given altered muscle activation patterns in individuals with chronic neck pain, there have been efforts to examine the effect of focused exercise programs on strengthening the deep neck flexors to decrease pain and increase function [27, 28, 29, 30]. Most work to date has focused on the craniocervical flexion test (CCFT) to promote increased activation and recruitment of deep neck flexors [31, 32, 33, 34]. The CCFT is a precise and controlled performance and maintenance of a series of low load craniocervical flexion exercises in a supine position [33]. Assessment of muscle activation and validation of exercise prescription using exercises like the craniocervical flexion has traditionally relied on electromyography (EMG) [35, 36]. However, assessment of neck flexor muscle activation has either relied on surface EMG to assess activation of superficial neck muscles as an inverse of deep neck flexor activity [25, 37, 38] or an invasive EMG approach using swallowed electrodes to place them in the pharynx [39, 40]. Lacking thus far are studies which directly assess deep neck flexor muscle activation in a non-invasive way during neck exercises to help determine the effectiveness of different exercises in targeting increased activation of deep neck flexor muscles in individuals with neck pain. A clearer understanding of how different exercises can progressively target deep neck flexor muscle activation could promote more effective rehabilitation strategies for individuals with chronic neck pain.
Real time ultrasound imaging (RTUS) is a non-invasive and valid tool to measure muscle activation, including activation of the deep neck muscles [18, 19, 41, 42, 43]. Ultrasound imaging can be used to assess changes in muscle morphology (e.g., thickness, cross sectional area) in real time as an individual completes specific exercises. Increased activation of a muscle is associated with increased muscle thickness [18, 44]. There have been no studies that have compared changes in deep neck flexor muscle morphology using RTUS with different exercise that are designed to require increasing levels of muscle activation. The purpose of the current study was to compare LC and SCM activation between individuals with and without neck pain using real time ultrasound (RTUS) during a series of craniocervical exercises designed to progressively increase the need for deep neck flexor muscle activation in individuals with neck pain compared to controls. Results of this study will help to inform rehabilitation activities and exercise prescription in individuals with neck pain.
Methods
Design
A prospective cross-sectional cohort study was conducted from September 2021 to September 2022 in the Physical Therapy Institute at Midwestern University. The study was approved by the Institutional Review Board at Midwestern University (approval code 20031), and study procedures followed the ethical standards of the Declaration of Helsinki.
Participants
Adults 18–65 years old with and without neck pain were recruited for participation by posted flyer. Twenty participants (14 female, 6 male) were recruited and completed the study: 10 individuals with nonspecific mechanical neck pain previously (8 female, 2 male) and 10 individuals without neck pain (6 female, 4 male). Neck pain has previously been diagnosed by a physician and participants met the criteria of neck pain with mobility deficits [45]. An a priori power analysis was conducted using G*Power version 3.1.9.2 [46] for sample size estimation based on data from 3 subjects with neck pain and 3 subjects without neck pain (mean thickness 11.1 and 12.8 mm respectively with a standard deviation of 1.0). With a significance criterion of
The mean intensity of neck pain was assessed on a numerical scale (NPRS, 0: no pain, 10: maximum pain). Functional disability was determined using the Neck Disability Index (NDI) which addresses activities of daily living, pain, and concentration. The numeric score for each item was totaled for a score between 0–50 with a higher score reflecting a higher level of disability. The neck pain participants were eligible for inclusion if they were 18–65 years old, scored 5 or more points on the NDI [47], and had chronic intermittent neck pain within the previous 3 months. They were excluded if they had undergone cervical spine surgery, presented with any neurological signs, or were currently involved in a neck exercise physical therapy program. Control group participants were included if they were between 18–65 years old, scored under 5 points on the NDI and had not experienced chronic intermittent neck pain.
