Abstract
INTRODUCTION:
Desmoid fibroma (DF) is a disorder characterized by strong clonal proliferation of myofibroblasts and fibroblasts. We describe a case of DF that mimicked a breast tumor, along with a review of the literature on the clinical manifestation, diagnostic process, and course of therapy for this combative disease.
CASE REPORT:
A 34-year-old female patient with breast lump at the junction of the upper quadrants of the left breast. After the diagnosis of DF, it was decided to perform a sectorectomy of the left breast associated with post-quadrant reconstruction, with immunohistochemistry and findings compatible with DF.
DISCUSSION:
Clinically manifests as a solid mass that is often painless and occasionally adherent to the chest wall. A treatment strategy should be idealized for each patient. Thus, there is the possibility of performing radical surgery for resection and/or radiotherapy, and surgery may be followed by radiotherapy.
Keywords
Introduction
Desmoid fibroma (DF) is a condition of clonal proliferation of fibroblasts and myofibroblasts, which occurs aggressively in mainly muscle, fascia and aponeurosis. Its reported incidence is about two to four cases per million people per year, corresponding to approximately 0.3% of all solid tumors. The DF also has association with familial adenomatous polyposis (FAP) and Gardner’s Syndrome, which represents almost 85% of cases [1,2]. Also DF has a high chance to develop after local trauma, such as surgical scars, especially after cesarean sections and abdominal resections [3]. However it rarely presents in an extra abdominal manner, especially in the breast region, representing only 0.2% of breast tumors, as described in this case [4,5]. They are more frequently observed in women than in men, with a 2:1 preference for women, mainly between 25 and 35 years old [6–8].
Among the first steps of management and diagnosis is a detailed anamnesis physical examination, looking mainly for history of axillary lymph node, trauma, previous surgeries and personal or family history of FAP or Gardener’s Syndrome [1,6,7].
The most common clinical presentation of the desmoid fibroma is a painless mass [3], which can probably lead to undiagnosed cases. Even though the DF presents a low probability of metastases, this pathology has a high chance of recurrence and local invasion [1,3], requiring its early diagnosis for proper management.
Here we report a case of DF that simulated a breast neoplasm, together with a literature review about the clinical presentation, diagnostic and treatment approach of this aggressive subject.
Materials and methods
This study was approved by the Research Ethics Committee of the University of Fortaleza (CAAE 50617421.6.0000.5052), and the patient provided written informed consent for collection and publication of clinical and molecular data.
Case report
A 34-year-old female patient, pre-diabetic, with celiac disease, complained of a lump in the intersection of the upper quadrants in the left breast that was painful on palpation for 7 months. Reported relief after the period, without aggravating factors and previous treatment and also increasing 2 cm in the last 2 months. Upon examination: breast lump at the junction of the upper quadrants of the left breast, with slight bulging of the skin on inspection, without changes in its color, painful, slightly adherent to deep planes, firm, with irregular contours, about 4 cm and no palpable axillary lymph nodes. She denied self-examination of the breasts and papillary discharge. Menarche at age 12, G4P2C2A2, had her first childbirth at age 16, and breastfed her 2 children until 1 year and 8 months. She used oral contraceptives for less than 2 years and denied a personal history of trauma, breast or chest wall surgery, and a family history of breast cancer or other sites.
She brought along a breast ultrasound requested during a routine consultation that identified a solid, ellipsoid, spiculated nodule with imprecise limits, with the longest axis parallel to the skin, emitting posterior acoustic shadowing, located in the left breast, at the transition from the medial quadrants, 9 o’clock axis (Fig. 1). Furthermore, it measured 1.9 × 0.9 × 1.6 cm, 1.6 cm from the skin and 6 cm from the nipple, and had no vascularization by Doppler, being classified as BI-RADS US 4. Although the main diagnostic hypothesis was low-grade fibromyxoid sarcoma, the differential diagnoses of the tumor according to tissue analysis were desmoid fibromatosis, solitary fibrous tumor, and spindle-shaped variant well-differentiated liposarcoma.
