Abstract
BACKGROUND:
Inspired by natural bones, many organic components were added to Calcium Phosphate Cements (CPCs) to improve their mechanical strength. However, the strength of these composite CPCs is limited by the low strength of organic components itself and the weak interaction between organic components and CPCs.
OBJECTIVE:
Firstly, a composite CPC containing mussel-inspired adhesive, Poly-(Dopamine Methacrylamide-co-2-methoxy Ethylacrylate) (pDM) was developed. Secondly, the interactions between pDM and CPC and their effect on mechanical properties were investigated.
METHODS:
The interactions between pDM and CPC were performed by Nuclear Magnetic Resonance, Laser Raman, X-ray Photoelectron Spectroscopy, Fourier Transform-Infrared Spectroscopy and X-ray Diffraction Analysis.
RESULTS:
The toughness and compressive strength of pDM-CPC scaffold were both significantly enhanced, because of the enhanced interface binding strength among CPC and pDM due to their interaction and the improved mechanical strength of pDM owing to its self-oxidation cross-linking. The toughness of pDM-CPC scaffolds increased with the increased contents of pDM, while pDM-CPC scaffold containing 35 wt.% pDM had the highest compressive strength of all, which the latter was more than five times compared to that of CPC.
CONCLUSION:
The mechanically strong pDM-CPC scaffolds has potential application in bone regeneration as well as in craniofacial and orthopedic repair.
Introduction
The bone defect caused by trauma, tumor, deformity, degeneration, and an aging population has significant effects on patients’ quality of life, prompting an enormous demand for bone repair materials [1,2]. However, in the common bone repair materials, autograft (from the patient) is in short supply; allograft (from a donor) is widely available but there is a risk of immune-rejection and disease transmission [3]. Thus, the development of synthetic materials for bone repair is important at present. Calcium Phosphate Cement (CPC) is promising for clinical applications in bone repair due to its advantageous properties including bioactivity, osteoconductivity, injectability, moldability, and avoiding sintering [4,5]. However, the most significant drawback of CPC scaffolds is their brittle and low mechanical strength, which limited their utilization to only non-load-bearing bone sites [6]. Additional reinforcement of CPC is usually necessary to meet the mechanical requirements for bone regeneration.
The high strength and toughness of the human bone stem from the organic-inorganic composite structure, which can be imitated to enhance the mechanical strength of CPC scaffolds [7,8]. The biocompatibility components such as gelatin [9], silk fibroin [10], and chitosan [11] are added to CPC to obtain the composite CPC. However, the compressive strength of these compound CPC scaffolds is enhanced limitedly after adding the aforementioned compounds [12,13]. Recent studies suggest that the mechanical strengths of composite CPC scaffolds are directly associated with the mechanical priority of polymers added to the CPC. Xu et al. use different fibers to fabricate composite CPCs and confirm that the mechanical strengths of composite CPCs are linearly proportional to the strengths of fibers [14].
On the other hand, the interactions between organic components and CPC play an extremely crucial role in determining the mechanical strength of composite CPC scaffolds. Zou et al. find that the toughness of CPC is observably promoted by introducing polycaprolactone and poly-L-lactic acid which have relatively high mechanical strength. But the bending strength and elastic modulus of those composite CPC were still lower than that of pure CPC due to the lack of interaction between CPC and organic components [15]. Both the preparation and setting of CPCs are in a humid environment and thus the main interactions between CPC and organic components are disabled due to the presence of the molecular layer of water. Neuendorf et al. found that the binding strength between CPC and polymer is significantly reduced by about 80--90% after being placed in a humid environment for a short time (less than 30 h), due to the diffusion of interfacial water and the degradation of the polymer [16].
