Abstract
Thoracoabdominal (TA) flaps are a good option for primary closure of small and medium defects after mastectomy for locally advanced breast tumours. Although they have a higher rate of necrosis than myocutaneous flaps, they can be easily performed by breast surgeons. Few studies on this procedure have been reported, and we have been unable to identify any prior publications reporting breast reconstruction with TA flaps.
Introduction
Locally advanced breast cancer (LABC) continues to be a challenging disease in clinical practice. When undergoing surgical treatment, many patients with this condition will need either myocutaneous or thoracoabdominal (TA) flaps to close the thoracic defect after mastectomy [1–3]. There are few publications describing the use of TA flaps for this condition, with only 15 studies describing 310 cases [1–3]. In addition, due to the unfavourable behaviour of LABC, there is a low probability of late secondary breast reconstruction [3].
Breast reconstruction (BR) after mastectomy improves the patient’s quality of life [4], and when clinical conditions allow this procedure, it should be offered to patients with breast cancer. However, reports of BR for patients with LABC who have undergone previous mastectomy with TA flaps are scarce [1,5], and no previous publication has reported secondary breast reconstruction with a second flap.
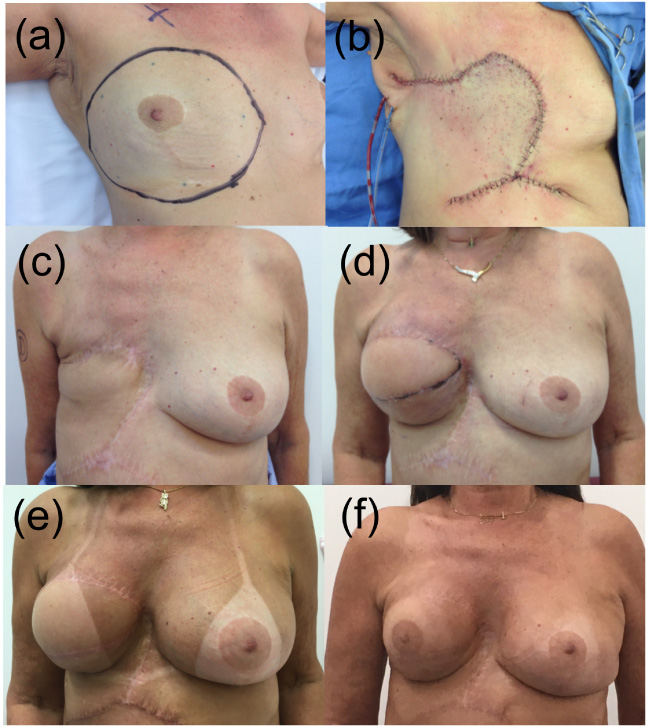
Case evolution: (a) Breast resection area; (b,c) ITADE flap; (d,e) Reconstruction with the latissimus dorsi; (f) Tattoo.
We present a 69-year-old female patient with secondary inflammatory LABC. The tumour measured 7.5 × 7.0 cm and was associated with Peau d’orange (20.0 × 12.5 cm) and an axillary tumour block (5.0 × 2.5 cm). Histological analysis revealed that it was a ductal invasive carcinoma, luminal B, Her2 negative tumour (Ki67 70%), clinical stage IIIb (T4bN2M0). The initial tumour area was marked. She received neoadjuvant ACT (adriamycin/cyclophosphamide + Taxol®/paclitaxel) with a partial clinical response and underwent modified radical mastectomy (MRM) with wide skin resection (covering the entire area preneoadjuvant chemotherapy, Fig. 1a). To close the defect in the chest wall, a surgical oncologist used an Ipsilateral ThoracoAbdominal DErmofat (ITADE) flap [1] (Fig. 1b and 1c; Fig. 2). Pathologic evaluation of the resected specimen reported a partial reduction (tumour 3.6 × 2.4 cm, 1/14 lymph nodes), representing a yT2N1M0 tumour. Adjuvant radiotherapy was performed in the plastron, armpit, and supraclavicular fossa. Hormone therapy with anastrozole was initiated and planned for 5 years.
She expressed a desire for breast reconstruction 17 months after the initial surgical treatment. In the absence of comorbidities and disease recurrence, breast reconstruction was performed with a myocutaneous flap of the latissimus dorsi (LD) muscle and a 330 cc silicone implant on the right side (Fig. 1d). At 25 months, she underwent contralateral symmetrisation and additive mammoplasty with a retroglandular silicone implant measuring 125 cc (Fig. 1e). Reconstruction of the right areolar complex was performed using a 3D skin tattoo 49 months after initial treatment (Fig. 1f).
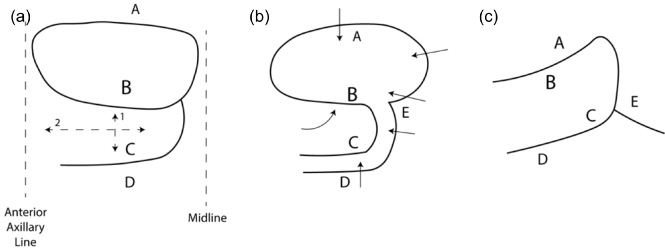
ITADE flap representation: (a) flap design; (b) flap detachment; (c) final results. Arrows: flap dissection and rotation. Letters: reference points.