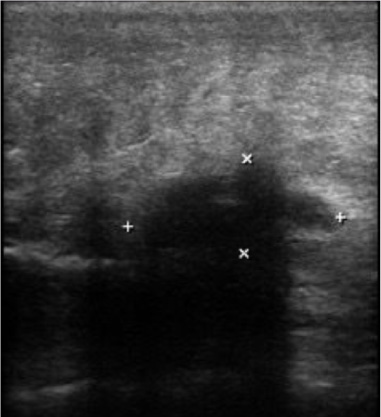
Ultrasonography demonstrating a solid lump in the left breast measuring 1.9 × 0.9 × 1.6 cm and 6 cm from the nipple.
Approximately 20 days later, a magnetic resonance imaging (MRI) of the breast, another breast ultrasound, and preoperative tests were performed. The MRI found an irregular lesion in the left breast (Fig. 2), measuring 2.6 × 2.2 × 1.9 cm, 9.5 cm from the papilla, with no signs of enhancement in the pectoralis muscle fibers (BI-RADS 5). Located medially to the dominant lesion, there was a non-neutral, focal enhancement in medial upper quadrant. A new breast ultrasound revealed the same findings as the first, with no progression of the lesion.
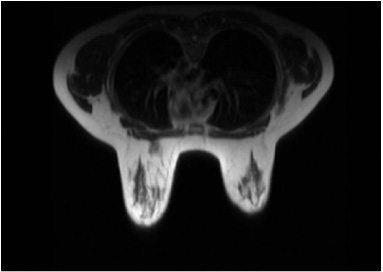
Magnetic resonance imaging indicating an irregular lesion in the superomedial quadrant of the left breast measuring 2.6 × 2.2 × 1.9 cm, 9.5 cm from the papilla.
We opted for a sectorectomy of the left breast associated with post-quadrant reconstruction. During the surgery, an infiltrative lesion was seen up to the pectoralis major muscle about 8 cm in the upper medial quadrant of the left breast. Two fragments were removed with characteristic yellowish, irregular fibroadipose and elastic features at macroscopy, whose measurements are 4.7 × 4.5 × 2.6 cm and 4.0 × 3.3 × 2.8 cm, respectively. Histopathology described spindle cell mesenchymal neoplasm with infiltration of adipose tissue, breast skeletal muscle tissue and surrounding ducts and breast lobules (Fig. 3). Presence of 3 mitosis figures/50 CGA, extravasation of red blood cells, some intralesional collagen bands, rare intratumoral mast cells, lipoblastic-like cells in the lesion, predominantly in the myxoid component. Absence of lymphoid aggregates peripherally or within the lesion and no hemangiopericytoma vessels or hyalinization in the vessel wall.
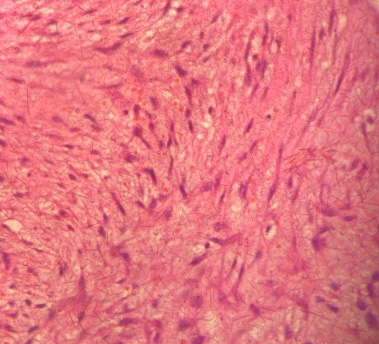
Microscopic characteristics of histopathology in the center of the lesion showing parallel collagen fibers and moderately cellular neoplasm with spindle cells arranged in long fascicles associated with a myxoid component hematoxylin-eosin staining.
Therefore, the neoplasm carried differential diagnoses such as desmoid fibromatosis, solitary fibrous tumor, solitary fibrous tumor, low-grade fibromyxoid sarcoma and well-differentiated liposarcoma fusiform variant. Immunohistochemistry was recommended as a diagnostic aid, which revealed the expression of smooth muscle actin, desmin and beta-catenin 1 (Table 1; Fig. 4), these findings being compatible with desmoid-type fibromatosis and indicating a segmental resection.
Immunohistochemical study of lump in the left breast at 12 h ray
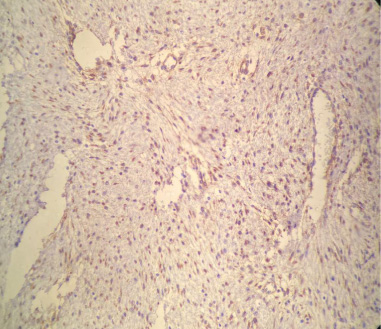
Immunohistochemistry expression of beta-catenin 1 in the lesion.
