Abstract
The identification of mutations in FGFR3 in bladder tumors in 1999 led to major interest in this receptor and during the subsequent 20 years much has been learnt about the mutational profiles found in bladder cancer, the phenotypes associated with these and the potential of this mutated protein as a target for therapy. Based on mutational and expression data, it is estimated that >80% of non-muscle-invasive bladder cancers (NMIBC) and ∼40% of muscle-invasive bladder cancers (MIBC) have upregulated FGFR3 signalling, and these frequencies are likely to be even higher if alternative splicing of the receptor, expression of ligands and changes in regulatory mechanisms are taken into account. Major efforts by the pharmaceutical industry have led to development of a range of agents targeting FGFR3 and other FGF receptors. Several of these have entered clinical trials, and some have presented very encouraging early results in advanced bladder cancer. Recent reviews have summarised the drugs and related clinical trials in this area. This review will summarise what is known about the effects of FGFR3 and its mutant forms in normal urothelium and bladder tumors, will suggest when and how this protein contributes to urothelial cancer pathogenesis and will highlight areas that may benefit from further study.
FGFR3 STRUCTURE AND ACTIVATION
FGF receptors are transmembrane receptor tyrosine kinases consisting of three extracellular immunoglobulin (Ig) domains, a single-pass transmembrane domain and an intracellular split kinase domain (Fig. 1A). There are four full-length receptors that conform to this structure (FGFRs 1–4). A fifth receptor (FGFRL1) lacks the intracellular domain and is thought to antagonise FGFR signalling by acting as a decoy receptor [6]. There are 22 known human fibroblast growth factors (FGFs), 18 of which are secreted and interact with FGFRs [7]. These bind to the extracellular domain of the receptor, with differential ligand-binding specificity determined by alternative splicing of FGFRs in Ig domain III. Although there are only 4 FGFRs, alternative splicing and the large number of FGFs provide highly variable signalling potential. In FGFR3, alternative inclusion of exons 8 or 9 generates isoforms 3b and 3c respectively (Fig. 1A). FGFs in the extracellular matrix interact with heparan sulphate proteoglycans (HSPGs) and binding of HSPGs by FGFRs helps to stabilise the ligand-receptor interaction. FGFR3-3b, expression of which is confined to normal epithelial tissues, binds FGF1 strongly and FGFs 9, 16 and 20 with weaker affinity, whereas FGFR3-3c can bind FGF2 and many more FGFs [8]. Binding of an FGF to a monomeric FGFR induces receptor dimerization, autophosphorylation of multiple tyrosine residues in the kinase domain and activation of downstream signalling pathways [9]. The activated receptor binds and phosphorylates FRS2 which recruits GRB2 and SOS to activate the RAS/MAPK pathway [10]. Phosphorylation of GAB1 leads to recruitment and activation of phosphatidylinositol 3-kinase (PI3K) [11], and PLCγ binding to FGFR3 independently of FRS2 leads to activation of protein kinase C [12]. Activation of STAT [13, 14] and RSK2 [15] can also occur. Signalling output, which is highly dependent on cell context, can lead to a range of changes including proliferation, migration and differentiation [7].
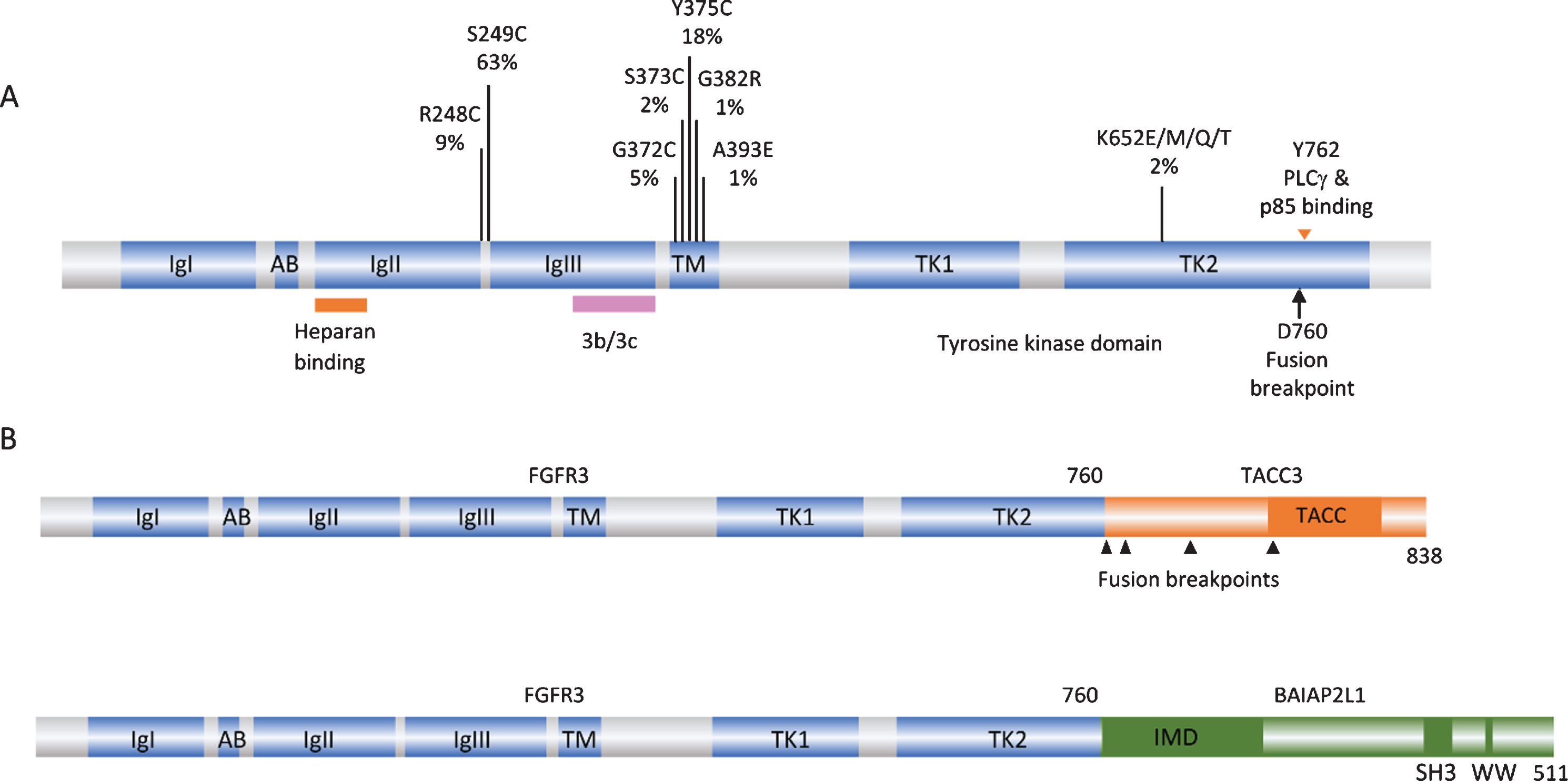
FGFR3 point mutations and translocations in bladder cancer. A. Structure of FGFR3 isoform 3b protein showing position of point mutations. Mutation data taken from tumors of all grades and stages (COSMIC, June 2020). IgI, IgII, IgIII: immunoglobulin-like domains. AB: acid box. TK1 and TK2: split tyrosine kinase domain. 3b/3c: region of exons 8 and 9 where alternative splicing generates isoforms 3b and 3c. B. Examples of FGFR3 fusion proteins identified in bladder tumors. TACC: transforming acid coiled-coil. IMD: IRDp53/MIM homology. SH3: src homology.
FGFR3 EXPRESSION IN THE NORMAL UROTHELIUM AND CULTURED NORMAL UROTHELIAL CELLS
In human urothelium, FGFR3 is the most abundantly expressed FGF receptor at both mRNA and protein levels [16, 17]. Normal human urothelial cells are highly proliferative in culture and can be maintained for multiple passages before senescence [18]. During this finite lifespan, FGFR3 mRNA and protein levels increase when cells are proliferatively quiescent at confluence and there is a major increase in expression as cells approach senescence [16]. These findings in normal urothelial cells provide clues that regulation of FGFR3 may be linked to urothelial contact inhibition and/or senescence.
