Abstract
BACKGROUND:
Sleep quality in patients with chronic low back pain (CLBP) may affect quality of life (QoL), possibly due to worsening pain, central sensitization (CS), and cognitive factors. However, causal relationship among the factors has not been confirmed yet.
OBJECTIVE:
The purpose of this study was to test the hypothesis that sleep quality in patients with CLBP is attributable to pain, cognitive factors, and CS, and influences QoL, by structural covariance analysis.
METHODS:
This is a cross-sectional study. Participants were recruited from six health care facilities and 101 patients with CLBP were included. Structural covariance analysis assessed the fit of data to the model using goodness of fit index (GFI), adjusted goodness of fit index (AGFI), comparative fit index (CFI), and mean squared approximation error (RMSEA).
RESULTS:
The structural covariance analysis showed that the goodness-of-fit indices were high (GFI
CONCLUSION:
This study suggests that sleep quality in patients with CLBP is indirectly mediated through multiple pathways, including cognitive factors and CS, which may influence QoL.
Introduction
Chronic low back pain (CLBP) is one of the most common musculoskeletal disorders [1, 2]. Patients with CLBP experience prolonged pain, which affects their quality of life (QoL) [3]. CLBP is not only a physical problem but also comprises various factors that can decrease the QoL of patients. The role of psychosocial factors and central sensitization (CS) in patients with CLBP has been proposed [4, 5].
Psychosocial factors can influence negative emotions in the descending pain pathway, which is involved in increasing or decreasing pain [6, 7]. In particular, cognitive factors of pain, such as self-efficacy and catastrophic thinking, may affect pain exacerbation and QoL in patients with low back pain [8, 9, 10]. CS is defined as a symptom of dysfunctional regulation in the central nervous system in response to the normal or subthreshold afferent input signal [11, 12]. It amplifies not only chronic pain, but also other clinical symptoms, such as headache, fatigue, and muscle stiffness, and reduces QoL [5, 13, 14, 15]. Thus, simultaneous management of cognitive factors and CS is necessary to improve QoL in patients with CLBP. Recently, it was noted that sleep disturbances affect cognitive factors and CS. Sleep disturbance in patients with CLBP causes a decrease in the pain threshold [16, 17, 18, 19, 20]. Sleep quality is thought to negatively influence the prognosis of low back pain and QoL [16, 21, 22]. Previous studies have reported that quality of sleep can affect self-efficacy and catastrophic thinking regarding pain perception [23, 24]. It has also been suggested that sleep disturbances cause nerve inflammation and increase susceptibility to CS symptoms [25, 26, 27].
Therefore, we hypothesized that sleep quality in patients with CLBP would be affected by pain, CS, and worsening cognitive factors, which would subsequently affect QoL. Although sleep quality, pain, CS, cognitive factors, and QoL have been studied separately, the causal relationships among them are currently unclear.
The purpose of this study was to test the hypothesis that sleep quality in patients with CLBP is attributable to pain, cognitive factors, and CS, and influences QoL, using structural covariance analysis.
Materials and methods
Participants
This was a cross-sectional study. The subjects were recruited from August 2020 to December 2021, from six medical facilities. Participants were personally recruited by research collaborators at the six facilities via posters. Orthopedic surgeons at each of the facilities used X-rays and magnetic resonance imaging to diagnose cases of lumbar spinal diseases. The inclusion criteria were (1) patients with a primary complaint of low back pain persisting for
Sample size
The appropriate sample size is five times the free parameters based on a structural covariance analysis [28]. The free parameters of the present study were 17 (including residual variables), thereby requiring at least 85 participants.
Evaluation items
Basic information
Patient questionnaires and medical records provided details of age, height, weight, sex, duration of illness, and diagnosis. Height and weight were used to calculate body mass index (BMI).