To sum up, the key factors influencing the mechanical strength of composite CPC are represented by the mechanical strength of the organic components and the interfacial adhesion between CPC and organic components in water. Mussel Foot Proteins (Mfp) enable the mussel to adhere to various solid surfaces in salty and humid conditions [17]. The role of the DOPA in Mussel and their inspired adhesive materials have been intensively researched especially in the wet environment [17–19]. The coexistence of catechol and amine (lysine) groups may be crucial for achieving adhesion to a wide spectrum of materials. Both Dopamine (DOPA) and the product of its self-polymerization (polydopamine) show extraordinary adhesive properties due to abundant catechol moieties [18]. The compressive strength of CPC is enhanced by adding dopamine and polydopamine in our previous research [19]. The composite CPC which contains dopamine or polydopamine shows enhanced mechanical properties. But the toughness and elasticity of dopamine and polydopamine were still poor because of their low molecular weight of them [20]. As a result, the mechanical properties of composite CPC are still limited [21]. To address these challenges, dopamine is grafted into the side chain of the polymers to form the mussel-inspired adhesive [22]. The toughness and elastic modulus of these polymers are enhanced due to the increased molecular weight given by graft copolymerization.
Thus, the objective of this study was to develop a novel reinforce CPC composite scaffold using mussel-inspired adhesive, poly-(dopamine methacrylamide-co-2-methoxy ethylacrylate) (pDM), for bone repairing (pDM-CPC). The interactions between pDM and CPC and its effect on the compressive mechanical properties of the pDM-CPC were investigated. The schematic of the present study is shown in Fig. 1.
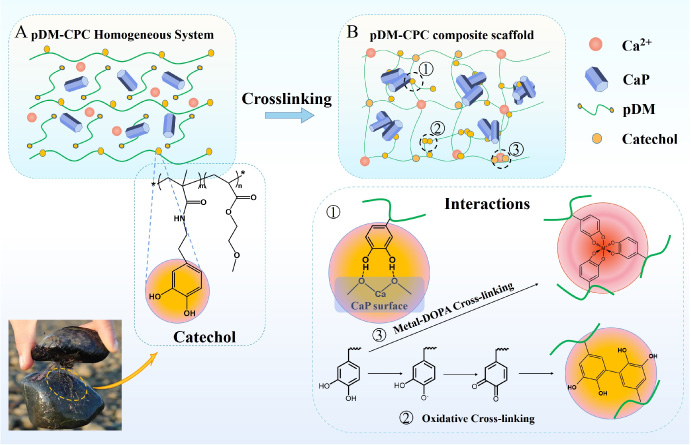
Schematic of the preparation for mechanically strong pDM-CPC scaffolds (A, B).
Preparation of CPC and adhesion pDM
The Biocement-D CPC was prepared, which was originally formulated by Driessens [23]. To recapitulate briefly, the CPC powder was mixed with 58% α-TCP (Sichuan University, Chengdu, China), 25% CaHPO4⋅2H2O (Kelong, Sichuan, China), 8.5% CaCO3 (Kelong, Sichuan, China), and 8.5% hydroxyapatite (HA) (Sichuan University, Chengdu, China). Mechanical activation was employed to process the aforementioned CPC powders to enhance their physical and chemical reactivity [24]. The mechanical activation course was as follows: 30 g CPC staring powders and 50 mL ethanol were mixed in a ball milling tank (QM-1SP4, Qixin, Sichuan, China) at speed of 250 r/min for 2 h. The mixture slurry was dried at room temperature and ground to obtain the final CPC powders. In this course, the mechanical energy was transformed to the internal energy of CPC powder [24].