The 3b isoform is the predominant full length mRNA FGFR3 isoform in normal urothelial cells. However, an isoform lacking exons 8–10 (Δ8–10) was found to dominate in cultured normal cells and, like the full length form, is upregulated at confluence. This isoform, which lacks the region encoding the second part of the third Ig-like loop and the transmembrane domain, is translated, glycosylated and secreted. It can bind FGF1 and dimerise, and was able to block the response to FGF1 in cells expressing full-length FGFR3. As Δ8–10 is expressed at lower levels in tumor cell lines, this implies that by binding and sequestering FGFs, or by binding and inhibiting signalling by the full-length receptor, this isoform may perform an important negative regulatory role in the normal urothelium [16].
ABERRANT ACTIVATION OF FGFR3 IN BLADDER CANCER
Alterations affecting FGFR3 signalling are found more frequently in bladder than in any other cancer type. Aberrant activation of the receptor can occur via several mechanisms. These are related to tumor stage and grade, and have different effects on downstream signalling and phenotypic consequences. Such aberrant activation may occur at different stages during the pathogenesis of NMIBC and MIBC and may cooperate with other events in a context-dependent manner.
FGFR3 point mutation
The first and most common mechanism of activation is missense point mutation [1, 19–24]. Mutations are located in several hotspots in the protein (Fig. 1A). By far the most common (63% of reported mutations) is S249C, followed by Y375C, these two mutations accounting for >80% of all mutations detected. These point mutations show a strong relationship to low tumor grade and stage [25]. Up to 85% of papillary urothelial neoplasms of low malignant potential (PUNLMP) and stage Ta tumors have a mutation, stage T1 tumors have lower frequency (∼20–40%) and MIBC lower still (12–15%) [17, 26–31].
When present in the germline, the common mutations found in bladder cancer cause severe, autosomal dominant and lethal forms of skeletal dysplasia. R248C, S249C, G370/372C and Y373/375C (3c/3b isoform numbering) cause thanatophoric dysplasia type I (TDI), and the kinase domain mutation K650/652E causes thanatophoric dysplasia type II (TDII) [32]. In this context, FGFR3 activation leads to premature cessation of proliferation of chondrocytes in the long bone growth plates [33].
Mutations resulting in replacement with a cysteine residue were assumed to cause formation of a disulphide bond leading to constitutive receptor activation. These mutant forms do indeed lead to constitutive receptor phosphorylation and ligand-independence [34], though when the strength of the dimers formed was measured by Förster resonance energy transfer (FRET) it was found to be relatively modest, suggesting that other structural perturbations in the dimer may contribute to the strong downstream signalling induced by these mutant forms [35]. In contrast, the K650/652E mutant form exists as a ligand-independent constitutively phosphorylated monomer [34, 36], and G380/382R and A391/393E remain predominantly ligand-dependent for activation [37, 38].
Although S249C is the most common mutation found in bladder cancer, R248C is much more common in the germline. These mutations are also found in the benign skin lesions seborrheic keratosis and epidermal nevi where, as in the germline, R248C is the more common mutation [39]. What determines the difference in prevalence in these situations? Several factors might contribute; there may be differential tolerance of certain mutations in the entire organism versus specific somatic tissues, somatic selection pressures may select for one rather than the other depending on context-dependent function, or the mutational processes at work may differ. An elegant study recently examined the relationship of S249C mutations and APOBEC-mediated mutagenesis in bladder cancer [40]. A large proportion of both NMIBC and MIBC have elevated expression of APOBEC enzymes and their mutational profiles contain a high load of predicted APOBEC-mediated mutational events [30, 41]. Of all FGFR3 point mutations found in bladder cancer, only S249C (TCC > TGC) shows similarity to an APOBEC-type mutation (TCN > T[G/T]N) where N is usually A or T. When the distribution of S249C mutations was related to APOBEC mutational load, a clear relationship was found in both NMIBC and MIBC [40]. APOBEC mutagenesis preferentially targets lagging strand ssDNA templates and it was shown that the coding strand of FGFR3 is mainly replicated as a lagging strand. The sequence context of S249 is predicted to allow a hairpin structure to form, also favoured by APOBEC enzymes. This study also demonstrated that the S249 codon sequence can be deaminated by APOBEC3A, providing a highly persuasive explanation for the excess of S249C mutations in bladder tumors. The differential mutation frequency in tumors of different stages, despite higher APOBEC activity in MIBC, likely reflects distinct pathogenesis pathways of these tumor groups.
Whether there are any differences in the selective advantage of the two most common mutations is not entirely clear. Although the most common mutations, S249C and Y375C, have similar oncogenic potency in immortal mesenchymal cells [40, 42], it is not certain that both confer the same advantage in urothelial cells. Knockdown of FGFR3 in bladder tumor cell lines bearing these mutations was shown to have similar transcriptional effects [40]. However, in cultured telomerase-immortalized normal urothelial cells (TERT-NHUC), a difference in the potency of these mutations has been found. Both mutations drive cells to a higher cell density at confluence, but S249C has the more potent effect [42]. Y375C is less strongly dimerised and retains some ligand dependence, suggesting that it may rely on stromal or autocrine ligand for maximal effect. Whether ligands are available in vivo at the time of receptor mutation is currently unknown. Thus S249C predominance can be explained as both APOBEC target and possibly in provision of a more potent selective advantage.
The strength of the phenotype of increased proliferation and cell viability at confluence in TERT-NHUC expressing point mutant forms of FGFR3 is in the order of mutation frequency found in tumors (S249C>Y375C>K652E) despite the fact that K652E is the most highly phosphorylated form in these cells [42]. This lack of contact inhibition was reflected in changes in cell-cell and cell-substrate adhesion, with the appearance of looser cell-cell junctions in cells expressing S249C- and Y375C-FGFR3 compared to controls and cells expressing high levels of wildtype FGFR3 [43]. The cells detached more readily from culture vessels coated with collagen IV, collagen I and fibronectin, and changes in expression of genes involved in mediating cell-cell and cell-substrate adhesion and extracellular matrix remodelling were found. This included downregulation of EPCAM and genes encoding structural components of desmosomes (DSC2, DSC3, DSG1, PKP1, PKP3), adherens junctions (CDH1, CDH16) and focal adhesions (PXN, ZYX) and upregulation of several integrins (ITGA2, ITGAV, ITGB5, ITGB6) and genes involved in extracellular matrix remodelling (HAS3, PLAT, PLAU, PLAUR, MMP10). Several of these genes were conversely altered when S249C-FGFR3 was knocked down in a mutant tumor cell line [43]. MMP10 has previously been identified as an easily-accessible pharmacodynamic biomarker for FGFR-targeted therapy in bladder cancer. It was also downregulated when FGFR3 was silenced, and both MMP1 and MMP10 levels were reduced in the urine of bladder cancer patients in a phase I trial of anti-FGFR3 monoclonal antibody [44]. Taken together, these data indicate that mutant FGFR3 has effects on regulation of the cell cycle in response to cell-cell contact, elicits changes in cell junctions and cell adherence to proteins found in the urothelial basement membrane and adjacent connective tissue [45], and induces changes in expression of potent modulators of the extracellular matrix, all functions predicted to provide a selective advantage to cells early in the process of tumor development.
FGFR3 fusion proteins
FGFR3 can also be activated by the generation of fusion proteins. These were initially identified when aberrant high molecular weight forms of FGFR3 were detected in cell lines and tumors [46]. Such fusions have subsequently been found in 2–6% of MIBC [30, 47–49]. FGFR3-TACC3 and FGFR3-BAIAP2L1 chimeric proteins have been identified, with FGFR3-TACC3 fusions appearing most common. Activating point mutations have not been found in these fusions. NMIBC have not been examined, though the very high frequency of point mutations in Ta tumors suggests that any fusions are likely to be found in T1 cases.
All of the fusions described to date contain the entire sequence of FGFR3 apart from the final exon (amino acids 1–760) fused to C-terminal regions of the fusion partner. Examples are shown in Fig. 1B. TACC3 lies 48 kb telomeric to FGFR3 on chromosome arm 4p and the common mechanism of fusion is tandem duplication and insertion so that the coiled-coil region of TACC3 is fused in-frame to FGFR3 exon 18. As a result of this mechanism, low-level gain of the region is detected in cases with translocation.