Now on she continues the follow-up once in a semester, maintaining asymptomatic after 2 years and 4 months of the surgery, without new lumps or disease relapse. The patient submitted to one MRI 6 months after the sectorectomy and an ultrasound of the breasts annually. No new findings were described.
In this respect, DF clinically presents as a firm palpable mass, usually painless, and may sometimes be adhered to the chest wall or even associated with skin retraction [10].
Following the imaging tests, mammography does not have great specificity, sometimes mimicking breast cancer, as in our patient. As well as MRI, which can have an auxiliary value, allowing in some cases a better definition of the size and type of tissue, however without absolute diagnostic value [10,11].
The diagnosis can be further aided with the help of a core, thick-needle biopsy, which still are not always successful in the differential diagnosis of mesenchymal tumors. However, the diagnosis can be made by hematoxylin and eosin staining of sections, which usually shows no malignant findings. It can also be aided by immunohistochemical staining for b-catenin, with nuclear positivity. However, there are no specific methods for identification of mammary DF [11].
Following the anatomopathological, macroscopically, it is characterized by a whitish or grayish stained, circumscribed mass with ill-defined borders. From the microscopic point of view, it presents parallel spindle cells, with scant cytoplasm, besides not having cells of the mammary parenchyma inside [12,13]. The study by Rosen et al. identified that some cases can present extensions in the fat near the glandular parenchyma, which can raise the rate of recurrences [12].
As for the risk factors of these tumors, studies have found that Familial Adenomatous Polyposis (FAP) and Gardner Syndrome (GS) may increase the risk of developing DF due to mutation of chromosome 5q21-22, which is responsible for the regulation of Beta-catenin, and may generate fibromatosis [14]. As for breast implants, especially silicone implants, it is believed that the fibrous capsules formed by the implant may trigger the formation of the tumor [15,16].
The study conducted by Ghanta et al. in 2019 proposed a way of classifying mammary desmoid fibromas into primary and secondary, in which the primary ones are related to genetic alterations, such as the cases already mentioned related to PAF and SG, and the secondary due to trauma, surgical procedures at the site, radiation, and silicone (mainly) or saline implants [12].
Treatment in most cases, as in the aforementioned case, is performed by total and wide resection of the tumor, involving the muscle fascia and often associated with a partial reconstruction of the pectoralis musculature, if necessary [4,14]. The use of ultrasound during surgery can be of great value in evaluating the extent of the tumor and defining the margins [9]. There is also the Duazo-Cassin et al trial, conducted in France, which raises the possibility of performing an active surveillance strategy, by means of MRI, indicating surgery only in cases that are really necessary. This study demonstrated a regression/stabilization rate of 88.2% [9].
In some specific cases, such as in young patients, as cited in Scheer et al., the drug approach may be an option, although less effective and riskier. This therapy can be done from cytotoxic agents such as Methotrexate or Vinblastine, cytostatic agents such as NSAIDs, hormonal agents and interferon-alpha or biologic agents such as Imatinib [10,14]. The adjuvant approach, on the other hand, can be performed when surgery would cause high morbidity, recurrence, or when margins remain affected [14]. In the article by Ghanta el al, radiotherapy demonstrated a reduction of up to 25% in local recurrence, and is strongly indicated when surgical resection is not possible [11].
Furthermore, in view of the high recurrence of 23–39% of cases in the first 3 years after excision, the follow-up of these patients should be closer during this period [14]. According to the prospective study by Grubstein et al, follow-up should start after 8–12 weeks, then it should be performed every 3 months in the first year, after that every 6 months until the 5th year, after that it should be yearly [11,17].
Footnotes
5.
The authors declare that there are no conflicts of interest.
Author contributions
Conception: Maria Stella Vasconcelos Sales Valente.
Interpretation or analysis of data: Bárbara Bezerra Ricciardi.
Preparation of the manuscript: Fábio Augusto Xerez Mota, Enzo Studart de Lucena Feitosa, Bárbara Matos de Carvalho Borges.
Revision for important intellectual content: Pedro Lucena de Aquino, Maria Stella Vasconcelos Sales Valente.
Supervision: Paulla Vasconcelos Valente.
