Abstract
BACKGROUND:
Metastatic breast cancer (MBC) is incurable. Systemic therapy is the standard treatment; however, an optimal sequence of chemotherapy has not been established.
OBJECTIVE:
Evaluating effectiveness and safety of eribulin in MBC treatment and comparing the results obtained with published literature.
METHODS:
Observational, descriptive and retrospective study of patients with MBC treated with eribulin from 01/12/2015 to 30/10/2021. Effectiveness was analysed using Kaplan–Meier-survival-curves, for the overall number of patients treated and stratified by treatment line. Safety was measured according to adverse events (AE) based on CTCAE v5.0. Data analysis was performed using R v4.0.1.
RESULTS:
They were included in this study 53 women who received eribulin (median age 58 years). Comparison of median survival from this study versus published data were: progression-free-survival (PFS) 3 (IC95%: 3–4) versus 3.7 months and overall-survival (OS) 8 (IC95%: 3–4) versus 13.2 months for the overall number of patients. For the 1–3 line treatment group, PFS was 6 (IC95%: 3-NA) and OS was 15 (IC95%: 6-NA). There were 322 AEs, the most frequent being blood disorders 16% (52), general disorders 12% (38), and gastrointestinal disorders 12% (38).
CONCLUSIONS:
The median PFS was similar to that reported previously, with lower OS. There was a tendency to achieve better results when eribulin was used earlier. Eribulin is a less well-tolerated drug than published literature.
Introduction
According to the World Health Organization (OMS), breast cancer (BC) is the accelerated and uncontrolled proliferation of cells of the glandular epithelium that have greatly increased their reproductive capacity. BC cells can spread through the blood and lymphatic vessels and reach other parts of the body, adhere to tissues and form metastases [1].
BC is the most common cancer in women worldwide. It is the third most common cancer diagnosed per year and the cancer with the highest 5-year prevalence. In Spain, 34,088 new cases were diagnosed in 2020, with ages ranging from 35 to 80 years, with the highest incidence between 45 and 65 years of age. In Spain, 5 years survival is 83%, although this drops to 20% in advanced stages [2,3].
Metastatic breast cancer MBC can occur in both women and men, however, more than 99% of cases happen in women [1,3]. The classification of breast tumours continues to evolve, with the integration of new knowledge from research rapidly being translated into clinical practice. Invasive breast cancers are still organised into chapters by their morphological subtypes, which remain clinically relevant, however, the majority of cases are of no special type [4].
The terminology for the most common type of breast cancer has changed from invasive ductal carcinoma, not otherwise specified (NOS) to invasive carcinoma of no special type (NST). This group of breast cancers comprises all tumors without the specific differentiating features that characterize the other categories of breast cancers. Standard prognostic indicators, such as tumour size, lymph node status, and Nottingham grade, continue to be highly relevant. As well as classic lobular carcinoma in situ (LCIS), the pleomorphic and florid subtypes are now recognised [4,5].
The overview acknowledges the treatment-relevant subtypes of invasive carcinoma [based on estrogen receptor (ER) and human epidermal growth factor receptor 2 (HER2) status], and new data are added to support the differences in pathogenesis, treatment response and prognosis of these clinically relevant groupings. In newer modality of management, the target therapy forms the basis of effective treatment. BC which are negative for ER, progesterone receptor, and HER2 are considered as triple-negative breast cancer (TNBC). These represent about 15%–20% of newly diagnosed cases [4,6].
MBC is an incurable disease, but survival has improved in the past years thanks to the development of new therapeutic strategies. Systemic therapy is the standard of care in advanced disease, but can be complemented with locoregional treatments. Re-exposure to drugs previously used in early BC is a widely used option, provided that the disease has had a disease-free interval of more than 12 months [1,6]. The optimal sequence of chemotherapy in MBC has not been established, taxanes and anthracyclines should be considered, especially in patients who have not received them previously or those who have had sustained responses over time [7,8].
The most commonly used pharmacotherapeutic regimens for systemic treatment in recurrent or stage IV breast cancer according to the NCCN (The National Comprehensive Cancer Network) treatment guidelines are [9]:
Anthracyclines: doxorubicin or liposomal doxorubicin Taxanes: paclitaxel Antimetabolites: capecitabine Microtubule inhibitors: eribulin Targeted therapy.