These fusion proteins show some constitutive dimerization and are constitutively phosphorylated, though they do retain some ligand-dependence [46, 50], and they are potently transforming in immortal rodent mesenchymal cells [46, 48]. The fusion partners are predicted to induce dimerization, as the coiled-coil domain of TACC3 is retained almost intact in all FGFR3-TACC3 fusions and the IMD domain, related to the BAR (Bin-Amphiphysin-Rvs) domain which can also promote dimerization, is retained in FGFR3-BAIAP2L1. As BAR domains are predicted to interact with membranes [51], FGFR3-BAIAP2L1 may show altered cellular localisation. Early experiments in glioblastoma, where similar FGFR3-TACC3 fusions are found, indicated increased aneuploidy when FGFR3-TACC3 was expressed [52]. TACC3 normally provides stability to the mitotic spindle and altered levels of expression are associated with changes in mitotic progression and chromosome segregation [53]. As FGFR3-TACC3 could be detected at the spindle poles, it was proposed that this directly caused defects in chromosome segregation [52]. However, the majority of FGFR3-TACC3 fusions lack TACC3 Ser558, phosphorylation of which is required for formation of the TACC3-clathrin-ch-TOG complex at the spindle [54]. More recently, it has been shown that the fusion protein recruits wildtype TACC3 away from the spindle and this was demonstrated in bladder tumor cell lines RT112 and RT4, which contain FGFR3-TACC3 fusions with different TACC3 components. The effect could be induced by the TACC3 part of the fusion protein alone and could be rescued by low-level overexpression of wildtype TACC3. Inhibition of the kinase activity of the fusion protein with the FGFR inhibitor PD173074 did not abrogate the mitotic defects, clearly indicating a role for the fusion partner component alone [55]. Thus the contribution of the FGFR3-TACC3 fusion proteins to the neoplastic phenotype comprises not only FGFR3 kinase-regulated functions but also a contribution to genomic instability via the TACC3 component.
Upregulated expression and isoform switching
Precise regulation of expression of appropriate FGFR3 isoform and ligands is required for normal physiological processes. Studies of protein expression by immunohistochemistry in stage Ta and T1 bladder tumors show upregulated expression of FGFR3 in 70–80% of Ta and 40–70% of T1 tumors [17, 56–59]. In Ta tumors this directly relates to mutation frequency but in T1 tumors exceeds the known mutation frequency and indicates likely upregulation of wildtype protein. Although activating point mutations in FGFR3 are relatively uncommon in MIBC, upregulated expression of the protein is found in 30–50% of cases [17, 59–62], though high-level amplification has not been reported. Whilst a few of the tumors expressing high levels might contain fusion genes, the majority are likely to be wildtype. Figure 2 shows the relationship of expression to mutation status in relation to tumor stage in a one-year cohort of tumors diagnosed at a single Institution [17]. Studies that have examined expression in matched tumor and lymph node metastases [60–62] have found good concordance, suggesting that upregulated FGFR3, particularly if related to FGFR3 mutation, could be a valid therapeutic target in such cases. However, some evidence suggests that upregulated expression in the absence of mutation may not denote FGFR3-dependence. Thus it is notable that upregulated expression in FGFR3 wildtype tumors in a series of patients treated by cystectomy was not associated with prognosis in the same way as mutation [63] and in a trial of Dovitinib (a multi-kinase inhibitor that inhibits FGFRs 1–3) in BCG-unresponsive NMIBC, complete responses at 6 months were only observed in patients with mutant and not over-expressing wildtype tumors [64].
In bladder tumor cell lines, expression of FGFR3 was found to be restricted to those that also expressed E-cadherin and TP63, both of which are “epithelial” characteristics, whereas cell lines expressing ZEB1 and vimentin, features of epithelial-mesenchymal transition (EMT), expressed FGFR1 rather than FGFR3. Compatible with the relationship of FGFR1 to the EMT phenotype, inhibition of FGFR1 led to decreased invasion in these cell lines [65]. In bladder tumor cell lines with epithelial phenotype, ectopic expression of FGFR1 and treatment with FGF2 can elicit an EMT [66], suggesting that there are highly distinct roles for these two receptors in bladder cancer. This is supported by studies of bladder tumors, where a non-overlapping pattern of mRNA expression of FGFR3 and FGF2, a potent ligand for FGFR1, is found, with FGF2-expressing tumors showing EMT characteristics [67]. As expected, at the protein level FGFR1 is upregulated in tumors of higher grade and stage, in which FGFR3 mutations are less common [68, 69]. However, the relative expression of FGFR1 and FGFR3 has not been examined systematically in a large tumor series.
In addition to changes in expression level, isoform switching to the 3c isoform, which binds a wide range of FGFs, is also predicted to play a role, potentially facilitating autocrine or paracrine signalling. This has been identified in MIBC-derived tumor cell lines [16] but has not been examined in tumor tissues. However, upregulated expression of FGF2 is common in advanced bladder tumors [67, 71] and increased levels of FGF1 and FGF2 can be detected in the urine of bladder cancer patients [72–75]. Increased levels of FGFs 19, 21 and 23, all of which bind FGFR3-3c, have been reported in the serum of patients with bladder cancer [76], though the full repertoire of FGFs that bind FGFR3-3c remains to be examined in tumor tissues.
REGULATION OF FGFR3 EXPRESSION
As indicated above, although expression of FGFR3 mRNA and protein is strongly related to mutation status in NMIBC [17, 78], this is not the case in MIBC (Fig. 2). Understanding the mechanism for this upregulation, and of any differential significance of mutant versus upregulated wildtype FGFR3, is of clinical interest, particularly in MIBC where FGFR3 inhibitor therapy may be used. As amplification of the FGFR3 region on 4p has not been identified and low-level gains are uncommon, other mechanisms of upregulation must exist.
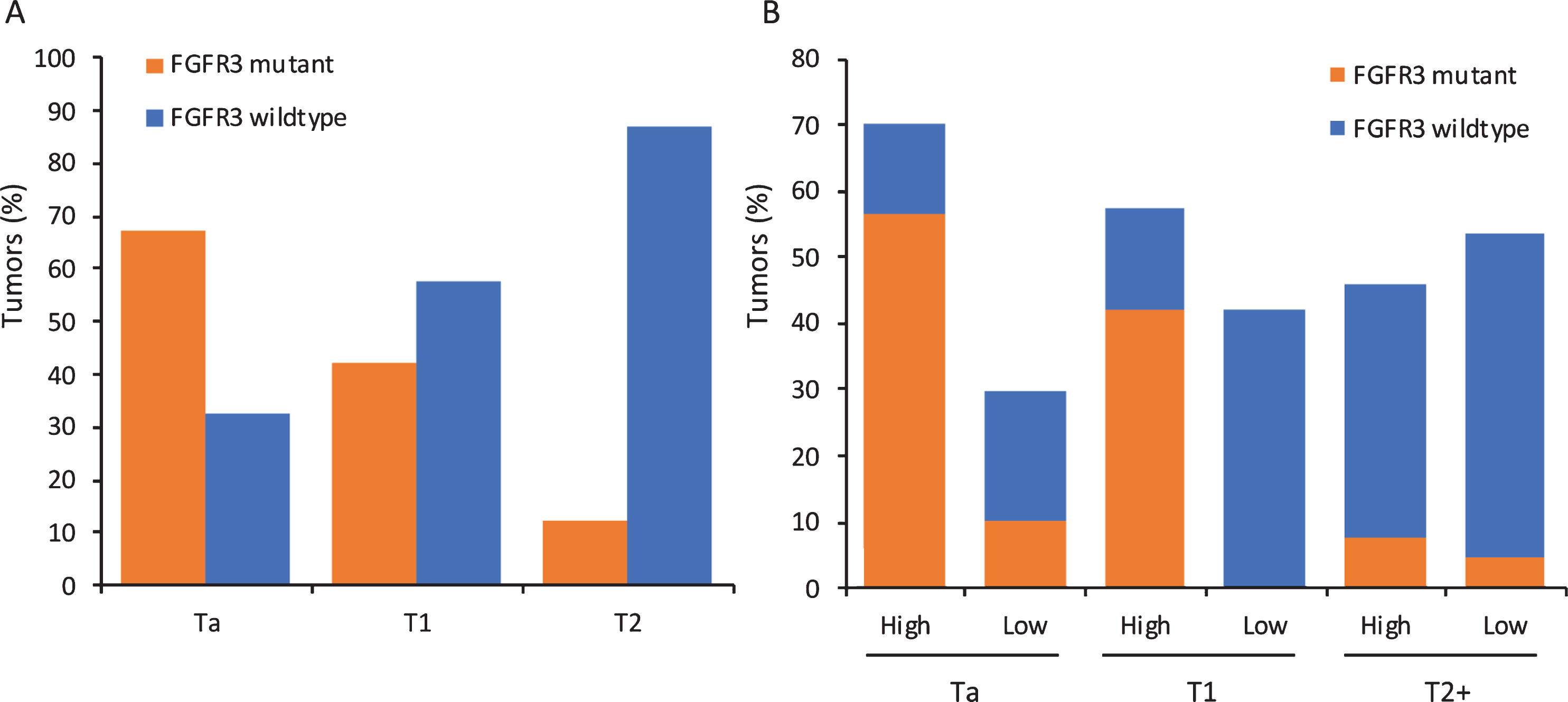
Mutation and expression of FGFR3 in relation to tumor stage in a one-year cohort of tumors from a single Institution. A. Distribution of activating point mutations according to tumor stage. B. Expression of FGFR3 protein according to tumor stage. “High” denotes expression above that found in normal urothelium and “Low” absence of expression or levels lower than in normal urothelium. Data from [17].
