Abstract
BACKGROUND:
Infections related to joint prosthesis are still a major concern for orthopedic surgeons. Hydroxyapatite (HA) is a useful biocompatible material because of its good osteocompatibility. Antibacterial HA coatings have been fabricated with addition of antibacterial agents such as Ag to HA using the plasma spraying method. However, the plasma-sprayed HA coating suffers from fractures at large thicknesses. The sputter-coated HA thin film has a high density, and has been applied clinically for dental implants. However, there are no extensive studies on Ag-containing HA thin films.
OBJECTIVE:
The aim of this study was to prepare an Ag-containing HA thin film by sputtering, and evaluate its antibacterial effect and cytotoxicity.
METHODS:
The Ag-containing HA thin films were prepared by radio-frequency magnetron sputtering. The films were characterized using X-ray photoelectron spectroscopy, X-ray diffractometry, and scanning electron microscopy (SEM). The antibacterial activities and cytotoxicities of the films were also evaluated.
RESULTS:
The Ag/Ca molar ratio of the films increased with the Ag ratio in the target. The SEM observation of the hydrothermally treated films showed surfaces covered with globular particles. All Ag-containing HA films exhibited an antibacterial effect against E. Coli. The number of cells of the films decreased with the Ag ratio. The films subjected to the hydrothermal treatment exhibited a higher number of cells than the as-sputtered films.
CONCLUSION:
The Ag-containing HA thin film was effective in terms of antibacterial effect, and had a low cytotoxicity in the proliferation of osteoblast cells at a low Ag ratio in the target.
Introduction
Implant infections are associated with biofilm formation. Bacterial cells growing within the biofilm exhibit an increased resistance to antibacterial agents [1]. Bacterial biofilms can form on any artificial surface. The infection rate of total knee arthroplasties is between 2.2% and 2.9% in primary total knee arthroplasty [2,3].
Infections related to joint prosthesis and fixation hardware are still a major concern for orthopedic surgeons. Hydroxyapatite (Ca10(PO4)6(OH)2, HA) is a useful biocompatible material because of the beneficial osteoconductive effect [4]. Recent studies demonstrated antibacterial HA coatings with added antibacterial agents to HA. Ag has a broad antibacterial spectrum, strong antibacterial activity, and low toxicity [5,6]. Ag-containing HA coatings have been produced using the plasma spraying method with good antibacterial effects [7–9]. However, this method has a few drawbacks, including the thermal decomposition of HA by the high-temperature plasma and fractures occurring in film coatings with thicknesses larger than 40 μm [10–15]. A radio-frequency (RF) sputtering technique has been used to coat homogeneous thin films with thicknesses below 1 μm with a high adhesion strength to the substrate [16]. The sputter-coated HA thin film exhibited a superior bone bonding strength, and has been applied clinically for dental implants [17,18].
In this study, we aimed to prepare a Ag-containing HA by sputtering. We propose a mixture target of HA and Ag to prepare the Ag-containing HA thin film using the sputtering technique. The film was characterized using X-ray photoelectron spectroscopy (XPS), X-ray diffractometry (XRD), and scanning electron microscopy (SEM). Escherichia coli bacterial strain and osteoblast cells were chosen to evaluate the in-vitro antibacterial activity and cytotoxicity effect of the film, respectively.
Materials and methods
Materials
The substrates were titanium plates (10 × 10 × 1 mm3). Powdered HA (Taihei Corp.) and Ag (99.9%, FUJIFILM Wako Chemicals Corp.) were used as sputtering targets.
Four series of compounds were prepared at Ag/(Ag + HA) weight ratios of 0, 5.0, 10.0, and 20.0%. Disk-shaped targets (diameter = 50 mm, thickness = 4 mm) were fabricated using the powder mixture by pressing at 3 MPa.
Methods
Deposition
RF magnetron sputter coating was performed using a L-210HS-D magnetron sputter system (Canon Anelva Corp., Japan). The distance between the target and substrate was approximately 60 mm. Both target and substrate were water-cooled during the sputtering process. An RF generator operating at 13.56 MHz was coupled to the target electrode (cathode) through an impedance matching network. The sputtering chamber was evacuated to a pressure below 1 × 10−5 Pa using an oil-diffusion pump equipped with a liquid nitrogen trap. An argon gas (99.999%) was then introduced into the chamber using a mass flow controller at a constant flow rate of 10 sccm. The substrate was at floating without any extra heating or biasing during the deposition process. Before the deposition, the target was covered with a shield and pre-sputtered using Ar ions for 10 min. The deposition was performed using an Ar gas pressure of 0.5 Pa and discharge power of 50 W for each target. Each film was coated onto the titanium plate to fabricate 1-μm-thick film samples from each target (Ag/(Ag + HA) = 0, 5.0, 10.0, and 20.0%). The films fabricated from the four series of compound targets were labeled as Ag0, Ag5, Ag10, and Ag20.