The pDM was synthesized according to our previous studies [22,25]. Briefly, Methacrylate anhydride (Sigma, Steinem, Germany) was grafted to dopamine-HCl (Sigma, Steinem, Germany) to get dopamine methacrylamide (DMA) under the protection of nitrogen. Methoxy ethylacrylate (MEA, Aladdin, Shanghai, China), DMA and azobisisobutyronitrile (AIBN, Kelong, Chengdu, China) were added to tetrahydrofuran (THF, Kelong, Chengdu, China) and stirred until all the solids were dissolved under the protection of nitrogen. The concentration of DMA is 0.137 mol/L and the molar ratio of MEA: DMA: AIBN is 18.8:1:1.3. Above solutions were heated to 60 °C and then stirred 3 h under the protection of nitrogen. The reaction mixtures were diluted with 50 mL methanol and then added to 400 mL diethyl ether to precipitate the polymers. After being precipitated twice the precipitation was dried in the vacuum desiccator, pDM (a thick brown liquid) was obtained.
Fabrication of pDM-CPC scaffold
The pDM was dissolved in dichloromethane to have a final concentration of 100, 140, and 180 mg/mL, respectively. These pDM solutions were termed the liquid phase of the CPC paste. The CPC pastes with different contents of pDM were prepared by mixing CPC and the aforementioned pDM solutions based on the weight ratio of pDM to CPC in these pastes was 0.25:1, 0.35:1, and 0.45:1 (w/w), respectively. The CPC pastes whose liquid phase was deionized water were used as the control. Then the pDM-CPC pastes were mixed by magnetic stirring in the ice bath for 3 hours to distribute pDM uniformly in CPC. After that, these pDM-CPC pastes were put into the cylindrical mold tube (∅ 4.8 mm × 18 mm) and centrifuged for 10 min at 4000 rpm to get cylindrical scaffolds. The cylindrical scaffolds were dried at room temperature for 12 h to volatile the solvent completely, hydrated in 100% humidity at 37 °C for 24 h, and then immersed in deionized water at 37 °C for 24 h in turn. Four groups of specimens were thus fabricated and named 25%pDM-CPC, 35%pDM-CPC, 45%pDM-CPC, and CPC without pDM (CPC), respectively.
Characterization of pDM-CPC
Compressive mechanical test
The mechanical strength of pDM-CPC scaffold was characterized in the same manner in our previous research [26]. In brief, the compressive strength of the aforementioned four groups of specimens (n = 5 for each set) was tested by a universal mechanical testing machine (5567, Instron, Norwood, USA), respectively. The loading speed was 0.5 mm/min. The highest pressure was divided by the integration area of the stress curve to calculate the compressive toughness (106 J/m3) of this series of pDM-CPC scaffolds [27].
Nail test
The 25%pDM-CPC was used for the remaining test in 2.3 to investigate the interaction of pDM and CPC due to the highest contents of HA of all. The toughness of pDM-CPC scaffolds was characterized in the same manner in the literature [28]. The toughness of CPC and 25%pDM-CPC was evaluated by the nail test with a commercial steel nail (∅1.5 mm), which penetrated the samples (∅10 × 5 mm) at a constant rate of 1 mm/min−1 using the universal mechanical testing machine (5567, Instron, Norwood, USA) until the nail was inserted in the wood underneath the samples.
Nuclear magnetic resonance (1H NMR analysis
1H NMR spectra of a series of adhesives were performed by a Bruker 1H NMR spectroscopy at 600 MHz (AV spectrometer, Bruker, Germany) at room temperature. Deuterated methanol (CD3OD) was used as the solvent. Both the pure pDM and the pDM in pDM-CPC scaffolds were used for 1H NMR test to analyze the cross-linking of pDM during the hydration of CPC. The specimen for 1H NMR was prepared as follows: the aforementioned 25% pDM-CPC scaffold was ground into powders, these powders were washed with dichloromethane (3 times), then the dichloromethane solution was filtered and evaporated to separate pDM form 25%pDM-CPC.
Laser Raman analysis
Laser Raman spectrometer (DXi, Thermo Fisher Scientific, Waltham, USA) was used to characterize the structure of CPC, 25%pDM-CPC before hydration and after hydration. The samples were ground into powders and pressed for characterization. The laser wavelength was 780 nm and the spectrum range was 200--4500 cm−1.
