Abstract
BACKGROUND:
This study on gait biomechanics is based on a functional test (FT) performed at free and fast walking speeds.
OBJECTIVE:
We investigated the pattern of changes in gait biomechanical parameters and the knee function in patients after anterior cruciate ligament (ACL) injury or its reconstruction.
METHODS:
The study included 51 patients (33 males, 18 females) with a confirmed recent or old history of ACL tear, before or after reconstruction (26 patients). Mocap data was obtained using an inertial system. All patients were divided into three groups: 41 patients with physiological response (compensation), 6 patients with signs of decompensation, and 4 non-assessable patients.
RESULTS:
Increase in gait speed was associated with decrease in the walking cycle duration, stance and double support phases. In the compensation group, the physiological response of the knee was manifested in increased amplitudes. In the decompensation group, the amplitudes remained unchanged or decreased. In the compensation group, there were increases in the symmetrical muscle activity. The decompensation group was characterized by electromyography asymmetry.
CONCLUSION:
The observed universal physiological mechanism limiting the load on the damaged joint can be used for accurate assessment of the knee functional state in various periods of rehabilitation.
Introduction
Medical rehabilitation widely uses stress tests [1]. Human gait is important for efficient and safe locomotion [2]. Gait analysis provides important information about the functional capacity of patients with musculoskeletal disorders, which is also necessary for preoperative planning and further treatment [3, 4]. Timely assessment of biomechanical parameters during treatment response monitoring helps reduce the risk of developing pathological patterns of locomotion [5, 6]. Since gait abnormalities and asymmetries may indicate underlying musculoskeletal problems, their identification and elimination are of paramount importance [7].
Patients with anterior cruciate ligament (ACL) injury or ACL reconstruction often exhibit persistent changes in gait biomechanics [8, 9, 10], which may contribute to the development of post-traumatic knee osteoarthritis [11, 12]. ACL injury often leads to a neuromuscular deficit of the lower extremities [13, 14, 15]. The study [16] found a decrease in the knee flexion amplitude which lasted for up to 1.5 years after ACL reconstruction. Follow-up assessments found no asymmetries at 3.5 years post-surgery. At 8.5 years, however, a decrease in the amplitude was noted again. Another study [17] revealed a relationship between the strength asymmetry of quadriceps femoris and the biomechanical asymmetry of the lower extremities during walking.
Gait speed is a predictor of vital changes such as cognitive decline, cardiovascular complications, mortality, and the onset of depression symptoms [18]. Gait speed, one of the most important vital parameters, was termed as “the sixth vital sign” [18]. Repeated gait speed assessments can be used to evaluate functional changes in gait over time [19].
Spatiotemporal gait parameters, including step length and cadence, are the key factors that determine the gait speed during locomotion [20]. In healthy adults, both young and old, the stride length and cadence increase with gait speed [21]. In healthy individuals, the speed of gait significantly affects all its biomechanical variables: joint kinematics, ground reaction forces, muscle activity and spatiotemporal parameters [22, 24, 25]. Some authors describe a decrease in support phase with increasing gait speed [21, 23, 20], which is consistent with previous classical studies [24, 25].
The kinematic parameters of the joints and support reaction force also show a linear dependence on gait speed [26, 27, 28, 29, 30]. These relationships are described as coupled motion [31, 32, 33]. In healthy individuals, increase in gait speed is associated with proportional increase in muscle activity [26, 34].
In this study we investigated the possibility to use a functional walking test (FT) performed at free and fast speed to evaluate the response from the injured joint. This is important for subsequent rehabilitation management and other activities.
Materials and methods
Patient characteristics
The study population included a total of 51 patients (33 males, 18 females) with a confirmed recent or old history of ACL tear, either before or after reconstruction. The number of operated patients was 26, they were enrolled in the study 6 or more months after the surgical reconstruction. The period of time from the injury to the beginning of the study (in non-operated patients) varied greatly, from a week to 18 years, with a median of 4 years and 4 months. The median patient age was 38 years (20–55 years). The patient height ranged from 159 cm to 190 cm, with a median of 171.3 cm. The body mass index ranged from 17.58 to 35.73 kg/m
The study was conducted according to the principles of the Declaration of Helsinki and was approved by the local ethics committee of the Federal Research and Clinical Centre of Russia’s Federal Medical-Biological Agency (FNKC FMBA), Moscow, Russia (number: 1_6_21, date: 26 January 2021). All patients provided written informed consent before the tests.
