Abstract
BACKGROUND:
Fibroadenomas are the most common benign breast lesions in women. They present as a unilateral mass and can rapidly enlarge in size through hormonal changes. Fibroadenomas could be classified as small or giant, and as simple or complex. They are classified as ‘giant’ when the size exceeds 5 cm and/or weight 500 gram; and as ‘complex’ if one of the following characteristics is present: cysts with a size >3 mm, epithelial calcifications, sclerosing adenosis and papillary apocrine metaplasia. Giant fibroadenomas can cause compression of surrounding breast tissue or breast asymmetry, requiring surgical excision in order to preserve a normal breast shape.
CASE:
A 26-year-old pregnant woman was referred with a palpable mass of her right breast. The mass rapidly increased in size to a diameter of 13 cm during the second trimester of her pregnancy. A tru-cut biopsy confirmed a fibroadenoma. The rapid growth and compression of normal breast tissues indicated a lumpectomy during her pregnancy. The mass was easily excised without any consequences for the pregnancy. Pathological examination showed a complex giant fibroadenoma.
CONCLUSION:
A unique case of a pregnant woman with rapid progression of a fibroadenoma that met the criteria of a complex and giant fibroadenoma, was presented. This case emphasizes the importance of timely surgical intervention, even during pregnancy, to prevent permanent breast tissue damage.
Introduction
Fibroadenomas are the most common benign breast lesions in women, with a peak incidence in the second and third decade of life [1–3]. In 0.5–2% of the cases, the mass is classified as giant fibroadenoma with a size greater than 5 cm and/or a weight of more than 500 gram [4–6]. Fibroadenomas usually present as a unilateral, smooth, solid and mobile mass in the upper outer quadrant of the breast [3,7]. The lesion can be asymptomatic, but can also induce complaints as pain and rapid enlargement of the breast, leading to asymmetry [3,7]. The growth of fibroadenomas is stimulated by hormonal changes in estrogen, progesterone and prolactin [2,5,8–10]. Therefore, fibroadenomas enlarge during pregnancy and lactation, and reduce in size during the menopause [2,3,5,7–9]. Compression of surrounding breast tissue may occur due to accelerated growth, leading to possible inflammation, infarction, hemorrhage and ulceration [1,3,7,11,12]. Furthermore, rapid growth and these symptoms of compression challenge the discrimination between a fibroadenoma and phyllodes tumor, malignancy or an inflammatory lesion [1]. Indications for surgical excision of presumed benign mammary lesions are rapid growth, compression of surrounding breast tissue, breast asymmetry and suspected malignant lesions [3,6,7,12].
In this case report, we present a patient with a rapidly growing breast lesion in the second trimester of her pregnancy. This lesion met the criteria of a giant and a complex fibroadenoma which makes this case unique.
Case description
A 26-year-old woman, 15 weeks pregnant, presented at our outpatient department with a unilateral mass of the right breast. She became aware of the lump two weeks before. Ultrasonic examination of the right breast showed a homogenic solid hypoechoic mass of 6.3 × 5.2 × 2.8 cm with vascularization in the upper region of her right breast. Differential diagnosis based on the ultrasound images included a fibroadenoma or phyllodes tumor. The mass was determined as BIRADS 3. No abnormalities were seen at the left breast. A tru-cut biopsy of the lesion showed low cellularity in the stromal component, with ducts in an organoid pattern. The ducts were covered with two layers of epithelial cells and the stromal cells showed no atypia; matching with fibroadenoma (Fig. 1). In accordance with the patient, we decided to wait and discuss treatment after her pregnancy if needed.
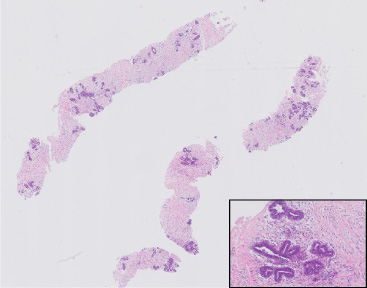
The first biopsy shows an organoid configuration of a bland stromal and a benign epithelial component (H&E, 10×. H&E 200×).
After 12 weeks, she returned to our clinic with complaints of pain and significant growth of the mass. On physical examination, a swollen breast was noted and the tumor was no longer distinguishable from surrounding tissues. The growth caused significant breast asymmetry (Fig. 2). A repeat ultrasound showed a markedly increase of the size of the lesion of 13 cm, with compression of the surrounding breast tissue and skin. A biopsy was repeated to exclude malignancy; this showed a fibroadenoma again, this time with lactational changes in the epithelial component (Fig. 3). Due to the complaints and critical deformity of the breast, the patient was scheduled for an urgent lumpectomy under general anesthesia.