Exercises
Participants completed 4 exercise activities which aimed to target progressive activation of the LC while RTUS images were taken. All exercises were performed in supine with an inflatable cuff (Stabilizer™, Chattanooga
Ultrasound imaging
Ultrasonograms from one participant demonstrating measurement of (a) sternocleidomastoid and longus colli thickness in long axis, and (b) cross sectional area of longus colli in short axis. Abbreviations: LC (longus colli), SCM (sternocleidomastoid), CA (carotid artery), RPS (retropharyngeal space), VB (vertebral body).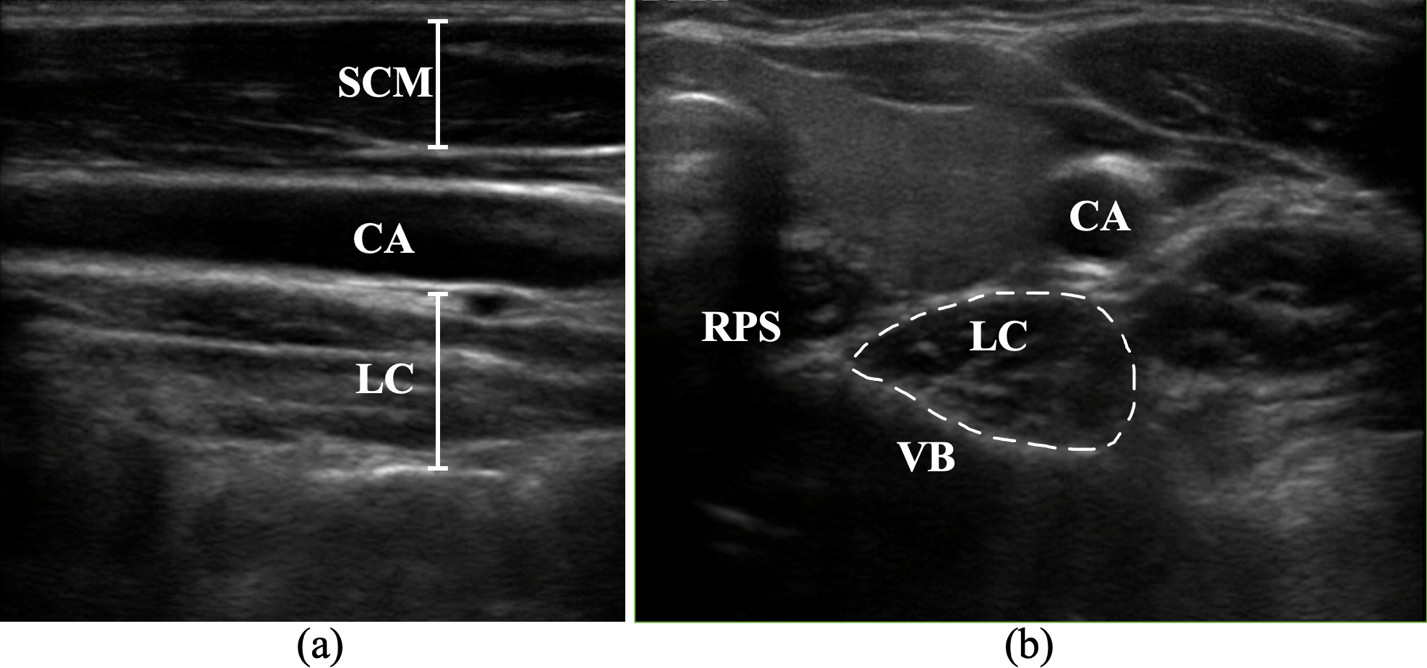
Percent change of longus colli muscle thickness (a), longus colli cross sectional area (b), sternocleidomastoid muscle thickness (c) between control (grey diamonds) and neck pain group (black squares). Data shown are the mean and standard error of the mean. Abbreviations: LC (longus colli), SCM (sternocleidomastoid), CSA (cross-sectional