Abstract
BACKGROUND:
The effects of stretching exercises in fibromyalgia (FM) deserves further study.
OBJECTIVE:
To evaluate the effectiveness of a Physical Self-Care Support Program (PSCSP), with emphasis on stretching exercises, in the treatment of FM.
METHODS:
Forty-five women with FM were randomized to the PSCSP (
RESULTS:
Nineteen and 21 patients completed the trial in PSCSP and control groups, respectively. After 10 weeks, the PSCSP group showed significantly better FIQ (difference between adjusted means,
CONCLUSIONS:
A PSCSP emphasizing stretching exercises significantly improved FIQ and SRT scores, and may be a helpful therapy for FM.
Introduction
Fibromyalgia (FM) is a musculoskeletal condition of unknown etiology, characterized by chronic and widespread pain lasting longer than 3 months [1, 2]. It is frequently associated with depression and anxiety [1, 2, 3]. Neurobiological mechanisms involved in the pathogenesis include greater activation of painful afferent pathways, decreased serotonin levels, and consequent reduction in the activity of the Pain Inhibitory System [1]. FM treatment aims to control pain, reduce functional limitations, and improve quality of life through comprehensive strategies involving a multidisciplinary approach [4, 5, 6, 7].
Among the non-pharmacological therapeutic approaches to FM, physical exercise is an important step to improve the disease symptoms [8, 9]. In general, evidence has favored aerobic and, to a lesser degree, strengthening exercises [8, 9]. However, stretching, a kinesiotherapeutic exercise, enables the recovery of functional muscle length, range of motion, and flexibility, relieving stress and realigning posture [10, 11]. Given their easy execution and tolerability, muscle stretching exercises can be easily included as part of a self-care strategy to obtain and maintain improvements in FM symptoms [4].
There is evidence that supported self-care, an intervention where the individual plays a central role in determining his own health care, may effectively improve symptoms of chronic diseases. This approach implies close collaboration between the health care team and the users to define the problem jointly, set the goals, institute care plans, and solve problems that arise during chronic condition management. Supported self-care utilizes group dynamics to educate through didactic actions, information, and physical means. The goal is to inform the patient about his disease, making him capable of managing his symptoms autonomously, without depending on the permanent assistance of the health team. It involves, to some extent, principles of behavioral therapies because it requires the implementation of new lifestyle habits in the daily routine to promote health [12, 13, 14].
Considering the potential benefits of supported self-care for chronic diseases and the effects of stretching exercises on musculoskeletal pain syndromes, we developed a Physical Self-Care Support Program (PSCSP) emphasizing stretching exercises, relaxing, wellness, and posture techniques for the treatment of FM. Hence, we designed a randomized controlled trial to test the effect of the PSCSP on the impact caused by the disease (measured with the Fibromyalgia Impact Questionnaire) and on measures of pain and flexibility in FM patients.
Methods
Study design and ethical approval
This is a single-blind, parallel-group, superiority randomized controlled trial conducted from September to December 2012. The study was done in accordance with the principles of the Declaration of Helsinki, and its protocol was approved by the Research Ethics Committee of the Health Science Center at Universidade Federal de Pernambuco (UFPE) and registered under the CAAE (‘Certificado de Apresentação de Apreciação Ética’, Certificate of Presentation of Ethic Appreciation) number 00701512.7.0000.5208. This study did not receive external financial support, and the institution where the study took place (Hospital das Clínicas da Universidade Federal de Pernambuco) did not have any influence on the analysis, reporting, or interpretation of results. The study was retrospectively registered in ReBec (‘Registro Brasileiro de Ensaios Clínicos’, Brazilian Registry of Clinical Trials), identification code: RBR-6c7tns.
Subjects
Potential eligible candidates for participation in the study were patients attending the Outpatient Fibromyalgia Clinic at Hospital das Clínicas da Universidade Federal de Pernambuco (HC/UFPE). They were identified by one of the principal investigators (SL) through a review of medical charts. SL enrolled the patients into the study, inviting them to participate during medical consultations or by phone contact. To be included, the patients had to meet all the following criteria: women aged 30–55 years; fulfillment of the American College of Rheumatology 1990 and 2010 criteria for diagnosis of FM [3]; residence in the metropolitan area of Recife, Pernambuco; stable psychological, physical, and drug therapy in the preceding month. Exclusion criteria were patients attending physiotherapy sessions, using gait assistance devices, suffering from associated autoimmune rheumatic diseases, subjects with physical and/or cognitive deficits, or presenting uncontrolled comorbidities.