Several transcriptional regulators have been implicated, though their relative importance in the normal urothelium and in specific tumor settings has not yet been explored in detail. The p53 family members p63 and p73 have been shown to regulate FGFR3 [79]. p63 is expressed from two promoters to give rise to two major isoforms, Transcriptionally Active p63 (TAp63) with a transactivation domain at the N terminus and ΔNp63 which has an activation domain at the C terminus. p63 is expressed in basal and intermediate layers of the normal urothelium and is required for urothelial differentiation [80, 81]. ΔNp63 has been shown to inhibit EMT [82]. Levels of ΔNp63 are upregulated in low-grade tumors [83] and mirror FGFR3 mutation in distribution. Overall, loss of expression is a poor prognostic feature [80, 84–87], with most invasive tumors apart from a subset of aggressive tumors with squamous differentiation showing low expression [88, 89].
Analysis of cell cycle regulators in a panel of FGFR-addicted cell lines including urothelial lines, identified MYC as a critical determinant of FGFR inhibitor sensitivity and response, and FGFR inhibition in sensitive cell lines led to phosphorylation and subsequent degradation of MYC by the proteasome. The importance of MYC in sustaining the effects of FGFRs was confirmed by inhibition of MYC expression with the BET inhibitor JQ1 which recapitulated the effect of FGFR inhibition [90]. A second study of the relationship of FGFR3 and MYC in FGFR3-driven bladder tumor cells [91], identified an FGFR3/MYC positive feedback loop whereby activated FGFR3 regulates MYC mRNA and protein levels via p38 and AKT and in turn MYC directly upregulates FGFR3 by binding to upstream enhancers. Disruption of this feedback loop though FGFR, p38, AKT or MYC inhibition was demonstrated. Importantly, whilst inhibition of FGFRs and the related phosphorylation of the adaptor protein FRS2 was independent of response outcome, downregulation of MYC in response to FGFR inhibition predicted response [90]. These findings present not only potential for therapy but a biomarker for FGFR3 dependence that may be applied in early assessment of treatment response and/or development of resistance.
FGFR3 expression is also regulated at the mRNA level by micro RNAs miR-99a/100, both of which are downregulated in low-grade NMIBC compared to normal urothelium. These miRs were shown to downregulate FGFR3 expression in cultured normal urothelial cells [92] and also to regulate the urothelial differentiation factor FOXA1 [93]. Loss of the 3’ end of FGFR3 containing the target sequence for these miRs in the FGFR3-TACC3 fusion transcript, is compatible with the high-level expression of these fusion transcripts [50]. As loss of expression of miRs-99a/100 was almost ubiquitous, and more frequent than FGFR3 mutation in low-grade tumors, and the presence of the S249C mutation was not associated with differential miR expression, it was suggested that altered miR expression may lead to FGFR3 upregulation before the acquisition of mutation in NMIBC [92]. It has been reported that under hypoxic conditions FGFR3 mRNA and protein is upregulated in a HIF1α-dependent manner in bladder tumor cell lines, whilst miR-100 is downregulated [94]. As developing papillary bladder tumors have a strongly hypoxic periphery, this may indicate that hypoxia could be an early mechanism by which FGFR3 expression is upregulated via changes in miR expression prior to acquisition of mutation.
In MIBC, expression of miRs 99a/100 is lowest in the luminal-papillary mRNA expression subtype (see below), where FGFR3 mutations are most common [30]. Upregulation of wildtype FGFR3 protein in other MIBC subtypes is unlikely to be regulated in the same way. Here, levels may be more strongly regulated by MYC, possibly related to common gain of 8q in MIBC. Although the predicted relationship of MYC and FGFR3 mRNA levels could be found in FGFR3-mutant tumor samples, this did not apply to FGFR3 wildtype tumors [91]. Indeed, overall levels of MYC expression in MIBC are highest in the basal/squamous subtype and not in groups with high frequency of FGFR3 mutation [95].
It has been shown that the position of mutations in FGFR3 has differential effects on phosphorylation, retention at the plasma membrane and ubiquitylation of the receptor, with presumed effects on duration of signalling. Thus, mutations such as R248C and Y373/375C, which form dimers at the cell surface, show reduced internalisation compared to the wildtype receptor [96] and G380/382R, which is ligand-dependent, is also retained at the membrane and escapes ligand-mediated internalisation even at saturating ligand concentrations [37].
FGFR3 SIGNALLING IN NORMAL AND TUMOR-DERIVED UROTHELIA
Whilst inhibition or knockdown of FGFR3 in cells containing point mutant or fusion proteins inhibits cell proliferation e.g.[97–102], cell cycle arrest rather than apoptosis is induced [100], and escape from inhibition can occur relatively rapidly [103, 104]. Thus, there is much interest in understanding the downstream effects of FGFR3 in order to identify combinatorial or second line approaches that can enhance the effects of FGFR inhibition and/or prevent the development of resistance.
Key to understanding how FGFR3 contributes to bladder cancer development is the critical role of cellular context in determining its signalling output. This is exemplified by the differential effect of point mutant forms on chondrocyte proliferation and in epithelial cancers [42, 105–107]. Signalling downstream of FGFR3 via RAS/MAPK, PI3K-AKT, PLCγ and STATs has been reported in chondrocytes and other non-epithelial cell types and malignancies such as multiple myeloma where FGFR3 is upregulated following a translocation event involving the immunoglobin heavy chain [15, 108–110]. Many studies using chondrocytes or immortalised mesenchymal cells have focussed on the K650/652E mutant form. An early study of the tyrosine phosphorylation sites in the receptor, and their relative importance for subsequent phenotypic and signalling effects, used the 3c isoform, its K650E derivative and assays in NIH-3T3 cells [13]. In this system STAT1/3 activation was shown to be a major consequence of FGFR3 activation [14]. Whether phosphorylation of these sites has similar consequences in urothelial cells is unknown.
In TERT-NHUC, RAS/MAPK and PLCγ signalling are activated following FGF1 stimulation of ectopically expressed wildtype FGFR3-3b. This contrasts with the situation in NIH-3T3 where there is strong additional activation of the PI3K pathway and src [42]. In these normal cells, MAPK and PLCγ signalling are also activated following ectopic expression of mutant forms of FGFR3, with no changes in the levels of activated p38, JNK, SRC, STAT1 and AKT [42]. In tumor cells, the situation is less clear. It cannot be assumed that the same pathways are activated as in normal urothelial cells and it is likely that responses depend on the availability of docking and effector proteins and the overall mutational and expression landscape. In FGFR3-altered bladder tumor cell lines, the MAPK pathway is active downstream of FGFR3 as demonstrated by major inhibition of p-ERK but not p-AKT following FGFR inhibitor treatment [103, 111]. This preferential activation of the MAPK pathway in urothelial cells provides a rationale for the finding that FGFR3 and RAS gene mutations are mutually exclusive in bladder tumors [112]. However, dominance of the MAPK pathway has not been confirmed by assessment of pathway activation status in tumor tissues. Indeed, p-AKT rather than p-ERK detected by immunohistochemistry was reported to be associated with FGFR3 mutation [113]. Possibly both pathways are activated in the majority of tumors. Plasticity in signalling via these pathways is now well-documented and is exemplified in cell lines by resistance to or escape from FGFR3 inhibition in RT112 via PI3K/AKT signalling [111, 114]. Maintenance of MAPK pathway activity via activation of EGFR or ERBB2/3 signalling have also been reported as mechanisms of escape from or resistance to FGFR inhibition in bladder tumor cell lines [103, 115].