Hypothetical model: Sleep Quality mediates each factors, leading to lower QoL.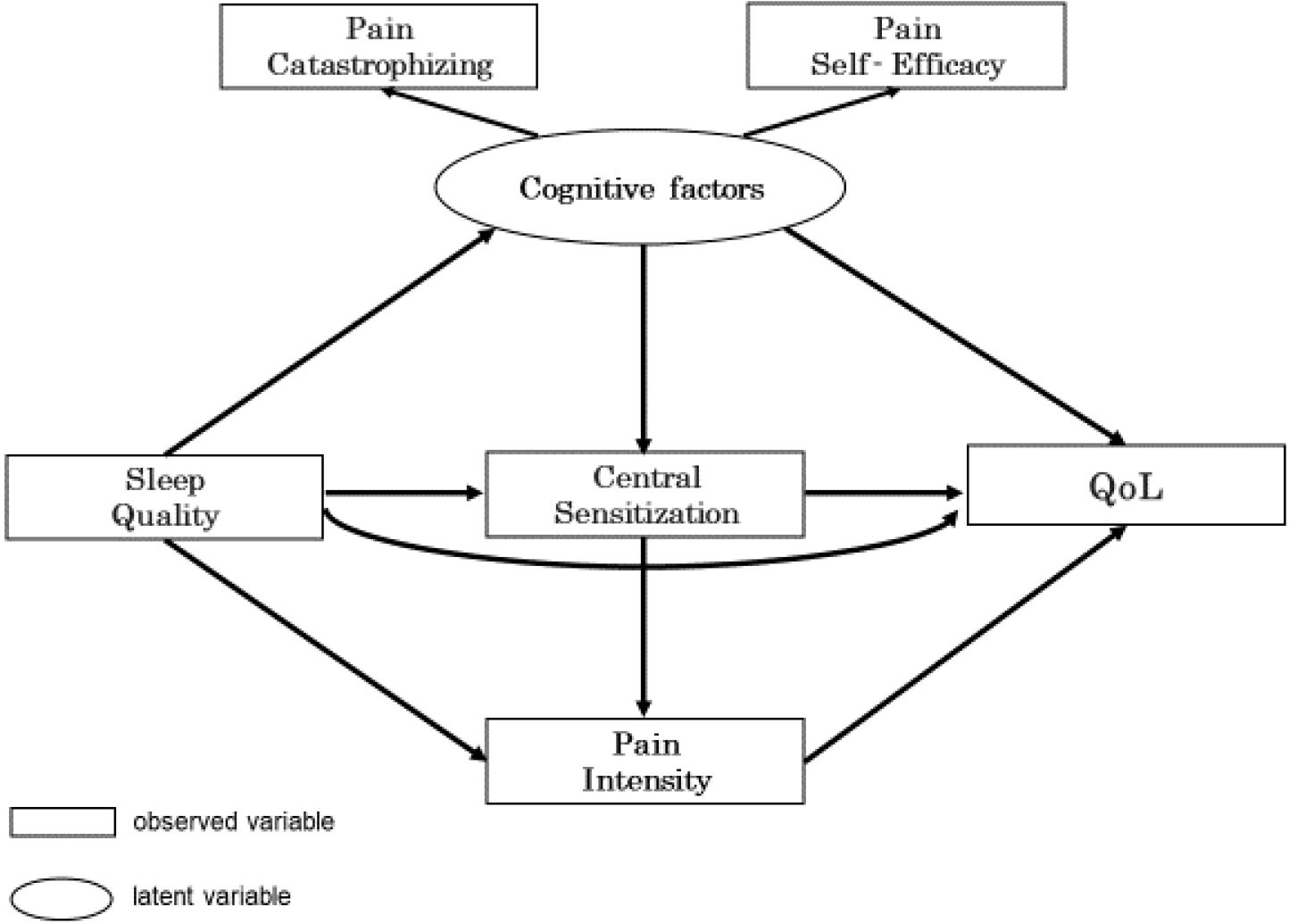
This study used the Japanese version of the ODI to assess specific QoL in patients with CLBP [29]. The total score of the 10 items was divided by 50, and the percentage was calculated; 0% indicated no disability and 100% indicated maximum disability. The higher the total score, the greater the severity of impairment.