Inclusion criteria: confirmed history of ACL tear, either before or after reconstruction; fully restored walking function [35, 36]. Previous studies have shown that for most cases of ACL tear, walking is fully restored within 6 (sometimes more) months after injury or after surgery [16, 35, 36].
Exclusion criteria: any pathological condition of the lower extremities that affect the walking ability, knee osteoarthritis grade II or higher according to Kellgren-Lawrence, inability to walk freely at a normal or fast speed.
The study group was homogeneous according to the analyzed biomechanical parameters. However, the patients’ clinical characteristics varied significantly. More than half of the patients (26 out of 51) had undergone ACL reconstruction, with a varying length of post-surgery period. The rest of them were not reconstructed, and the time period after injury significantly varied as well. However, all subjects had one essential common feature: the ability to walk without visible gait abnormalities and discomfort, both at a free and fast speed, which was critical in this study.
Inertial sensors and EMG electrodes are attached to the patient.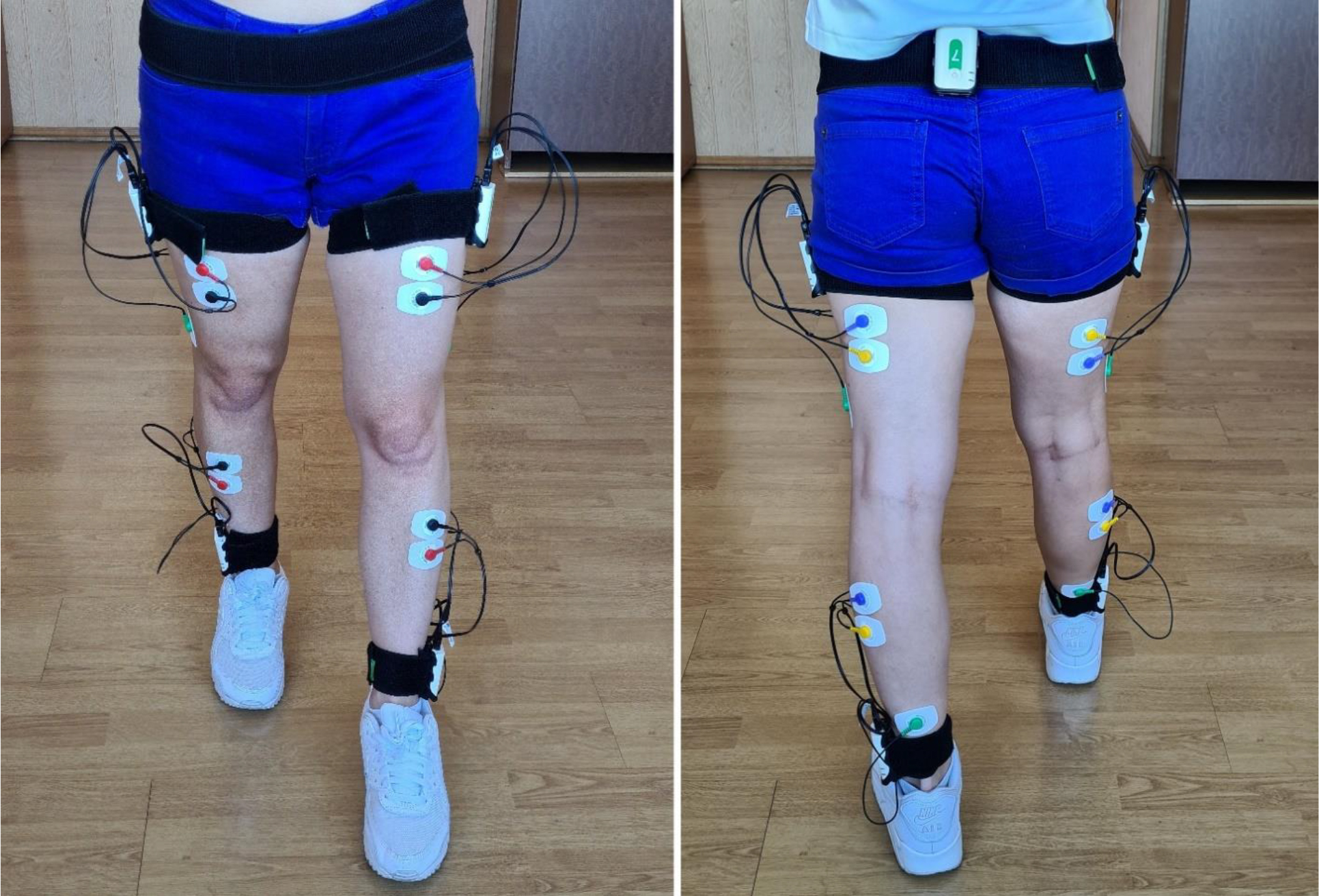
Biomechanical analysis of gait was performed using a Stadys system (Neurosoft, Ivanovo, Russia). Inertial sensors were attached to the patient’s sacrum, the outer middle third of the thigh, and the outer ankle on both sides (Fig. 1). Patients were instructed what type of footwear they should use in the FT. The research methodology was similar to that described in detail in our previous study [35, 37]. We used a recommended coordinate system [38]. Each sensor had two channels for electromyography (EMG) data. The thigh-located sensors captured the EMG of m. Rectus and Biceps femoris. The lower leg-located sensors captured the EMG of m. Tibialis anterior ?m. Gastrocnemius lateralis. Disposable Medico electrodes were placed according to the SENIAM guidelines [39].
The patient’s upright standing position with straightened hips and knees was assumed as neutral (calibration position). Biomechanical parameters were recorded while walking on a flat floor for a distance of 10 m with a turn at the end and continued walking. Steps with unsteady parameters (acceleration or deceleration) were automatically excluded from analysis. The remaining walking cycles (WC) were calculated. On average, a patient completed 30 or more walking cycles.