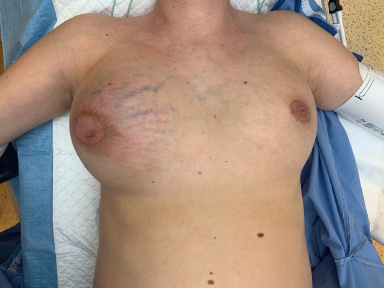
The swollen breast resulting in breast asymmetry.
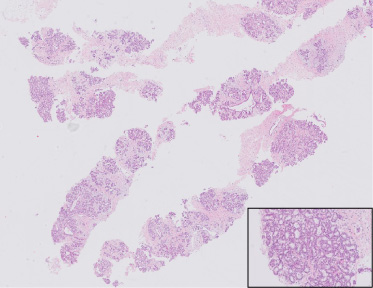
In the second biopsy, the epithelial component is more prominent, with lactational changes (vacuolization of lobuli and secretions in the lumina), (H&E, 10×. H&E, 200×).
The mass was easily excised with an overhead para-areolar incision with a small extension to the lateral side (Fig. 4). A redon drain was left behind, which was removed the following day. No signs of inflammation were seen during surgery and no antibiotics were given. The condition of the 30-weeks-old fetus was monitored by cardiotocography before and after surgery. No effects on the fetus were observed and the patient was discharged. The patient recovered well and a good cosmetic result was accomplished at the right breast (Fig. 5). The pregnancy was completed without any complications.
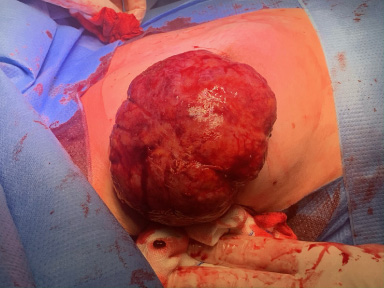
Excision of the tumor.
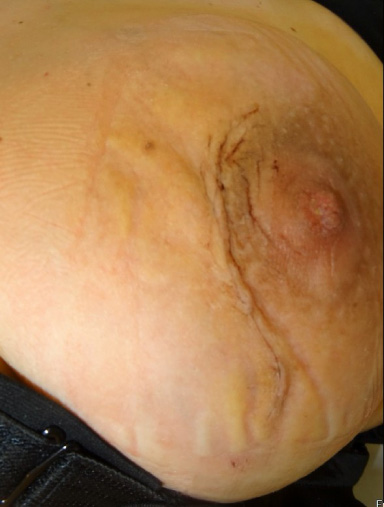
Postoperative outcome.
At pathological macroscopic examination, a nodular homogenous grey-brown encapsulated lesion with a size of 12 × 10.5 × 6.5 cm was determined (Fig. 6). Microscopically, lobular structures with proliferation of glandular ducts were seen, embedded in low stromal cellularity. Lactational changes were present in the epithelial component. The stroma component was homogeneous, composed uniform spindle cells without nuclear atypia nor mitotic figures. In some areas, the stroma showed mucoid changes. The ducts were partly cystically dilated. Focally, cysts exceeded the diameter of 3 mm (Fig. 7). No other features of a complex fibroadenoma were seen. Therefore, the tumor was diagnosed as a complex giant fibroadenoma with lactational changes.
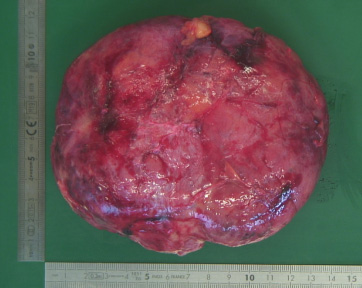
A macroscopic picture of the excised tissue.
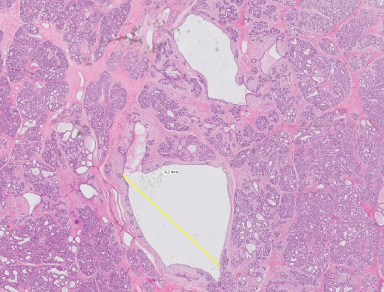
In the excised tumor, the epithelial component is more pronounced than the stromal component, with cysts measuring 4 mm diameter (H&E, 10×).
Nine months after surgery, ultrasonic examination of the right breast was repeated, showing no recurrence or rest abnormalities.
Fibroadenomas are the most common solid breast tumors in young female patients, and are found in up to 20% of biopsies [13]. They are classified as giant when the diameter exceeds 5 cm and the weight exceeds 500 grams. A subset of fibroadenomas are classified as ‘complex’ if at least one of the next pathologic characteristics are present: epithelial calcifications, sclerosing adenosis, papillary apocrine metaplasia and cysts >3 mm [2,5,9,14]. Giant fibroadenomas are rare and only a few case reports have been written about these fibroadenomas in pregnant women. In this case report, a pregnant patient is presented with a rapidly growing fibroadenoma that met the criteria of a giant and a complex fibroadenoma, leading to challenges in clinical decision making.