FGFR3 can interact with both p85α, the negative regulatory subunit of PI3K, via the PLCγ interaction site Y762 [116] and TGFα-activated kinase 1 (TAK1) via amino acids 589–806 [117]. The consequences of p85α interaction in urothelial cells have not been studied but in the bladder cancer cell line MGH-U3, which contains a Y375C mutation, FGFR3 signalling via TAK1 was shown to positively regulate NF-κB activity [117]. The juxtamembrane domain of FGFR3 has also been shown to interact with the N-terminal region of the kinase domain of EphA4 leading to mutual transactivation and ephrin-A1 potentiation of the FGF response of wildtype FGFRs [118]. It has also been shown that wildtype FGFRs 1–3 can form all possible stable heterodimers in the absence of ligand. Interestingly, largest effects were found for heterodimers of mutant FGFR3 (G380R and A391E) with wildtype FGFR3 [119]. These experiments were carried out using ectopically-expressed proteins in HEK293 and CHO cells and thus confirmation in urothelial models is required. It will be of particularly interest to examine the effects of common mutant forms of FGFR3 found in bladder cancer in this context.
Although S249C, Y375C and K652E mutant forms are all able to activate both MAPK and PLCγ during active proliferation of normal urothelial cells, only S249C and Y375C forms show strong activation of PLCγ at confluence. S249C with the PLCγ binding site (amino acid Y762)[120] mutated to phenylalanine shows a major reduction in this response and in continued proliferation and viability at confluence. K652E fails to activate PLCγ and is unable to elicit this phenotype [42]. This may indicate that there is positive selection for mutant forms with this capability and explain the relatively infrequent finding of K652E mutations in bladder tumors.
In urothelial and other cell types, FGFR3 fusions activate MAPK signalling [46, 121], but other aspects of downstream signalling are likely to be altered. The region of FGFR3 that is lost in these fusions (amino acids 761–806) contains Y762, which is implicated in PLCγ activation and binding of p85α [116], and part of the region implicated in interaction with TAK1 [117]. As the fusions fail to activate PLCγ, unlike point mutant forms, they are unable to elicit the overgrowth of normal urothelial cells at confluence [42, 46]. The consequences of altered interaction of the fusion proteins with p85α and TAK1 have not been examined.
ETV5, a member of the PEA subfamily of ETS transcription factors [122] is an FGF effector during embryonic development [123–125]. It is upregulated and has been implicated in regulation of several aspects of the malignant phenotype in other cancers [126–130] and was identified as a pharmacodynamic biomarker for inhibition of FGFRs 1–3 in a variety of FGFR-driven tumor cell lines [131]. In normal urothelial cells, ETV5 is upregulated downstream of MAPK activation by ligand-stimulated wildtype or point mutant forms of FGFR3, with S249C inducing the highest level of upregulation. In these cells, ETV5 had an effect on confluent cell density independent of FGFR3 expression, implicating it as an effector of this FGFR3-stimulated phenotype. In bladder tumor cell lines, it conferred a proliferative advantage and interestingly, in some MIBC-derived lines, genes previously implicated in ETV5-induced EMT in other tumour types were upregulated, implying a context-dependent effect of FGFR3/ETV5 in these cases [132].
In the FGFR3-driven tumor cell line 97-7, knockdown of ETV5 was found to modulate expression of WWTR1 (TAZ), and TAZ and its transcriptional targets were also upregulated in TERT-NHUC expressing mutant FGFR3 [132]. TAZ is a co-transcriptional regulator that together with the related protein YAP1 mediates transcription of pro-proliferative and anti-apoptotic genes. YAP/TAZ are negatively regulated through the Hippo pathway in response to cell-cell contact and cell density. Thus upregulation of TAZ as a consequence of FGFR3 signalling via ETV5 provides a rational explanation for FGFR3-induced loss of contact inhibition.
Downstream effects of signalling by ectopically-expressed wildtype FGFR3 and an FGFR3-TACC3 fusion have been examined using phosphoproteomics and network analysis in TERT-NHUC in the presence and absence of FGF1 stimulation. Pathways uniquely implicated by the fusion protein included chaperone activation pathways and stress response. Interestingly, two pathways related to TP53 expression and degradation were implicated and in both TERT-NHUC and RT112 tumor cells it was shown that TP53 is downregulated when FGFR3-TACC3 fusion and not wildtype FGFR3 is stimulated by FGF1 [133].
Further information on downstream effects of FGFR3-TACC3 has also come from examination of expression changes elicited in astrocytes by the active fusion protein or a kinase-dead version in the presence and absence of the FGFR inhibitor PD173074 [134]. This revealed that FGFR3-TACC3 activates genes involved in oxidative phosphorylation and mitochondrial biogenesis. Cells expressing the fusion showed increased oxygen consumption rate and mitochondrial inhibitors were shown to be inhibitory. PIN4 was identified as a phosphorylation substrate of FGFR3-TACC3 that triggered the biogenesis of peroxisomes and the production of intracellular reactive oxygen species (ROS) that in turn activated the transcriptional coactivator PGCIα, a regulator of mitochondrial biogenesis [134]. Dependence of tumors with FGFR3-TACC3 fusions on mitochondrial metabolism suggests that inhibitors of oxidative phosphorylation may be beneficial for this subset of patients. As this effect is driven by the kinase activity of the fusion protein, it is possible that other fusions and point mutant forms may have the same effect. Whether these effects are relevant in the urothelial context remains to be examined.
The ultimate effect of FGFR3 activation also depends on a range of feedback regulatory mechanisms [135]. These include the sprouty proteins (SPRY1-4) that are upregulated in response to FGFR signalling and bind to GRB2 and SOS1 to provide negative feedback [136] and SEF and DUSP proteins. Changes in other key proteins such as FRS2, or regulatory phosphatases [137, 138] may also have major effects on signalling. Ultimately all of these determine the context in which FGFR3 signals and may require consideration in interpreting the results of clinical trials of FGFR inhibitors.
FGFR3 ALTERATIONS AND BLADDER CANCER SUBTYPES
Several classification systems based on mRNA expression have been reported for bladder cancer. Most have focussed on MIBC [30, 139–141], one included two thirds NMIBC cases [142] and one has focussed on NMIBC [143, 144]. Broad classification of MIBC into two subtypes termed “luminal” and “basal-like”, showed that FGFR3 mutation was confined to the luminal type [139]. Further sub-classification into three [88], four [140] or more [30, 141] sub-groups has confirmed that mutation and upregulated expression of FGFR3 are largely focussed in subgroups of luminal tumors many of which have high levels of expression of markers of urothelial differentiation such as uroplakins, transcription factors (TFs) involved in urothelial differentiation (PPARG, FOXA1, GATA3, ELF3), ERBB2 and ERBB3. Recent evaluation of both mRNA and protein expression by immunohistochemistry (IHC) has allowed tumor cell phenotype to be determined independent of the overall mRNA features of the entire cellular population, and this defined 5 phenotypic classes of MIBC: urothelial-like, genomically unstable, basal/SCC-like, mesenchymal-like, and small-cell/neuroendocrine-like, with high FGFR3 expression within the urothelial-like group. This large group could be further subdivided into UroA, UroB and UroC, the latter two of which showed some features in common with basal/SCC-like and GU groups respectively [141]. An mRNA expression classifier (LundTax), that captures these IHC and RNA features was subsequently developed [95] and used to examine the large TCGA MIBC dataset [30]. The UroA subtype, which is hypothesised to represent UroA NMIBC that have progressed, has high frequency of FGFR3 mutations (44%), high expression of urothelial differentiation-associated TFs and high expression of uroplakins. UroB also has a high frequency of mutations (50%) but lacks expression of these TFs and markers of differentiation and has upregulated expression of the basal-type keratins KRT5 and KRT14 which are associated with squamous differentiation. In contrast, Uro C tumors contain few FGFR3 mutations (4%) and lack a previously-derived FGFR3 signature [142], but retain urothelial TFs and differentiation markers. This analysis reveals considerable heterogeneity in the “luminal” class of MIBC that is reflected in differential survival, with Uro A and UroC showing significantly better outcome than UroB. Given the high frequency of FGFR3 mutations in both UroA and UroB, it will be of great interest to compare the responses of these groups to FGFR inhibitors.
Three (1–3) [143] and more recently four (1, 2a, 2b and 3) transcriptional classes of NMIBC have recently been described by the UROMOL group [144]. Classes 1 and 3 contain mostly stage Ta tumors, with relatively stable genomes measured by SNP array analysis. These express high levels of FGFR3 and have a high frequency of FGFR3 mutation. Fewest mutations were detected in class 2a, which contained the largest proportion of T1 tumors and showed significantly reduced recurrence-free survival.