X-ray photoelectron spectroscopy (XPS) analysis
The compositions of CPC and 25%pDM-CPC were characterized in the same manner in our previous research [19]. The CPC and 25%pDM-CPC was ground into powders, pressed, and characterized by XPS (ESCALAB Xi+, Thermo Fisher Scientific, Waltham, USA) with X-ray source (Al Kα, 1486.6 eV). The high-resolution spectra of O1s were recorded.
Fourier transform-infrared spectroscopy (FTIR) analysis
The pDM is coated on the KBr chip and the CPC and 25%pDM-CPC mixed with KBr (the mass ratio of the sample to KBr was 1:100) were ground into powders, pressed, and characterized by FTIR (Nicolet 5700, Thermo Nicolet, Waltham, USA). Spectra were recorded in a range from 400 to 4000 cm−1 with a resolution of 4 cm−1.
X-ray diffraction (XRD) analysis
XRD (X
′
Pert Pro, Philips, The Netherlands) was carried out to identify the crystalline phases of 4 samples fabricated in 2.2, the CPC without hydration (Wh-CPC) was used as control. The diffraction patterns were collected with a scanning angle 2θ ranging from 4° to 50° in step-scan intervals of 0.02° with Cu K𝛼 radiation at 40 kV and 40 mA. Some organic compounds could be the template to chelate calcium ions and serve as nucleation sites for calcium phosphate. It was inferred that the mineralization of calcium phosphate was influenced by catechol [29]. To describe this influence, the contents of hydroxyapatite (HA) in CPC, Wh-CPC, 25% pDM-CPC, 35% pDM-CPC, and 45% pDM-CPC were calculated by the data of XRD. The contents HA in each sample were calculated according to Eq. (1) [30].
SEM (JSM 7800F, JEOL, Japan) was used to observe the morphology of CPC and 25%pDM-CPC. The fracture surfaces of samples were coated with Au by sputtering.
Statistical analysis
All quantitative data were tested quintuple and expressed as mean ± standard deviation (SD). Statistical analysis was performed using students’ paired t-test and ANOVA. Significant differences were assumed at p < 0.05. Mean values with different letters are significantly different.
Results
Mechanical properties
The effect of the pDM content on the mechanical properties of CPC was shown in Fig. 2. In Fig. 2A, the compressive strength (MPa) of 25%pDM-CPC, 35%pDM-CPC, and 45%pDM-CPC were 16.57 ± 2.61, 29.71 ± 3.5, and 20.59 ± 1.81 respectively. The compressive strength reached a high point when pDM content increased to 35% and then start to decrease. The compressive strength of 35%pDM-CPC was almost 5 times of pure CPC. As shown in Fig. 2B, the strain (%) at maximum compressive strength of 25%pDM-CPC, 35%pDM-CPC, and 45%pDM-CPC were 8.93 ± 1.42, 11.77 ± 0.76, and 20.69 ± 1.36, respectively. In Fig. 2C, the toughness of CPC was rapidly enhanced by adding a small account of pDM. The compressive toughness (MJ/m3) of 25%pDM-CPC, 35%pDM-CPC, and 45%pDM-CPC were 0.67 ± 0.15, 1.58 ± 0.37, and 2.33 ± 0.47 respectively. Figure 2D showed the stress-strain curve of CPC, 25%pDM-CPC, 35%pDM-CPC, and 45%pDM-CPC. The graph shows that there has been a steady rise of strain at the maximum compressive strength and compressive toughness with the increase of pDM in scaffolds. Compared with the control group, pDM-CPC significantly enhanced the mechanical properties. The stress-strain curves of 35%pDM-CPC and 45%pDM-CPC show several yield stress points lower than the highest points in comparison with pure CPC and 25%pDM-CPC.