The hydrothermal treatment was performed at 120 °C and 0.2 MPa in NaOH solutions in a stainless-steel vessel for 24 h to recrystallize HA. The NaOH solution (pH = 9.5) was used to reduce the film dissolution during the treatment [19]. After the treatment, the films were washed in distilled water to remove any residual NaOH solution. The hydrothermally treated films of Ag0, Ag5, Ag10, and Ag20 were labeled as Ag0-hyd, Ag5-hyd, Ag10-hyd, and Ag20-hyd, respectively.
The as-sputtered and hydrothermally treated films were identified using XRD (Ultima IV, Rigaku Corp., Japan) by a Cu K𝛼 radiation source operating at 40 kV and excitation current of 30 mA. The surface observation of the films was performed using SEM (VE-9800; KEYENCE, Japan) with an accelerating voltage of 15 kV. The Ag/Ca atomic ratios of the films were measured by XPS (JPS-9010TR; JEOL) with Mg K𝛼 radiation. The electrical resistances of the films were measured by an insulation resistance tester (IR4051-10, Hioki, Japan). The final result was obtained by the average of five samples.
Antibacterial effect
The survival rate of Escherichia coli (E. coli) was analyzed by the standard plate count method, by counting visible cells (colonies) in terms of colony forming units (CFUs). The bacteria strain was E. coli (IFO3972; NITE, Japan). E. coli was precultured in a Lysogeny Broth (LB) culture medium (Miller, Kanto Regents, Japan) for 24 h at 37 °C. The treated E. coli were resuspended and diluted to ∼2 × 105 CFU/mL with the LB culture medium. 20 μL of the diluted E. coli suspension was pipetted onto the films, and placed in a humidified atmosphere to prevent drying at 37 °C. The test specimens were Ag0, Ag5, Ag10, Ag20, Ag0-hyd, Ag5-hyd, Ag10-hyd, and Ag20-hyd. After 24 h, the E. coli suspension, which retained water droplets, was collected into a 0.15-M saline solution. An appropriate dilution of the collected E. coli was incubated at 37 °C for 24 h on nutrient agar medium plates (Nissui Corp., Japan) to determine the number of visible E. coli in terms of CFU. The antibacterial rate was calculated as a percentage of reduction of surviving E. coli in comparison to the control. Each final result was obtained by the average of three samples.
Cell proliferation
Osteoblast-like MC3T3-E1 cells (Riken BioResource Center, Japan) were cultured in a plastic cell culture flask (BD Bioscience, USA) in an alpha Modified Eagle Minimum Essential Medium (𝛼MEM; Gibco BRL, USA) supplemented with 10% fetal bovine serum (JRH Bioscience, USA), penicillin (100 U/mL), and streptomycin (100 μg/mL, Sigma) at 37 °C in 5% CO2. The cells were subcultured every three days with trypsin-ethylenediaminetetraacetic acid (EDTA) (0.25% trypsin with 1 mM of EDTA). The test specimens (titanium (control), Ag0, Ag5, Ag10, Ag20, Ag0-hyd, Ag5-hyd, Ag10-hyd, and Ag20-hyd) were sterilized using an autoclave, and then each specimen was placed in a 35-mm culture dish. The cells were seeded at a concentration of 5 × 104 cells on each dish.
After periods of 72 h, the cells were fixed with phosphate-buffered saline (PBS(-), Nissui, Tokyo, Japan) containing 3.7% formaldehyde for 10 min, permeabilized with PBS containing 0.5% Triton X-100 (ICN Biomedicals, Irvine, CA, USA) for 5 min, and rinsed with PBS containing 1% bovine serum albumin to block nonspecific protein binding. We also incubated the fixed cells with this blocking solution for 30 min before treatment with staining reagents. Actin filaments in the cells were stained fluorescently by incubation with Alexa Fluor 546-conjugated phalloidin (Molecular Probes, Eugene, OR, USA) at a concentration of ∼200 nM for 60 min. For fluorescence visualization of the nucleus, intranuclear deoxyribonucleic acid was stained with Hoechst 33342 (Invitrogen, Carlsbad, CA, USA) for 30 min. The stained nucleus was observed with a fluorescence microscope (IX-71, Olympus, Japan) for cell counting. Each reported experimental value was obtained using the average value from three wells.