The system software provides standardized reports containing average data for all WCs immediately after completion of data capture. For subsequent statistical processing, each assessment report was converted into a table.
The following biomechanical parameters were selected for the subsequent analysis.
Temporal parameters: WC duration (s); rhythm (symmetry) index (RI) (the ratio of longer to shorter stance phase). Individual WC phases were measured as % of WC: stance phase (SP), single support (SS), double support (DS), and the parameter of the beginning of the contralateral leg WC (the beginning of the second double support phase, (SDS)). Spatial parameters: Foot clearance (cm), defined as the foot’s height during the swing phase; gait speed (V), km/h. Kinematic parameters: For hip joints: maximum range of motion (ROM) while walking (degrees). For knee joints: first flexion amplitude (a1) and its phase (x1), extension amplitude (a2) and its phase (x2), swing flexion amplitude (a3) and its phase (x3) as at previous research [37].
Temporal parameters of walking cycle. Same limb parameters in free versus fast walking in the compensated group. Where are: WC – walking cycle, SP – stance phase, SS – single support phase, DS – double support phase, SDS – beginning of the second double support phase
Temporal parameters of walking cycle. Same limb parameters in free versus fast walking in the compensated group. Where are: WC – walking cycle, SP – stance phase, SS – single support phase, DS – double support phase, SDS – beginning of the second double support phase
Muscles EMG was analyzed for maximum amplitudes developed over WC, at
The functional test was performed as follows. The biomechanical parameters of gait were first recorded during free walking at a natural, self-chosen speed. The patient then repeated the routine at a faster, but still comfortable, speed.
The FT results were then used to assess changes in gait biomechanics. If the knee response to a faster walking was the same as in healthy subjects, it was assessed as compensation. Otherwise (e.g., the ROM of the joint decreased with higher speed), it was assessed as decompensation. Based on the assessments, all patients were divided into three groups: 41 patients with characteristic functional signs of compensation, 6 patients with signs of decompensation, and 4 non-assessable patients. The group with non-assessable response was excluded from analysis.
The compensation group included 12 female and 29 male patients, the decompensation group included 3 male and 3 female patients. There were 21 operated patients in the compensation group and 5 in the decompensation group.