area). Abbreviations: LC (longus colli), SCM (sternocleidomastoid), CSA (cross-sectional area); Yes, looking down with the eyes towards the feet; CCF Low, craniocervical flexion to 24 mmHg against the pressure cuff; CCF High, craniocervical flexion to 30 mmHg against the pressure cuff; CCF High arm, craniocervical flexion to 30 mmHg against the pressure cuff while also completing an arms extension against an elastic resistance band. 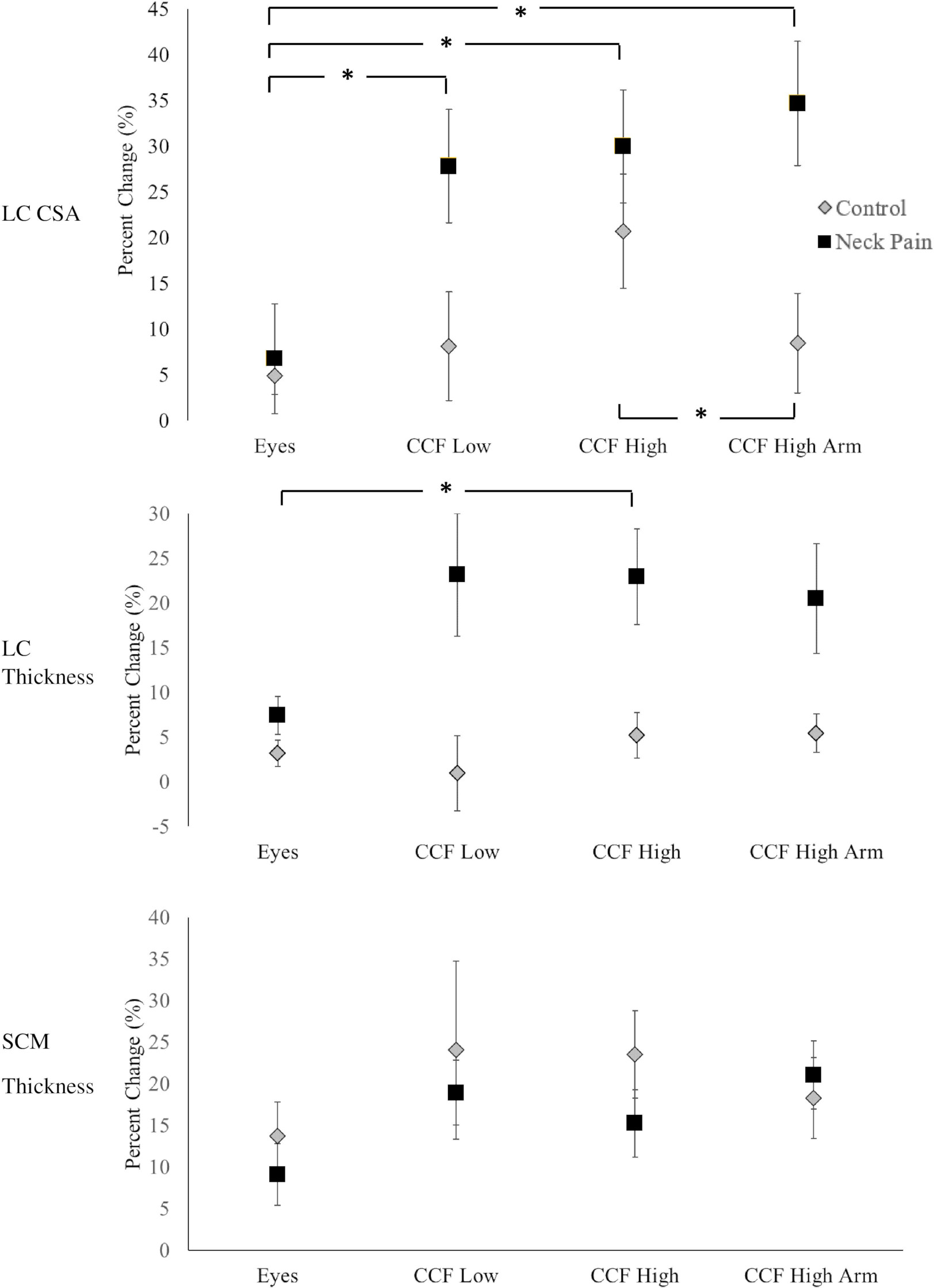
Baseline characteristics of control and neck pain groups
Abbreviations: LC (longus colli), SCM (sternocleidomastoid), CSA (cross-sectional area), NDI (neck disability index), BMI (body mass index), NPRS (neck pain rating scale).
Results of 2-way (Group by Exercise) Analysis of Variance on change of muscle thickness
Key: LC (longus coli), SCM (sternocleidomastoid), CSA (cross-sectional area), C (control group), NP (neck pain), CCF (craniocervical flexion).