Randomization
Patients who fulfilled the eligibility criteria and agreed to participate and sign a written informed consent form were included in the study. A closed list of these patients was organized sequentially and numbered following the alphabetic order to serve as base for the random sequence application. A professional not involved with patent care and unaware of patients’ names generated a computerized random sequence (with fixed block size equal to 2) with the aid of the R software (R Foundation for Statistical Computing, Vienna, Austria), allocating patients to PSCSP or control groups, with an allocation ratio of 1:1. SL was responsible for assigning, at once, all participants to one of the groups by using the alphabetically ordered list of complete patient names and the computer generated random sequence. A post-hoc verification made by the researcher responsible for the statistical analysis (MB), checking patient names and the random sequence, confirmed that all patients were allocated correctly to the group they should have been.
Interventions
All participants entered the study simultaneously and were followed during the same time frame. Over 10 weeks, patients allocated to active treatment underwent the PSCSP. The main objectives of the PSCSP were to promote knowledge of the syndrome, provide instruction on stretching techniques, and promote postural changes in the daily activities of volunteers. It consisted of 10 weekly 90-minute sessions in fixed groups of approximately 10 people and was based on active learning methodologies. Supervised and guided by the research physiotherapist (SL), the sessions usually began with chat circles where experiences were shared and questions regarding the previous session were clarified. There were moments when the floor was used as part of the practice field with mattresses made available to provide comfort and aid. At the sessions, participants were given leaflets with exercises they had learned (to keep on with the treatment at their homes), relaxing and wellness techniques, and postural guidelines for conducting everyday activities and preventing intensification of symptoms. The material was self-explanatory, containing a wealth of illustrations and written in plain language. Teaching resources and strategies included slideshows, whiteboards, demonstrations, games, and group dynamics. The PSCSP included 36 posture tips and 46 self-stretching exercises, of which 11 were for the spine, 16 for the lower limbs, 10 for the upper limbs, and 9 for body mobility and flexibility. At first, the stretching was done in 3 sets of 30 seconds holding the position. Later, this was extended to 45 seconds and afterward to 1 minute, which is the ideal duration for maximizing the technique’s benefits. Awareness-raising and/or myofascial tissue releasing practices were also presented, extending the soothing effects of stretching. Preparatory techniques for the stretching included diaphragmatic breathing, active muscle relaxation, self-massage, and warm compress. The contents of the program were progressively distributed over 10 weeks to ensure that participants learn them and become physically prepared. The PSCSP was developed in three stages, as follows: I – Information and physical preparation (sessions 1–3); II – flexibility gains and treatment of specific body areas (sessions 4–7); III – promoting independence in managing symptoms through knowledge integration (sessions 8–10). Participants received supporting material at the end of the PSCSP (self-care kits, folders, music CDs, spiky balls, tennis balls, pool noodles, bandage tapes, guides with posture tips, etc.) to maintain the benefits of the PSCSP. For further details, see Appendix Text and Appendix Table 1; the original, more detailed and illustrated description of the procedures (in Portuguese) taken in each session is available online at
Patients allocated to the (inactive) control group were monitored at three clinical outpatient appointments with a rheumatologist (AR) to supervise the use of medications and fulfillment of the pain medication diary, but no change in treatment was made. They were told to keep their usual routine but avoid starting a new physical exercise practice of any kind. All patients in the control group were included in a waiting list and informed that, if the intervention proves effective, they would be allowed to receive the same training given to the PSCSP group. These activities took place in the following 4 months after the study ended.