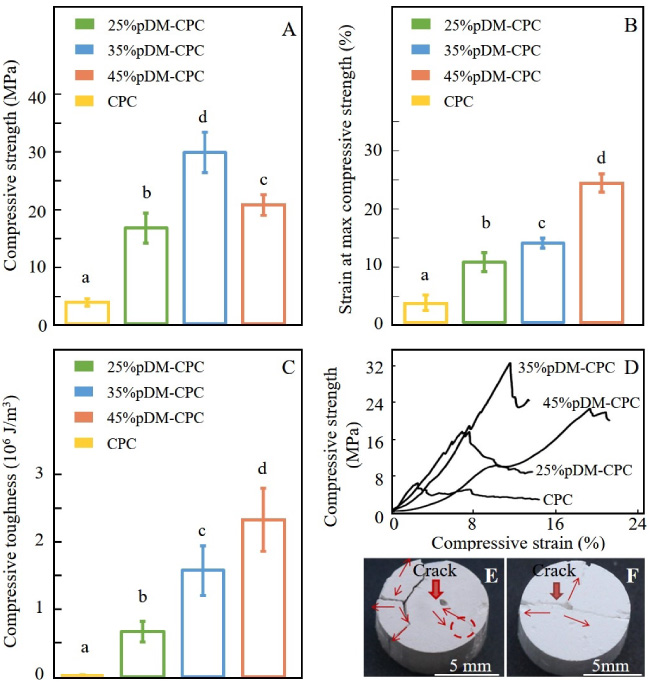
Mechanical properties of a series of pDM-CPC containing 25%, 35%, and 45% pDM, respectively, pure CPC was used as control. (A) The maximum compressive strength; (B) The strain at the maximum compressive strength; (C) The Compressive toughness; (D) The stress-strain curve; The images of surface crack growth, (E) 25pDM-CPC, (F) CPC.
Figures 2E and F show the crack propagation of 25%pDM-CPC and CPC. The crack propagation path of CPC was simple, and there was no obvious trend of bifurcation and deflection. On the contrary, the crack propagation path of 25%pDM-CPC had an obvious trend of bifurcation and deflection. The scaffold of 25%pDM - CPC was used for subsequent characterization. The reasons were that 25%pDM-CPC had a low hydration degree, which could comprehensively explore the interaction between CPC and pDM and this ratio is convenient to characterize due to the relatively low viscosity of pDM-CPC pastes.
The 1H NMR of pDM and 25%pDM-CPC are shown in Fig. 3A. The characteristic peak of pDM was consistent with that in the previous literature in Fig. 2A [22]. A total of 10 hydrogen signals were found in 1H NMR spectra of p(DMA-co-MEA), corresponding to the following hydrogen atoms [4]: [𝛿
H
6.65–6.75 (d, 2H, C6H
Figure 3B shows the Raman spectra of CPC and 25%pDM-CPC after and before hydration. All curves had characteristic peaks of P-O for CPC. Before hydration of 25%pDM-CPC, the peak at 2850–3010 cm−1 indicated organic C-H stretching vibrations, and the presence of peaks at 1450 and 1520 cm−1 was produced by the DOPA ring. After hydration of 25%pDM-CPC, the peaks at 506 cm−1 and 539 cm−1 could be attributed to the chelation between Ca2+ and phenolic oxygens of DOPA [12].