Results and discussion
Elemental analysis of the films
Figure 1 shows the Ag/Ca molar ratios of the as-sputtered films and films subjected to the hydrothermal treatment. The Ag/Ca ratio of the films increased with the Ag ratio in the target regardless of the hydrothermal treatment. The Ag/Ca ratios of the as-sputtered films tended to be higher than the Ag/Ca ratio of the target. For example, the Ag/Ca ratio of the film prepared at a Ag/(Ag + HA) ratio of 10 wt% in the target was 0.14, higher than the Ag/Ca ratio of the target (0.10). This indicates that Ag atoms are more sputtered from the target than Ca atoms. The sputtering yield of Ag is higher than that of HA because the deposition rate of Ag is higher than that of HA. In our laboratory sputtering system, the deposition rate from the Ag target was more than ten times higher than that from the HA target.
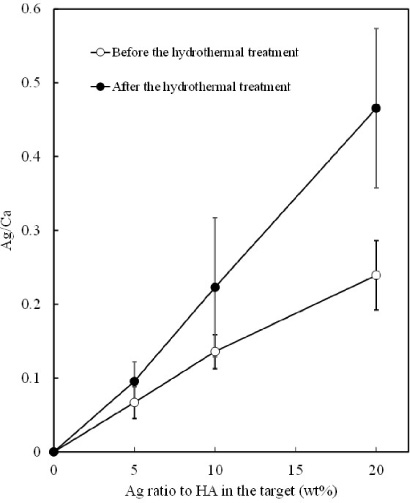
Ag/Ca molar ratios of the films as a function of the Ag ratio to HA in the target.
The Ag/Ca ratios of the films subjected to the hydrothermal treatment were higher than the Ag/Ca ratios of the as-sputtered films at any Ag ratio in the target. This indicates that more Ca atoms were released into the solution during the hydrothermal treatment than Ag atoms. The solubility of silver compounds varies largely depending on the partner element. However, the dissolution pattern of AgHA under hydrothermal conditions is not clear, and can be a subject for future research.
Figure 2 shows XRD patterns of the as-sputtered films of Ag0–Ag20. All films exhibit only three strong peaks at 2𝜃 = 35.1, 38.4, and 40.1°, corresponding to titanium because the as-sputtered films have a low crystallinity of HA [9].
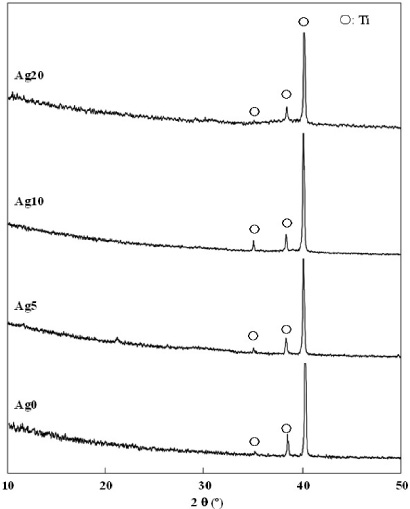
XRD patterns of the as-sputtered films (Ag0–Ag20).
Figure 3 shows XRD patterns of Ag0-hyd–Ag20-hyd, subjected to the hydrothermal treatment. Some peaks corresponding to HA appeared for all films, which indicates that the HAs of all films were crystallized. For Ag20-hyd, the (111) peak from Ag was observed at 2𝜃 = 38.1°, indicating growth of Ag crystallites. The substitution of Ag+ ions for Ca2+ ions occurs preferentially in the Ca(1) sites of HA, which leads to an increase with the amount of Ag added in the range of atomic ratio of Ag/Ca between 0 and 0.058 [20]. According to the XPS results, the Ag/Ca ratio of Ag5-hyd was 0.092, which exceeded 0.058 (Fig. 1). The excess of Ag ions could aggregate to metal Ag. Therefore, the Ag peak appeared for Ag20-hyd, which had a large amount of Ag crystallites.
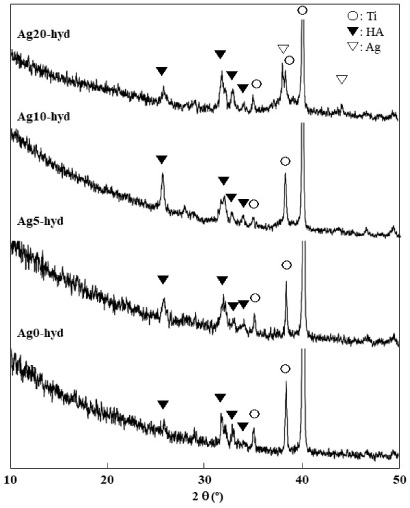
XRD patterns of the sputtered films (Ag0-hyd–Ag20-hyd) after the hydrothermal treatment.