Outcomes
At the baseline visit, patients were evaluated using a standard interview form collecting sociodemographic and clinical data by trained members of the research team (VF or EP). On the same occasion, the Fibromyalgia Impact Questionnaire (FIQ) [15], the Visual Analogue Scale (VAS) for pain [16], and the Sit and Reach Test (SRT) [17] were also applied. The FIQ is a targeted instrument for assessing FM patients’ current health status, encompassing functional capacity, employment status, psychological distress, and physical symptoms. It consists of 19 questions organized into 10 items; higher scores (the maximum possible score is 100) indicate a worse condition [15]. The VAS for pain is a tool that evaluates the level of self-perceived pain, and scores range from 0 to 10, where zero reflects the complete absence of pain and 10 means maximum pain [16]. The Sit and Reach Test is used to assess muscular flexibility and is performed using a Wells bench. Subjects are asked to remain seated on the floor with their legs stretched straight ahead and the soles of their feet flat against the box. They are then directed to reach forward as far as possible with their hands on top of each other while breathing out, moving the bench’s scalimeter as far as they can. The maximum value obtained after three repetitions is used [17]. Patients were also asked about pain in specified body areas (left and right mandibles, shoulders, arms, forearms, hips, thighs, and legs, and cervical, dorsal, lumbar, thoracic, and abdominal regions) marking yes or no in a questionnaire. After an initial evaluation, patients received a pain medication diary, an instrument developed for this study to monitor the use of medication, where participants recorded the number of analgesic tablets taken each day.
At the end of 10 weeks, all patients were reassessed over two weeks (weeks 11 and 12) using the same instruments applied in their first evaluation. The study’s primary outcomes were the FIQ, the VAS of pain, and the SRT scores after 10 weeks of training. The FIQ sub-items measured semi-quantitatively or on VAS, the frequency of falls, the intake of analgesics, and the number of painful body areas served as secondary outcomes. The same researcher (VF or EP) who evaluated each patient at baseline applied the tests at the end of the study; these researchers were blinded to the treatment the patient was allocated to. Patients were explicitly instructed not to give any information that could permit the identification of the group they belong. All patients, independently of completing or not the training program, should return for the final evaluation. The number of analgesic tablets taken during the study period was calculated using the pain medication diaries.
Statistical analysis
The sample size was calculated considering a mean FIQ score of 70 in the control group and 55 in the PSCSP group at the end of the study, with a pooled standard deviation of 15 [18]; statistical power was set at 80% for detecting a significant difference with a P value less than or equal to 0.05. Considering the possibility of a 30% loss to follow-up, the estimated sample size was 21 individuals in each group.