Statistical analysis
The obtained data were analyzed using ANOVA in Statistica 12 software. Medians and quartiles (the 25th and 75th percentiles) were calculated. The data distribution differed from normal, therefore the significance of differences between free and fast walking was assessed using the nonparametric Wilcoxon test at
Hip Flexion-extension ROM and Knee kinematics in free and fast walking. Where are: H
– amplitude of Hip joint, Knee joint – the first flexion amplitude (a1) and its phase (x1), extension amplitude (a2) and its phase (x2), swing flexion amplitude (a3) and its phase (x3)
Hip Flexion-extension ROM and Knee kinematics in free and fast walking. Where are: H
Muscle EMG amplitudes in free and fast walking. Where are muscles: TA – tibialis anterior, GA – gastrocnemius lateralis, RF – rectus femoris and BF – biceps femoris
The results of the biomechanical analysis are presented in Tables 1–3.
In the compensation group: WC of both limbs in fast walking was 0.2 s (
SS of both limbs in fast walking was longer than in free walking (
In the decompensation group: WC in fast walking decreased by 0.2 s (
SS in fast walking increased compared to free walking (
In the compensation group, the hip ROM of both limbs in fast walking was 7 degrees (
In the decompensation group in fast walking, the hip ROM of the intact and affected limbs was, respectively, 9 and 8 degrees larger than in free walking (
In the compensation group, the first flexion amplitude (a1) in fast walking increased by 2.9 degrees in the intact, and by 3.7 degrees in the affected knee (
Goniograms of hip and KJs on the affected and intact sides in the compensation and decompensation groups. Along the vertical axis: flexion, degrees. Along the horizontal axis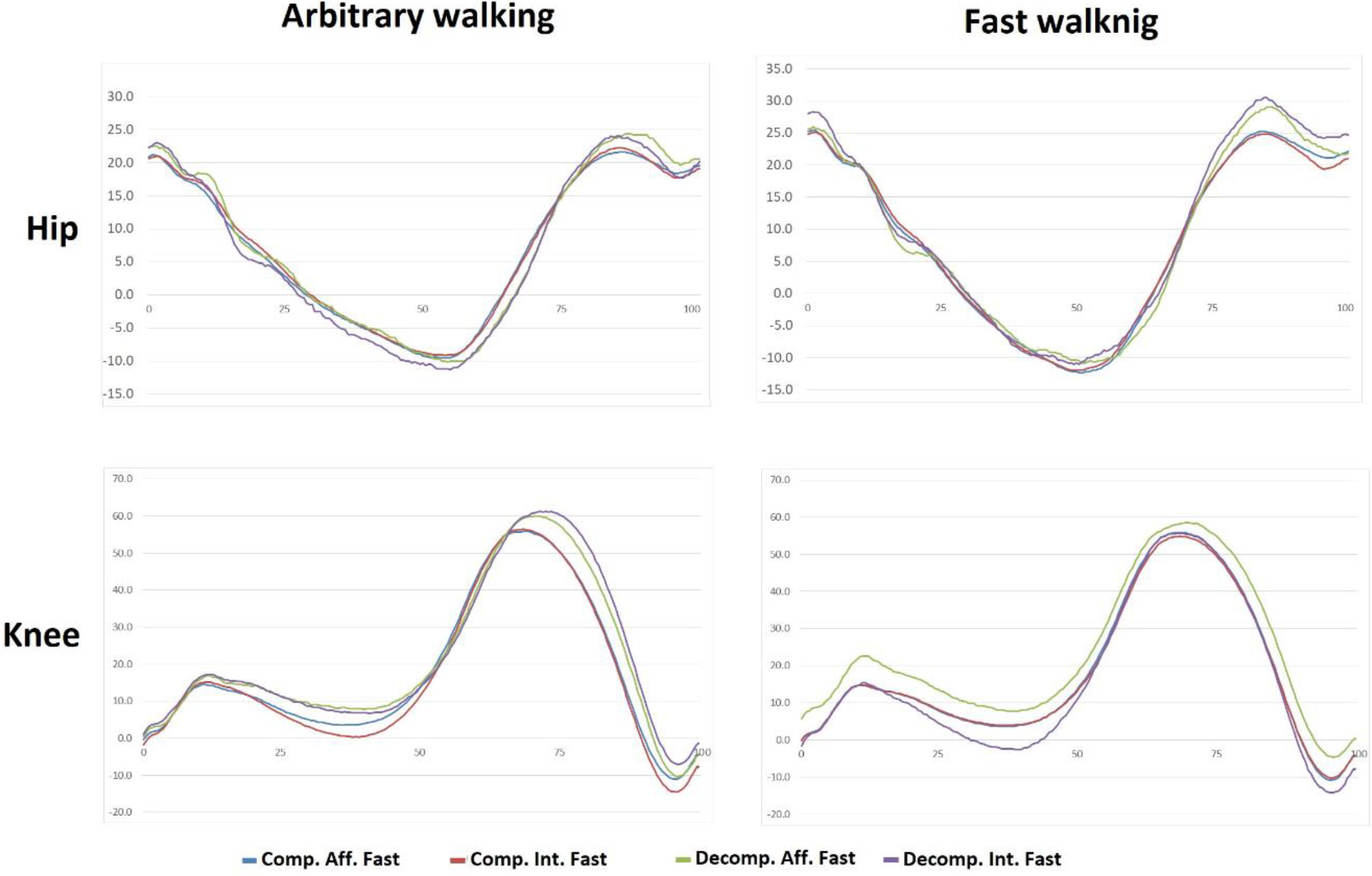