Figure 4 shows SEM images of the as-sputtered and hydrothermally treated films. In all as-sputtered films, a few particles were observed. After the hydrothermal treatment, the surface morphology changed. The surface was covered with columnar particles. The size of the particles increased with the Ag content in the target. A driving force of crystal growth is the degree of supersaturation. An Ag-containing HA had a lower solubility than HA [21]. The decrease in the solubility in the Ag-containing HA film leads to an increase in the supersaturation. Consequently, the crystal growth rate of the Ag-containing HA increased with the Ag content.

SEM images of the films before and after the hydrothermal treatment.
Table 1 shows the electrical resistances of the as-sputtered and hydrothermally treated films. The as-sputtered film had a resistance of over 100 MΩ at an Ag ratio in the target below 15%, which decreased to 3.45 MΩ at an Ag ratio in the target of 20%. After the hydrothermal treatment, the resistances of the films significantly decreased, compared to that of the as-sputtered film. The large crystallite size of Ag reduced the conductivity of the film. The XRD patterns indicated that the crystallite size of Ag increased after the hydrothermal treatment because the Ag peaks emerged after the hydrothermal treatment (Fig. 2). The heat energy promotes silver agglomeration, increase in the crystallite size, and connection for charge carriers to flow yielding the resistivity decrease [22,23].
Electrical resistances of the as-sputtered and hydrothermally treated films
Electrical resistances of the as-sputtered and hydrothermally treated films
In all films, the resistances decreased with the increase in the Ag ratio in the target because of the increase in the content of silver as a conductor.
Figure 5 shows the spread plate result of E. coli collected from the droplet on the films. The antibacterial rates of the films were calculated by the number of E. Coli (Fig. 6). All Ag-doped films exhibited antibacterial effects against E. Coli. For Ag5-hyd, the antibacterial effect of the film subjected to the hydrothermal treatment had a value of 98.8%, slightly lower than that of the as-sputtered film (Ag5). The as-sputtered film had a higher solubility than the films subjected to the hydrothermal treatment because the as-sputtered film had a low crystallinity. The higher solubility led to a higher concentration of Ag in the E. coli droplet on the film, compared to the film subjected to the hydrothermal treatment.
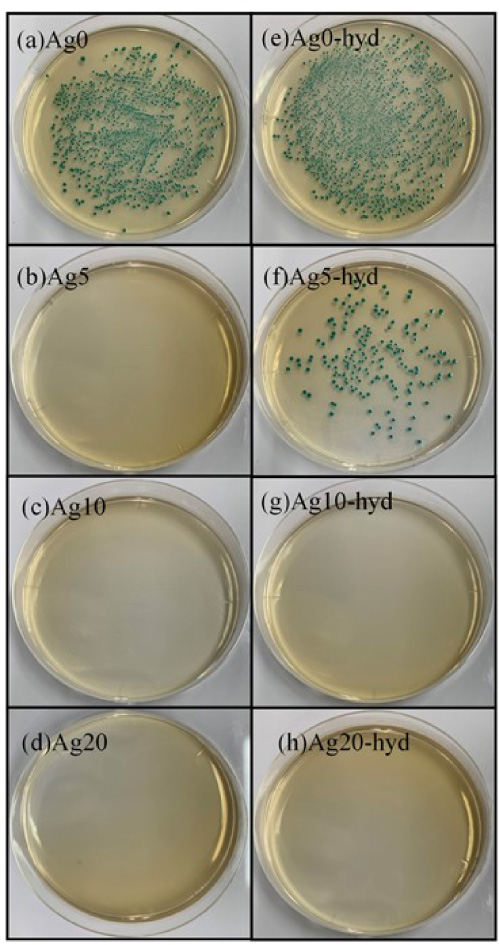
Optical images for the E. coli colonies cultured in an agar plate after incubation with different films.
The antibacterial mechanism of the action of Ag ions to bacterial cells is very complex. Several proposals have been reported to explain the antibacterial effect of Ag ions. First, Ag ions bind to the cell envelope, causing changes in their structure and permeability and inactivation of proteins. The Ag ions penetrate the cell, inhibit various proteins in the cytoplasm and ribosome, and interact with nucleic acids preventing replication and translation processes, causing cell death [24]. In this study, the mechanism of action of the Ag-containing HA film involves adhesion of bacteria cells to the surface of the films and penetration of Ag ions in the cellular envelope. The caused structural changes in the cellular envelope, particularly in the cytoplasmic membrane, might be the cause of bacteria cell death.