Eribulin is indicated for the treatment of adult patients with locally advanced or MBC who have progressed after at least one chemotherapeutic regimen for advanced disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting unless patients were not suitable for these treatments. The efficacy of eribulin in BC is primarily supported by two randomized phase 3 comparative studies, Studies 305 and 301 [10].
Study 305 met its primary endpoint with an overall survival (OS) result that was statistically significantly better in the eribulin group compared to treatment of physician’s choice (TPC) at 55% of events. The results of the main analysis in 301 Study showed no statistically significant differences in terms of OS for eribulin and capecitabine and neither in terms of PFS as assessed by the study investigators [10].
The most commonly reported adverse reactions related to eribulin are bone marrow suppression manifested as neutropenia, leucopenia, anaemia, thrombocytopenia with associated infections. New onset or worsening of pre-existing peripheral neuropathy has also been reported. Gastrointestinal toxicities, manifested as anorexia, nausea, vomiting, diarrhea, constipation, and stomatitis are among reported undesirable effects. Other undesirable effects include fatigue, alopecia, increased liver enzymes, sepsis and musculoskeletal pain syndrome [10].
The objectives of the work are
To evaluate the effectiveness of eribulin in the treatment of locally advanced MBC over a six-year study period (12/2015–10/2021) at a 3rd grade hospital. To assess the toxicity of eribulin in routine clinical practice. Compare the results obtained in this work (Real World Data) with the results provided in the literature and in published clinical trials (CCTs) [2,7,10–15].
Due to the long period of time since the marketing of eribulin (16) without further review, the results obtained will allow for improved clinical decisions related to the optimisation of its pharmacotherapy in patients with MBC.
Material and methods
We conducted an observational, descriptive and retrospective study of the use of eribulin in a 3rd grade hospital for the treatment of locally advanced MBC. In this hospital, eribulin is used according to the autonomic recommendations and also following the summary of product characteristics (SmPC). If liver failure (LF) due to metastases is present, the dose should be adjusted according to the indications set out by SmPC and in case of several LF or cirrhosis, use should be closely monitored [10,17].
Patients diagnosed with MBC with progression of the disease after receiving an anthracycline, a taxane and capecitabine in the adjuvant or metastatic setting, unless they were not suitable for the patient [17]. Patients with MBC who received at least one cycle of eribulin during the period from December 2015 to October 2021 were included in the study. The cut-off date for data analysis was 30-10-2021 (study period 6 years).
The presence of brain metastases, unless they were treated and stable, was considered an
The electronic medical record software (Cerner Millennium®) and the oncohaematological patient management software (Farmis-Oncofarm®) were used to obtain the necessary data. Demographic variables (sex, age and date of death), biometric variables (height and weight), disease-related variables (diagnosis, morphological subtypes, presence of mutations in hormone receptor expression and HER2) and treatment safety variables (reason for discontinuation, adverse events (AE) and degree of each one) were obtained from the electronic medical records.
AEs developed following eribulin administration were classified according to the NCI (National Cancer Institute) Common Terminology Criteria for Adverse Events (CTCAE) criteria (18). AE grade 3 and 4 were considered as severe. AEs observed in CCTs and/or reported post-marketing of eribulin are described in its SmPC and grouped according to the classification system of organs or system affected and by their frequency [10].
The study collected AEs related to infections, disorders of the blood and lymphatic system, metabolism and nutrition, hepatobiliary, psychiatric, nervous system, respiratory and cardiovascular system, general disorders, gastrointestinal, skin and subcutaneous tissue, musculoskeletal, ocular, ear and labyrinth, renal and urinary disorders.
Treatment-related variables were obtained from the oncohaematology patient management programme: number of patients treated with eribulin, dosing regimen, dose per cycle and cumulative dose, dose reductions, number of cycles, duration of treatment (start and end date) and previous lines of treatment.