Ultrasound images were captured using B-mode, 2-D ultrasound imaging (Sonosite M-Turbo
Morphological activation changes were measured offline using ImageJ software [49] by a rater blinded to group and exercise. Measurements were assessed for LC muscle thickness and SCM muscle thickness in long-axis view (Fig. 1a) and LC cross-sectional area (CSA) in short-axis view (Fig. 1b). The percent change in activation from rest was computed and assessed for LC CSA (Fig. 2a), LC thickness (Fig. 2b), and SCM thickness (Fig. 2c). The borders of the LC in short-axis were determined by the most distant fascial borders using the following landmarks: inferiorly and medially by the body of the C6 vertebrae, laterally by the carotid artery, and superiorly by the retropharyngeal space [10]. The borders of LC in long axis were determined by the most distant fascial borders using the carotid artery anteriorly and the body of the C6 vertebrae posteriorly. C6 was chosen because of its clear visualization and less overlap between the longus colli and longus capitis muscles. The borders of SCM in long axis were determined by the most distant fascial borders between the carotid artery posteriorly and the superficial fascia anteriorly in the same segment of the image where LC was measured. Data were averaged over 3 trials. Absolute values of thickness and CSA were obtained and used to compare baseline differences. Percent change in activation from rest to activation was computed to assess changes in activation during each exercise and to compare activation between groups.
Data analysis
Statistical analysis was conducted using SPSS version 28.0 (SPSS Inc., Chicago, IL, USA). Independent t-tests were used to compare baseline differences between groups (control vs neck pain). Two-way repeated measures analysis of variance (ANOVA) was used to examine differences in percent change activation between individuals with and without neck pain (Group) and across exercise conditions (Eyes, CCF Low, CCF High, and CCF High arm). A statistical significance level alpha was set as
Results
Baseline differences
The neck pain group had significantly higher NDI scores (
Longus colli activation differences between control and neck pain groups
There was an interaction effect between exercise and group for change in activation of CSA for LC (Table 2). The neck pain group showed a significantly larger increase in LC CSA across exercise compared to the control group (
When considering differences within each group across exercises, there were significant main effects of group in change of LC thickness but not CSA (Table 2, Fig. 2). The neck pain group showed a significantly higher percent change in LC muscle thickness activation compared to the control group across exercise (
When considering how exercise changed muscle activation regardless of group, there was a main effect of exercise on percent change LC CSA (
Sternocleidomastoid activation differences between control and neck pain groups
Consistent with no baseline differences between neck pain and control groups in SCM thickness, both groups showed similar activation levels of the SCM throughout all exercises and there were no between group differences in SCM activation between exercises (Table 2, Fig. 2c).
Discussion
The purpose of this study was to compare deep and superficial neck flexor muscle activation between individuals with and without neck pain using real time RTUS during a series of craniocervical exercises designed to progressively increase the need for deep neck flexor muscle activation in individuals with neck pain compared to controls. The novel findings of this study were that individuals with neck pain were able to robustly activate the LC, a deep neck flexor muscle, without activating the SCM, a superficial neck flexor muscle using this series of supine exercises. However, using a deliberate craniocervical flexion action rather than a secondary craniocervical flexion action through an eye gaze task (cranial flexion only) was needed to produce the most robust change in deep neck flexor activation. Across group, adding arm resistance to craniocervical flexion was a robust way of increasing cross-sectional area of the deep neck flexor muscles beyond just inducing craniocervical flexion. Results of this study will help to inform rehabilitation activities and exercise prescription in individuals with neck pain.
The results from the current study showed that, compared to individuals without neck pain, individuals with neck pain had approximately a 20% reduction in size of the LC at rest (Table 1). Neck pain has been associated with small, atrophic deep neck flexors leading to cervical stabilization concerns [7, 50, 51] and the results of the current study support atrophy of LC in individual with neck pain. It is unclear however if the deep neck flexor atrophy contributes to neck pain or is a consequence of it. Regardless of atrophy, neck pain participants in the current study were able to activate their LC during each of the exercise activities. Indeed, the neck pain group showed significantly larger changes in LC activation compared to the control group across exercise. This demonstrates that the supine craniocervical flexion exercises actively engaged the deep neck flexors in individuals with neck pain despite their lower resting thickness.