Overall, the luminal/urothelial-like subtypes of MIBC have relatively low levels of immune cell infiltration and stromal markers. T-cell infiltration is very low in FGFR3-mutant tumors [145] and this led to speculation that FGFR3 may be causally related to poor infiltration and that such tumors may show reduced response to immune checkpoint inhibitors. Whether FGFR3 status directly influences the associated non-infiltrated phenotype and response to checkpoint inhibitors is under debate. During induction of tumors by BBN in a genetically engineered mouse model expressing S249C in the urothelium, no difference in T-cell infiltration compared to that in wildtype mice was reported, though an early decrease in neutrophil infiltration prior to the development of tumors in S249C mice was evident [146]. Although some initial studies reported that luminal phenotype tumors responded less well to immune checkpoint inhibitors [147, 148], analysis of response in direct relationship to FGFR3 status in two large trials has recently found no statistically significant relationship. Data from this study suggests that stromal TGF-β signals, which are known to adversely influence response [149], are lower in FGFR3-altered cases, potentially balancing out the effect of low T-cell infiltration [150].
TIMING OF FGFR3 MUTATION AND MUTATIONAL LANDSCAPE OF FGFR3-ALTERED TUMORS
When does FGFR3 mutation occur during bladder cancer pathogenesis? Although there is considerable information for NMIBC, the overall picture is not completely clear and there are several caveats that preclude straightforward interpretation of current data. Common mutational events in NMIBC that could represent initiating or very early events are mutations in the TERT promoter [151, 152], FGFR3 mutation and loss of heterozygosity (LOH) of 9q [153, 154], with mutation frequency in the order TERT > FGFR3 >9q (∼80% >70% >50%). Does this imply an order of events? One major caveat is that as discussed above it is now clear that several molecular subtypes of both NMIBC and MIBC exist and mutational profiles and potentially the preferred order of molecular events are also expected to differ.
One approach to investigate the timing of events has been to examine frequency of events in cohorts of samples that are predicted to represent “stages” in disease pathogenesis. Several studies have been made of morphologically “normal” urothelium from patients with bladder cancer and potential precursor lesions. Studies of histologically normal urothelium from NMIBC-bearing bladders have found LOH of 9q or deletion detected by FISH [155–158], suggesting that 9q deletion can precede the appearance of an overt tumor. To date only a single study has examined FGFR3 in such samples [159]. This study examined normal samples from 38 patients with FGFR3-mutant bladder tumors and found no mutations. 9q was not examined, though it seems likely that a significant number of the tumors would have had 9q LOH and that, as in other studies, the surrounding “normal” urothelium in such samples would contain some 9q deletions. Studies of flat and papillary hyperplasia, now defined as “urothelial proliferation of uncertain malignant potential” [160], which may represent precursors of NMIBC, also report 9q LOH [161–163] and in a single study that examined both FGFR3 and 9q, 9q LOH was found to be the more common event [164]. These studies suggest that 9q LOH precedes FGFR3 mutation, at least in a subset of NMIBC. However, there may be different requirements and timing of events in different NMIBC subtypes. Thus, it is noteworthy that two studies aimed at defining molecular subtypes of NMIBC have described one group of low-grade tumors with FGFR3 mutation but retention of 9q and a second with both FGFR3 mutation and 9q LOH [77, 165]. This indicates that the ordering of these so-called “early” events is not uniform, and points to two distinct subsets of tumors, only one with 9q loss as an early event but both with common FGFR3 mutation.
As indicated above, downregulation of miRs 99/100 and upregulation of FGFR3 may precede acquisition of mutations [92]. Compatible with this suggestion is the finding that a germline sequence variant close to FGFR3 is associated with low-grade and low-stage bladder cancer. This variant, at least in adipose tissue, is associated with increased FGFR3 expression and importantly, the frequency of the variant is higher in FGFR3-mutant tumors [166].
Urothelial papilloma and inverted urothelial papilloma are low-grade tumors with low frequency of recurrence, often occurring in young patients, and with debated relationship to urothelial carcinoma. In many of these cases, FGFR3 mutation is absent, with a high frequency of HRAS or KRAS mutations [167–169], indicating a separate molecular profile from both PUNLMP and low-grade non-invasive urothelial carcinoma for this benign lesion.
In flat dysplasia and carcinoma in situ (CIS) that are predicted precursors of non-papillary MIBC, TP53 mutation/17p LOH and 9q LOH are common, but FGFR3 mutations are absent [170, 171], indicating that FGFR3 has no early role in the development of these tumors. TP53 mutations and/or upregulated expression are detected frequently in flat dysplasia and CIS [171–173] and appear to precede deletion of chromosome 9 [171, 174].
The distinct mutational profiles of NMIBC and MIBC and their precursor lesions provided evidence for the early concept that bladder tumor development may follow two distinct pathways, the non-invasive/papillary pathway with FGFR3 alterations as a major feature and the pathway initiated in CIS, containing many TP53 mutations [171–173]. This concept remains, and in general is supported by more profound current understanding of tumors of both types. However, it now seems clear that papillary/luminal/urothelial-like MIBC with FGFR3 alteration as a major feature represent NMIBC that have progressed. Several studies indicate that loss of the 9p21 locus CDKN2A, which encodes the RB1 and TP53 regulators p16INK4A and p14ARF, may allow progression of FGFR3-mutant NMIBC. p16INK4A is a regulator of cell cycle arrest and senescence via binding to CDK4/6, which retains the retinoblastoma protein RB1 in a hypophosphorylated and active state. It is upregulated in response to various oncogenic stimuli, inducing a state termed “oncogene-induced senescence” [175], and in the setting of an FGFR3 mutation may be partially induced and restrain tumor growth. CDKN2A homozygous deletion is found more frequently in FGFR3-mutant tumors [30, 176] and strikingly, this was found in 79% of FGFR3-mutant MIBC [176]. The role of CKDN2A/RB1 inactivation in FGFR3-mutant tumor pathogenesis has also been indicated in a genetically-engineered mouse model with inducible expression of Fgfr-S243C (mouse equivalent of human S249C) in the urothelium. Although increased levels of p-AKT and p-MAPK were induced, there was no increase in urothelial proliferation and p16, p19 and p53 protein levels increased. However, when crossed with mice with urothelial expression of SV40T, which inactivates both p53 and Rb1, high-grade papillary tumors developed [177]. Thus, loss of CDKN2A/RB1-regulated cell cycle arrest and/or the oncogene-induced senescence function appears essential in allowing tumor formation in the Fgfr3-driven mouse model and for tumor progression in human NMIBC. This has important implications for follow-up of FGFR3-mutant NMIBC with deletion of CDKN2A, as these may represent a high-risk group.
Examination of the TCGA data supports this concept. Separation of MIBC based on CDKN2A deletion status reveals that FGFR3 mutation is more common in samples with deletion, compatible with this predominantly luminal-papillary subtype tumors representing progressed NMIBC. These tumors also contain a higher frequency of mutations that are common in NMIBC such as PIK3CA and STAG2. TP53 mutation is more common in tumors with retention of CDKN2A that lack FGFR3 mutation and is skewed towards the basal-squamous and other subtypes (Fig. 3A).