Currently, at the 70-month follow-up, 53 months after reconstruction, there is no evidence of oncological disease, and a certain degree of atrophy of the LD has been observed (Fig. 1f), but the patient has expressed satisfaction with the final cosmetic result.
Thoracoabdominal flaps allow skin closure of large mastectomy defects after LABC resection. Although the risk of skin necrosis is higher after TA flap use, a team with experience in breast reconstruction is not needed, and it can be easily performed by surgical oncologists or breast surgeons [3]. They can be used to close small and medium defects, complementing myocutaneous flaps, which are used to close medium and large defects [3]. A plastic surgeon or a surgeon with specialized training in oncoplastic surgery is necessary to close a defect with a myocutaneous flap.
Recently, an ITADE flap was described for closing large skin defects, and although it has a higher risk of skin necrosis, it provides extra time before secondary reconstruction. The ITADE flap technique was described in a previous study [1]. Briefly, the ITADE flap consists of a random flap with blood supply based on posterior intercostal, subcostal and lumbar vessels. The upper limit is the lower border of the mastectomy, the medial limit is 1 to 2 cm from the midline, and the lower limit depends on the area between the anterior axillary line and the medial limit but should be determined at a minimum proportion of 2:1 (2:1 or more). The flap is performed with upwards rotation [1] (Fig. 2). In our service, the ITADE flap is the flap of choice for closing small and medium defects after mastectomy. For large defects, we use a myocutaneous flap, and the LD is the initial option, but for larger defects, we use a modified external oblique myocutaneous flap [2,6]. At the time of initial surgical treatment of the present case, our team was composed of 4 surgical oncologists and 2 breast surgeons, and a specific plastic surgery team was not available for support. ITADE flaps can be used to close large areas, similar to myocutaneous flaps, but can be performed by a breast surgeon, although it is associated with higher rate of tip necrosis [1]. In many parts of the world, a team of plastic surgeons is not available to support advanced cases, for whom the ITADE flap is a feasible option initial for skin closure.
Patients who need flaps for skin closure have larger tumours and a higher risk of local recurrence than patients who undergo mastectomy [3]. These flaps are only used for skin closure, and aesthetics are a secondary consideration after cancer survival. In the present case, the tumour resection was extensive, requiring extra skin for primary closure of the resection area. The surgeons opted for primary closure using the ITADE flap technique. Another option would be the use of the LD muscle for primary skin closure, making this flap unfeasible for future use. We had skin limitations, and primary reconstruction with the LD muscle and a breast expander was not feasible. The absence of local recurrence one year after radiotherapy and the desire of the patient led us to perform reconstruction. By controlling the disease, the patient will have time for future reconstruction.
The use of thoracoabdominal or myocutaneous flaps in the primary closure of postmastectomy defects tends to be reserved for special cases, with the goal of locoregional control. The risk of recurrence is high [3], and our primary goal is locoregional control alongside adjuvant radiotherapy. Unfortunately, we were unable to find support in the literature for primary breast reconstruction with flaps when they are necessary for skin closure, after extensive defects.
There are very few cases in the literature of patients with LABC treated with TA flaps for closure of the postmastectomy thoracic defect [1–3]. Even rarer is breast reconstruction of patients initially treated with TA flaps [7]. This patient was initially treated with an ITADE flap, which enabled the future use of an LD myocutaneous flap during breast reconstruction. Care was taken not to compromise the intercostal/subcostal vessels that direct served as the blood supply for the ITADE flap.
The rarity of the use of the ITADE flap, the absence of recurrence and a favourable oncological and physical condition allowed breast reconstruction to be performed for this patient, who was satisfied with the final result. There was only one reconstruction performed after the TA flap, which involved use of a prosthesis [7]. It is important to describe this new option because more patients are surviving, and we have to be able to offer them breast reconstruction options. The ITADE flap allows the closure of extensive defects, eliminating the need to use the LD muscle in the first stage and allowing it to be used for breast reconstruction with a secondary myocutaneous flap, a procedure described for the first time in this publication.
Footnotes
Ethical approval
Written informed consent was obtained from this patient for publication of this case report and any accompanying images. This study was approved by the Research Ethics Committee of the Barretos Cancer Hospital under number 2222/2021. It was conducted in accordance with the Helsinki Declaration.
Conflict of interest
The authors declare no conflicts of interest.
Funding
Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPQ) supported this study.
Author contributions
Study concepts: Vieira RAC, de Oliveira-Junior I; Study design: Vieira RAC, de Oliveira-Junior I; Data acquisition: Vieira RAC, de Oliveira-Junior I; Data analysis and interpretation: All authors; Manuscript preparation: All authors; Manuscript editing: All authors; Manuscript review: All authors.