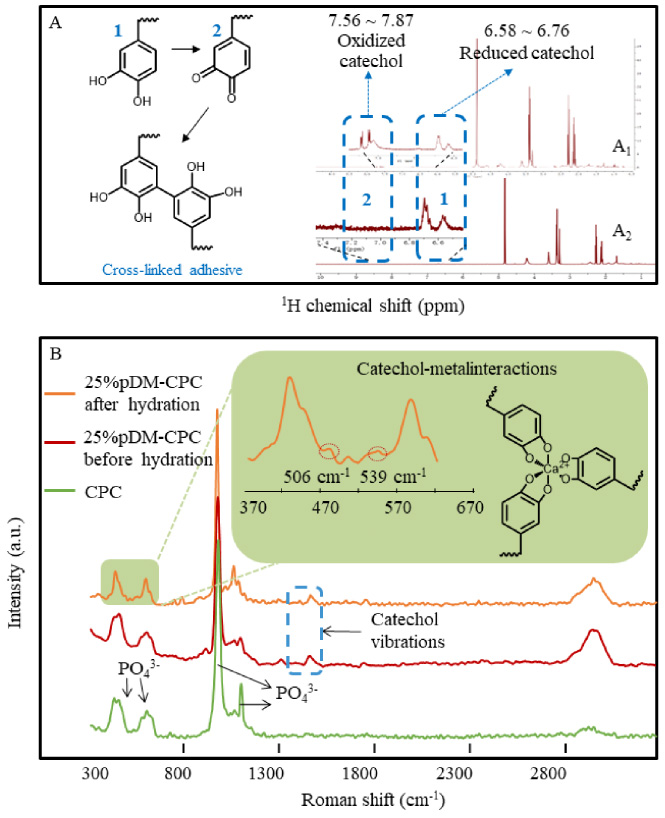
The cross-linking of pDM in composite scaffolds; (A) 1H NMR spectrum of pDM (A1) and pDM in 25%pDM-CPC (A2); (B) The Raman spectrum of CPC and 25%pDM-CPC before hydration and 25%pDM-CPC after hydration.
Figure 4A and B show the high-resolution XPS spectra of O1s in CPC and 25%pDM-CPC, respectively. Peak I and II in O1s indicated P-O (C=O) and P=O (C-OH), respectively [31]. Peak I of CPC and 25%pDM-CPC were located at 530.3 and 530.5, respectively. Peak II of CPC and 25%pDM-CPC were located at 531.8 and 532.2, respectively. In contrast with CPC, peaks I and II of 25%pDM-CPC shifted toward the higher binding energy.
Figure 4C shows the FTIR of CPC, pDM, and 25%pDM-CPC. In the FTIR of pDM, the peaks in 1731 and 1260 cm−1 were attributed to the presence of C=O and C-N, respectively. The peak in 3390 cm−1 belongs to -OH. In the FTIR spectra of CPC, the peaks in 580 and 1065 cm−1 belong to PO4 3−, the peak in 1429 cm−1 belongs to CO3 2−, the peak in 877 cm−1 belongs to HPO4 2−; the peaks in 1645 and 3435 cm−1 belong to -OH. In addition to the characteristic peaks of CPC, the existence of the C=O peak in 25%pDM-CPC contributed to pDM. Compared with pDM, the intensity of -OH peak in 25%pDM-CPC was weaker and the bands became wider, and the wavenumber of -OH peak shifted from 3411 cm−1 to 3437 cm−1. Compared with CPC, the PO4 3− and CO3 2− peaks in 25%pDM-CPC shifted from 580 cm−1 to 582 cm−1 and from 1429 cm−1 to 1431 cm−1, respectively.
Some organic compounds could be the template to chelate calcium ions and serve as nucleation sites for calcium phosphate. It was inferred that the mineralization of calcium phosphate was influenced by catechol [32]. To describe this influence, the contents of hydroxyapatite (HA) in CPC, 25% pDM-CPC, 35% pDM-CPC, and 45% pDM-CPC were calculated by primary maximum peak integrated intensity of HA and all phases, respectively. Figure 4D shows the relative content of HA in a series of pDM-CPC. After hydration, all crystalline phases of CPC have been translated to HA, a thermodynamically stable phase, and the HA percentage of 25%pDM-CPC, 35%pDM-CPC and 45%pDM-CPC was approximately 73%, 70% and 65%. Compared to CPC, the HA percentage was significantly reduced. And the HA percentage decreased with the increase of pDM content.