Envelope EMG for muscles under study, on affected and non-affected sides. Vertical axis: EMG amplitude, 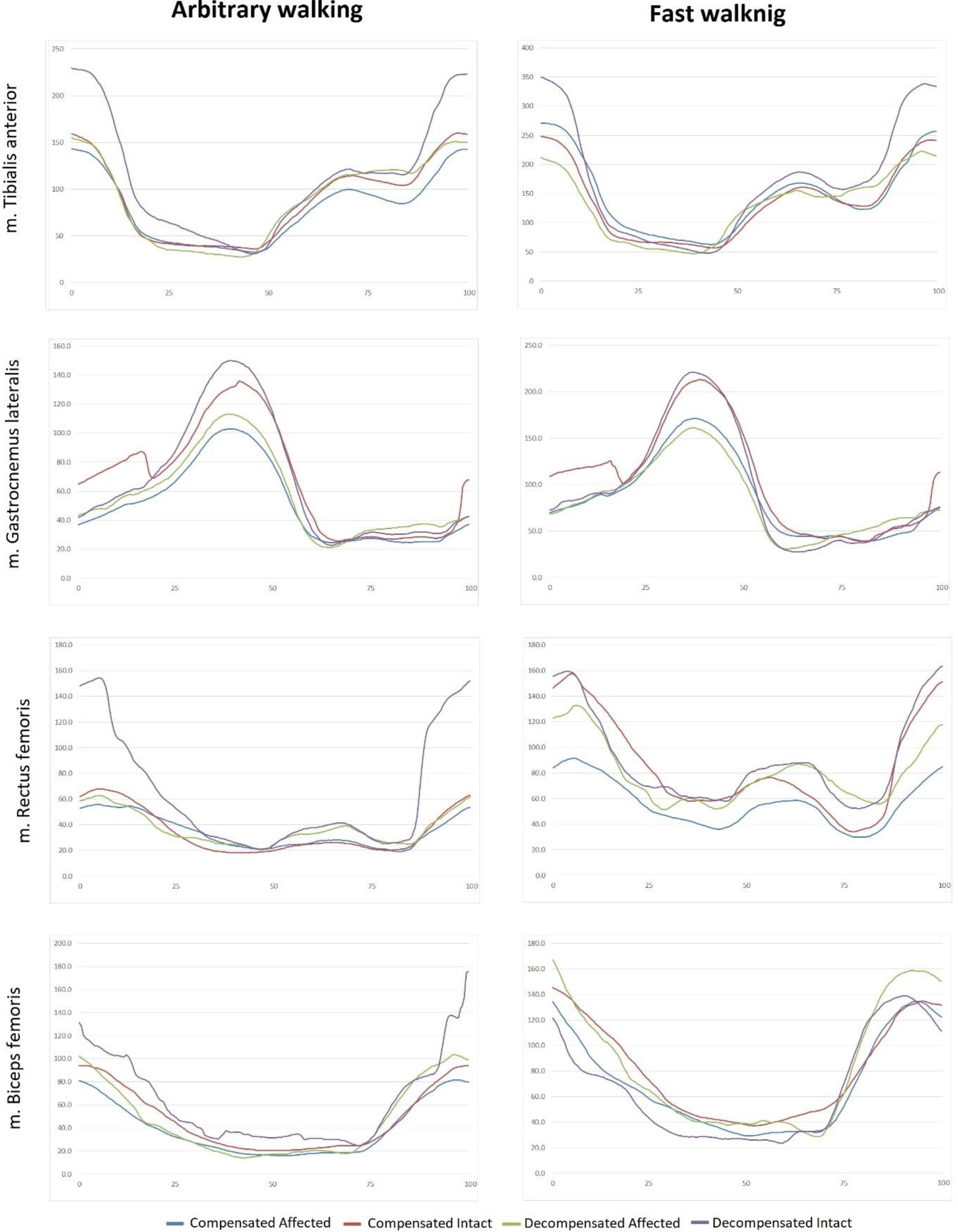
In fast walking, the extension amplitude (a2) on both sides decreased (
The swing amplitude (a3) showed no significant changes, yet the amplitude phase of the intact limb in fast walking was 3.6% shorter (
In the decompensation group, the first flexion amplitude of the knee (a1) showed no significant changes. The extension amplitude in both knees (a2) in fast walking decreased (
In the compensation group, a faster walking speed was associated with a symmetrical increase in the activities of the TA, RF, and HM muscles (Table 3). In addition, there was asymmetry in the GA function between the affected and intact sides. In fast walking, the affected-intact side symmetry was retained only for GA.
In the decompensation group, there was a marked asymmetry between the affected and intact sides in free walking. Thigh muscles showed a symmetrical increase in EMG activity in fast walking, and their EMG amplitude significantly differed from free walking. The asymmetry in the function of lower leg muscles, already present in free walking, increased in fast walking.
A faster gait speed was associated with shorter WC, SP, and DS, and increased SS and RI, the latter, however, in the compensation group only. The hip ROMs increased in both groups. In the compensation group, this parameter increased symmetrically. In the decompensation group, however, fast walking was associated with asymmetric ROMs.
All types of the above-discussed responses, with the exception of asymmetries, are physiological and associated with walking speed changes in healthy individuals [20, 24, 25, 28].