Treatment effectiveness was assessed in terms of OS and progression-free survival (PFS), calculated using Kaplan–Meier non-parametric survival analysis. For the effectiveness and safety of eribulin treatment, continuous variables were presented as median and interquartile ranges (IR) and categorical variables as number of observations and frequency (percentage). Data analysis of the collected variables was performed using the statistical programme R version 4.0.1. Effectiveness and safety results were compared with the results obtained in pivotal CCTS and with the published literature [2,7,10–16].
The sources of information searched were: Pubmed, UpToDate, ClinicalKey, Cochrane, Centre for Information on Medicinal Products (CIMA), European Medicines Agency (EMA), NCI, American Society of Clinical Oncology (ASCO), NCCN guidelines, European Society for Medical Oncology (ESMO) and Spanish Society of Medical Oncology (SEOM). The keywords used in the literature search were: eribulin, Halaven®, BC, MBC, EMBRACE study. Mendeley was used as bibliographic reference management software [1–3,7,8,10,15,16].
Results
The study included 53 patients with MBC who were treated with eribulin during the study period, 100% female, with a median age of 58 (37–75) years. The morphological diagnosis was: infiltrating ductal carcinoma 77% (41), infiltrating lobular carcinoma 11% (6), one (2%) atypical medullary carcinoma, one (2%) mixed phenotype infiltrating carcinoma and 8% (4) breast carcinoma not pathologically confirmed.
The presence of hormone receptor expression mutations and HER2 status are listed below. The Classification of patients included in the study according to hormone receptor and HER2 expression are:
RH + HER2 – → 35 (66%) TNBC → 9 (17%) RE + RP – HER2 – → 7 (13%) RE + RP – HER2 + → 2 (4%) RH – HER2 + → 0 (0%).
The median starting dose of eribulin received by the patients included in the study was 1.01 mg/m2 (0.87–1.31) expressed per body surface area, and being the median starting dose 1.70 (1.23–2.6) mg. Dose reductions were required in 28.30% (15) of the patients, 15.09% (8) dose increases and one patient who initially required a dose increase had to have her dose reduced due to toxicity. Dose modifications were not required in 54.72% (29) of the patients. The median cumulative dose for the total number of cycles received was 12.5 (1.45–143.12) mg.
The median number of cycles received was 4 (1–30) cycles, 15% (8) of patients discontinued treatment after receiving only one cycle. The median duration of treatment was 4 (0–24) months, three patients (5.66%) exceeded one year of treatment and only one exceeded two years. Eribulin was used in the majority of cases in the sixth line of treatment or later (64.15%), as shown in Fig. 1.
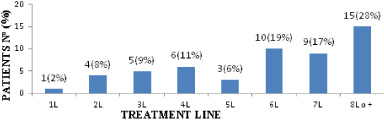
Number and percentage of patients who were treated with eribulin according to the indicated line of treatment.
At the end of the study, 77.36% (41) of the patients treated with eribulin had progressed to worsening disease or new metastases and 18.87% (10) had died. At the end of the study, only two patients (3.77%) were still on eribulin treatment, as shown in Fig 2. The reason for discontinuation of treatment was due to disease progression in 66.04% (35) of patients, 18.89% (10) due to death and 11.32% (6) due to toxicity, as shown in Fig 3.
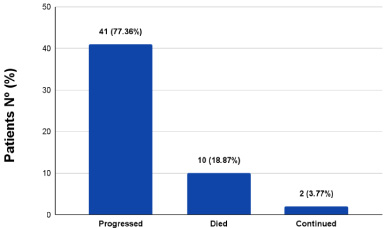
Number and percentage of patients who progressed, died and continued treatment with eribulin at the end of the study.
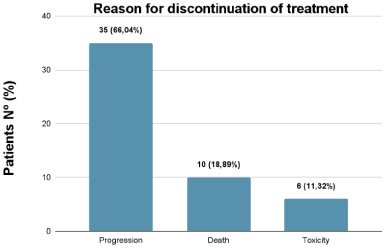
Reason for discontinuation of eribulin treatment, number and percentage of patients who progressed, died or suffered toxicity.
The median PFS was 3 months (95%CI: 3–4) and median OS was 8 months (95%CI: 6–11). Survival curves are shown in Fig. 4.