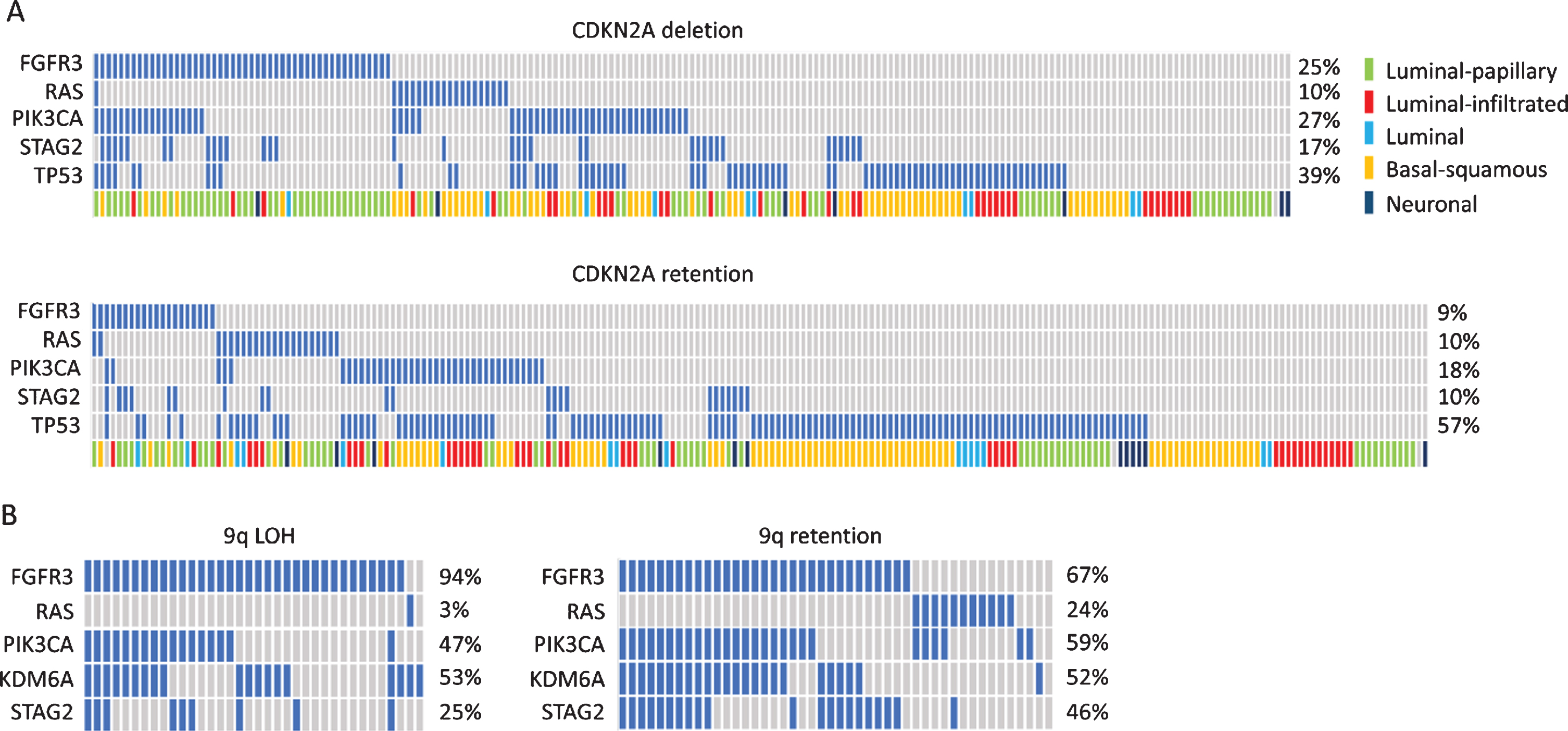
Oncoplots showing distribution of common mutations according to chromosome 9 status. A. Oncoplot for selected genes in muscle-invasive bladder tumours with and without deletion of the CDKN2A locus. Data from [30]. B. Oncoplot for selected genes in stage Ta tumors with and without 9q loss. Data from [165].
An alternative mechanism of progression for FGFR3-mutant tumors is suggested by studies that have shown the presence of mutation in a non-invasive/superficial part of a tumor and lack of mutation in a deeper or invasive component [17, 62]. It is possible that cases with discordant mutation status contain more than one tumor clone but alternatively, loss of FGFR3 mutation may have occurred, potentially alleviating oncogene-induced upregulation of p16 and related cell cycle checkpoint genes and allowing tumor progression.
Where does mutation of the TERT promoter fit within these pathogenesis pathways? Such mutations are more common than FGFR3 mutation in NMIBC and have been reported in the “normal” urothelium of tumor-bearing bladders [178] and in urine samples up to 10 years before diagnosis of bladder cancer [179]. As mutations are equally common in NMIBC and MIBC (70–80%), these mutations may precede all other molecular events identified to date and represent an essential and potentially initiating event. Telomerase is known to allow cells to overcome oncogene-induced senescence [180] and thus early mutation may allow cellular tolerance of later FGFR3 mutations.
Recent findings suggest potential complex interplay between FGFR3 and several other genes early in NMIBC pathogenesis. In thyroid cancer, ETV5 is upregulated downstream of mutant BRAF and binds to the TERT promoter [181]. Interestingly, binding is preferentially to the –124 bp(T) TERT promoter mutation that is the most common mutation found in both thyroid and bladder tumors [182]. It is also known that MYC can activate TERT [183] and recent data show that ETV5 and MYC can cooperate in de-repressing the TERT promoter [184]. Taken together, this could indicate a perfect storm between TERT, FGFR3, ETV5, MYC and TAZ early in bladder cancer pathogenesis with FGFR3 mutation playing a central role (Fig. 4).
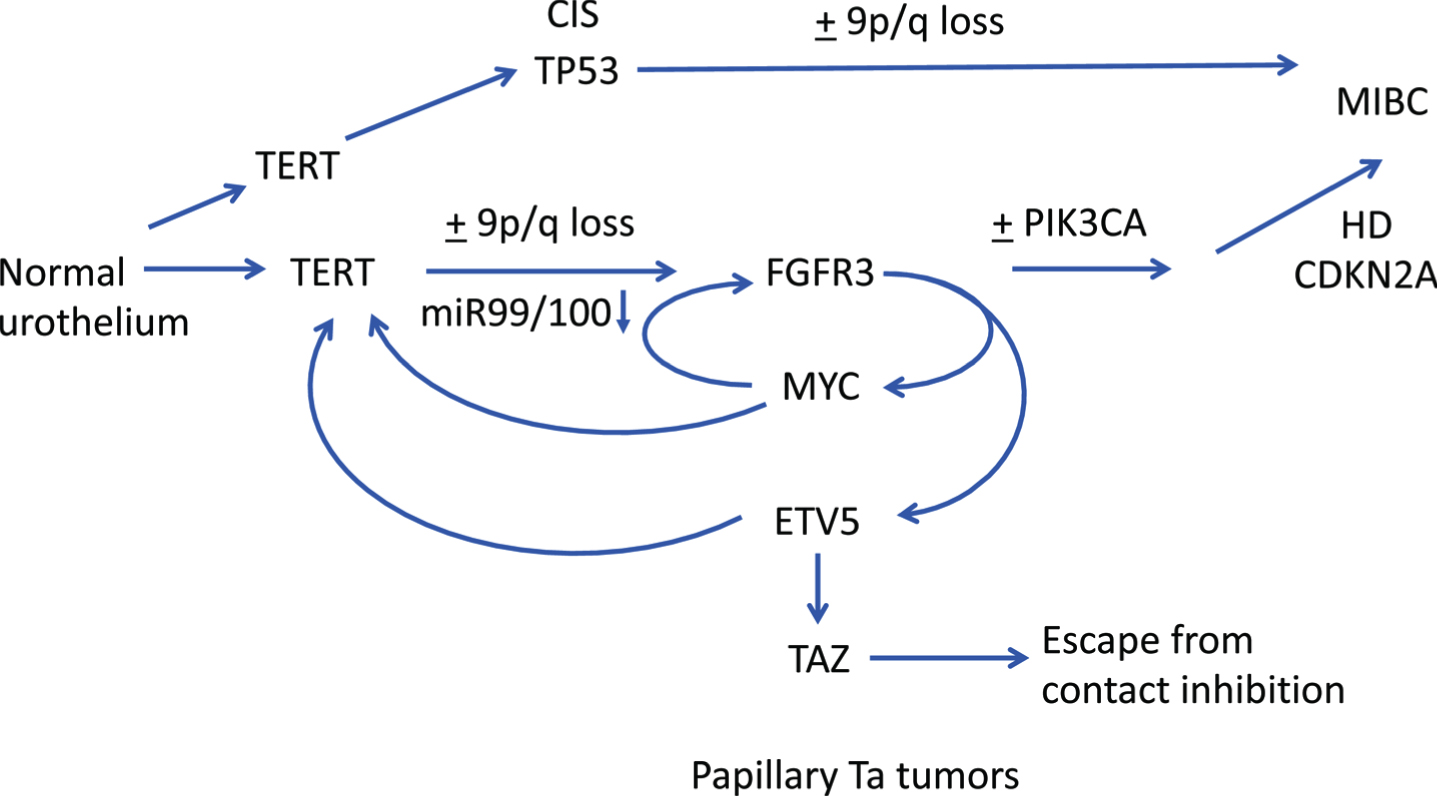
Hypothetical pathways of pathogenesis of non-invasive and invasive bladder cancer. Straight arrows show potential timing of selected events during development of NMIBC and MIBC based on data from dysplasia, CIS and urothelial hyperplasia and subtype analysis of NMIBC and MIBC. FGFR3, PIK3CA: activating point mutation. TERT: promoter point mutation. TP53: inactivating mutation. HD: homozygous deletion. Curved arrows indicate interrelated regulation of expression of FGFR3, TERT, MYC, ETV5 and TAZ.