The knee kinematics in the compensation group showed a physiological response, which manifested in increased amplitudes of the first flexion and the subsequent extension in the stance period along with unchanged (with a tendency to decrease) flexion amplitude in the swing phase. The affected side mirrored the movements of the healthy side. We assume that this is also a physiological mechanism to reduce the functional asymmetry between the left and right sides through compensatory capabilities of the healthy side. Such response of the knee to increased functional load can be characterized as physiological. The first two amplitudes change exactly as they normally do. The fact that the joint is in the process of recovery and its function is limited is manifested by the absence of changes in the swing flexion amplitude (a3). It is probably the ‘ceiling’ amplitude in the current functional state (Fig. 2).
In the decompensation group, we observed unchanged first flexion amplitude, increased joint extension in the SS phase, and decreased flexion in the SP phase. Here, the response was of a different type. The first thing that draws attention is the unchanged amplitude of the first flexion. Obviously, the joint is not yet able to adapt its function in this phase of locomotion. The phase involves the body weight acceptance by the leg put forward and the weight transfer. Actually, the first flexion of the knee in this phase plays a very important part as shock absorber and recuperator of energy. At that moment, it is probably maximum the patient can afford without a significant discomfort. Much more demonstrative is the decrease in the flexion amplitude (a3) compared to that in free walking. This is an obvious sign of the fact that fast walking cannot be supported by an adequate amplitude. The decrease is asymmetric. In fast walking, the asymmetry increased from 2 to 5 degrees. Finally, the extension amplitude in the single support phase was the only parameter that demonstrated a physiological response to a change in the walking speed [28, 29].