Pittsburgh Sleep Quality Index (PSQI)
This study used the Japanese version of the PSQI to evaluate sleep disorders [30]. The PSQI evaluates the items of “sleep quality,” “time to fall asleep,” “sleep duration,” “sleep efficiency,” and “sleep difficulty” in the last month; the total score (0–21) was calculated. The higher the score, the worse the sleep quality.
Central Sensitization Inventory (CSI)
This study used the Japanese version of the CSI to assess the severity of CS [31]. The CSI consists of Part A (CSI score), which asks about health-related symptoms common to CS, and Part B, which asks about the presence of a diagnosis of a disease characteristic of CS. Part A consists of 25 items. The higher the score, the higher the severity of CS.
Pain Catastrophizing Scale (PCS)
The Japanese version of the PCS was used in this study [32]. The PCS consists of 13 items, each of which has a scale of 0 to 4, with “Ruminating,” “Helplessness,” and “Enlargement” Patients select numbers according to their symptoms. The total score was 52. The higher the total score, the greater were the catastrophic thoughts of pain.
Pain Self-Efficacy Questionnaire (PSEQ)
This study used the PSEQ to assess pain self-efficacy [33]. The PSEQ is a 10-item questionnaire with a score of 0 to 6 for: “How confident are you in your ability to answer questions during the pain phase”? The mean total score was 60. A higher score indicates higher self-efficacy for pain.
Numerical Rating Scale (NRS)
This study used the NRS to assess the intensity of back pain [34] , where 0 indicates no pain and 10 indicates the worst possible pain. A higher NRS score indicates more intense pain.
Statistical analyses
Pearson’s correlation coefficient analysis was selected after confirming that each variable followed a normal distribution using the Shapiro-Wilk test. Structural equation modeling was then used to evaluate the fit between the hypothetical model and the data. In the hypothetical model, PCS and PSEQ were latent variables of the cognitive factors. The causal model was designed such that sleep quality would affect pain, cognitive factors, and CS, leading to a decrease in QoL (Fig. 1). The goodness of fit was assessed using the goodness of fit index (GFI), adjusted goodness of fit index (AGFI), comparative goodness of fit index (CFI), and root mean square error of approximation (RMSEA) to evaluate the data model. The goodness of fit was based on GFI