The data were analyzed using SPSS 20.0 software (IBM Corporation, Armonk, NY, USA), jamovi v.1.8 (The jamovi project, Sidney, Australia), and the ’rcompanion’ package of R v.4.0.1 (R foundation for statistical computing, Vienna, Austria). Quantitative variables were graphically and statistically tested (with the Kolmogorov-Smirnov goodness-of-fit test) for normality of distribution. Variables with normal distribution were presented as mean
Baseline sociodemographic and clinical features of the participants who completed the study (numbers represent number and percentage, except where indicated otherwise)
Baseline sociodemographic and clinical features of the participants who completed the study (numbers represent number and percentage, except where indicated otherwise)
PSCSP: Physical Self-Care Support Program; SD: standard deviation; FIQ: Fibromyalgia Impact Questionnaire; VAS: Visual Analogue Scale; SRT: Sit and Reach Test.
Missing data of continuous outcome variables were handled using complete case analysis (CCA, assuming that missing data occur completely at random [MCAR]) and multiple imputation (under the assumption that data were missing at random [MAR]). For multiple imputation, baseline variables (those reported in Table 1, except for active medications) and (non-missing) values of FIQ, VAS of pain, and Sit and Reach Test after 10 weeks were used to predict the missing values of the primary outcomes. Multiple imputation was performed separately for the PSCSP and the control groups; 20 imputed databases were generated using the Markov Chain Monte-Carlo (MCMC) method with linear regression [20], allowing a maximum of 100 iterations. In sensitivity analysis, considering that data may be missing not at random (MNAR), we performed delta-adjusted multiple imputation adding 15 to the imputed final FIQ score, assuming that non-completers would have a significant clinical worsening. In a post-hoc analysis, we categorized patients according to response (change in FIQ
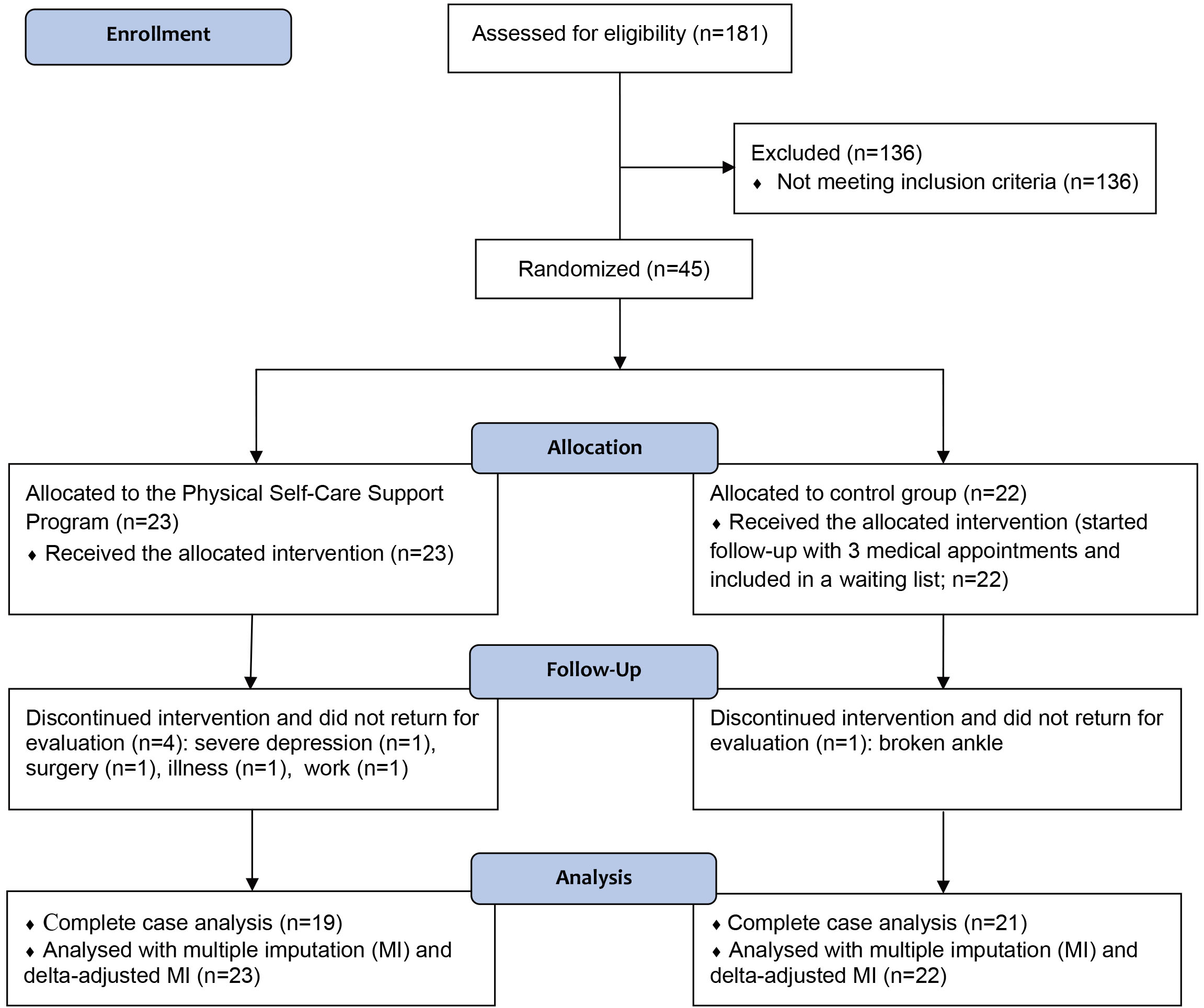
Out of 181 patients attending the Outpatient Fibromyalgia Clinic at HC/UFPE in August 2012, 45 individuals fulfilled the criteria and agreed to participate in the study; 23 were allocated to the PSCSP group and 22 to the control group. Four patients in the PSCSP group and 1 in the control group dropped out during follow-up due to reasons unrelated to the study (see Fig. 1); these individuals could not return for the evaluation of outcomes in due time, despite the efforts of the research team. The clinical and demographic features of individuals who completed the study and were included in the final analysis are depicted in Table 1. Most of these patients were middle-aged low schooling women; almost all patients (except for one individual) were on active medication treatment for FM. Half were professionally employed, and few were engaged in regular physical activity. Appendix Table 2 describes the characteristics of completers and non-completers of this study.
Results for the primary outcomes (FIQ, VAS of pain, and flexibility at weeks 11–12) using analysis of covariance (complete case analysis)*
Results for the primary outcomes (FIQ, VAS of pain, and flexibility at weeks 11–12) using analysis of covariance (complete case analysis)*
Footnotes
Conflict of interest
The authors declare that they have no competing interests.