The diagnostic process of fibroadenomas can be difficult. Complex fibroadenomas cannot be distinguished from non-complex fibroadenomas based on clinical or radiologic findings [2,9]. However, the diagnostic procedure is more challenged by the overlap in characteristics of giant fibroadenomas and phyllodes tumors. This distinction is essential because a conservative treatment is justified in fibroadenomas, while phyllodes tumors should be excised with wide clear margins due to the possibility of development of malignant recurrent disease [8,10]. In general, phyllodes tumors occur in older patients than fibroadenomas. These tumors are rare and are only present in 1% of all breast tumors and 2–3% of all fibroepithelial tumors [15,16].
Patient's history often reveals a rapidly growing unilateral palpable breast mass. However, there are no clear criteria defining a fibroadenoma as ‘growing’ or ‘rapidly growing’ [17]. The growth of fibroadenomas is often biphasic where a lesion that has been stable for years, suddenly exhibits rapid growth. The tumor of the patient in our case showed fast growth but the patient reported no previous lesion. An ultrasound was ordered by her general practitioner, which showed a large homogeneous hypoechoic solid lesion. Needle biopsy was conducted immediately in accordance with practice guidelines at our hospital. Histology showed no signs of hypercellular stromal fragments and indeterminate features, and the diagnosis of phyllodes tumor could be rejected based on the Paddington Clinicopathologic Suspicion Score [16,18].
Complex fibroadenoma are associated with a two to three fold increased risk of developing invasive breast cancer [13]. This is a higher risk than in non-complex fibroadenomas [5,9]. However, recent studies have shown that the increased risk is caused rather by proliferative and atypical hyperplastic aspects in the fibroadenoma or a significant positive family history for breast cancer than the presence of a (giant) fibroadenoma [19].
Treatment for non-giant sized fibroadenoma is either conservative or surgical excision. Treatment choice is based on age, family history for (breast) cancer, proliferative characteristics of the adenoma and patient’s preference. For giant fibroadenomas, surgery should always be considered due to asymmetry or deformity of the breast, and due to the possibility of a malignancy. However, surgical options vary and there are no clear guidelines on the recommended practice. A review of Sosin et al. that examined surgical techniques for giant juvenile fibroadenoma, showed that all surgically treated tumors underwent excision and a small minority underwent mastectomies [20]. Preserving the breast parenchyma and nipple areolar complex is one of the main goals in breast surgery. Although there is a defect at the former place of the excised giant fibroadenoma, reconstructive surgery is rarely indicated [7]. As in our patient, the compressed breast tissue tends to expand to fill the defect. Literature about the management of complex fibroadenomas is scarce and its management varies among surgeons. Nevertheless, in case of a rapid growth of a breast mass it is paramount to exclude a (pre)malignant lesion.
There is no consensus on the best treatment in pregnant women, and, in particular, the best timing of surgery remains unclear. Surgery could be postponed because of the peri-operative risks for the mother and fetus, the benign character of fibroadenomas and because of the possible shrinking of the fibroadenoma postpartum [21–23]. However, a surgery could be justified in pregnant women if a malignant lesion cannot be excluded or the breast mass jeopardizes normal breast tissue by rapid expansion [21–25].
The recurrence rate for giant fibroadenomas is considered to be low, although individual cases have been described [26–28]. Progesterone seems to have a greater influence on the growth of fibroadenomas than estrogen [29]. Hypothetically, a new rise in hormone levels caused by a subsequent pregnancy could induce remaining fibroadenoma tissue to grow. However, since most fibroadenomas form during menarche [4] due to hormone induced changes in breast tissue, it is unlikely that a significant secondary (giant) fibroadenoma will form in our patient.
Conclusion
A rare case of a pregnant woman with a rapidly growing complex giant fibroadenoma is described. Surgical excision of these breast lesions may be justified, even during a pregnancy, in case of rapid growth, signs of compression and/or asymmetry of the breast.
Footnotes
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
The patient gave an informed consent for publication of this case report including the images.
Conflict of interest
The authors declare that they have no conflict of interest.
Author contributions
I.M. Brouwer de Koning: writing-original draft and editing. H.C. van Heusden: writing-original draft and editing. S.J.J. Mol: writing-original draft and review. M.L. Rots: direct patient care, conceptualization, writing-review. W.A. Draaisma: writing-review, supervision. K. Bosscha: direct patient care, conceptualization, writing- review, supervision.