The characteristics of the participants
The characteristics of the participants
Sleep quality was not directly influenced by QoL but rather Central Sensitization and Cognitive factors.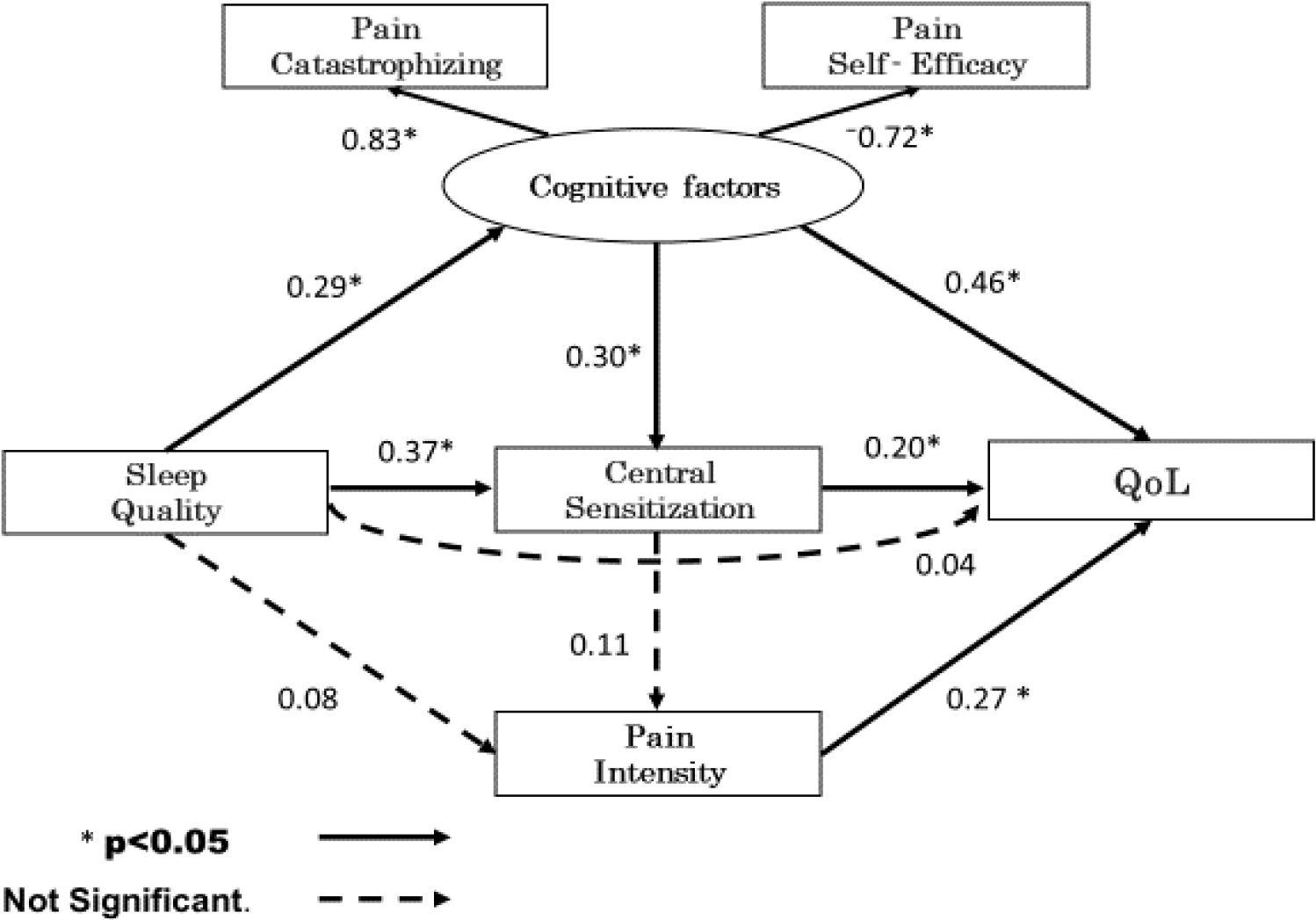
Correlation coefficient results for measurements
The data from 101 subjects were analyzed after excluding six subjects whose self-report questionnaire data were missing. The characteristics of the subjects are shown in Table 1. Correlation analysis showed that the ODI, PSQI, CSI, PCS, and PSEQ were all significantly associated. The NRS was correlated only with the ODI Age, sex, and pain duration were not associated with other factors (
Figure 2 shows the results of the hypothetical model, which omits the residual variables. The
Discussion
This study investigated the effects of sleep quality on pain, cognitive factors, CS, and QoL in patients with CLBP. The results revealed that sleep quality did not directly influence QoL, however it mediated cognitive factors and CS.
Sleep quality mediates cognitive factors and affects QoL. Previous studies have reported that sleep disturbances are associated with catastrophic thoughts regarding pain and self-efficacy. Sleep disturbances in knee osteoarthritis have been reported to aggravate catastrophic pain thoughts, and affect the sense of self-efficacy, and QoL [35]. A prospective cohort study of fibromyalgia patients reported that the longer the duration of total sleep, the less catastrophic thinking was observed [36]. Cognitive factors, such as increased catastrophic thinking and decreased self-efficacy, have been suggested to lead to excessive avoidance thinking, excessive muscle activity in the lumbar back muscles, poor variety of postures, and narrower range of motion [37, 38, 39, 40, 41]. Since the ODI reflects functional aspects of the lumbar region [42], sleep quality may influence cognitive aspects and induce lumbar avoidance behaviors and limitations in daily activities.