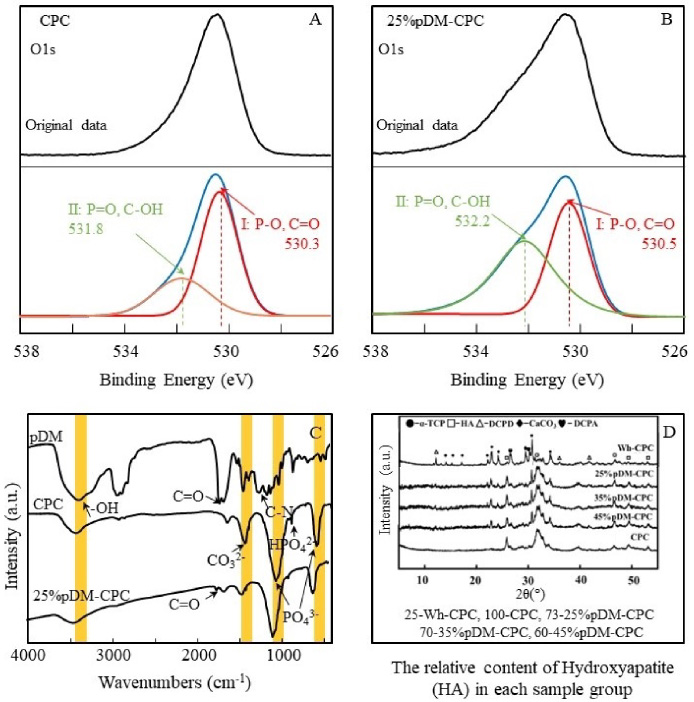
The interaction between CPC and CPC in pDM-CPC; (A, B) The O1s XPS spectrum of CPC and 25%pDM-CPC; (C) Fourier infrared spectra of pDM, CPC and 25% pDM-CPC; (D) The XRD data of 25%pDM-CPC, 35%pDM-CPC, 45%pDM-CPC, and CPC, the CPC without hydration (Wh-CPC) was used as control, the values before the samples (25, 100, 73, 70 and 60) were the relative content of HA after being hydrated for 24 hours.
SEM images are acquired to characterize the morphology of CPC and 25%pDM-CPC. Figure 5A shows the presence of flocculent HA crystals and microporous on CPC surface. But, on the surface of 25%pDM-CPC, there were many granular agglomerates (white square) in Fig. 5B.

SEM observation of (A) CPC and (B) 25%pDM-CPC.
CPC is an important bone substitute for the clinic and the mechanical properties are one of the key factors to be considered [33]. Strength is stress representing the intrinsic capability of a material to resist irreversible deformations, while toughness refers to the energy required to induce a fracture [7]. Usually, the toughness of polymer was higher than that of CPC but the compressive strength was just the opposite [34]. The energy that induces a fracture in the scaffold could be absorbed by the plastic deformation of the ductile phases [35], as a result, the toughness of CPC could be enhanced by adding pDM. The compressive strength of pDM was lower than that of CPC. Because of this, with more CPC being replaced by pDM, the compressive stress of 45%pDM-CPC scaffold began to decrease. The influence of pDM on the strain at the maximum compressive strength and compressive toughness of CPC has been shown to have a similar profile. Typically, the addition of fibers or ductile phases in composites acts to limit an initiated crack and reduce the stress and strain fields at the crack tip, preventing further opening [36]. On the other hand, in 25%pDM-CPC, the pDM was dispersed in the whole scaffold supporting that the initiated cracks were spread to every direction of the scaffold leading to the further opening of these cracks being inhibited.