As evident from the hip and knee goniograms (Fig. 2), there is little difference between fast and free walking. The observed changes are both numerically and visually much smaller than in classic studies [20, 24, 25, 28].
Some persistent changes were observed in the electric activity of the muscles. The changes accompanied increase in walking speed (Fig. 3).
In the compensation group, they included increases in the muscle activity, symmetrical activity of the TA, RF and HM muscles, both at a free and fast walking speed. Only the GA muscle showed an asymmetric activity, with a decrease on the affected side in both walking modes. In the decompensation group, there was also increase in amplitude in fast walking, while the inter-side asymmetry was retained only for TA and GA muscles. For BF and RF muscles, asymmetry appeared only in fast walking. Thus, increase in the EMG amplitude asymmetry can be interpreted as a pathological response to increased load. In the compensation group, the asymmetry was not typical and did not increase in fast walking. In the decompensation group, however, it was present in free walking and increased even more in fast walking.
In decompensated patients, faster walking increased the asymmetries of biomechanical parameters. The same effect was noted in some studies in similar patient populations, but for other biomechanical parameters [32, 33, 40]. In two papers [32, 33], the authors came to the conclusion that studies of biomechanical parameters at different walking speeds can provide useful information for rehabilitation management.
In our study, the free walking speed was 3.96 km/h and 4.35 km/h in the compensation and decompensation groups, respectively. The fast-walking speed in the two groups was 5.63 and 6.02 km/h, respectively. In earlier publications, the free walking speed ranged 3.8 to 5.1 km/h in young healthy adults of a median age of 27.1 years [41, 42]. In an old-age group (mean age
This study was aimed to evaluate the type of response of the musculoskeletal system, physiological or pathological. The FT was used to assess the ability of the knee to accept load in fast walking only in patients with a relatively preserved knee function and adequate walking function. In addition, all patients in the study had a unilateral joint damage. We significantly expanded the inclusion criteria, by enrolling patients both with operated and non-operated ACL rupture, in order to be able to assess the response to increased loading in clinically diverse but functionally homogeneous patients. Another limitation of the study was its relatively small sample size. Four of the patients could not be referred to any of the two categories considered in the study. Hence, there can be more types of FT response than the two analyzed in the study. The design of the study did not include a detailed physical exam of the patients because none of them had any evident complaints at the time of the study. And one more limitation was that we did not employ a healthy control group.
Future studies should provide for a more rigorous targeted selection of clinically homogeneous subjects, including different recovery phases after ACL injury and reconstruction. This will help identify the limits within which the FT can provide useful information for rehabilitation management.
Conclusion
We identified two main types of response to increased walking speed in patients after ACL tear. In our study, most patients showed physiological response in the FT, with a single exception: there were no increases in the maximum swing amplitude in the affected knee. This exception was the sign that the knee joint had suffered damage in the past (even if the ACL was reconstructed). The other type of observed response was characterized by decreasing activity of the injured joint with increasing walking speed. Thus, a biomechanical gait study using FT can provide valuable information not available clinically. Further studies are needed to understand how the FT can be used in the process of rehabilitation.
Ethics statement
The study was conducted according to the principles of the Declaration of Helsinki and was approved by the local ethics committee of the Federal Research and Clinical Centre of Russia’s Federal Medical-Biological Agency (FNKC FMBA), Moscow, Russia (number: 1_6_21, date: 26 January 2021). All patients provided written informed consent before the tests.
Funding
The study was funded as part of the government assignment to Federal Research and Clinical Centre of Russia’s Federal Medical-Biological Agency (state registration no. 20.004.21.800).
Author contributions
DS – conception and design of the study, investigation of patients, data analysis, draft article writing, final article writing. AA – patient research, data analysis, statistical analysis, draft article writing, final article writing. SK – patient research, data analysis, organization of admission of patients for examination, editing draft. AA2 – selection of patients for the study, clinical examination, data analysis, draft editing. All authors contributed to manuscript revision and read and approved the final version.
Footnotes
Acknowledgments
The authors have no acknowledgments.
Conflict of interest
The authors declare that they have no conflict of interest.