In this study, sleep quality mediated CS and affected QoL. In contrast, poor sleep quality did not affect pain intensity via CS. These are symptoms of CS in which sleep deprivation leads to a low-grade inflammatory response mediated by glial cells [26, 27]. It has been suggested that the symptoms of CS increase sensitivity to pain. On the other hand, CS has been reported to cause tension, headache, and fatigue regardless of pain [43]. Furthermore, there is a group with high CSI but low pain values, suggesting that symptoms such as tension, fatigue, and muscle stiffness can lead to decreased QoL [44]. CS has been reported to be weakly to moderately associated with functioning [45]. That is, exacerbation of CS due to poor sleep quality may affect QoL through multifaceted symptoms such as muscle hypertonia and fatigue rather than pain intensity.
Our study revealed that sleep quality did not directly affect QoL and did not directly mediate pain intensity. Some groups of researchers insist on the association between sleep quality, functional impairment, and lower pain thresholds. A previous study revealed a mean PSQI score of 8.1
Several studies [7, 10, 26, 35, 45, 46] have revealed that sleep quality in patients with CLBP is influenced not only by pain but also by cognitive and CS factors. Conversely, most of these reports emphasized that the influence of sleep quality in patients with CLBP was a complex phenomenon and that related factors needed to be simultaneously considered. Previous studies revealed no relationship among these factors; therefore, this study is the first to demonstrate that sleep quality in patients with CLBP affects QoL via CS and cognitive factors. Physical therapy for sleep disturbance is often recommended for patients with CLBP with no pharmacotherapy responses [27, 47, 48]. This study indicates that sleep-focused treatment in patients with CLBP may improve cognition and CS and help improve QoL.
This study has several limitations. First, data were obtained from outpatient records of CLBP requiring physical therapy. The treatment duration was not limited because the study was conducted at six sites. Furthermore, spinal disease diagnosis was not considered. Therefore, caution must be exercised in making generalizations, as differences in treatment duration and diagnosis may have influenced the results. Second, a self-administered questionnaire was used. Thus, the interference of recall bias and the potential overestimation is undeniable. Third, this is a cross-sectional study, which is not suitable for assessing a strict causal relationship. Lastly, a lack of perspective focusing on the emotional aspects, such as anxiety or depression, can affect the results, and the inclusion of these factors may be necessary for future studies. A longitudinal study that includes these emotional factors is recommended if a stronger causal relationship is sought.
Conclusion
In this study, we found that sleep quality in patients with CLBP is mediated indirectly through multiple pathways including cognitive factors and central sensitization, which may influence QoL.
Ethics statement
The study was conducted after obtaining approval from the Ethics Committee of the Graduate School of Health Sciences, Hirosaki University (Reference number 2020-009). In accordance with the Declaration of Helsinki, the purpose and methods of the study were fully explained to the subjects and informed consent was obtained.
Funding
No funding from any source was received for this study.
Author contributions
Study design and conceptualization: KM and HO. Data collection and analysis: KM, KY, KK and TS. Analysis and interpretation of data: KM, HO and RE. Manuscript drafting: KM, KY and HO. All authors read and approved the final manuscript.
Footnotes
Acknowledgments
The authors would like to express their deep appreciation to Dr. Eiki Tsushima, Graduate School of Health Sciences, Hirosaki University, for his comments on this paper. They would also like to thank Yuki Arihara (Omi Orthopedic Clinic) and Takafumi Kusamori (Doukikai Tomakomai Hospital) for their help in collecting the data.
Conflict of interest
None of the authors have any conflict of interest to declare.