The adhesion mechanism of pDM was analogous to that of the adhesive protein secreted by mussels [37]. The interaction of DOPA in pDM with Ca2+ occurs mainly through coordination bonds between the oxygen atoms of the hydroxy groups and Ca2+ of the surface on CPC [38]. Previous studies showed that DOPA produced hydrogen bonds with polar sites on mineral surfaces, such as oxygen atoms [39]. In the FTIR of pDM, the blue shift of the peaks belongs PO
The yield stress points before the highest points stress-strain curves of 35%pDM-CPC and 45%pDM-CPC indicated that micro-cracks generate in the samples during compression. Owing to inter particulate van der Waals forces and hydrogen bonding between the surface hydroxyl groups, CPC particles tended to agglomerate and form clusters under non-solvent conditions [41,42]. Additionally, CPC has a poor capability of dispersion in organic solvents and tends to agglomerate in a polymer matrix when added by blending [43]. In this research, pDM was dissolved in dichloromethane and mixed with CPC to get the pDM-CPC pastes. Such a method would inevitably lead to the agglomeration of CPC in pDM and the uniformity of pDM-CPC were limited. Agglomeration means that particles are combined loosely which can be simply broken by mechanical forces, which induce early failure at the interface and thus significantly limits the compressive strength of pDM-CPC [44]. Therefore, it is of great significance to tune the surface properties of CPC and improve its phase compatibility with the pDM to enhance the dispersion of CPC particles in the pDM.
In addition, DOPA-DOPA interactions contribute to the adhesive ability of pDM through their cross-linking [45]. Oxidative cross-linking of pDM itself resulted in an increased molecular weight due to high activity in the oxidative state [46]. In contrast, higher molecular weight generates supplementary chain entanglements to enhance cohesive bonding between chains [38]. The data of 1H NMR showed oxidative cross-linking of pDM (Fig. 1 reaction 2). This is one of the reasons for the enhanced mechanical property of pDM-CPC, which is consistent with our previous studies [22]. The pDM-CPC being prepared in a humid environment, free Ca2+ dissolved from CPC could form a coordination bond with DOPA [45]. The data of the Raman spectrum showed that pDM could form a network structure through metal-DOPA cross-linking (Fig. 1 reaction 3). Harrington et al. pointed out that due to the metal-DOPA cross-linked structure of the mussel byssus cuticle, the high cross-linking density region maintains the hardness of the mussel byssus cuticle, while the low cross-linking region maintains its ductility [47]. The pDM with network structure can effectively dissipate external energy under external force to increase toughness.
The CPC particles were anchored by pDM and the hydration ensued. During the hydration process of CPC, calcium phosphate salts are transformed into HA, a thermodynamically stable phase. The anchored CPC particles favored the overlap between particles to form chemical bonding and improve compressive strength. Moreover, the data of XRD indicated that the addition of pDM inhibited the hydration of CPC [12]. The loose CPC particles transformed into dense flocculent crystals due to the inhibited hydration.
Conclusions
A mechanically-strong composite CPC scaffold with improved compression strength and toughness was developed by using mussel-inspired adhesion for the first time. The toughness of the CPC scaffold increased with the content of pDM and the scaffold which contain 35% pDM had the highest compression strength of all. The mechanical strength of scaffolds was improved due to the enhanced binding strength between the CPC crystals given by the adhesion of pDM and the interaction between CPC and pDM. The pDM in the scaffold was cross-linked as a result of the oxidation of pDM and the interaction between pDM and CPC. Consequently, the mechanical strength of pDM in the scaffold was enhanced and the composite CPC scaffold was further reinforced.
The mechanically-strong CPC scaffolds which contain mussel-inspired adhesive have potential in bone regeneration, craniofacial and orthopedic repair. However, further studies should investigate the influence of pDM to the biological property of CPC by in vitro cell culture experiments.
Footnotes
Acknowledgements
This study is supported by the following programs: National Natural Science Foundation of China (Grant No. 52035012), Fundamental Research Funds for the Central Universities (Nos. 2682020ZT91, 2682021ZTPY026), the Basic Research Foundation Key Project of Sichuan Province (No. 2021JY0046), and Basic Research Foundation of Sichuan Province (No. 2022JDRC0088). We would like to thank the Analytical and Testing Center of Southwest Jiaotong University for their assistance with morphology and phase composition analysis.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
The authors declare that all data supporting the findings of this study are available within the article.