Figure 7 shows the proliferation of the osteoblast cells on the films. The Ag0-hyd film exhibited the highest cell proliferation rate, higher than that of the control (titanium). Ag0 exhibited a value of 6.3 cells, lower than that of Ag0-hyd, which was subjected to the hydrothermal treatment, because of the cytotoxicity of the as-sputtered film. The as-sputtered film includes calcium oxide and increases the pH of the medium [25].
For the films subjected to the hydrothermal treatment, the cell proliferation rate decreased with the increase in the Ag ratio in the target because of the cytotoxicity of the Ag ions. However, 10.5 cells existed even at an Ag ratio in the target of 20% (Ag20-hyd). The Ag-containing HA film exhibited an antibacterial ratio of 98.8% even at an Ag ratio in the target of 5% (Fig. 6). This indicates that the Ag ions exhibit an antibacterial effect at a low concentration.
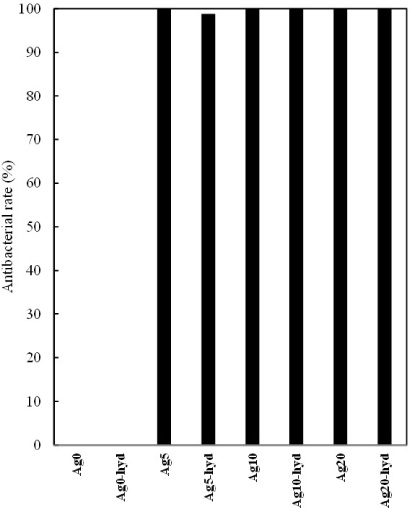
Antibacterial rates of the films against E. coli.
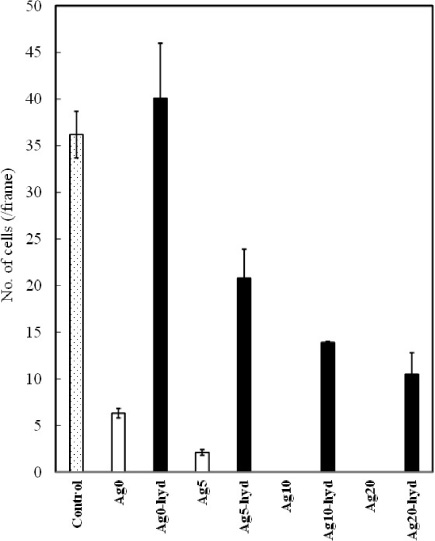
Proliferation of the MC3T3-E1 cells on the films.
In general, the antibacterial effect of Ag ions occurs at significantly lower concentrations than that of the cytotoxicity of Ag ions. Prokaryotic cells such as bacteria cells are significantly smaller than eukaryotic cells. Furthermore, prokaryotic cells have a lower structural and functional complexity than eukaryotic cells. Consequently, lower Ag ion concentrations are sufficient to achieve the same toxic effects as in eukaryotic cells [26]. According to the above results, the Ag-containing HA thin films have a potential for an antibacterial surface to avoid infection for artificial orthopedic implants.
The Ag-containing HA thin films were prepared successfully using the sputtering technique from mixture targets of HA and Ag. The following conclusions were obtained:
In the XPS analysis, the Ag/Ca ratio of the films increased with the Ag ratio in the target. The Ag/Ca ratios of the films subjected to the hydrothermal treatment were higher than the ratios of the as-sputtered films at any Ag content. According to the XRD patterns of the as-sputtered films, Ag0–Ag20 had non-crystalline HA. After the hydrothermal treatment, all films were crystallized, and the peak of Ag was observed for Ag20-hyd. In the SEM observation of the hydrothermally treated films, the surface was covered with globular particles, with sizes increasing with the Ag content. The as-sputtered films had higher electrical resistances than the film subjected to the hydrothermal treatment. All Ag-doped films exhibited an antibacterial effect against E. Coli. For Ag5-hyd, the antibacterial effect of the film subjected to the hydrothermal treatment had a value of 98.8%, slightly lower than that of the as-sputtered film (Ag5). In the cell proliferation of osteoblast cells, the number of cells of the films decreased with the Ag ratio in the target. The films subjected to the hydrothermal treatment exhibited a higher number of cells than the as-sputtered films.