Neck pain is the target population for the craniocervical flexion test from which the exercises in the current study were drawn [15, 25, 33, 34]. Previous studies have shown that individuals with chronic neck pain may compensate for weakness or dysfunction of the deep neck flexor muscles by utilizing superficial neck flexors to produce craniocervical flexion [25]. In the current study, individuals with neck pain were able to effectively activate their deep neck flexors without activating the SCM, a superficial neck flexor muscle during the exercise series. The craniocervical flexion test was designed to isolate activation of the deep neck flexors, and utilizes a pressure cuff to target activation of the deep neck flexors progressively from lower levels of required force (22–24 mm Hg) to progressively higher levels of required force (28–30 mm Hg) and it is from this validated test that the high and low pressures used in the current study were drawn [33]. However, there was not a linear response in deep neck flexor muscle activation as anticipated across the series of exercises in the current study. The lowest force demand on the deep neck flexors was the eye gaze only. Previous studies suggested that changing eye gaze may engage the deep neck flexors since gazing down is accompanied by slight cranial flexion [52, 53], and an optimal gaze angle of 20 degrees has been recommended to promote deep neck flexor activation [52]. However, just moving the eyes without an active attempt at craniocervical flexion in the current study was the least effective at activating the LC in both groups in the current study: eye gaze produced significantly smaller changes in longus colli activation compared to all the other exercises. The results of this study suggest that to robustly activate the deep neck flexors, a clear craniocervical component is needed for an exercise. When comparing the craniocervical flexion exercises (CCF Low, CCF High and CCF High with arm), activation results did not differ between the craniocervical flexion with a lower flexion force requirement (24 mm Hg) and the exercise that had a higher flexion force requirement (30 mm Hg). Adding resistance to the arm from a resistance band in the CCF High arm condition was an attempt to increase the complexity of the task beyond the highest level of the craniocervical flexion series. However, while adding arm resistance significantly increased activation across group, supporting that it was a more challenging exercise, it did not increase deep neck flexor muscle activation in the neck pain group more than craniocervical flexion alone. Adding a secondary task during craniocervical flexion, however, may be useful for other neuromuscular reeducation goals not captured by changes in muscle thickness. Dual-tasking, either cognitive motor interference or motor-motor interferences, can alter kinematic spatial-temporal parameters during movement [54, 55]. This potentially is a higher level of motor control not captured by observing changes in muscle thickness with exercise.
The deep neck flexor muscles, which include the LC, are more technically challenging to image than the neck extensor muscle using RTUS. Despite this, the ultrasound scanning protocol used in the current study was effective at capturing activation changes of the LC in both neck pain and healthy participants, despite resting measurement differences between groups. A prior study validated ultrasound CSA of the LC against MRI measurements in asymptomatic subjects and reported an average LC CSA of 1.22 cm SD
Strengths of the current study include powering the study based on pilot data using the exercises chosen in this study, having age matched groups to minimize age related group differences, and bias reduction efforts by having a rater blinded to group complete the offline measurements. Additionally, using ultrasound imaging provided a practical, non-invasive measurement method to assess deep neck muscle activation. However, several limitations exist, including the sample size. Despite being adequately powered based on an a priori power analysis, power was assessed using absolute thickness values and not percent change in activation which raises a concern about the study being underpowered. Given the importance of obtaining valid ultrasound measurements, adding a formal intrarater reliability component to the study would increase confidence in the results. Another limitation is that the exercises chosen to study were all in supine. Future studies are recommended to include non-supine activities to promote muscle activation in more functional upright positions. Additionally, expanding the sample population to include other forms of neck pain, particularly whiplash associated disorders would be beneficial.
Conclusion
Despite apparent atrophy in the deep neck flexor muscles, individuals with neck pain can activate their deep neck flexor muscles appropriately without activating their superficial neck flexor muscles in a supine series of craniocervical flexion exercises. However, adding a higher force load or introducing a secondary arm task does not proportionally increase muscle activation in individuals with or without neck pain. Instead, using a deliberate craniocervical flexion action rather than a cranial flexion action produces the most robust deep neck flexor activation.
Funding
None to report.
Ethical approval
The study was given ethical approval from the institutional review board at Midwestern University (approval code 20031).
Footnotes
Acknowledgments
The authors acknowledge all participants in this study.
Conflict of interest
The authors declare that they have no conflict of interest.