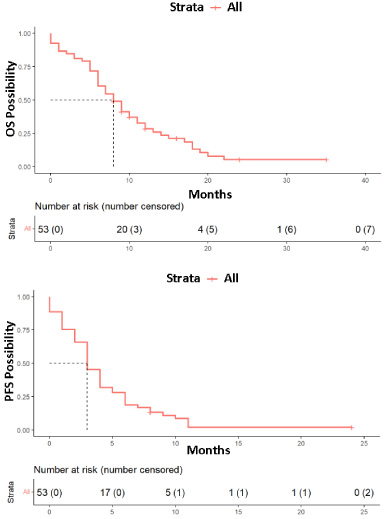
PFS and OS curve of the disease obtained from data provided by eribulin-treated patients included in the study. Red: All patients.
Statistically significant differences in PFS were obtained when the data were analysed according to the line of treatment in which eribulin was used. Thus, when stratified into groups according to the number of treatment lines in which eribulin was used, the median PFS was 6 months (95%CI: 3-NA) when eribulin was used in the first to third line of treatment, 3 months (95%CI: 3–6) when it was used in the fourth to sixth line of treatment and 3 months (95%CI: 1–4) when it was used in the seventh or later lines. PFS curves stratified according to the treatment line occupying the eribulin treatment are shown in Fig. 5.
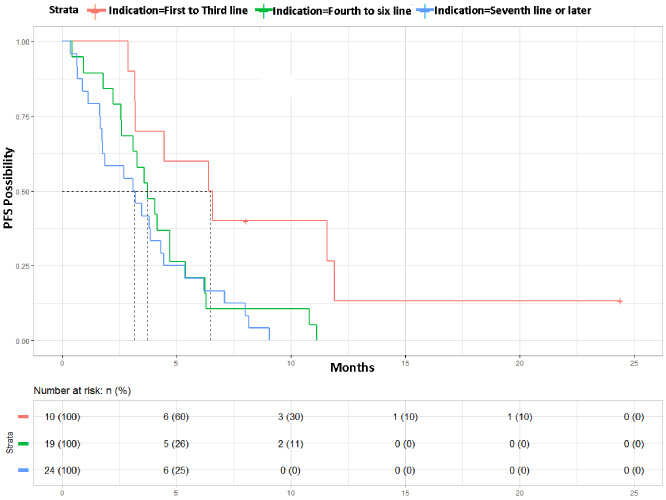
PFS curve of the disease obtained from the data provided by the patients included in the study stratified according to the line of treatment occupied by the use of eribulin. Red: First to third line; Green: Fourth to sixth line; Blue: Seventh line or later (p = 0.0098).
When the analysis was performed using OS, differences were also obtained, but were not statistically significant. Thus, median OS was 15 months (95% CI: 6-NA) when eribulin was used from first to third line of treatment, 8 months (95% CI: 6–13) when it was used between the fourth and sixth line of treatment and 8 months (95% CI: 6–12) when eribulin was used in treatment lines equal to or following the seventh. OS curves stratified according to the treatment line occupying the eribulin treatment are shown in Fig. 6.
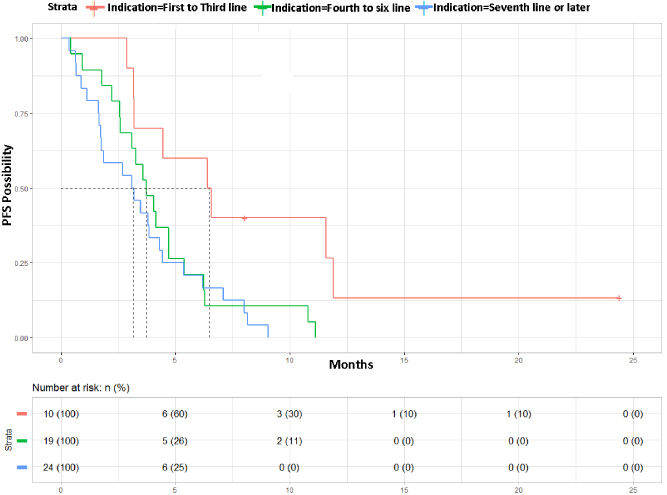
OS curve of the disease obtained from the data provided by the patients included in the study stratified according to the line of treatment occupied by the use of eribulin. Red: First to third line; Green: Fourth to sixth line; Blue: Seventh line or later (p = 0.068).