What is the final mutational context of FGFR3-altered tumors? FGFR3 alterations are generally found in genomically stable tumors with low mutational burden. When examined across the entire bladder cancer spectrum, mutations in FGFR3 and TP53 mutation are almost mutually exclusive [27, 185–188]. Similarly, few FGFR3-altered tumors have mutations in RB1, PTEN or high-level amplification of E2F3 [30], all features of non-luminal MIBC. As indicated above, FGFR3 and RAS gene mutations are mutually exclusive [112] and FGFR3 and ERBB2 mutations are reported to be mutually exclusive in high-grade NMIBC [29].
Common features accompanying FGFR3 mutation in stage Ta tumors are mutations in PIK3CA, KDM6A and STAG2 [165]. When the mutational profiles of Ta tumors with and without loss of 9q are examined, both groups have high frequency of FGFR3 mutation (Fig. 3B), but interestingly, those with no loss of 9q have a greater number of RAS gene mutations and a higher frequency of STAG2 mutation [165]. It is assumed that, as tumors with 9q LOH usually have hemizygous deletion of the entire chromosome, this group contains those at risk of progression to MIBC via loss of the second allele of CDKN2A. How these different mutational constellations contribute to phenotype and whether this affects clinical outcome remains to be evaluated. Larger studies of NMIBC including methylation and non-coding RNA profiles may elucidate exact requirements and interrelationships more fully in the future. However, it is clear that in Ta tumors mutations in PIK3CA almost invariably occur in tumors with FGFR3 or RAS mutation [113, 189–192] (Fig. 3B). Why PIK3CA mutation in the absence of FGFR3 mutation is common in MIBC (Fig. 3A) but not found in NMIBC remains an unanswered question.
PROGNOSTIC AND PREDICTIVE VALUE OF FGFR3 ALTERATIONS
As FGFR3 point mutations are most common in tumors of low grade and stage, there is a strong relationship of these mutations with good clinical outcome. Several studies have examined the relationship of point mutation or FGFR3 protein expression to recurrence in NMIBC and most report no significant relationship [59, 193–195], though when Ta tumors were stratified by grade, an association was reported in TaG1 tumors only [24]. In univariate analysis of data from all NMIBC (Ta and T1), FGFR3 wildtype or tumors expressing low levels of FGFR3 protein have higher risk of progression, though this has not been found to be an independent prognostic factor in multivariate analyses [59, 195]. In the subset of FGFR3-mutant tumors, hemizygous or homozygous deletion of the CDKN2A locus is a predictor of progression that is independent of tumor grade and stage [176]. When FGFR3 mutation is assessed in combination with MIB-1 expression as a measure of proliferative index this “molecular grade” showed independent significance with FGFR3 mutant, MIB-1 low tumors having best outcome. When used with EORTC risk score [196], this provided improved prediction of progression [195]. When primary T1 tumors alone were evaluated, a significant association of FGFR3 mutation or expression status with disease progression has been reported in multivariate analysis in some [197, 198] but not all studies [27]. Overall, these data indicate that FGFR3 mutation identifies patients with favourable NMIBC disease, though only grade and stage remain single independent predictors of outcome.
In a very large cohort of patients with MIBC or high-risk NMIBC that were treated with radical cystectomy, mutation was related to longer disease-specific survival [63]. Interestingly, although over-expression of wildtype protein was associated with lower tumor stage and grade, it was not associated with outcome, potentially indicating a functional distinction between mutation and over-expression of wildtype FGFR3, with a driver role for mutant FGFR3, and possibly a passenger role only for upregulated expression of wildtype protein. This provides additional evidence that FGFR3 mutation rather than upregulated expression may represent the better predictive biomarker for FGFR inhibitor therapies. A response rate to the FGFR inhibitor Erdafitinib of 40% has been reported in patients whose tumors contained FGFR3 point mutations or FGFR2/3 fusions [199] leading to FDA approval for this drug in locally advanced or metastatic bladder cancer. Whilst this response rate is very encouraging, it will be important to explain the lack of response in the remaining 60% of patients. Although inhibition of FGFR3 in cell lines with mutation has been widely demonstrated, as discussed above, it is clear that several mechanisms allow escape from inhibition or development of stable resistance. Detailed discussion is beyond the scope of this review but it will be important to understand these mechanisms if FGFR inhibitors are to be applied appropriately and relevant combination therapies developed in the future. Lack of response may also indicate loss of dependence on FGFR3 during tumor progression or intratumor heterogeneity with outgrowth of a non-addicted variant in these advanced tumors. It is hypothesised that heterogeneity and cellular signalling plasticity may be much less prominent in low-grade NMIBC. Thus FGFR3 as a therapeutic target may be more relevant in these patients if localised means of targeting can be developed.
The significance of FGFR3 status in predicting response to chemotherapy has not yet been examined extensively. As previous studies have indicated that better response to cisplatin-based chemotherapy is associated with basal rather than luminal expression subtype [200, 201], worse response of FGFR3 mutant tumors might be expected as mutations and upregulated expression of FGFR3 are far more common in the luminal subtype. Findings to date suggest that this is the case. One study that evaluated FGFR3 mutation status and protein expression in 72 MIBC patients, 42 of whom were treated with adjuvant chemotherapy, reported significantly shorter overall survival and borderline significance for disease-free survival related to FGFR3 overexpression. This remained a significant independent prognostic factor in multivariate analysis. The relationship to mutation was not significant, but few samples in the chemotherapy group contained mutation [202]. A recent retrospective study that examined three groups of patients treated with cisplatin-based chemotherapy supports these findings. Lower rates of pathologic response to neoadjuvant chemotherapy and reduced recurrence-free survival were recorded in patients whose tumors contained FGFR3 point mutations or fusions. In patients included in the TCGA study of MIBC who received adjuvant chemotherapy reduced RFS was also found in FGFR3 altered tumors. In a third group of metastatic patients treated with first line platinum-based chemotherapy FGFR3 alteration was associated with a different pattern of metastatic spread including higher rates of pulmonary metastases and with lower response rate. Patients with FGFR3-altered tumors that did not receive chemotherapy had superior clinical outcome [203]. Overall this suggests that despite the relationship of FGFR3 status to better outcome in MIBC, chemotherapy may be detrimental to this group and that other treatment options including FGFR inhibitor therapy may be more suitable. Interestingly, an in vitro study has also reported decreased sensitivity to cisplatin of a bladder tumor cell line (97-7) containing an S249C mutation compared to wildtype cell lines. In this case, a high level of p-AKT in the mutant line was decreased following FGFR3 inhibition and this was associated with increased cisplatin sensitivity [204]. Larger studies of patients treated with cisplatin-based neoadjuvant chemotherapy in combination with molecular profiling may allow the basis for these effects to be elucidated.
CONCLUSIONS
Whilst a great deal has been learnt about FGFR3 and its role in the pathogenesis of NMIBC and MIBC, much remains to be clarified if it is to be optimally targeted in these settings. Current data suggests that it is an early event in NMIBC, though we lack understanding of exactly how it contributes during the initial development of urothelial hyperplasia and the elicitation of a branching vasculature and papillary tumor architecture. More detailed analyses of these early stages and the development of mechanisms for localised FGFR3 targeting or targeting of the FGFR3-related phenotype have potential for major impact on the clinical management of the very large NMIBC population. In vitro and in vivo models may make a major contribution to understanding these early processes and more complex models may improve understanding of any influences of FGFR3 signalling on tumor infiltration. Unlike other epithelial tissues, the ability to culture normal urothelial cells with relative ease is a major advantage and this should allow the hypothetical interactions of TERT, FGFR3 and MYC in NMIBC pathogenesis hypothesised here to be tested. In MIBC, the excitement related to responses to FGFR inhibitors is tempered by lack of good predictive biomarkers. More detailed molecular profiling that takes into account some of the broader mechanisms of FGFR3 regulation and cross-talk should facilitate improved biomarker-driven treatment selection.
Footnotes
ACKNOWLEDGMENTS
I am extremely grateful to past members of my group, Darren Tomlinson, Erica di Martino and Sarah Williams for their practical and intellectual contributions over many years that have shaped my thinking about this most interesting protein, and to Julie Burns for her critical reading of this manuscript.
FUNDING
Funding of our past work on FGFR3 has been from Cancer Research UK (C6228/A5433; C6228/A12512) and Yorkshire Cancer Research (L376PA).
ETHICAL CONSIDERATIONS
As a review, this work is exempt from any requirement for Institutional Review Board approval.
CONFLICT OF INTEREST
Consulting/Advisory Role-Janssen Oncology, Bioclin Therapeutics.