OS and PFS curves were constructed by stratifying patients into two age groups, taking 58 years as the cut-off point. The analysis showed that there were no significant differences in OS and PFS between the two groups (younger and older than 58 years) (Fig. 7).
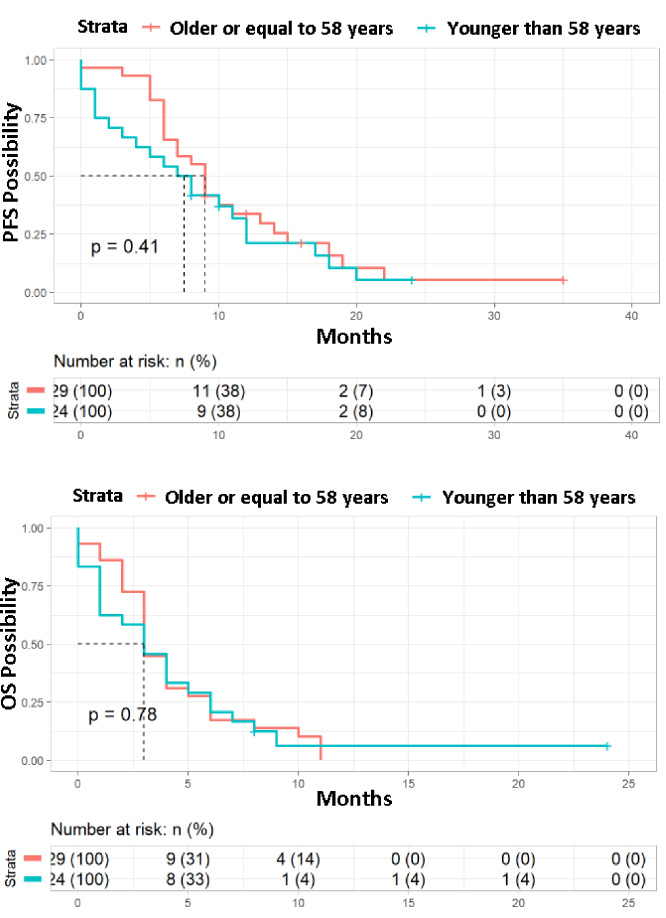
PFS and OS curves of the disease obtained from the data provided by the patients included in the study stratified into age groups (<58≥). Red: Older or equal to 58 years; Blue: Younger than 58 years.
The percentage of patients who developed AEs, the total number of AEs developed, the mean number of AEs per patient and their severity are described below:
Patients treated with ERIBULIN: Patients who developed some degree of AE: Total number of AEs: Average AE/patient: CTCAE Degrees:
Grade 1: 43% (139/322) Grade 2: 33% (107/322) Grade 3: 17% (54/322) Grade 4: 7% (22/322) Patients with AE ≥ grade 3: 60.37% (32/53)
In Table 1 are described the AEs observed in the patients included in the study classified by organ or system affected. Table 1 lists the most frequent alterations in each of the groupings and describes the toxicity grades according to CTCAE v5.0 (only toxicity grades ≥2 are listed, the rest being grade 1)
Number and percentage of patients included in the study who manifested disorders in the indicated organs and systems after treatment with eribulin, and the AE classified according to the degree of toxicity (≥2)
∗AE according to organ or system affected grade 2–4 according to CTCAE v5.0. and percentage of total.
Table 2 shows the comparison of the grade 3 and 4 AEs observed in the patients included in the study with those described in the eribulin SmPC [10]. The remaining AEs not listed in the table are grade 1 and 2, as they are not considered severe. Only the data published in the literature with the highest frequency are available.
Comparison of the percentage of patients included in the study who reported AE after eribulin treatment with those described in the TF
∗RA not described in the data sheet for eribulin.
Patients with MBC use a wide variety of therapeutic lines including HER2-targeted therapies, hormone therapy and chemotherapy. There is a disparity in treatment response rates, with only 50% of patients with MBC responding to systemic chemotherapy, compared to 90% of patients with localised tumours. Women with HR negative or HR positive tumours refractory to hormonal treatment are candidates for systemic chemotherapy regardless of the site of metastasis [14,21].
According to the NCCN treatment guidelines for MBC, sequential monotherapy is recommended over combination therapy as it is associated with lower toxicity and longer survival. The recommended first-line treatments are anthracyclines and taxanes. Many of the patients treated in the first line of treatment with these drugs become resistant to them and other available drugs have to be used [1,6]. Eribulin is one of the drugs used in later lines of therapy for the treatment of MBC, its efficacy and safety was evaluated in two randomised phase III comparative CCTs [12,13,16].
The pivotal
The second study consulted was a phase III study (
The median PFS for the overall number of patients included in our study (n = 53) was similar to the median PFS of
In our study, more than 60% of patients received eribulin in the sixth or subsequent line of treatment, one of the inclusion criteria being the maximum number of previous lines of treatment received, three for study 301 and five for study 305. This is confirmed when assessing PFS and OS by subgroups according to the line of treatment in which they received eribulin. Statistically significant differences were obtained for the first to third line of treatment subgroup, with median PFS and OS being higher for these patients. Patients who received eribulin in the fourth or subsequent line of treatment had a similar PFS and OS to that obtained for all patients included in the study.
Studies 305 and 301 assessed OS of patients based on HER2 overexpression, in our study it was not possible to perform such stratification because not enough patients diagnosed with HER2 receptor-positive MBC were available. This is because the emergence of new HER2-targeted therapies, such as the monoclonal antibodies trastuzumab or pertuzumab, against the HER2 receptor or trastuzumab-emtansine, a conjugate of trastuzumab with a cytotoxic agent (emtansine), means that less specific treatments, such as eribulin, are reserved for later lines of treatment or may not even be used.
For this reason, the largest group of patients included in the study were patients with HR-positive and HER2-negative carcinomas, accounting for more than 60% of patients, followed by the TNBC group. These groups of patients have the least therapeutic resources available to treat the disease. This is why the trend at this 3rd grade hospital is to reserve eribulin for later lines of treatment, prioritising the use of more specific targeted therapies with less toxicities early on, especially in patient groups with specific therapeutic targets.
Ductal in situ carcinoma, the most prevalent in the population, may progress in more advanced stages to invasive carcinoma, requiring systemic treatments, such as those evaluated in this study [13]. Consistent with the epidemiology described, about 80% of the patients included in the study were diagnosed with infiltrating ductal carcinoma.
The recommended dosage of eribulin in the data sheet is 1.23 mg/m2 administered IV over 2–5 minutes on days 1 and 8 of each 21-day treatment cycle [10]. The median starting dose of eribulin, expressed per body surface area, received by the patients included in the study was lower than the dose described in SmPC. In the study, as mentioned above, the patients were more polytreated and had more sequelae from previous treatment, which is the most likely reason for starting treatment with lower doses of eribulin.
This also meant that about 30% of patients had to reduce their dosage after initiation of treatment due to the toxicities developed. However, more than 50% of the patients did not require subsequent dose modifications. This is also observed in the median number of cycles received, duration of treatment, dose per cycle and cumulative dose. In all these variables there is a large difference in the IR, due to discrepancies between patients with a single cycle and patients with longer treatment times.
The reason for discontinuation of treatment was mostly due to disease progression, less than 20% due to death and in a few cases due to toxicity. Although a few patients had to discontinue treatment due to toxicity, all of them suffered some degree of AE, with a large number of AEs described (n = 322).
The most frequently reported AE in the literature is bone marrow suppression [10,12,13,16], mostly present as neutropenia, leukopenia, anaemia and thrombocytopenia, associated with infections. Peripheral neuropathy, gastrointestinal toxicities and other AEs including fatigue, alopecia, increased liver enzymes, sepsis and musculoskeletal pain syndrome have also been reported [10]. Some AEs are very rare, but can result in a remarkable rate of residual or fatal symptoms, such as febrile neutropenia or pleural effusion.
The AEs described with the highest incidence in the technical data sheet were observed to a greater extent in our study, with blood and lymphatic system disorders being the most numerous, followed by gastrointestinal disorders and general disorders, in agreement with the published literature. When the severity of AEs was classified according to the CTCAE v5 scale, it was found that more than 40% of AEs were grade one, considered mild, and the most severe AEs, grade 3 and 4, accounted for around 20% of the AEs recorded in the patients’ medical records. However, despite the lower number of severe AEs, 60% of patients had some AE of this grade.
When analysing the most severe AEs, blood disorders continue to be the most numerous, mainly due to the high incidence of neutropenia and anaemia. The AEs in decreasing order of frequency both overall and grade 3 and 4 were: asthenia, neutropenia, anaemia, dyspnoea, increased liver enzymes, upper respiratory tract infections, bone pain and nausea. Comparing the frequency of AEs described in the literature with those reported in our study, all of them show a higher proportion in both overall incidence and grade 3 and 4 frequency, except for neutropenia and gastrointestinal disorders.
However, the AEs described in our study population are not comparable to those of the CCTs consulted due to the use of eribulin in later lines and the use of lower doses in patients with more polytreatment and greater sequelae of previous therapies.
Six of the patients suffered pleural effusion during the treatment with eribulin, a complication not described among the AEs in the SmPC and of considerable severity. In all cases, invasive intervention was required to drain the pleural fluid, which can lead to many complications and in some cases to a delay in the eribulin cycle administration. Other grade 3 and 4 AE not described in the literature that occurred in the study were ascites, mucositis and seizures. All of these required some form of intervention or treatment for their management.
Conclusions
The median overall PFS of the patients included in the study was similar to that published in the CCTs; however, the median OS obtained in our study was lower.
PFS and OS were slightly higher for the subgroup of patients who received eribulin in the first to third line of treatment. Taking into account that more than half of the patients included in the study received eribulin in subsequent lines of treatment to those described in the CCTs consulted, it is possible to conclude that the PFS and OS results obtained in our study are consistent with those published in the literature.
The results provided in our study seem to indicate that there is a tendency to achieve better PFS and OS outcomes when eribulin treatment selection is made at earlier lines of treatment, as long as they cannot be treated with targeted therapies against their disease.
The results on safety in our study show that eribulin is worse tolerated than described in the CCTs consulted. Approximately 30% of the patients were required to reduce their dosage as a result of toxicities developing after the commence of treatment; however, more than half of the patients did not need to make any further adjustments to their dosage.
In addition, 11% of the patients experienced pleural effusion during treatment with eribulin, a complication not reported among the AEs in the SmPC and had significant severity. Other unreported grade 3 and 4 AEs that were observed during the study included ascites, mucositis, and seizures. All of these require some form of intervention or treatment for their management. The AEs described in our trial that are consistent with the CCTs, do not match in the frequency of occurrence, this comparison being conditional on the use of eribulin under different conditions to those published in the literature.
Footnotes
Ethics approval
The Research Ethics Committee of the Principality of Asturias, has reviewed the research project CEImPA Code 2022.038 with title “Effectiveness and safety of eribulin in metastatic breast cancer”. The Committee has accepted that the research meets the ethical conditions necessary for it to be carried out and has therefore given its authorisation.
Conflicts of interest/Competing interests
The authors whose names are listed in the manuscript have no affiliations with or involvement in any organization or entity with any financial interest, or non-financial interest in the subject matter or materials discussed in this manuscript.
Data availability
The datasets generated during the current study are not publicly available but are available from the corresponding author on reasonable request.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
All authors meet the ICMJE authorship criteria. Clara Luz Fernández Laguna was the chief investigator and contributed in every process. Ivan Maray Mateos was part of the idea development, made a literature review, contributed with data extraction and analysis, scientific writing and final approval. Lola Macia Rivas and Cristina Álvarez-Asteinza helped with literature review, data analysis, and scientific writing. Sergio Fernández-Lastras and Veronica Velasco Durántez reviewed the manuscript. Finally, Ana Lozano Blázquez and Mónica Carbajales Álvarez reviewed the manuscript and finally approved it.
